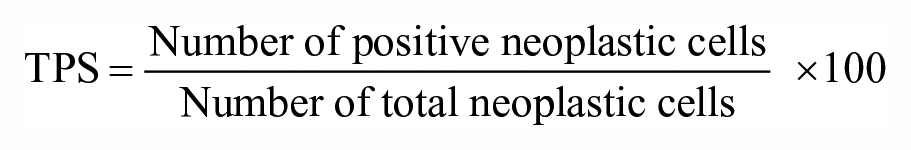Abstract
Immuno-oncology research has brought to light the paradoxical role of immune cells in the induction and elimination of cancer. Programmed cell death protein 1 (PD1), expressed by tumor-infiltrating lymphocytes, and programmed cell death ligand 1 (PDL1), expressed by tumor cells, are immune checkpoint proteins that regulate the antitumor adaptive immune response. This study aimed to validate commercially available PDL1 antibodies in canine tissue and then, applying standardized methods and scoring systems used in human pathology, evaluate PDL1 immunopositivity in different types of canine tumors. To demonstrate cross-reactivity, a monoclonal antibody (22C3) and polyclonal antibody (cod. A1645) were tested by western blot. Cross-reactivity in canine tissue cell extracts was observed for both antibodies; however, the polyclonal antibody (cod. A1645) demonstrated higher signal specificity. Canine tumor histotypes were selected based on the human counterparts known to express PDL1. Immunohistochemistry was performed on 168 tumors with the polyclonal anti-PDL1 antibody. Only membranous labeling was considered positive. PDL1 labeling was detected both in neoplastic and infiltrating immune cells. The following tumors were immunopositive: melanomas (17 of 17; 100%), renal cell carcinomas (4 of 17; 24%), squamous cell carcinomas (3 of 17; 18%), lymphomas (2 of 14; 14%), urothelial carcinomas (2 of 18; 11%), pulmonary carcinomas (2 of 20; 10%), and mammary carcinomas (1 of 31; 3%). Gastric (0 of 10; 0%) and intestinal carcinomas (0 of 24; 0%) were negative. The findings of this study suggest that PDL1 is expressed in some canine tumors, with high prevalence in melanomas.
Research in immuno-oncology has shaped the ontological approach to tumor progression, revealing the paradoxical role of immune system cells in both the induction and elimination of cancer. 3 Inflammation associated with neoplasms can contribute to genomic instability, tumor angiogenesis, epigenetic alterations, and increased proliferative and antiapoptotic activity. 3
Traditionally, cancer therapy research has deployed immunodeficient mice to engraft human cancer tissues. These models are primarily designed to investigate tumor targets, while minimizing the impact of the host immune system, such as innate and adaptive immunity in NOD/SCID (nonobese diabetic/severe combined immunodeficiency) mice. However, given that studies on immune checkpoint inhibitors aim to investigate host-tumor interactions, especially at the immune system–tumor interface, novel humanized mouse models have been developed to investigate this interaction. 33
In contrast, the utility of canine models of human cancers has been underestimated compared with mouse models, despite their potential advantages. The study of the interface between the host immune system and tumors, in spontaneous animal models that share high genetic homology with humans, 16 can more closely replicate human cancer. This approach also offers the benefit of parallel studies for tumor therapies in pets. The International Immuno-Oncology Biomarker Working Group has recently recognized the value of comparatively studying tumor-infiltrating lymphocytes and immuno-oncology in domestic animals as a spontaneous model for humans. 26
Programmed cell death protein 1 (PD1) is an inhibitory receptor belonging to the CD28 family and is mainly expressed in activated T lymphocytes. Its ligand, programmed cell death ligand 1 (PDL1), is expressed in various cell types, including cells of the hematopoietic system, which interact with T lymphocytes through the PD1/PDL1 immune checkpoint axis, inducing immune modulation and preventing an excessive effector response. 24 The same immune-evasion mechanism is exploited by tumor cells to silence the immune response.10,17
The Food and Drug Administration (FDA) has approved multiple tests to evaluate the expression of PDL1 for clinical use in humans. 6 PDL1, evaluated by immunohistochemistry as the gold standard test, is currently the biomarker that guides the selection of human patients eligible for anti-PD1 and anti-PDL1 therapy. 6 Anti PD1/PDL1 immunotherapy has dramatically increased the survival of patients with advanced pulmonary carcinoma, becoming the first-line strategy for non-small cell lung cancers without driver gene alterations.8,22 In addition, the advent of new checkpoint inhibitors (nivolumab and pembrolizumab) has significantly improved the outcome of patients with advanced melanoma, which, prior to the “immunotherapy era,” was considered to have a poor prognosis and was disappointingly unresponsive to conventional chemotherapy. 31 However, the use of immunohistochemistry or immunofluorescence for PDL1 as a biomarker for predicting response to therapy is affected by several critical points, such as the antibody used, the reference cutoffs, the sampling time, and the number of biopsy sites tested, considering the possible focal distribution of PDL1. 39
In dogs, PDL1 expression has been tested using different techniques (western blot, immunohistochemistry, RNA in situ hybridization, ELISA (enzyme-linked immunosorbent assay), and flow cytometry2,18,27,37) in different tumor types, such as melanoma, urothelial carcinoma, renal cell carcinoma, mammary carcinoma, hepatocellular carcinoma, squamous cell carcinoma, nasal adenocarcinoma, anal sac gland carcinoma, gastric adenocarcinoma, soft tissue sarcoma, osteosarcoma, histiocytic sarcoma, diffuse large B cell lymphoma, and transmissible venereal tumor.17,27 Pantelyushin and colleagues recently evaluated the cross-reactivity and functionality of FDA-approved human immune checkpoint inhibitors in dogs. 23 However, a lack of standardization in the diagnostic evaluation of PDL1 still exists. There is variability in the types of antibodies used and/or in the criteria used when evaluating labeling for PDL1 in canine tumors through immunohistochemistry.5,17,21,35 Achieving a uniform consensus in assessing cellular PDL1 immunolabeling, similar to what has been accomplished in human studies, is crucial.1,19 In human medicine, only membrane expression is considered positive and tumor cells with cytoplasmic granular and nuclear expression are considered background labeling and fixation artifacts.1,19,28 In human patients, the use of standard scoring systems that rely upon specific cutoff values for a specific tumor type and antibody clone have emerged as crucial parameters in consistently identifying positivity. Such uniform definitions are instrumental in determining the therapeutic efficacy of a given treatment and identifying cases that may be subject to ineffective therapy and its associated side effects.1,20
To standardize the preanalytical and analytical methods, this study aimed to validate the cross-reactivity of commercial anti-PDL1 antibodies in dogs and evaluate the immunohistochemical expression of PDL1 in different histotypes of canine tumors through the use of specific scoring systems.
