Abstract
Lymphoglandular complexes are components of the gut-associated lymphoid tissue that are characterized by submucosal lymphoid aggregates invested by projections of mucosal epithelium. Reports of pathology involving these structures are rare in both human and veterinary literature. Here, the authors report 2 cases of rectal masses excised from dogs following a period of tenesmus and hematochezia. In both animals, the masses were composed of lymphoid tissue closely encompassing tubuloacinar structures. Immunohistochemistry and polymerase chain reaction antigen receptor rearrangement testing demonstrated that the lymphoid population was polyclonal, comprising T and B cells arranged in loosely follicular aggregates centered on the epithelial foci. In light of these findings, a diagnosis of lymphoglandular complex nodular hyperplasia was reported. To the authors’ knowledge, this is the first report of this condition in dogs.
Keywords
Lymphoglandular complexes are components of the gut-associated lymphoid tissue, formed from a close association between lymphoid follicular tissue and epithelial diverticula projecting from the overlying mucosa. They are predominantly located within the distal small intestine, cecum, and colon and have been described in a wide range of mammalian species, 6 including humans, 14 dogs, 2 cattle, 10 horses, 11 pigs, 13 dolphins, 5 and echidnas. 16 While undoubtedly involved in immune surveillance of the gastrointestinal tract, the specific role of these structures in mucosal immunity is unclear. Some researchers have speculated that they may be mammalian analogues of the bursa of Fabricius. 16 The clearest indication of their function is derived from studies in pigs, where they are the predominant site of IgA production during Campylobacter jejuni infection. 12
A range of proliferative lesions involving the gastrointestinal lymphoid tissue are recognized in humans and animals. Lymphoid follicular hyperplasia is a common endoscopic and histological observation in many chronic inflammatory enteropathies, reflecting expansion of the mucosa-associated lymphoid tissue (MALT) in response to persistent antigenic stimulation. While this is a non-specific finding seen with a variety of infectious and hypersensitivity diseases, 1 gastrointestinal MALT hyperplasia is also closely associated with several immunodeficiency syndromes in humans, 8 indicating that genetic predisposition may be a factor in some cases. Although hyperplastic foci are typically small, they can sometimes form larger masses up to several centimeters in size, and distinguishing these lesions from MALT lymphoma can be challenging. Indeed, progression from hyperplasia to neoplasia has been noted to occur in humans. 7 Hyperplastic lesions of the lymphoglandular complexes are rarer still, 19 and the incorporation of an epithelial component into these lesions introduces the risk of a misdiagnosis of both carcinoma and lymphoma. The following report describes nodular hyperplasia of lymphoglandular complexes within the rectum of 2 dogs.
In the first case, a 12-year-old male Chinese crested dog was presented for acute large bowel diarrhea and tenesmus after 3 weeks of boarding. Similar episodes of diarrhea occurred over the previous 2 years after antibiotic or non-steroidal anti-inflammatory treatment, with 1 episode of hematochezia. The dog was regularly treated with anthelminthics.
Rectal endoscopic examination identified 2 large masses (each over 1 cm in diameter) and 3 small masses (less than 5 mm in diameter), arising from the dorsal and ventral rectal walls, 3 to 7 cm cranial to the anus. The masses were broad-based with a sunken center and intact overlying epithelium (both with white light and narrow-band imaging). The colon and distal ileum were assessed and no other masses were noted. Needle aspirates were collected from one of the masses for cytology. The samples contained cuboidal epithelial cells displaying mild to moderate pleomorphism and arranged in tubular-type structures within a background of lymphocytes and plasma cells. An epithelial malignancy was considered unlikely but could not be entirely excluded.
In the second case, a 9-year-old male neutered cross-breed dog presented with frequent bleeding during defecation and tenesmus. The dog was originally treated with antibiotics for a suspected anal gland infection, but the problem persisted. Further endoscopic investigation of the rectum revealed a small pale raised polypoid lesion arising from the rectal mucosa.
