Abstract
Interface dermatitis or lichenoid interface dermatitis refers to a cutaneous inflammatory pattern in which keratinocyte cell death is the essential feature. These terms have evolved from the originally described lichenoid tissue reaction. These lesions are the basis for an important group of skin diseases in animals and people where cytotoxic T-cell-mediated epidermal damage is a major pathomechanism. Yet, for largely historical reasons these commonly used morphological diagnostic terms do not reflect the essential nature of the lesion. An emphasis on subsidiary lesions, such as the presence of a lichenoid band, and definitions based on anatomical features, such as location at the dermo-epidermal location, may cause confusion and even misdiagnosis. This review covers historical aspects of the terminology, including the origin of terms such as “lichenoid.” The types of cell death involved and the histopathologic lesions are described. Etiopathogenesis is discussed in terms of aberrations of immune/inflammatory mechanisms focusing on cutaneous lupus erythematosus, erythema multiforme, and Stevens-Johnson syndrome/toxic epidermal necrolysis. Mechanisms have most extensively been studied in humans and laboratory animals and the discussion is centered on these species. As interface dermatitis is firmly entrenched in dermatological parlance, rather than using “cytotoxic” as its substitute, the terminologies “interface cytotoxic dermatitis” and “panepidermal cytotoxic dermatitis” are recommended, based on location and extent of epithelium affected.
Keywords
Interface dermatitis, also known as lichenoid interface dermatitis and more recently cytotoxic dermatitis, is the most controversial inflammatory skin pattern in veterinary dermatology. The term interface dermatitis was first introduced in human dermatopathology in the 1970s. 4 The term was adopted by veterinary pathologists soon after. The discrepancies and exceptions inherent in this terminology are obvious to anyone involved in teaching veterinary dermatopathology. To understand this conundrum, it is useful to know how we arrived at the status quo. This review traces the history of the various terminologies and describes the type of cell death involved and the major histological lesions. The etiology and pathomechanisms of cytotoxic diseases of the skin in animals are discussed with reference to the more extensive literature on similar human conditions.
History of Lichenoid-Interface-Cytotoxic Dermatitis Terminology
The “Lichenoid Tissue Reaction”
The lichenoid tissue reaction refers to a stereotypic group of histological lesions with the fundamental feature of epidermal cell death. 95 Lichenoid refers to lichen planus—a scaling dermatosis in people that is the archetypal disease of this group. 119 The lichenoid tissue reaction was defined as “epidermal basal cell damage and the chain of histobiologic events resulting from that damage”(Fig. 1a, c). 95 Histologically, lichen planus is typified by basal and suprabasal cell death, including both apoptosis (originally termed Civatte bodies or hyaline degeneration) and hydropic degeneration or vacuolar change (also termed liquefactive necrosis). The heavy lymphocytic infiltration at the dermo-epidermal junction became known as the “lichenoid band.”
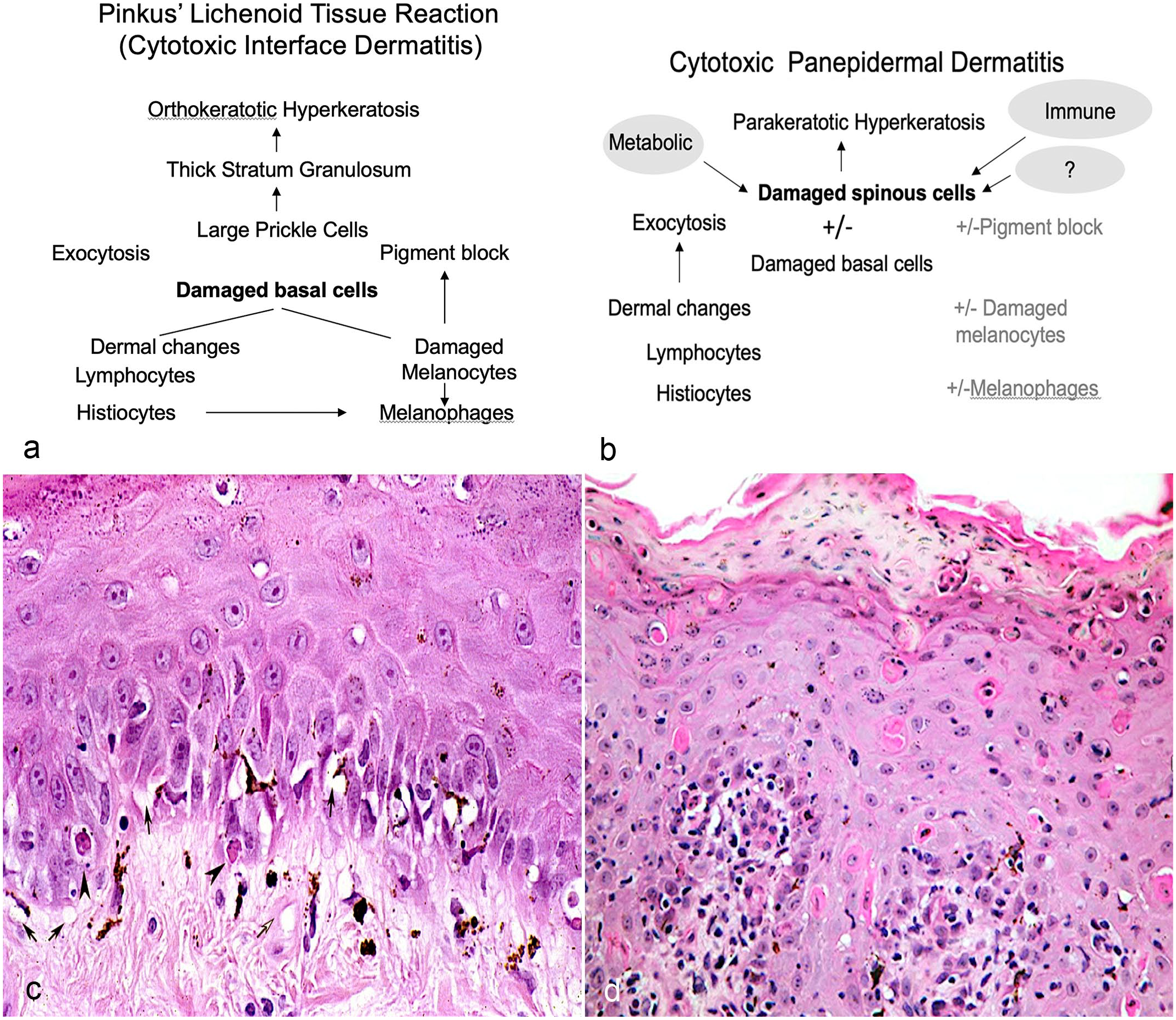
Cytotoxic interface dermatitis and panepidermal cytotoxic dermatitis. (a) Pinkus lichenoid tissue reaction (most consistent with cytotoxic interface dermatitis) is described as “epidermal basal cell damage and the chain of histobiologic events resulting from that damage” 95 (“prickle cell layer” is equivalent to stratum spinosum). (b) Panepidermal cytotoxic dermatitis with cytotoxicity throughout epidermis includes various etiologies (immune-mediated, metabolic diseases, and a group of diseases with unknown or poorly understood etiopathogenesis). Damage of melanocytes will result in loss of pigment, referred to as pigment block. (c) Example of cytotoxic interface dermatitis with multiple vacuolated basal cells (arrow) and few apoptotic basal cells (arrowhead). Hematoxylin and eosin (HE). (d) Example of panepidermal cytotoxic dermatitis with numerous apoptotic keratinocytes in all layers of the epidermis and retained apoptotic cell within the layer of parakeratosis. HE.