Materials and Methods
Case Selection
Cases of pulmonary carcinoma, cutaneous and oral squamous cell carcinoma, mammary carcinoma, urothelial carcinoma, renal cell carcinoma, nodal lymphoma, and cutaneous or oral melanoma, collected from 2011 to 2023, in which FFPE (formalin-fixed paraffin-embedded) samples were available, were selected from the database of the Pathology Service of the Department of Veterinary Medical Science, University of Bologna; the Veterinary Pathology Unit of the Department of Agricultural, Food, Environmental, and Animal Sciences, University of Udine; and the Department of Veterinary Science, University of Turin. Hematoxylin and eosin sections were reviewed by two veterinary pathologists, one board certified (L.V.M.) and one an experienced pathologist (G.S.), to confirm the diagnosis.
These tumor types were selected because, in humans, they can express PDL1 and are considered for treatment with anti-PD1/PDL1 therapies. Both incisional and excisional biopsies were included, but tumors that did not have sufficient archival material for the study were excluded.
To evaluate PDL1 expression in normal tissues, the relevant human and murine literature was consulted,7,11,12,38 and it was found that PDL1 expression varies across different human tissues, with the highest levels observed in tissues involved in immunotolerance, the tonsil and placenta, which were used as positive controls to validate the method. Kidney was selected as the negative control, given the known negative expression in humans.11,37
Western Blot
Normal canine placental, tonsillar, and renal samples were used for western blot analysis, and two commercial antibodies were tested to demonstrate the cross-reactivity in dog tissues. The commercial antibodies used included a rabbit polyclonal anti-PDL1/CD274 antibody (ABClonal, catalog no. A1645) for which the immunogen used has a sequence identity of 80.54% between human and canine; and a mouse monoclonal anti-human PDL1 antibody (clone 22C3, DAKO Agilent Technologies, REF M3653), which is used in human pathology for the selected histotypes. 20 A rabbit polyclonal anti-GAPDH (glyceraldehyde-3-phosphate dehydrogenase) antibody that is specific for human, rat, and mouse (Sigma Aldrich no. 4595, Saint Louis, MI, USA) was used as a housekeeping protein loading control.
Tissue samples were weighed and mechanically processed in RIPA (radio immunoprecipitation assay) lysis buffer supplemented with protease inhibitor cocktail (catalog no. P8340, Sigma-Aldrich, 1:100) and 1 mM of Na3VO4. Protein extracts were quantified with DC Protein Assay (Bio-Rad, Hercules, USA), resolved using 15% gel electrophoresis, and transferred to nitrocellulose membranes. After blocking, membranes were incubated overnight with the indicated primary antibodies, followed by incubation for 1 hour with horseradish peroxidase–conjugated antirabbit secondary antibodies and probed through a chemiluminescent reaction with Amersham ECLTM detection reagents. Signals were detected using the ChemiDoc Imaging System (Bio-Rad) and images were acquired through ImageLab Software.
Immunohistochemistry
Immunohistochemistry was performed manually. Three-µm-thick sections were dewaxed in xylene substitute solvent and rehydrated. Endogenous peroxidase was blocked by immersion in 3% H2O2 diluted in methanol for 30 minutes. Antigen retrieval was performed using a pH-6.0 citrate buffer (Sigma-Aldrich, Merck KGaA, Darmstadt, Germany) heated for 10 minutes in a microwave oven at 750 W. Slides were then incubated for 10 minutes in a commercial blocking solution (Super Block, Scytek Laboratories, Logan, Utah, USA). Sections were incubated overnight at 4°C with antibody against PDL1 (PD-L1/CD274 Rabbit pAb, ABClonal, catalog no. A1645, dilution 1:800). Antibody binding was visualized using an anti-Polyvalent HRP Polymer (Scytek Laboratories, Logan, Utah, USA). The chromogen DAB (3,3′-diaminobenzidine 0.05% w/v, catalog no. ACB999, Histo-Line Laboratories, Pantigliate, MI, Italy) was used to visualize the reaction. Slides were counterstained with Harris’s hematoxylin (catalog no. 01HEMH2500, Histo-Line Laboratories). Normal canine tonsil and placenta were used as positive controls. For negative controls, the primary antibody was replaced with an irrelevant, isotype-matched antibody to control for the nonspecific binding of the secondary antibody (Rabbit IgG Isotype control, catalog no. 02-6102, Invitrogen-Thermo Fisher Scientific, Waltham, Massachusetts, USA).
Images were acquired with an optical microscope (Eclipse E600; Nikon, Shinjuku, Japan) equipped with the Imaging Source “33” Series USB 3.0 Camera (cat. no. DFK 33UX264; Bremen, Germany).
