Abstract
Conjoining of the major pancreatic duct and common bile duct at the major duodenal papilla (MDP) is suspected to predispose cats to the clinical syndrome of “triaditis.” However, microanatomy of the MDP or presence of lesions at the MDP has not been assessed in cats with or without triaditis. The aims of this study were to characterize feline MDP histomorphology and to identify associations between MDP anatomy/disease and the presence of biliary, pancreatic, or intestinal inflammation or neoplasia. Histologic assessment was prospectively performed on the MDP, duodenum, jejunum, ileum, liver, and pancreas from 124 client-owned cats undergoing postmortem examination. The majority of cats (104/124, 84%) had a complex ductular network at the MDP, with no distinction between pancreatic and common bile ducts. Lymphoid aggregates at the MDP were common (63/124, 51%). Inflammation of the MDP (MDPitis) was present in 35 of 124 cats (28%) and was often concurrent with cholangitis, pancreatitis, or enteritis (32/35, 91%), but was only associated with enteritis (19/35, 54%, P < .05). Triaditis was less common (19/124, 15%), but was associated with both conjoined MDP anatomy (19/19, 100%, P < .05) and MDPitis (12/19, 63%, P < .05). Neoplasia was present in 37 of 124 cats (29%), with lymphoma (28/37, 78%) predominating. Enteropathy-associated T-cell lymphoma type 2 (EATL2) was most common (n = 16/37, 43%) and was associated with triaditis and MDPitis (P < .05). These findings suggest that anatomy, immune activation, and/or inflammation of the MDP may play a role in the pathogenesis of triaditis. Further studies are needed to elucidate the relationships between triaditis, MDPitis, and EATL2.
Concurrent pancreatitis, cholangitis, and enteritis, colloquially referred to as “triaditis,” can cause significant morbidity in domestic cats.12,20,26,30 Antemortem diagnosis of this syndrome is hindered by a variety of factors, including nonspecific clinical signs and insensitive clinicopathologic data as well as reliance upon histologic evaluation of affected tissues for definitive diagnosis.12,20,30 Inflammation in these organs can often be subdivided by the type of inflammatory infiltrate, with either neutrophilic or lymphoplasmacytic inflammation being the most common.9,35 Neutrophilic inflammation is most often associated with bacterial infections, while the cause of lymphoplasmacytic inflammation is more nebulous and is variably attributed to immune-mediated disease, chronic infection, or idiopathic processes. Ultimately, the underlying pathogenic mechanisms causing triaditis are currently unknown.
Historically, cats are thought to be predisposed to triaditis due to the anatomy of their major duodenal papilla (MDP), in which the common bile duct and pancreatic duct conjoin at the ampulla of Vater prior to entering the duodenum (Supplemental Figure S1). However, the frequency of this conjoined anatomy in cats is unknown. To our knowledge, detailed histologic characterization of the MDP in cats is currently limited to studies that only examined a total of 5 asymptomatic cats.2,32 Histology of the MDP has not been described in cats with pancreatic, intestinal, or hepatobiliary disease. Therefore, a contributory effect of MDP pathology in facilitating extension of disease (eg, inflammation, infection, and neoplasia) between the pancreas, intestine, or hepatobiliary system remains unknown.
Given the paucity of data regarding the feline MDP and the clinical significance of this location in triaditis, further characterization of this site is warranted. The aims of this study were 3-fold. First, we characterized the histologic anatomy of the MDP in cats presented for routine postmortem examination. We hypothesized that the majority of cats would exhibit joining of pancreatic and common bile ducts prior to entry into the duodenum. Second, we performed histologic evaluation of the MDP and associated organs (liver, pancreas, and intestine) for the presence of concurrent inflammation. We hypothesized that cats with pancreatitis, enteritis, and/or cholangitis would have concurrent inflammation of the MDP (herein referred to as major duodenal papillitis, or MDPitis). We also hypothesized that cats with triaditis would be more likely to have MDPitis than cats without triaditis. Finally, given the propensity for cats to develop neoplasia in the systems of interest, we evaluated cats for (1) the presence of multi-organ neoplasia in the pancreas, small intestine, hepatobiliary system, and MDP and (2) any association between identified inflammatory lesions and enteropathy-associated T-cell lymphoma type 2 (EATL2). We hypothesized that the spread of neoplasia to one or more of the examined organs would be common and that cats with inflammatory lesions in examined tissues would be more likely to have concurrent EATL2.