In the same publication, Pinkus described histological lesions in which basal epithelial cell death occurred in the absence of, or with, minimal lymphocytic infiltration. 95 He named this cell-poor lesion, with prominent vacuolar interface changes, the “poikilodermatous tissue reaction.” Poikiloderma refers to a constellation of clinical lesions, including cutaneous atrophy, vascular ectasia, and macular pigmentary changes. 84
Interface Dermatitis
In the mid-1970s, Ackerman 4 proposed an alternative method to classify inflammatory skin diseases in people, which he called “pattern analysis.” This approach is strictly morphological. While the seminal lesions of the lichenoid tissue response are listed, namely, epidermal cell death, the defined criteria for the interface pattern emphasized the obscuring of the interface between the epidermis and dermis, whether by lymphocytic infiltration or by vacuolar change. Interface dermatitis therefore included elements of both the lichenoid tissue reaction, as “lichenoid interface” dermatitis, and the poikiloderma tissue reaction, as “vacuolar interface” dermatitis.
Cytotoxic Dermatitis
In 1995, Murphy characterized “cytotoxic dermatitis” as one of a finite number of reaction patterns that may occur in human skin consequent to the infiltration of reactive T lymphocytes. 79 Cytotoxic dermatitis includes lesions in which keratinocyte necrosis occurs throughout the epidermis. In many diseases currently diagnosed as interface dermatitis, for example, erythema multiforme (EM), the keratinocyte damage is panepidermal (Fig. 1b, d). Cytotoxic dermatitis has been largely ignored in human dermatopathology as any PubMed search will establish.
Terminology Used in Veterinary Dermatopathology
Interface dermatitis has been the widely accepted terminology in veterinary dermatology since the 1980s. However, the vacuolar and lichenoid subdivisions of the human classification were renamed “cell-poor” and “cell-rich” interface dermatitis.40,131 The division between cell-rich and cell-poor categories is arbitrary. Some diseases, such as feline thymoma-associated and nonassociated exfoliative dermatitis (FTA(NA)ED), have been described as both.97,104 Cell-poor interface dermatitis, which occurs in ischemic dermatopathies in dogs,9,77 equates to the poikilodermatous tissue reaction. 95
The term lichenoid was used to describe some scaling clinical diseases in dogs in the 1980s but not usually in histological diagnosis. 103 However, the pathomechanism of the lichenoid tissue reaction pattern is accepted as the basis for most diseases within the interface pattern.40,131 The meaning of the term lichenoid has mutated in veterinary dermatopathology usage to describe a dense band of mononuclear cells in the superficial dermis, whether or not there is evidence of epidermal cell death. 40 This usage diminishes the diagnostic power of the lichenoid tissue reaction as originally described. 95 This often has practical consequences leading to misdiagnosis of an autoimmune disease such as canine discoid lupus erythematosus (DLE) as opposed to mucocutaneous pyoderma, based on the presence of a lichenoid inflammatory population in the absence of keratinocyte cytotoxicity.86,126
While cytotoxic dermatitis has not made inroads into the human dermatopathology lexicon, it has been adopted increasingly by veterinary dermatopathologists, particularly for resident training and teaching in specialist veterinary dermatology courses. The “cytotoxic reaction pattern” is now used in one standard veterinary pathology textbook, 124 emphasizing keratinocyte cytotoxicity as the primary histopathologic feature. When keratinocyte cell death is largely restricted to the basal levels of the epidermis, the modifier “interface” is used, whereas “panepidermal” cytotoxic dermatitis describes lesions with keratinocyte death throughout epidermal layers (Fig. 2a, b). There are rare examples where the basal layer is not targeted, for example, a subset of canine superficial necrolytic dermatitis. Examples of diseases that can present as cytotoxic interface dermatitis or panepidermal cytotoxic dermatitis or both are listed in Table 1.
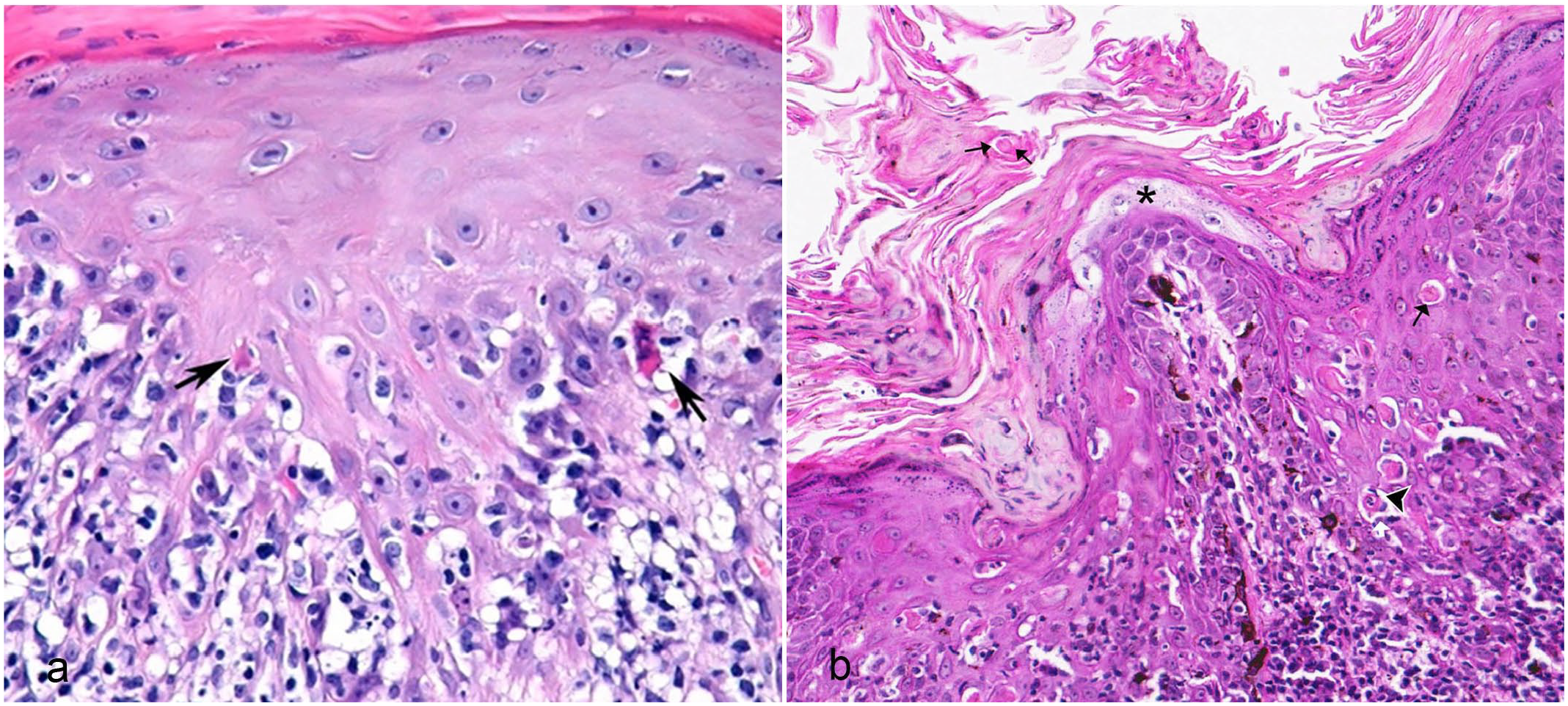
Histopathologic features of cytotoxic dermatitis. Hematoxylin and eosin. (a) Chronic lupus erythematosus, dog. “Cytotoxic interface dermatitis” characterized by basilar keratinocyte apoptosis often with lymphocytic satellitosis (arrows) and hydropic degeneration that obscures the dermo-epidermal junction. (b) Hyperplastic erythema multiforme, dog. “Panepidermal cytotoxic dermatitis” characterized by apoptotic cells across the depth of the epidermis from basal/suprabasal layer (arrowhead), stratum spinosum, and stratum corneum composed of parakeratotic keratin (black arrows). Superficial “pallor” (black asterisk) in the spinous layer mimics lesions usually associated with the metabolic skin disease such as superficial necrolytic dermatitis. The interface of the epidermis and dermis is obscured by dense lymphocytic infiltrates. Hyperpigmentation and pigmentary incontinence are present.