Immunofluorescence
To avoid interference of visualization of the reaction due to the presence of melanin, immunofluorescence was performed on melanomas instead of immunohistochemistry. Three-micron-thick sections of melanomas were dewaxed in xylene and antigen retrieval was achieved by using citrate buffer at pH 6.0 for 10 minutes in microwave 750 W. Blocking of nonspecific antigenic sites was achieved by incubating the slides in a solution of 3% of bovine serum albumin, 3% of fetal bovine serum, and 0.25% TritonX100 in phosphate-buffered saline for 1 hour at room temperature. Sections were incubated overnight at 4°C with the anti-PDL1 primary antibody (PD-L1/CD274 Rabbit pAb, ABClonal, catalog no. A1645) diluted 1:100. Detection of primary antibodies was visualized with a secondary goat antirabbit antibody conjugated to Alexa Fluor 555 (catalog no. ab150078, Abcam). Sections were counterstained with DAPI (4’,6-diamidino-2-phenylindole; catalog no. D9542-5MG) and mounted with Fluorshield histology mounting medium (catalog no. F6182-10MG, Sigma Aldrich).
Normal canine tonsil and placenta were used as positive controls. For negative controls, the primary antibody was replaced with an irrelevant, isotype-matched antibody to control for the nonspecific binding of the secondary antibody (Rabbit IgG Isotype control, catalog no. 02-6102, Invitrogen-Thermo Fisher Scientific). The slides were examined using a Nikon Eclipse Ni microscope equipped with the appropriate filter cubes to distinguish the fluorochromes employed. The images were recorded with a Nikon DS-Qi1Nc digital camera and NIS Elements software BR 4.20.01.
Scoring Systems
Approved criteria in human medicine, consisting of tumor proportion score (TPS) and combined positive score (CPS),1,32 were used to assess PDL1 expression. For both scores, neoplastic cells were considered positive only when they had membranous expression; cytoplasmic and nuclear expression was considered nonspecific in neoplastic cells. Cytoplasmic expression was only accepted in the tumor-infiltrating immune cells, along with the membranous expression, considering the high nuclear-to-cytoplasmic ratio for small lymphocytes. 1
TPS is the ratio of the number of positive neoplastic cells to the total number of neoplastic cells expressed as a percentage. Tumor-infiltrating immune cells are not included in the score:
CPS is the ratio of the number of total positive cells (including neoplastic cells and tumor-infiltrating immune cells) to the total number of neoplastic cells expressed as a percentage:
The glass slides were examined at low power (4× objective), and the areas with the highest density of tumor cells and tumor infiltrating immune cells (lymphocytes and macrophages) were selected. In the selected areas, the counts were performed at higher power (20× objective; field of view 1.1 mm diameter; area 0.950 mm2) in at least 500 total tumor cells, using the cell counter plugin in the ImageJ software (http://imagej.nih.gov/ij/). The scoring was performed by a single board-certified pathologist (L.V.M.). Any tumor with a TPS or CPS value >0% was considered positive.
Results
Antibody Specificity
By western blot, cross-reactivity with canine tissue was demonstrated for both antibodies tested (ABClonal and DAKO), with a specific band observed at the expected molecular weight of 40 kDa. However, signal specificity was higher for the polyclonal antibody (ABClonal, A1645, Fig. 1a) than for the monoclonal antibody (Dako, Clone 22C3), which showed several nonspecific background bands (Fig. 1b). Hence, the antibody A1645 was selected for the immunohistochemical and immunofluorescence analyses.
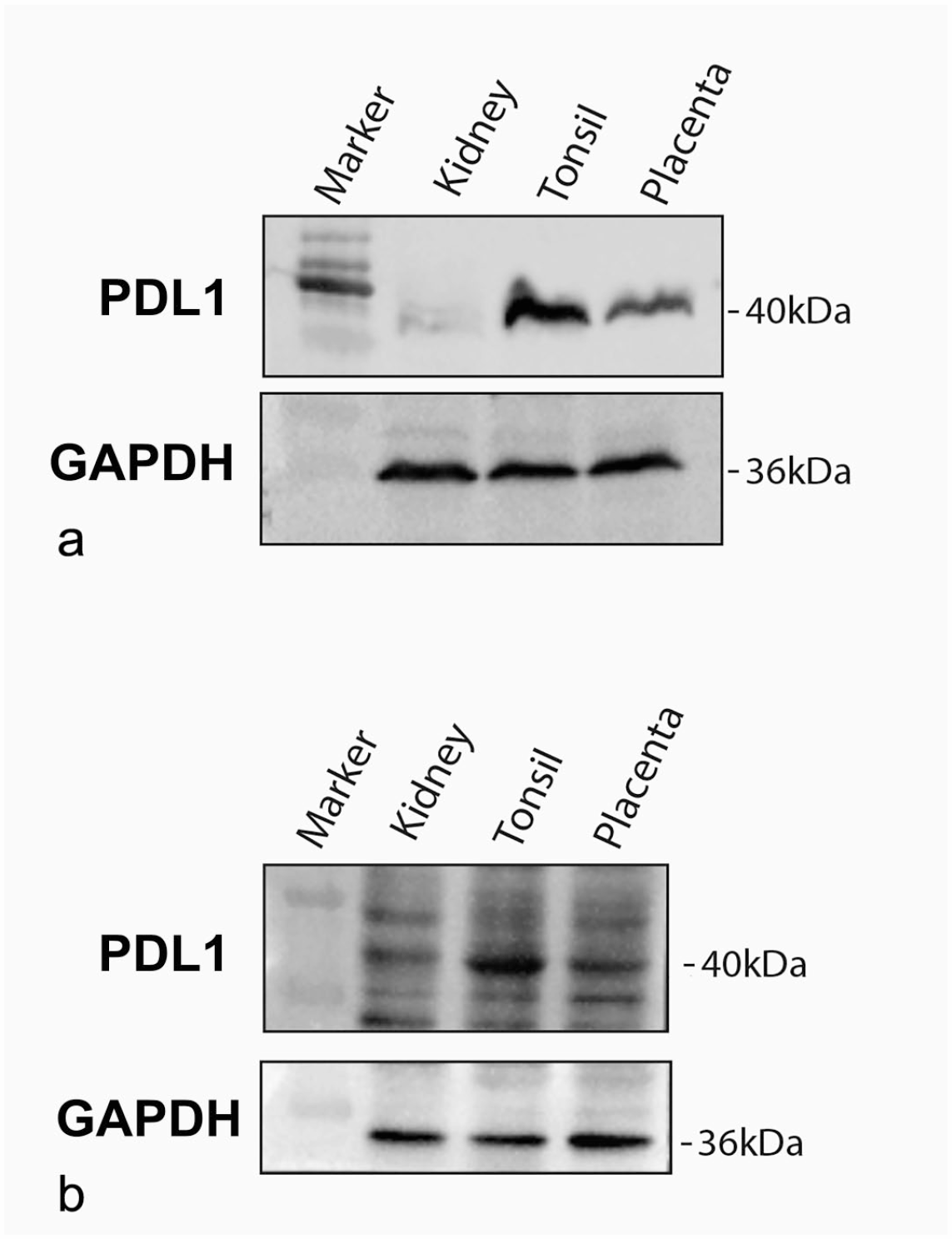
Western blots on tissue cell extracts of canine kidney, tonsil, and placenta probed for PDL1 protein with (a) ABClonal, and (b) DAKO antibodies. GAPDH was used as a housekeeping protein. PDL1, programmed cell death ligand 1; GAPDH, glyceraldehyde-3-phosphate dehydrogenase.
PDL1 Immunoexpression in Normal Canine Tissues
In normal canine tonsil, PDL1 was expressed in squamous epithelium with intense membrane labeling (Fig. 2a, b); furthermore, PDL1 expression was observed in lymphoid follicles with increased labeling in the cytoplasm of macrophages and centrofollicular lymphocytes (Fig. 2a). In the placenta, membranous immunopositivity was present in syncytiotrophoblasts and vascular endothelium (Fig. 2c, d).
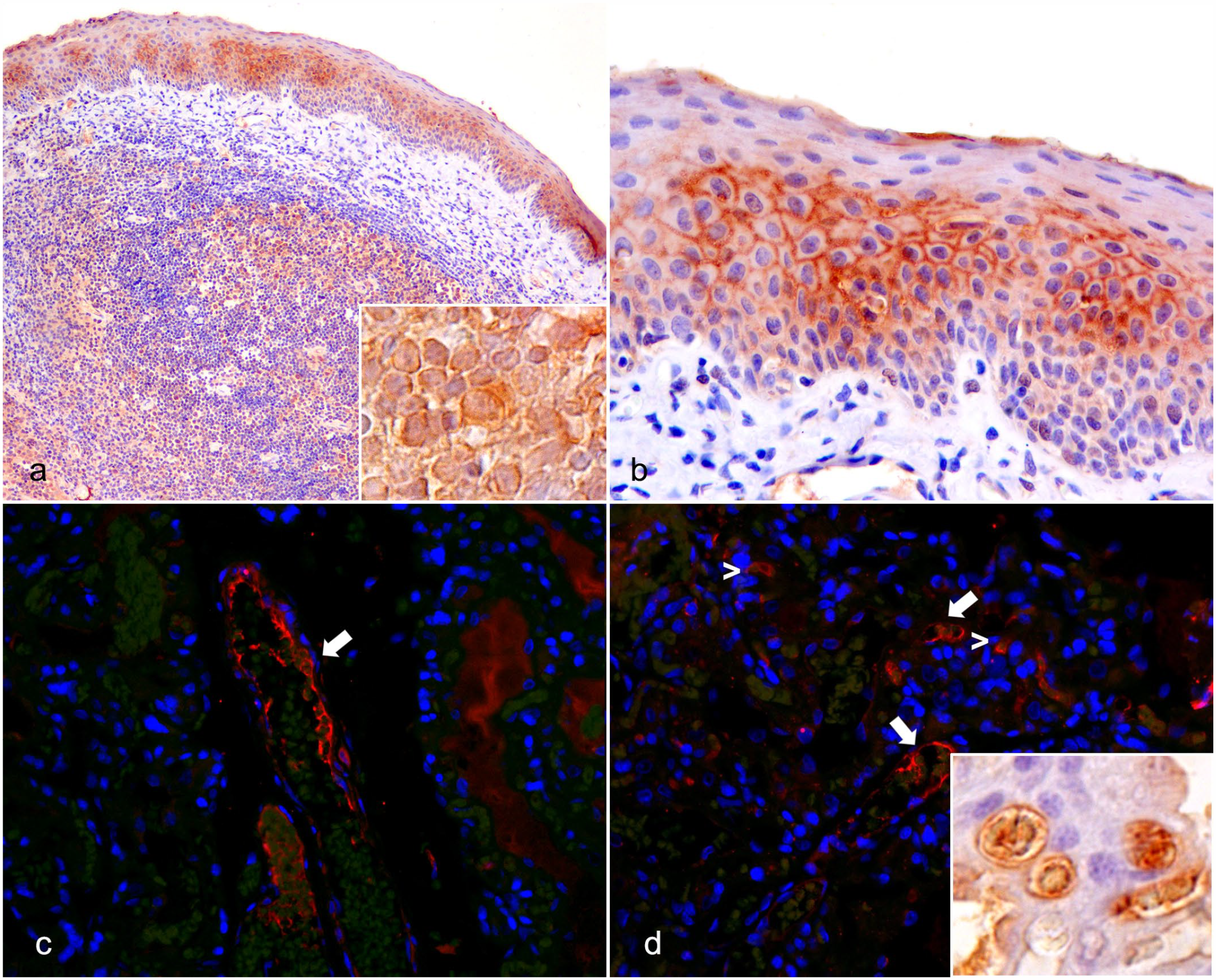
Immunoexpression of PDL1 in canine control tissues. (a) Tonsil. Labeling of PDL1 in overlying squamous epithelium. Inset: PDL1 labeling of centrofollicular lymphocytes and dendritic cells. Immunohistochemistry (IHC) for PDL1. (b) Tonsil. Detail of diffuse and intense membrane labeling in tonsillar squamous epithelium. PDL1 IHC. (c) Placenta. Membrane labeling in endothelial cells of medium-sized placental vessels (arrow). Immunofluorescence for PDL1 (red signal). (d) Placenta. Membrane labeling in endothelial cells of medium and small caliber placental vessels (arrows) and scattered syncytiotrophoblasts (arrowheads). There is green autofluorescence of erythrocytes in the vessel’s lumen. Immunofluorescence for PDL1 (red signal). Inset: detail of syncytiotrophoblast (right) and endothelial cells in small caliber vessels (left) using PLD1 IHC. PDL1, programmed cell death ligand 1.