Masses from both cases were excised for histological examination. The lesions were well-circumscribed and largely confined to the submucosa (Fig. 1a, b). Histologically, the masses were essentially identical in composition, containing irregularly shaped tubuloacinar structures lined by tall columnar epithelium. The epithelium was crowded and sometimes displayed loss of polarity (Fig. 1c, d). The epithelial cells displayed mild anisokaryosis, sometimes with hyperchromatic nuclei and prominent nucleoli, and frequent mitoses were present. Surrounding these glands, there were lymphocytes arranged in loosely follicular architecture with occasional germinal centers intermixed with plasma cells and histiocytes. The lymphoid cell population displayed a high mitotic rate. Immunohistochemical phenotyping of the lymphocytes was performed as previously described,3,17 using normal canine lymph node as control tissue. The lymphocytes immediately surrounding and adjacent to the glandular structures were predominantly CD20 and Pax-5 positive, consistent with a B-cell phenotype, and these foci were encircled by a population of CD3-positive cells, consistent with T cells (Fig. 2a, b). Low numbers of both T and B cells infiltrated the epithelium of the glandular structures, and occasional intraepithelial clusters of B cells were identified. In some areas, the epithelial lining of the tubuloacinar structures was discontinuous, with apparent protrusion of the lymphoid tissue into the lumen. In the first case, the overlying mucosa was inflamed and mildly eroded, while the mucosa in the second case was unremarkable. No infectious agents were identified in the tissues histologically, and bacterial culture of the tissue in the first case yielded only light growth of mixed gastrointestinal flora.
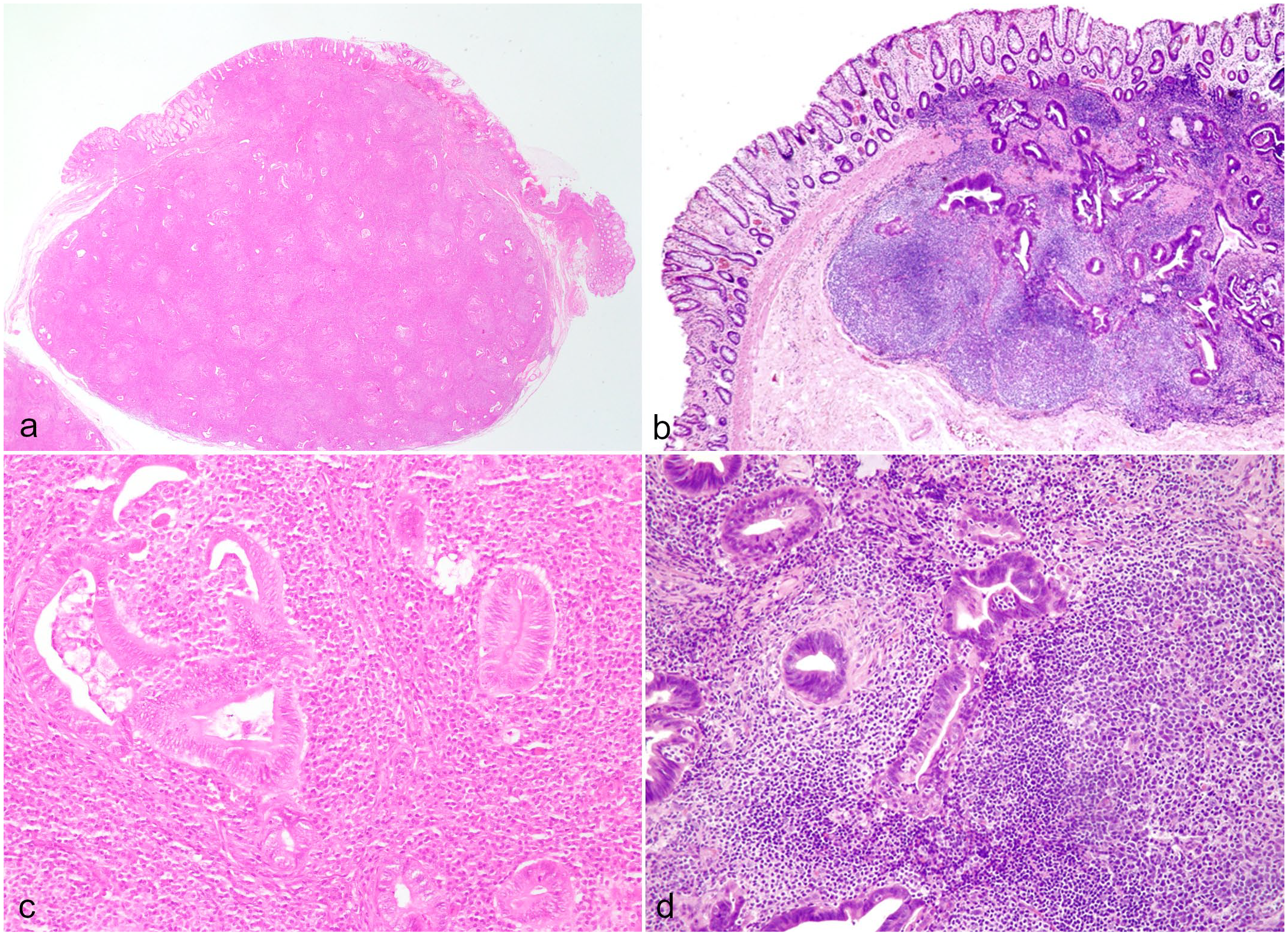
Lymphoglandular complex nodular hyperplasia, rectum, dog. Excisional rectal biopsies from case 1 (a and c) and 2 (b and d), both demonstrating well-demarcated submucosal masses containing a dual population of lymphoid and epithelial tissue. Hematoxylin and eosin.
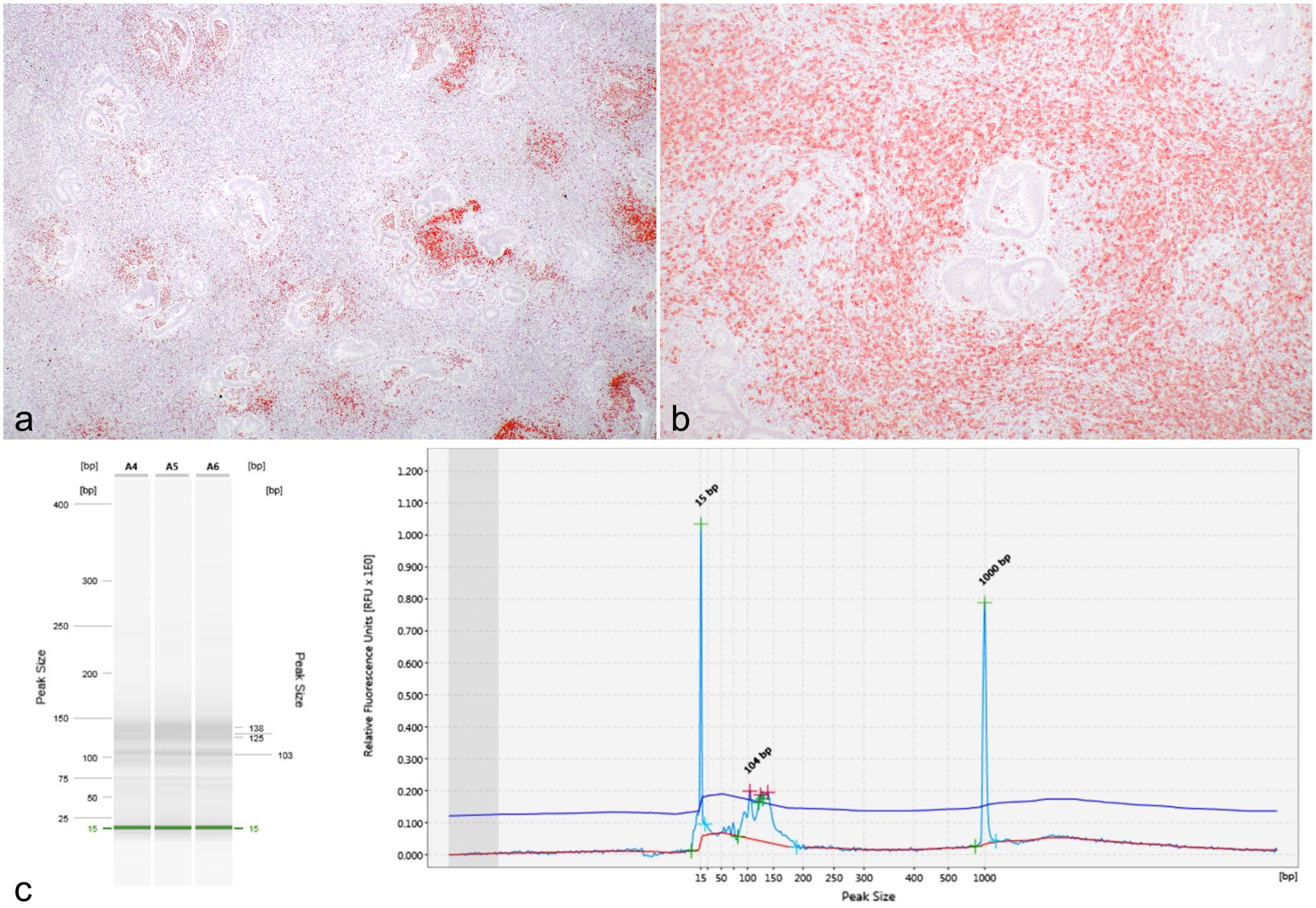
Lymphoglandular complex nodular hyperplasia, rectum, dog. (a) Photomicrograph of Pax-5 immunohistochemistry within the rectal mass, displaying aggregates of strongly Pax-5-positive cells surrounding tubuloacinar structures. Case 1. 3,3’-diaminobenzidine (DAB) chromogen, hematoxylin counterstain. (b) Photomicrograph of CD3 immunohistochemistry within the rectal mass, demonstrating a cuff of CD3-positive cells that surround the central foci of B cells and tubuloacinar structures. Case 1. DAB chromogen, hematoxylin counterstain. (c) Polymerase chain reaction (PCR) to detect antigen receptor rearrangement (PARR) in the T-cell receptor gamma gene for case 1. The non-neoplastic lymphocytic (polyclonal) population presents as a smear (A4-A6) between 125 and 138 bp in the PCR gel and in the electropherogram produce multiple small, broad peaks rather than a single tall peak that is observed in clonal populations. Size markers are represented as peaks at 15 bp and 1000 bp in the electropherogram and green lines in the PCR gel.
Polymerase chain reaction for antigen receptor rearrangement (PARR) testing was performed for both cases at the Michigan State University Veterinary Diagnostic Laboratory by extracting DNA from 5 serial 5-μm-thick sections (25 μm thickness total) from formalin-fixed, paraffin-embedded tissues. Rearrangements of the T-cell receptor gamma and the immunoglobulin heavy chain gene variable regions were assessed by amplifying the complementary determining region 3 (CDR3), as previously described.4,18 Clonality was not detected for either receptor and based on this result and the histological appearance, the masses were diagnosed as nodular hyperplasia of lymphoglandular complexes (Fig. 2c).
In both cases, the tenesmus resolved soon after surgery, with only a single small nodule 2 to 3 mm in size palpated on rectal examination 4.5 months after surgery in the first dog and no further progression over a 4-year follow-up, while the second dog was clinically unremarkable through to 6-month follow-up.