Disease processes that present with cytotoxic dermatitis as their primary lesion and some diseases that may show cytotoxicity as a minor pattern (in parenthesis).
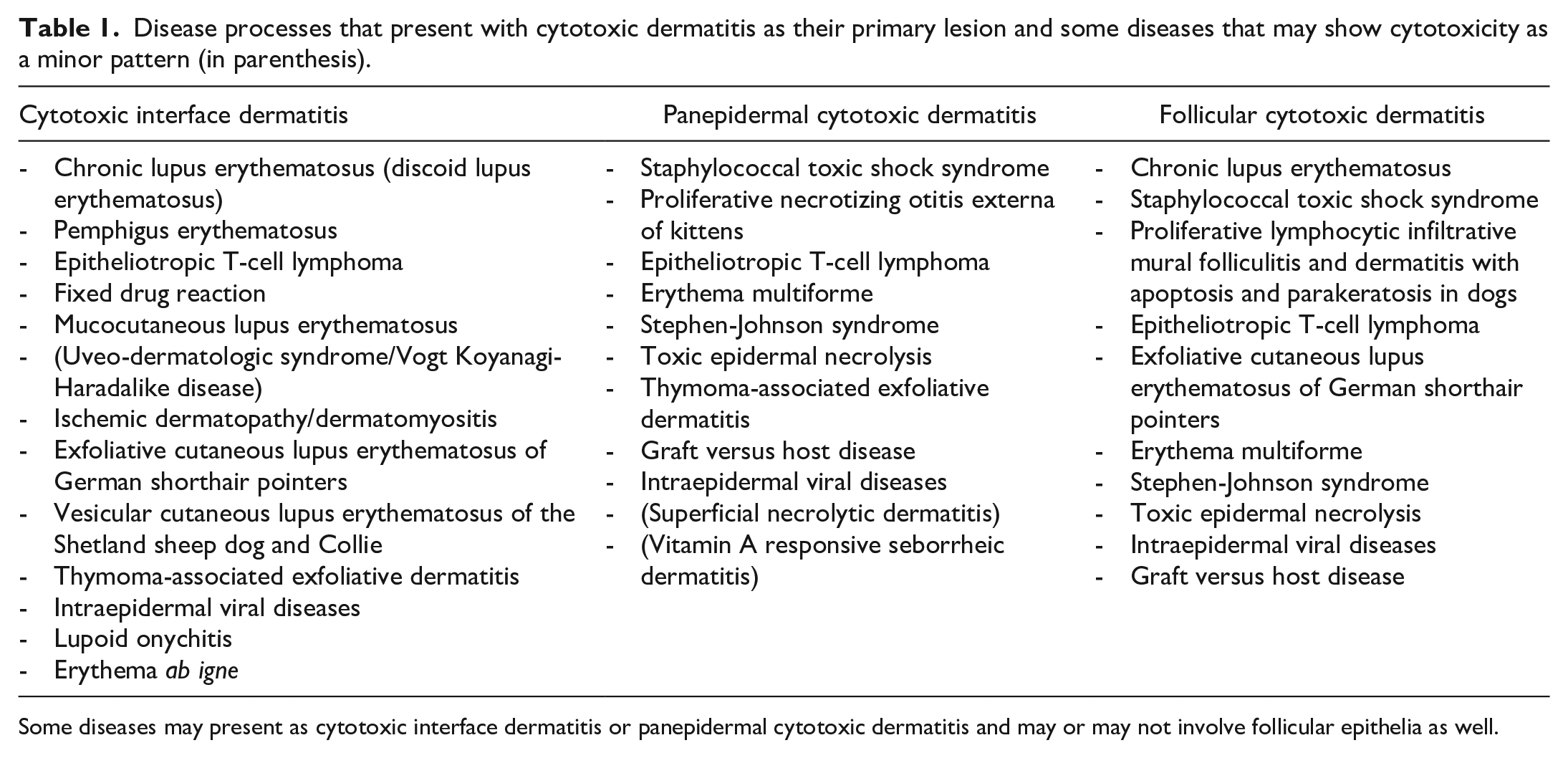
Some diseases may present as cytotoxic interface dermatitis or panepidermal cytotoxic dermatitis and may or may not involve follicular epithelia as well.