PDL1 Immunoexpression, Subcellular Localization and Scoring Systems in Canine Tumors
A total of 168 tumors were examined, including 20 pulmonary carcinomas (13 papillary adenocarcinoma, 2 solid adenocarcinoma, 2 lepidic adenocarcinoma, 2 squamous cell carcinoma, and 1 adenosquamous carcinoma), 17 squamous cell carcinoma (13 cutaneous and 4 oral), 11 gastric carcinoma, 23 intestinal adenocarcinoma (16 small intestine and 7 colorectal), 31 mammary carcinoma, 18 urothelial carcinoma, 17 renal cell carcinoma, 17 melanoma (13 oral and 4 cutaneous), and 14 nodal lymphomas.
PDL1 expression was demonstrated by immunohistochemistry or immunofluorescence in the tumors (Fig. 3), albeit with variable frequency, both in tumor cells and tumor-associated immune cells. Melanomas showed the highest prevalence of PDL1 expression, with all examined cases (17/17, 100%) demonstrating multifocal and consistent immunofluorescence. The prevalence of PDL1 immunopositivity in melanomas was followed by that of renal cell carcinomas (4/17, 24%), squamous cell carcinomas (3/17, 18%), urothelial carcinomas (2/18, 11%), lymphomas (2/14, 14%), pulmonary carcinoma (2/20, 10%), and mammary carcinoma (1/31, 3%). On the contrary, gastric and intestinal carcinomas showed no PDL1 immunopositivity in any of the examined cases (0/10 and 0/24, respectively, 0%; Fig. 4).
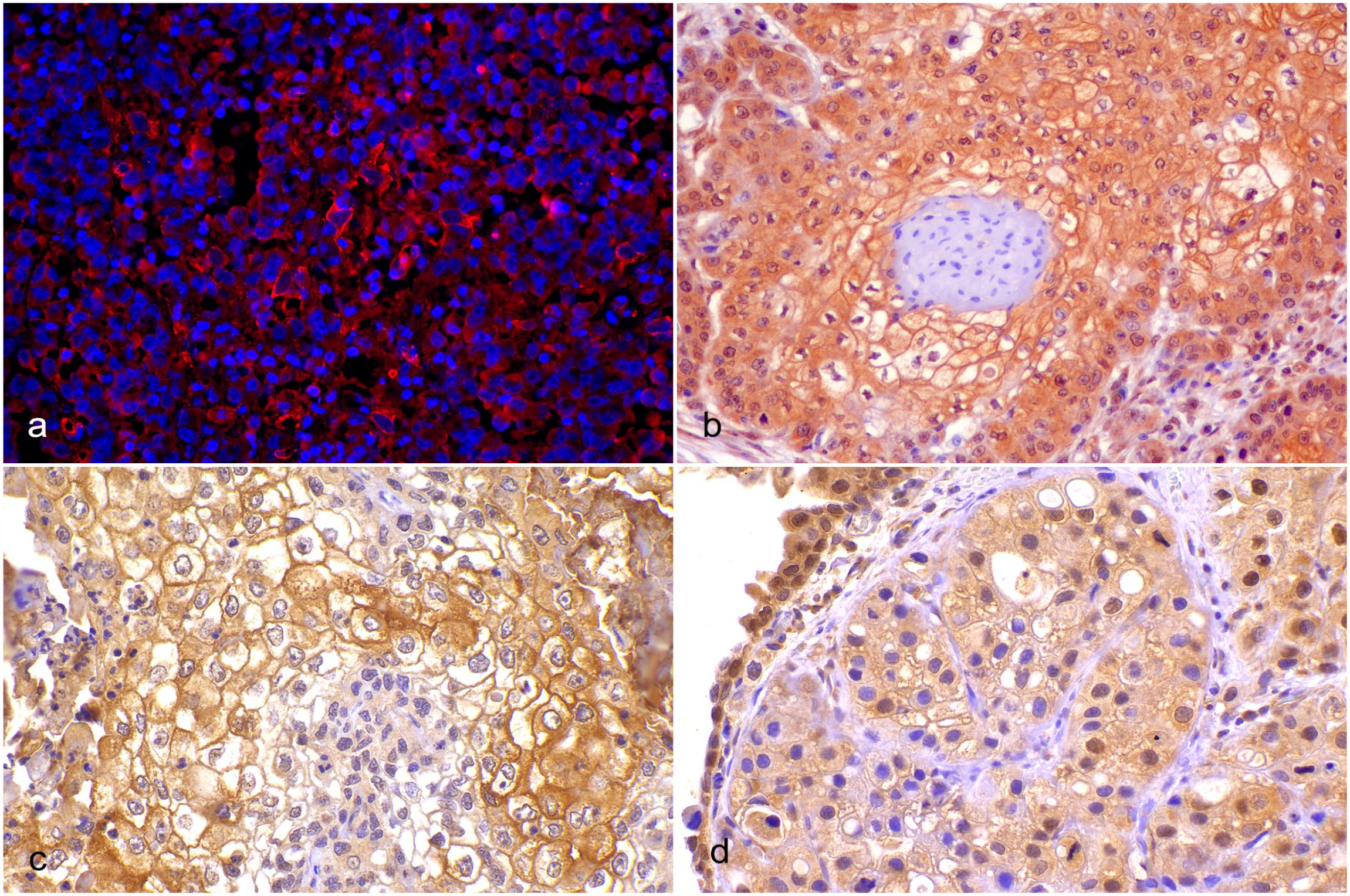