Lymphoglandular complexes have been found throughout the intestinal tract of dogs, but occur with particular abundance in the duodenum, colon, and rectum. 6 Two architectural forms of lymphoglandular complex have been described: Strauchdrüsen (shrub glands), which form from branching projections of a single epithelial tubule, and Büscheldrüsen (tufted glands), in which multiple adjacent epithelial tubules independently invest the lymphoid tissue. 6 Büscheldrüsen were reported as most prevalent form in dogs, but it is uncertain which form of lymphoglandular complex these cases most closely resemble, as the communication between the epithelial structures and the overlying mucosa was unclear from the histological sections. Nevertheless, the masses described in this report displayed features broadly consistent with lymphoglandular complexes reported in dogs and other species—albeit of greater size and complexity—and are considered to represent hyperplastic enlargement of these structures. Lymphoid proliferation with prominent follicle formation has been reported in porcine lymphoglandular complexes in association with C. jejuni infection, 12 but no evidence of campylobacteriosis or any other infectious disease was identified histologically or by bacterial culture in the present cases. Based on the history of previous episodes of acute diarrhea after medical treatment, the neutrophilic inflammatory component found on endoscopic biopsy and resolution without specific treatment, an episode of acute colitis was suspected to be the trigger for hyperplasia in the first case. No further information about the cause was determined as the dog was boarded when the tenesmus and diarrhea initially developed. However, the presence of multiple concurrent and persistent masses in the first case suggests this dog may have had an underlying predisposition to development of such lesions.
Similar masses containing varying proportions of epithelial and lymphoid components have been reported rarely in the human literature,9,15,19 though some of these cases are more consistent with inverted adenomatous polyps or submucosal herniation of colonic glands than primary proliferation of the lymphoglandular complexes. The case documented by Zhou et al, designated an inverted lymphoglandular polyp, bears closest resemblance to the lesions described here, but the arrangement of the epithelial component was slightly different, comprising sparse, irregular ectatic glandular structures rather than the small, contorted tubuloacinar foci observed in these dogs. This discrepancy may reflect anatomical differences in human lymphoglandular complexes, which typically contain relatively few, superficially distributed epithelial projections. 14
The distinctive histological features of this condition were rectal submucosal masses that were well-circumscribed and displayed integration of glandular structures into lymphoid tissue. Lymphoid follicles were centered on these glands, and there was extension of the lymphocytic population into the gland lumina. Several notable differentials were considered for this case. Submucosal herniation of crypts as seen in focal colitis cystica profunda could have a similar appearance, but while there may be secondary inflammation with this condition due to compression or rupture of crypts, it lacks the intimate association between lymphoid and glandular tissues seen in these cases. Furthermore, goblet cells typical of crypt mucosa were absent from the epithelium in the lymphoglandular complex. Lymphoma arising within a lymphoglandular complex was a concern due to the nests of lymphocytes in the glandular epithelium. However, the mixed population of inflammatory cells throughout the lymphoid tissue, the presence of recognizable follicular architecture, the lack of clinical progression, and the absence of detectable clonality with PARR testing makes this diagnosis highly improbable. An invasive adenocarcinoma was a further consideration, but the dispersed distribution of glands within the lymphoid tissue, close association between lymphoid and epithelial components, lack of stromal reaction or invasion outside of the mass, retention of basement membrane surrounding glands, absence of marked anaplasia within the masses or the overlying mucosa, and the lack of progression all refute a diagnosis of invasive adenocarcinoma. Failure to account for these factors engenders considerable risk of a misdiagnosis of malignancy, particularly as the submucosal location could make the procurement of truly representative samples problematic.
These 2 cases of lymphoglandular complex nodular hyperplasia demonstrate a non-neoplastic disease in the rectum that must be considered as a potential diagnosis for any submucosal mass containing both lymphoid and epithelial components. Considering the prognostic implications of possible differentials, such as adenocarcinoma or lymphoma, it is imperative that biopsy samples are of sufficient depth and quality to allow careful scrutiny and recognition of this condition.
Footnotes
Acknowledgements
We thank Annie M Zimmerman for her contribution to pathological assessment of these cases.
Authors’ Note
Julien R. S. Dandrieux is now affiliated to Royal (Dick) School of Veterinary Studies, Edinburgh, Scotland.
Author Contributions
AWS, MK, RL, and MMB performed pathological evaluations; MK and MMB performed PARR analysis; JRSD performed clinical investigations; the manuscript was written by AWS with contribution from the other authors.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