The focus of this review is cell-rich cytotoxic interface dermatitis and panepidermal cytotoxic dermatitis, which reflect the lichenoid tissue response. Cell-poor cytotoxic dermatitis typically occurs in the context of chronic vasculopathy, reflecting Pinkus poikilodermatous tissue response. Interesting archetypal diseases in each of these categories (cutaneous lupus erythematosus [CLE] in the former and dermatomyositis in the latter) are related pathogenically coming under the banner of type 1 interferonopathy. 24
Diagnostic Histological Features
Cell Death in the Epidermis
The dominant histological feature of cytotoxic dermatitis is the presence of mostly individual, but occasionally clustered, dead keratinocytes of the epidermis and, in some instances, the follicular epithelium and adnexa (Fig. 2a, b), the latter resulting in alopecia. These keratinocytes have pyknotic or karyorrhectic nuclei, hypereosinophilic cytoplasm, and are often surrounded by a clear halo resulting from the condensation of nuclear and cytoplasmic contents. These features cause the characteristic cell shrinkage identified ultrastructurally in the seminal paper on apoptosis. 57 Before their true nature was understood, these cells were known as Civatte cells or bodies, which subsequently have been identified as apoptotic cells.58,120
In cytotoxic interface, dermatitis apoptotic keratinocytes are concentrated in the basal layers of the epidermis, with occasional extension into the lower stratum spinosum (suprabasal). Apoptotic keratinocytes can also be found in the upper levels of the epidermis. These “high” apoptotic cells may occur in diseases classified in both human and veterinary medicine as “interface,” for example, CLE and EM, but are typically seen in canine hyperkeratotic EM and other idiopathic diseases such as proliferative and necrotizing otitis externa in cats (Fig. 2b).13,43,86,92 Apoptotic keratinocytes may be retained in the stratum corneum and surface scales and crusts. As corneocytes cannot undergo caspase-dependent apoptosis, it is assumed that these dead cells have been passively carried up to the stratum corneum. Normally, such cells undergo efferocytosis in the epidermis, consistent with the fourth phase of apoptosis, or undergo apical extrusion.19,62 Often called dyskeratotic cells, another hangover from the 20th century when hypereosinophilic keratinocyte cytoplasm was equated with terminal differentiation, their provenance is poorly understood. Cornification has been considered an example of programmed cell death. 68 However, the 2018 report of the Nomenclature Committee on Cell Death (NCCD) recommended that programmed cell death and terminal differentiation should be conceptually well discriminated as terminal differentiation of keratinocytes involves caspase 14, but not executioner caspases. 34
By the end of the 1970s, apoptosis was considered the major pathological event in many human lichenoid interface skin diseases, including cutaneous adverse drug reactions, solar damage, and tumor regression. 123 It is interesting that apoptosis was studied so early in skin diseases, while it was largely ignored in other areas of pathology until the late 1980s with the discovery of biochemical and genetic processes that implement it.114,115
With the development of terminal deoxynucleotidyl-transferase–mediated deoxyuridine triphosphate nick-end labeling (TUNEL), Civatte cells were shown to have the characteristic DNA fragmentation seen in apoptosis. 63 Assays for cleaved caspases demonstrate that the cells with an apoptotic morphotype were, generally, undergoing caspase-dependent regulated cell death. Apoptotic keratinocytes have been described in many epidermal cytotoxic diseases in animals, not always with biochemical confirmation.11,43,83,85,118 The drawbacks in using histological morphotypes of cell death to predict any particular molecular modality of regulated cell death was emphasized in the 2012 report of the NCCD. 35 Nonetheless, the 2016 International Harmonization of Nomenclature and Diagnostic Criteria (INHAND) guidelines for toxicological pathologists use morphotypes and these are the only practical option in routine veterinary diagnostic dermatopathology. 29
In addition to single or grouped dead epidermal cells, large areas of epidermis may undergo confluent cell death, for instance, in cutaneous adverse drug reactions (cADRs), particularly in lesions at the extreme end of the Stevens-Johnson syndrome/toxic epidermal necrosis (SJS)/(TEN) spectrum (Fig. 3a).14,93 Partial or full thickness epidermal necrosis leads to the epidermal detachment that characterizes the most severe, often fatal clinical lesions and differentiates SJS/TEN from EM in people. 15 Severe burns may be misdiagnosed as TEN by inexperienced pathologists failing to recognize the significance of the unaffected dermis in the latter. 130 Confluent cell death in SJS/TEN was originally interpreted as secondary necrosis (unregulated cell death) although regulated loss of membrane permeabilization leading to cell lysis recently has been demonstrated in late apoptosis. 26 Given that apoptosis has been demonstrated as a mechanism of epidermal cell death in SJS/TEN in people and dogs,14,93 many attempts were made to explain the mechanisms of confluent apoptotic cell death. In people, suggested factors include perforin, granzyme and granulysin (present at high levels in TEN blister fluid), soluble FAS ligand, tumor necrosis factor alpha, and annexin-1.1,22,82,90,117 As anti-apoptotic caspase-8 regulator cellular FLICE-like inhibitory protein (cFLIP) is reduced in lesions of SJS/TEN, the possibility of reduced regulation of apoptosis was proposed as a contributing mechanism. 89