PDL1 expression in canine tumors. (a) Oral melanoma. Multifocal membranous PDL1 labeling in neoplastic melanocytes; nonspecific cytoplasmic labeling is also present. Immunofluorescence for PDL1 (red signal). (b) Cutaneous squamous cell carcinoma. Strong membranous PDL1 labeling in cells with squamous differentiation; nonspecific cytoplasmic labeling is also present. Immunohistochemistry (IHC) for PDL1. (c) Pulmonary squamous cell carcinoma. Strong membrane PDL1 labeling in neoplastic cells with squamous differentiation; nonspecific cytoplasmic labeling is also present. PDL1 IHC. (d) Urothelial carcinoma. Moderate membrane labeling in urothelial neoplastic cells; nonspecific cytoplasmic labeling is also present. PDL1 IHC. PDL1, programmed cell death ligand 1.
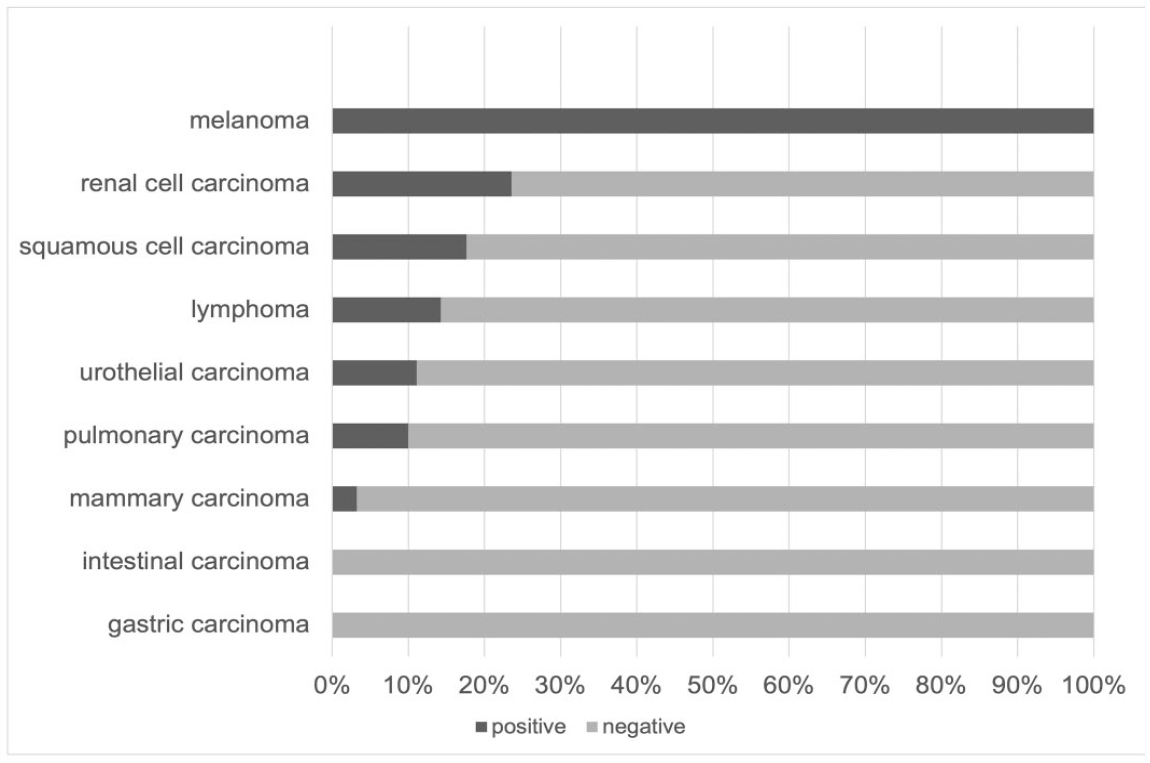
Horizontal histogram showing the percentage of positive and negative cases in the different tumor types examined.
PDL1-positive pulmonary carcinomas had a diffuse expression pattern in almost all tumor cells present. Immunopositivity in tumor-infiltrating immune cells was rare, as demonstrated by a small increase in the CPS compared with the TPS. PDL1 immunopositivity in the immune population associated with this tumor was limited to macrophages and was never present in lymphocytes. In squamous cell, mammary, and urothelial carcinomas, PDL1 immunopositivity was heterogeneous and multifocal. In these carcinomas, membranous to cytoplasmic labeling in the immune component was present in variable percentages of both lymphocytes and macrophages, with a moderate increase in the CPS compared with the TPS. In renal cell carcinoma, the expression pattern was multifocal in cell clusters to diffuse, but little to no inflammatory infiltrate was observed, with no increase in the CPS versus the TPS.