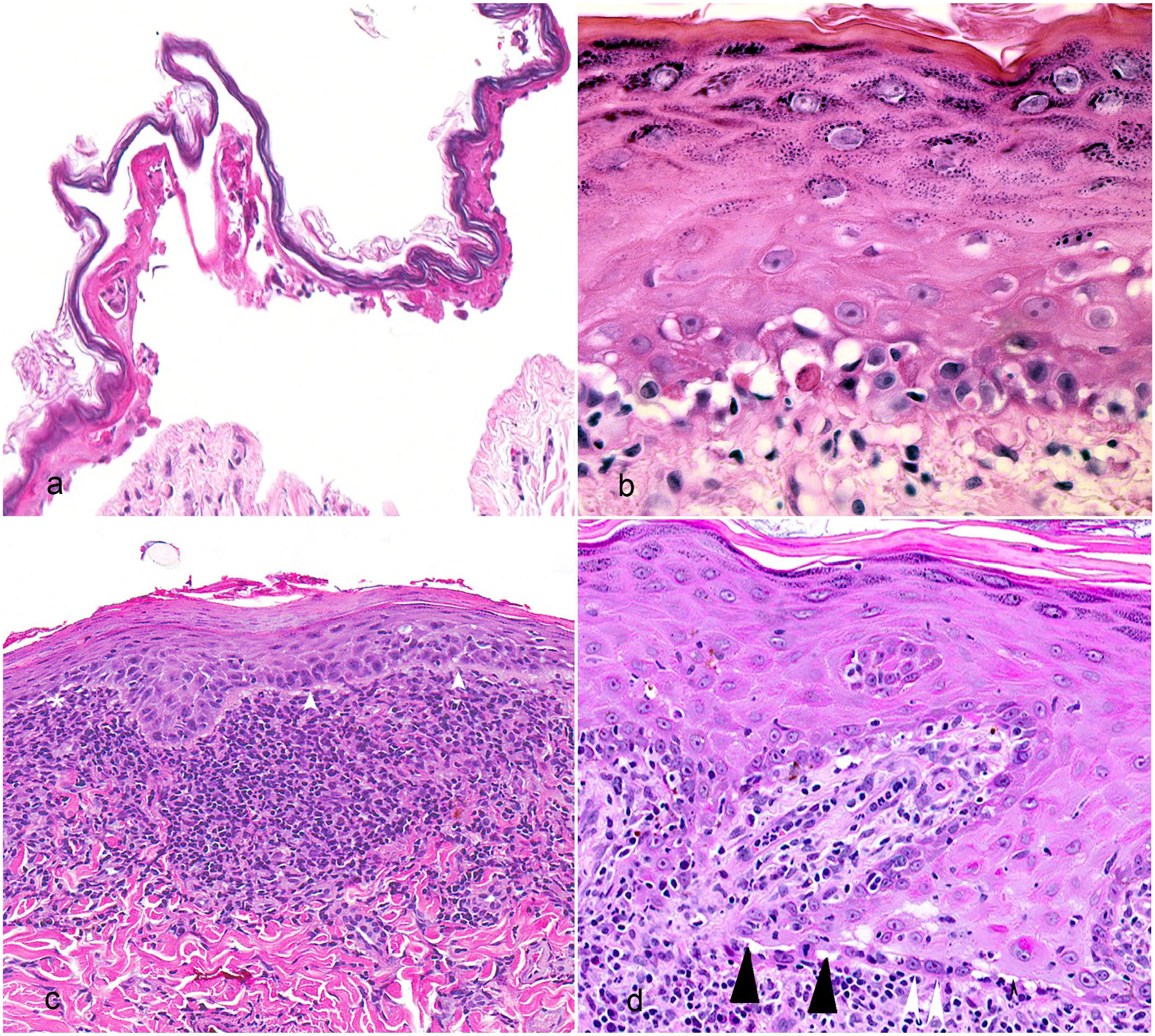
Histopathologic features of cytotoxic dermatitis. Hematoxylin and eosin. (a) Toxic epidermal necrolysis, dog. A detached dead epidermis referred to as epidermolysis. Note the dermo-epidermal cleft and superficial dermis lack inflammatory cells. (b) Thymoma-associated exfoliative dermatitis, cat. Interface cytotoxic dermatitis characterized by hydropic degeneration at the dermo-epidermal junction, patchy keratinocyte apoptosis, and minimal lymphocytic dermatitis and exocytosis. (c) Discoid lupus erythematosus, dog. Prominent lymphocytic “lichenoid” band, a thickened basement membrane zone (white arrowheads), and patchy hydropic change (asterisk). (d) Chronic lupus erythematosus, dog. Squamatization of the basal cell layer (white arrowheads) indicating loss of normal basal cells. There is hydropic degeneration (black arrowheads) and moderate dermal lymphocytic infiltration with some lymphocyte exocytosis.
It now appears that necroptosis, an RIPK3-dependent and sometimes RIPK1-dependent regulated cell death pathway that typically exhibits a necrotic morphotype, is responsible for confluent epidermal lesions of SJS/TEN in people.34,59,88,98 Moreover, NETotic cell death, associated with extrusion of chromatin and histone-containing fibers bound to granular and cytoplasmic proteins, known as neutrophilic extracellular traps (NETs), is implicated in human SJS/TEN. 60
Hydropic Degeneration/Vacuolar Change
Hydropic degeneration/vacuolar change was initially described as “minute spaces along the basement membrane zone.” 4 The consequent “holey” appearance is evident at low magnification. Hydropic degeneration of basal keratinocytes occurs in concert with apoptosis and is often more prominent than apoptosis (Fig. 3b).50,108 It is not limited to the basal cell layer and the area of basement membrane zone, an example being prominent swollen keratinocytes with pale-staining cytoplasm present in the stratum spinosum in lesions of canine SJS/TEN. 10 Sontheimer equated the cellular change to oncotic necrosis. 108 To the author’s knowledge, the pathogenesis of vacuolar degeneration, particularly on the dermal side of the dermo-epidermal junction, has received little attention.
The triggering mechanisms initiating hydropic degeneration may differ in cell-rich and cell-poor forms of cytotoxic dermatitis.50,108 In the former, cytotoxic T cells (CTLs) and natural killer cells (NK cells) are present at the dermo-epidermal junction where they mediate apoptotic cell death, but whether they have a role in hydropic change is unknown. 101 In cell-poor cytotoxic dermatitis, such as dermatomyositis, hypoxia due to chronic vascular damage likely contributes to the loss of basal keratinocyte viability by inducing endoplasmic reticulum stress and autophagy-associated cell death. 50
In addition, the vacuolar change along the dermo-epidermal junction, leading to loss of keratinocyte adhesion to the basement membrane zone, may represent anoikis, which in itself is not a cell death modality, but rather a pathway to intrinsic apoptosis.34,41 When basal keratinocytes detach from the basement membrane zone in the process of normal differentiation, they are protected from apoptosis by basement membrane proteins such as b1-integrins.68,96 Following disruptions to the basement membrane by edema or deposition of immune complexes, apoptosis is triggered as b1-integrin sites become vacant. A loss of basal keratinocytes may contribute to the epidermal “collapse” described in the original lichenoid tissue reaction. 95
Cellular Infiltrates Associated With Cytotoxic Dermatitis
Cytotoxic dermatitis is predominantly lymphocytic, with variable numbers of other mononuclear cells, including myeloid dendritic cells and macrophages.3,5,51,55,70,94,102,111 With the exception of mucocutaneous or mucosal lesions, plasma cells are uncommon.
Lymphocyte exocytosis into the epidermis often blurs the dermo-epidermal junction, as reflected in the term interface. The density of the lymphocytic infiltrates at the dermo-epidermal junction is highly variable. Despite typically being classified as cell-rich diseases, sparse lymphocytic infiltrates may be seen in EM or FTA(NA)ED and some human drug reactions.92,97,104 The accumulation of a heavy band of lymphomononuclear inflammatory cells in the superficial dermis is termed a “lichenoid band” (Fig. 3c). It is a feature of chronic cytotoxic interface diseases, for example, DLE. 86 Heavy inflammatory infiltrates often extend around the adnexa. Similar changes can be seen with mucocutaneous pyoderma. 126 The absence of keratinocyte cell death assists in differentiating leukocytic exocytosis and transmigration from cytotoxic interface dermatitis or panepidermal cytotoxic dermatitis.
The close approximation of small lymphocytes to apoptotic keratinocytes in the epidermis and/or follicular epithelia is known as “satellitosis” (Fig. 2a) and is a morphological representation of CTL-mediated cytotoxicity. In contrast, “sunburn” cells, which are a feature of human and animal photodermatoses (including CLE), are isolated apoptotic keratinocytes, typically occurring in the stratum spinosum, unassociated with lymphocytes. 113
The lymphocyte population generally reflects Th1 immunity comprising interferon-gamma producing CD4+ T cells, CD8+ CTL, and type 1 innate lymphoid cells, including NK and NK-T cells.3,16,48,55,70,111 The contribution of Th17 cells has been reported in some human diseases but data are not consistent.53,69 There are few additional data about subpopulations of T lymphocytes in veterinary cutaneous cytotoxic diseases as it was shown in 1998 that CD8+ T cells predominate canine EM and graft versus host disease. 5 In 2021, Tanaka et al 111 documented a junctional population of CD3+, CD8+, and granzyme B+ T cells in a single case of canine EM. Targeted transcriptomic analysis of skin lesions from 4 dogs with CLE demonstrated upregulation of IFNG and interferon-related chemokine CXCL10. 6 While infiltrating lymphocytes were identified as the main source of CXCL10 in dogs with associated systemic signs, keratinocytes were the main source in dogs without systemic signs. 6
In general, B lymphocytes are not major players in cytotoxic dermatitis. B-cell-enriched lesions of DLE in humans suggest a link to systemic disease. 2 Some CD21+ B cells were observed in the superficial dermis of dogs with EM. 5
Other mononuclear cells, including macrophages, monocytes, and various populations of dendritic cells, including myeloid dendritic cells and plasmacytoid dendritic cells (pDCs), may make up a significant proportion of the inflammation.5,13,90 Of particular interest are pDCs, as they are numerous in human cytotoxic interface diseases, as reported in CLE, dermatomyositis, lichen planus, and graft versus host disease.20,27,30,129 Plasmacytoid dendritic cells have not been characterized in domestic animals to date. While not residing in the skin, pDCs play crucial roles in antiviral defense as “professional” producers of type 1 interferons and in diseases resulting from interferon excess in people (the interferonopathies). 24 Plasmacytoid dendritic cells also promote specific immunity by activating myeloid dendritic cells, enhancing cross-presentation and T-cell survival, as well as supporting the induction of proinflammatory cytokines. 129 The interferon-gamma inducible chemokines, including CXCL10, attract effector cells, such as CXCR3+ CTLs, into the affected epidermis.3,125 In CLE in people, dermal pDCs mediate recruitment of CD4+ Th1 cells, while junctional pDCs recruit CTLs.30,116
Granulocytes are typically sparse in hematoxylin and eosin–stained sections. Their presence is usually attributed to erosions, ulcerations, or complications such as superficial pyoderma. However, recent studies revealed significant roles for NET forming neutrophils in diseases for a long time considered to be the sole purview of CTLs, including CLE and the cutaneous adverse drug reaction SJS/TEN in people. 60 Numerous neutrophils clustering around apoptotic keratinocytes resembling lymphocytic satellitosis is typically seen in lesion of exotoxin-mediated staphylococcal toxic shock syndrome as a result of rigorous interleukin (IL)-17A production by memory T lymphocytes.64,110 Eosinophils are rare in cytotoxic dermatitis in domestic animals, but are a feature of drug-induced EM in people. 92
Additional Changes of Cytotoxic Dermatitis
Pigmentary incontinence: Keratinocyte cell death leads to failure of melanin transfer and depigmentation. As a result, the free melanin granules are phagocytosed by dermal macrophages (Fig. 2b), referred to as pigmentary incontinence. Pigmentary incontinence may also result from hyperpigmentation. This can occur concurrently with hypopigmentation, for example, in CLE and dermatomyositis in people and dogs.
Squamatization refers to a change in orientation of the normally cuboidal and vertically orientated basal epidermal cell layer to a flattened and horizontally oriented layer. This is a cue to check carefully for evidence of active epidermal cell death along the basal layer (Fig. 3d).
Thickening of the basement membrane zone typically reflects deposition of immunoglobulins and immune complexes. It is a very prominent lesion of chronic DLE in people and occurs in canine DLE, mucocutaneous lupus erythematosus, and canine hyperkeratotic EM (Fig. 3c).13,86
Epidermal atrophy, described as stratum spinosum “collapse” in the original description of the lichenoid tissue reaction, 95 is typical of chronic CLE in people and dogs, particularly DLE, generalized chronic lupus erythematosus, and mucocutaneous lupus erythematosus. Because the normal canine and feline haired skin epidermis are thin (only 2 to 3 nucleated layers), atrophy is difficult to appreciate unless lesions involve nasal planum or mucosal surfaces.
Hyperkeratosis and parakeratosis (Fig. 2b): The stratum corneum was described hyperkeratotic as a consequence of the basal cell damage in the original description of the lichenoid tissue response hyperkeratosis. 95 It is noted in the subacute and chronic cytotoxic skin diseases typified by clinical scaling, including the archetypal human disease lichen planus. 92 Orthokeratotic hyperkeratosis is prominent in FTA(NA)ED and exfoliative CLE.67,86,97,104 Parakeratotic hyperkeratosis (parakeratosis) is particularly common in idiopathic scaling panepidermal cytotoxic diseases, such as proliferative and necrotizing otitis externa in cats and hyperkeratotic EM in dogs, among others.13,43,75 In these diseases, there is a marked follicular targeting with infundibular parakeratosis and cytotoxicity often more severe than in the interfollicular epidermis.
Dermal mucinosis is a feature of human CLE, particularly in chronic forms like DLE, but is not obvious in the canine disease. A possible mechanism for the induction of mucinosis is the upregulation of inducers of versican 1 by keratinocytes following interferon stimulation, as recently demonstrated in vitro. 80
Dermal fibrosis occurs in CLE, particularly in DLE in people, which is considered to be a scarring dermatosis. It is also seen in dogs and occasionally forms a separating band between the dermo-epidermal junction and lichenoid inflammation.12,86
Etiopathogenesis
In 1988, Shiohara et al 106 suggested that the primary pathological event in the lichenoid tissue reaction, also known as the cytotoxic dermatitis reaction pattern, was an attack by autoreactive T cells on the epidermis. In 1993, Le Boit reported that antigen-specific cell-mediated cytotoxic immune responses play a role in this process. 65 In both instances, the infiltrating cells are CD8+ CTL and/or NK cells that mediate cell death, as described above. However, the exact mechanisms of keratinocyte damage in some diseases are more complex, as exemplified by recent advances in the understanding of the pathogenesis of CLE. 69
It is accepted that genetic predisposition and environmental factors, including ultraviolet radiation (UVB), infections, and drugs, cause malfunctions in innate and acquired immune responses, both centrally and in the skin. Lesions occur when normal surveillance mechanisms are overwhelmed or when these protective mechanisms are subverted to cause harm, for instance, in autoimmune skin diseases. With this background, it is possible to divide epidermal cytotoxic dermatitis in groups depending on contributions from epidermis, immune system, and environment.