Melanomas and lymphomas had a multifocal, heterogeneous labeling pattern in single cells or in clusters. In these neoplasms, the CPS could not be performed. Due to the lymph node site for lymphomas, it was not possible to distinguish the immune cell population infiltrating the tumor from the resident one. For melanomas, it was not possible to morphologically evaluate the tumor-infiltrating immune cells using immunofluorescence.
The ranges of TPS and CPS by tumor type are summarized in Table 1.
Tumor proportion score (TPS) and combined positive score (CPS) of PDL1 in canine tumors.
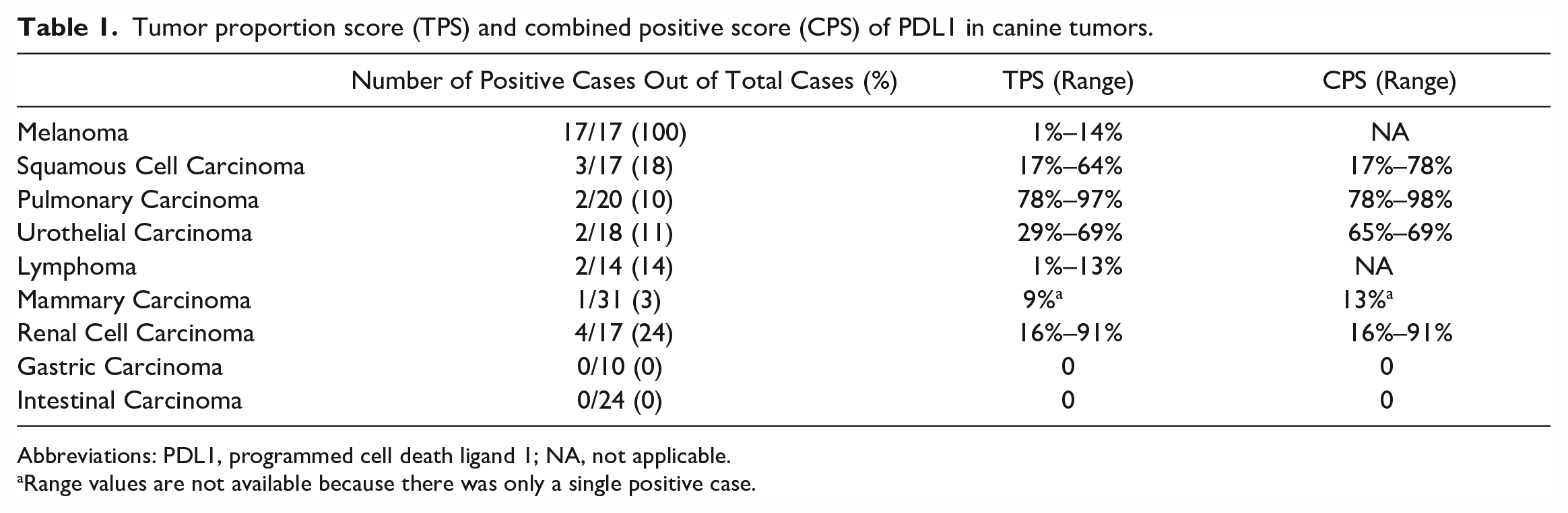
Abbreviations: PDL1, programmed cell death ligand 1; NA, not applicable.
Range values are not available because there was only a single positive case.
Discussion
Immunohistochemistry is currently the gold standard technique for evaluating PDL1 expression in human neoplasms. This method has been extensively studied and standardized, and, with the use of FDA-approved antibody clones, specific cutoff values have been defined, making it the most reliable evaluation system available. 20
The standardization of the method is crucial to obtain objective, reproducible, and comparable results and to lay the foundations for establishing specific cutoffs usable for therapeutic purposes. A fundamental factor in defining the reproducibility of results is the use of nonarbitrary evaluation systems and guidelines, thereby reducing differences in assessment methods among studies.18,21 In this work, we used the current human pathology evaluation systems. 1 TPS and CPS are scoring systems that provide quantitative data, which may prove helpful in future prognostic studies to establish specific cutoffs for therapeutic applications.
In published canine studies, custom antibodies from hybridomas are often used.18,21,35 Although this may be an advantage for the specificity of the reaction, with a view to future use for diagnostic purposes, it does not allow for their widespread availability. A possible alternative is the validation of commercial antibodies for specific use in canine tissues. To date, such commercial antibodies are not available; therefore, the first goal of this study was to validate commercial antibodies designed for human samples in the canine tissues. By western blot, the rabbit polyclonal antibody (PD-L1/CD274, ABClonal) showed cross-reactivity in extracts from canine tissues, with appropriate molecular weight, and excellent signal specificity in the positive controls by immunohistochemistry. In human pathology, the proper subcellular localization of PDL1 is highly stressed: only membrane expression should be considered as positive expression, considering that it is a transmembrane protein. 1 PDL1 immunohistochemistry can often result in cytoplasmic or nuclear labeling, which is considered nonspecific and inadequate for immunotherapy purposes. 1 Cytoplasmic labeling is accepted only in tumor-infiltrating immune cells. 1 In many of the studies previously investigating PDL1 expression in canine tumors by immunohistochemistry, cytoplasmic labeling was also considered positive, and a specific membranous subcellular localization was not considered mandatory.17,18,21,35 Conversely, in our study, we only considered tumor cells with membranous labeling positive, aligning with the evaluations used in humans. 1
A possible limitation of this study was that the scoring was performed by a single board-certified pathologist; future concordance studies are needed to evaluate differences at this assessment stage. In this study, the tumor that most consistently showed PDL1 positivity was melanoma, which labeled for PDL1 in all the examined cases, albeit its intratumoral labeling rate ranged from 1% to 14%. This is an expected result as melanoma is considered a highly immunogenic tumor with a good response to immunotherapy in human beings 29 and possibly in dogs. 17 Nevertheless, mechanisms of primary resistance to anti-PDL1 therapy have been recently reported in human patients. In primary resistance, the tumor shapes the immune response to prevent T cell activity, and this may occur through insufficient antigen immunogenicity, irreversible T cell exhaustion, the resistance of IFNγ, and the creation of an immunosuppressive tumor microenvironment. 15 Therefore, the study of PDL1 in the tumor microenvironment is essential for understanding the overall mechanism of immunomodulation.