Response of Normally Functioning Immune System to Pathogens and Altered Keratinocytes
In these cases, the immune system is operating within normal parameters against perceived threats. Skin homeostasis is maintained by constant removal of damaged keratinocytes.121,122 Examples include regressing viral papillomas and actinic keratoses, a common preneoplastic disease in heavily sun-exposed skin in people and in poorly haired and pigmented skin in animals.52,122 Cytotoxic interface lesions affecting the follicular infundibular epithelium have been reported as a rare manifestation of human herpesvirus infection. 18
The common cutaneous viruses in domestic animals rarely present with primary lesions of cytotoxic dermatitis because viral damage is overwhelming. An exception is a rare manifestation of canine parvovirus infection with lesions affecting skin and mucous membranes. Originally reported as canine parvovirus–associated “EM,” it represents an active viral infection, indicated by the presence of inclusion bodies confirmed by immunohistochemistry and intralesional virions demonstrated by electron microscopy.31,46,128
In contrast, the common form of EM in people, known as herpesvirus-associated EM, is not the result of an active viral infection. 33 Skin lesions occur after the initial infection with the human common cold sore virus and at distant sites from the primary viral-induced lesion. The characteristic target-shaped lesions result from CTL-mediated immune responses against keratinocytes whose antigenicity is altered following the transport of viral components (typically gene fragments) by progenitor dendritic cells to sites distant from the initiating infection. 8
Virus-associated “EM” has been described in other animal species, including horses, cats, and pigs.25,38,46,72 The clinical lesions are varied and, in the case of pigs, histological lesions of cytotoxic dermatitis were not shown. The clinical signs in 2 equine cases appeared consistent with hyperesthetic leukotrichia, a disease of unknown etiology, that also shares some histological features with human EM. It has been stressed in human dermatology that interface dermatitis is a tissue reaction pattern and not a disease. 92 To date, the closest animal counterpart to herpes-associated EM in people appears to be feline herpesvirus-1-associated “EM” in cats.25,130 This needs to be differentiated from feline necrotizing ulcerative dermatitis. 49
Immune-Mediated and Autoimmune Diseases
The second group, typified by immune-mediated and autoimmune diseases, occurs when presumably helpful cell-mediated immune responses, both innate and adaptive, are subverted to cause harm.23,28 Recent advances in the understanding of CLE in people reveal that environmental factors chiefly exposure to UVB, but also to drugs and some infectious agents, will induce a skewed type I interferon response in genetically predisposed individuals, resulting in lesions of chronic cytotoxic interface dermatitis.
Human CLE is manifest by an array of differing clinical presentations and may or may not be associated with systemic lupus erythematosus (SLE). 44 It is further subdivided into lupus-specific (including acute, subacute, and chronic categories) and nonspecific skin lesions, depending upon the presence of classic lymphocytic cytotoxic interface dermatitis.37,107 The task of unraveling the pathogenesis of CLE is made difficult by the multiple variants that may or may not share the same pathophysiology.
It is accepted that human CLE results from an excessive production of type 1 interferons, which qualifies it as a type 1 interferonopathy. 24 The ultimate result is CTL-induced keratinocyte apoptosis, which is desirable in the context of antiviral defense, but detrimental when delivered by autoreactive CTLs. 71 Significant advances have been made in understanding human CLE, but the exact sequence of events connecting triggering events and immune activation to the development of lesions remains unknown.36,55,69
In addition to interferon-alpha, dysregulation of another type 1 interferon, interferon-kappa, has been recently implicated in the pathogenesis of human CLE skin lesions. Interferon-kappa not only drives enhanced responses to interferon-alpha and stimulates activation of dendritic cells but also appears to plays a role in photosensitization through regulation of the apoptotic response of keratinocytes to UVB, which is not only an initiating but also a sustaining event. 99
In summary, keratinocytes acting as members of the innate immune system trigger uncontrolled reactions in genetically predisposed people, particularly in response to UVB irradiation. Second, type 1 interferon producing cells (particularly pDCs) drive innate and adaptive autoreactive immune reactions through a variety of mechanisms, including promoting release of interferon-gamma with induction of chemokines such as CXCL10 that attract CXCR3 CD8+ cytotoxic lymphocytes to the dermo-epidermal junction. The importance of the skin immunome has been further emphasized by recent studies in which nonlesional skin of CLE patients demonstrated a type 1 interferon–rich environment that skewed gene transcription in all the major skin cell types and distorted predicted cell-cell communication networks. 17 It is not surprising that the large array of genetic mutations that confer CLE susceptibility have functions related to type 1 interferon receptor signaling, nucleic acid sensing, or transducing DNA signals to innate immune responses among many others. 24
In veterinary medicine, CLE occurs most frequently in dogs, but unlike people canine CLE is only rarely associated with SLE. 86 The Sontheimer classification, in which CLE is subdivided into lupus-specific and lupus-non-specific categories based on the cytotoxic reaction pattern, has been adapted for canine CLE. 86 Like human CLE, the clinical and histological lesions of the canine subtypes of CLE are varied. 86 Canine CLE forms include DLE, its rare generalized variant generalized chronic lupus erythematosus, mucocutaneous lupus erythematosus, vesicular lupus erythematosus of collies and Shetland sheepdogs, the canine counterpart to human subacute CLE and exfoliative CLE of German shorthaired pointers, Vizslas, and closely related breeds.12,39,54,74,87 Acute CLE, which occurs in up to 70% of human SLE cases, is poorly documented in dogs with most reports predating the division into lupus-specific and lupus-non-specific skin lesions. 86 Lupus erythematosus panniculitis in humans is characterized by lymphocytic infiltrates and cell-poor cytotoxic interface dermatitis. Olivry et al 86 referred to a possible, previously reported lupus erythematosus panniculitis in a dog. 105 Canine lupus erythematosus panniculitis needs to be differentiated from rabies vaccine–induced inflammation and alopecia.45,127
Very little is known of the pathogenesis of CLE in dogs although the same triggers, UVB, drugs sensu lato, and infectious agents, likely apply to some variants. A missense variant of the UNC93B1 gene involved in regulating Toll-like receptor signaling has been identified as the underlying genetic defect in canine exfoliative CLE. 66 While it is important not to extrapolate between species, a recent retrospective study comparing gene expression in DLE lesions found that people and dogs shared 49 differentially expressed genes. 6 As mice only share 25 genes, it is possible that the dog may be a better model for the human disease than the mouse. 36
A second group of immune-mediated skin diseases that present with a cytotoxic interface reaction pattern fall under the category of cADR. Drug or drug metabolites alter the epidermis by covalently binding as a hapten to cell surface proteins, directly interacting with the major histocompatibility complex/T-cell receptor complex, or potentially altering endogenous peptides presented to T cells.81,112 Drug-specific CD8+ CTL and activated NK cells are the main mediators of keratinocyte death, but CD4+ T cells and dendritic cells play a role in promoting epidermal damage in human drug reactions.81,112 The exact mechanisms vary with drug and host factors, particularly HLA allotypes in people. 81 In fixed drug eruptions in people, the lesions recur at the same anatomic site because drug-specific CD8+ memory T cells remain in situ. 7 Fixed drug eruptions have been recognized in the dog. 73 The cytotoxic interface diseases most commonly triggered by drugs in people and animals are drug-induced EM and SJS/TEN.
The true prevalence of drug-induced EM in animals is not known largely because of the lack of studies in which proven drug causation has been established; recovery following withdrawal of a suspected drug and absence of another cause is insufficient. In one study where causation was assessed algorithmically, only 19% of dogs classified clinically as EM were definitely or highly likely to be reactions to a drug. 47 In general practice, because of a tendency to equate lesions of cytotoxic interface dermatitis with EM and to equate EM with “drug reactions,” the prevalence of EM in animals is likely overestimated because causality is seldom established conclusively. 130
In contrast, SJS/TEN lie on a spectrum of severe skin lesions that are mostly caused by cADR (occasionally by infectious agents). Extensive epidermal exfoliation and blister formation drive the high fatality rate of SJS/TEN in animals and people.102,130 Canine SJS/TEN lesions closely mimic those of the human disease and are highly likely to be drug-induced. 130 In the same study of canine cADR in which only 19% of dogs with EM like lesions were likely induced by drugs, 90% of dogs with clinical lesions of SJS/TEN had evidence supporting drug causation. 47
The pathogenesis of rapid and catastrophic epidermal death has been intensively studied in humans, as discussed previously. Most recently, a role for NETosis has been invoked in human TEN. Drug-specific CD8+ skin-seeking lymphocytes produce lipocalin-2, which triggers NETosis. 60 During the formation of neutrophil NETs, the antimicrobial peptide LL-37 is released and induces formyl peptide receptor 1 expression on activated keratinocytes. Necroptosis is triggered by annexin A1 binding to formyl peptide receptor 1.59,60,98 Keratinocyte death amplifies the LL-37 and formyl peptide receptor 1 loop, explaining the catastrophic nature of the ensuing epidermal cell death. 60 It appears so far that this NETosis-necroptosis axis is specific to SJS/TEN.
To the author’s knowledge, cell death modalities other than apoptosis have not been evaluated in SJS/TEN in animals, although necroptosis has been suggested as a possible mechanism in canine TEN. 11
Attack of Abnormal Immune System on Keratinocytes
In a third group comprising relatively few diseases, the normal epidermis becomes vulnerable to the action of an abnormal immune system. Alterations in the maintenance of tolerance can lead to an attack on normal keratinocytes. This has been documented in the human paraneoplastic syndrome, thymoma-associated multiorgan autoimmunity syndrome in which there is a marked reduction in regulatory T cells in the cutaneous lesions.42,78 It is not unreasonable to speculate that a diminution of tolerance may play a role in thymoma-associated exfoliative dermatitis in cats, rabbits, and goats.21,32,97 Occasionally, intraepithelial neoplastic CD8+ T cells may exhibit cytotoxic activity against keratinocytes, as recognized in lichenoid mycosis fungoides in people 132 and in canine epitheliotropic T-cell lymphomas. 76
Altered Environment Affects Both Epidermis and Immune System
The fourth group where both epidermis and lymphocytes are functioning normally but within an altered environment is typified by graft versus host disease, which histologically mimics EM. 5 The host epidermis is at particular risk of attack by allogenic engrafted lymphocytes because of the high expression of major histocompatibility complex I receptors on keratinocytes. 109 Graft versus host disease has been documented in dogs with bone marrow transplants.5,61,100
Cytotoxic Dermatitis of Yet to Be Identified Mechanisms
There are several, possibly unrelated diseases, mostly described in dogs, that are characterized histologically by cytotoxic panepidermal dermatitis with marked hyperkeratosis and often parakeratosis. The diseases include feline proliferative and necrotizing otitis externa, 75 infiltrative mural folliculitis and dermatitis with apoptosis and parakeratosis in dogs (black Labrador may be overrepresented), 43 hyperkeratotic EM previously called “old-dog” EM in dogs, 13 and a subset of cases of superficial necrolytic dermatitis also known as metabolic epidermal necrosis in dogs. 130 Similar histological lesions occurred in a black Labrador dog that developed generalized scaling early in life. This dog was not vitamin A deficient but the lesions resolved completely as long as the dog was treated with very high doses of the vitamin. 91
The clinical lesions in these canine diseases are generalized multifocal to coalescing papules and plaques covered by thick white to yellow-tan to brown crust often on pinnae, trunk, and abdomen, occasionally at mucocutaneous junctions. In feline proliferative and necrotizing otitis externa, the only feline example of this reaction pattern, lesions affect the inner surface of the pinna almost exclusively with facial and scattered, more generalized lesions. 75
These diseases are characterized histologically by (1) panepidermal apoptotic cell death, usually more prominent in the outer layers of the epidermis, sometimes sparing the stratum basale; (2) lymphocytic, and sometimes histiocytic or neutrophilic, satellitosis; (3) hyperproliferative changes in epidermis/outer root sheath epithelium of the follicular infundibulum; (4) severe, often markedly parakeratotic, hyperkeratosis; (5) the potential of follicular infundibular epithelia to be more severely affected than interfollicular epidermis; and (6) occasional epidermal pallor reaction in outer stratum spinosum.
These conditions are idiopathic and likely reflect a variety of pathogenic mechanisms and etiologies. These diseases extend the original concept of the lichenoid tissue response/interface dermatitis from a condition in which basal keratinocytes are the targeted cells to a more comprehensive version where cells throughout the epidermis are involved. Clinically, these conditions are chronic and typified by scaling (hyperkeratotic or parakeratotic) in distinction to the erosive, ulcerative atrophic or scarring lesions associated with many of the classic interface cytotoxic dermatoses. The pathogenesis of these conditions is likely quite varied. For example, in the case of the subset of superficial necrolytic dermatitis cases with high apoptosis (a lesion recognized in the equivalent human disease necrolytic migratory erythema), the cause is clearly metabolic damage to the epidermis rather than the result of an immunological reaction, yet to the pathologist the histological lesions of this form of superficial necrolytic dermatitis can be virtually indistinguishable from hyperkeratotic EM lesion.
Conclusions
While acknowledging the difficulty of dislodging entrenched terminology, when the central event is one of keratinocyte cell death, and not just the involvement of the dermo-epidermal interface, this should be reflected in the diagnostic language. An argument could be made to resurrect “epidermitis.” With exception of exudative epidermitis in pigs, the term fell out of favor because the epidermis was thought to be incapable of actively engaging in an inflammatory response. Starting with the discovery that keratinocytes could produce IL-1 in the 1980s, it rapidly became apparent that the epidermis is a very active player in immune and inflammatory processes, resulting in the concept of the “epimunome.” 56 In the author’s opinion, the adoption of cytotoxic dermatitis will be hard enough. A change to epidermitis would be a bridge too far.
Depending on the location of the keratinocyte cell death, the process can be subdivided into “cytotoxic interface dermatitis” and “cytotoxic panepidermal dermatitis.” The nature of the inflammatory response, alterations in pigmentation, and dermal changes can be then employed in the second stage of pattern analysis to narrow down the possible list of diseases and determine a pathological diagnosis. Ultimately, it is crucial to interpret the lesions in association with clinical data, emphasizing the importance of clinicopathologic correlations. This approach should simplify the teaching and practice of veterinary dermatopathology while facilitating understanding of emerging diseases by allowing comparison with other diseases of epidermal cytotoxicity.
Footnotes
Acknowledgements
My sincere gratitude to Dr Julie A. Yager for her invaluable support to create this manuscript. She contributed with discussions, schematics, and histological pictures as well as editing manuscript.
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