The prevalence of PDL1 immunolabeling in melanoma was high in our study (100%) and consistent with that previously reported in canine melanoma (95%). 17 Furthermore, a high prevalence is reported in humans, ranging from 24% to 49%, with higher levels of expression in UV-induced melanomas.14,29,40 Recently, the efficiency of immunofluorescence detection of PDL1 in human melanoma has been demonstrated, specifically using the multiplex immunofluorescent technology, ensuring reproducible results compared with immunohistochemistry. 4 Nonetheless, it should be stated, considering the results obtained in melanomas in our study compared with other tumors, that immunofluorescence may have been more sensitive than immunohistochemistry in detecting the signal. In our literature searches, no studies using immunofluorescence for PDL1 detection in dogs have been reported.
PDL1 was also expressed in squamous cell carcinoma, urothelial carcinoma, mammary carcinoma, and lymphoma in this study, as previously reported in the literature on canine tumors.17,18,35 However, variations were noted in the proportions of positive cases among these different tumor types, and these differences may be due to the not necessarily membranous, subcellular localization in the previous studies. 17
In our series, only 18% of the squamous cell carcinomas tested were positive for PDL1, whereas 90% positivity is reported for canine squamous cell carcinoma in the literature. 17 In the human pathology literature, cutaneous squamous cell carcinomas show a variable percentage of PDL1 expression, ranging from 20% to 70%, which increases as the metastatic risk increases. 36 On the contrary, in head and neck squamous cell carcinomas, an expression rate of 15% to 20% is reported.13,30
Similar to what is reported in the human pathology literature, 13 PDL1 expressing urothelial carcinomas in our study have a low prevalence (11% of the tumors), 12 but a prevalence of 95% is reported for canine urothelial carcinomas. 17
The difference in the prevalence of PDL1 in mammary carcinoma reported in the literature, compared with our study, is even more marked, with positive labeling in only 3% of cases in this work compared with reports of PDL1 expression in 100% 17 and 80% 18 of canine mammary carcinomas in the published literature. Indeed, this discrepancy, coupled with the utilization of different analytical methods, may be attributable to the heterogeneity of the tumors in terms of prognosis and molecular subtypes. An association between PDL1 and the molecular classification of breast tumors has been reported in humans, where PDL1 expression is associated with hormone receptor loss, HER2 overexpression, and triple-negative PTEN-mutated carcinomas. 13
Fourteen percent of the lymphomas in this work were immunopositive for PDL1, as opposed to 100% of the lymphomas in a previous study in dogs. 35 These discrepancies can be a consequence not only of the different antibodies used, but also of the different evaluation criteria applied. In fact, in previously published studies in dogs, evidence of cell labeling is considered the unique parameter, without specifying a defined subcellular localization;18,35 moreover, cells are described as having intracellular labeling, with surface positivity obscured by cytoplasmic labeling. A significant challenge in standardizing the PDL1 method is the elimination of cytoplasmic background, which can obscure positive membrane labeling. According to the human literature, only membrane labeling is considered positive.1,19
In this study, the evaluation criteria for human PDL1 were applied to various canine tumor types, resulting in data that closely resemble human tumor counterparts. This alignment may offer more precise correlation in predicting therapeutic response in dogs.
Expression of PDL1 in pulmonary carcinoma and renal cell carcinoma, to the best of our knowledge, has not previously been detected in dogs, but has been comprehensively described in humans as a predictive and prognostic marker for the same tumor histotypes.25,41 About 20% to 30% of non-small cell lung tumors express PDL1 in humans,9,13,34 which is fairly close to the prevalence in our series of cases, where we observed immunolabeling in 10% of the tumors.
TPS was the first proposed evaluation method for PDL1 scoring in humans and is currently the one used in human lung cancer. Subsequently, it was discovered that the expression of PDL1 in immune cells in addition to neoplastic cells held predictive-therapeutic and prognostic value. As a result, PDL1 was further assessed with the CPS in different tumor types, including urothelial carcinoma, head and neck squamous cell carcinoma, and gastro-esophageal carcinoma, with promising results. 20 We observed PDL1 labeling in some of the tested canine tumor types in both neoplastic cells and immune cells. However, selecting either scoring system necessitates its correlation with response to therapy and clinical outcomes.
In conclusion, the results of this study, supported by the use of a validated commercial antibody, objectively demonstrate that PDL1 labeling occurs in some canine tumors, with a high prevalence as determined by immunofluorescence in melanomas. Robust evaluation protocols and quantitative scoring systems can facilitate a more comprehensive and consistent approach to research. These data have significant implications, particularly in directing research efforts toward tumor types that express PDL1. The proposed immunohistochemical or immunofluorescence methodology may help in identification of canine oncological patients who could benefit from PD1/PDL1 inhibitory immunotherapy, similar to their human counterparts.
Footnotes
Acknowledgements
We thank Professor Luca Aresu for providing part of the lymphoma caseload.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.