Materials and Methods
Samples were collected prospectively from cats that were presented for postmortem examination at North Carolina State University College of Veterinary Medicine between August 2017 and March 2020, and ethical approval was given for the use of tissues for teaching and research for patients upon submission for autopsy. Any cat submitted for autopsy examination was considered eligible for study inclusion. Tissues collected included an en-bloc section of the duodenum at the level of the MDP, including adjacent the pancreas, pancreatic duct, and common bile duct; the tail of the pancreas; 1 section of jejunum; 1 section of ileum; and 2 sections of liver (left lateral lobe and right medial/quadrate lobe adjacent to and variably including gallbladder; Supplemental Figure S1). Tissues were fixed in 10% neutral buffered formalin and trimmed. For evaluation of the MDP, 2 to 4 serial transverse sections that were perpendicular to the longitudinal axis of the duodenum were made through the duodenum at the site of the MDP; the goal of this method of trimming was to capture multiple sections of the pancreatic and common bile duct prior to and at the entry point into the duodenum. The remaining tissues were routinely trimmed, processed, and stained with hematoxylin and eosin for microscopic examination. All slides were evaluated by one pathologist (MES), and a random subset of slides were further evaluated by a second pathologist (JMC) for confirmation of findings. Pathologists were not consistently blinded to the clinical histories of the patients.
Enteritis, cholangitis, and pancreatitis were diagnosed based on previously established guidelines as determined by the World Small Animal Veterinary Association (WSAVA; https://wsava.org/). Briefly, the criteria used for the diagnosis of enteritis included both expansion of epithelium and lamina propria by inflammatory cells (neutrophils, lymphocytes, plasma cells, or eosinophils; inflammatory criteria), as well as morphologic criteria (villus fusion and blunting, epithelial injury, crypt dilation, etc). A cat was considered positive for enteritis if inflammation was present in any of the 3 evaluated segments (duodenum, jejunum, or ileum), and the highest severity score of these 3 segments was recorded. Enteritis was differentiated from EATL2 using previously defined guidelines8,19,23,24,37 and included evaluation of the location of small lymphocytes, the presence of admixed leukocytes, density of lymphocytes, and the presence of intraepithelial nests and/or plaques of small lymphocytes. In cases with concurrent enteritis and “emerging” or “early” EATL2, cats were recorded as having both enteritis and neoplasia. For any neoplasm, small intestine was considered to be affected if neoplastic cells were present in any associated tissue layer (ie, mucosa, submucosa, muscularis, serosa, and/or intravascular). For cases of non-EATL2 lymphoma, neoplasia was classified based on cell size (ie, small vs large cell lymphoma) and/or any other identifying diagnostic features as applicable (ie, intravascular lymphoma). Large cell lymphoma was not further classified by immunohistochemistry (ie, T vs B cell origin was not determined). Cholangitis was defined by the presence of inflammatory cells specifically cuffing and/or infiltrating the biliary mucosa and was further supported by the presence of bile duct proliferation (type I ductular reaction) and/or biliary epithelial injury (attenuation, apoptosis, necrosis, hyperplasia, and/or ductopenia). Cholangitis was categorized as either neutrophilic or lymphocytic. Lymphocytic cholangitis was differentiated from biliary spread of EATL2 by the presence of ductular reaction, biliary epithelial invasion, biliary injury, and the presence of other leukocytes (eg, macrophages, plasma cells) and/or lipogranulomas, as previously described. 35 Additional liver lesions (eg, hepatic lipidosis, necrosis, non-specific reactive hepatitis) were also recorded. Pancreatitis was defined by the infiltration of the interstitium, serosa, or exocrine acini and ducts by inflammatory cells, and was further supported by saponification of fat and tissue necrosis (acute) or interstitial fibrosis, nodular regeneration, or acinar to ductal metaplasia (chronic). Other pancreatic lesions (eg, nodular hyperplasia, islet amyloidosis) were also recorded. MDPitis was defined as expansion of the submucosa by clusters of 5 or more inflammatory cells and was further supported by the presence of epithelial ulceration, necrosis, or exocytosis of inflammatory cells into the lumen. The criteria used to distinguish inflammation from EATL2 in the small intestine were also used for distinguishing these diseases in the MDP; the presence of neoplasia was also defined as for the small intestine. Triaditis was histologically defined as the presence of concurrent cholangitis, pancreatitis, and enteritis. For all inflammatory lesions, severity was subjectively scored as mild (1), moderate (2), or marked (3). Both enteritis and pancreatitis severity scores were adapted from previously published guidelines; guidelines utilized for enteritis were also applied for assessment of MDPitis.8,9 Briefly, for enteritis, mild = 20 to 50 lymphocytes and plasma cells per 40× field (field number 22, 0.237 mm2), up to 20 eosinophils per 40× field, or up to 20 scattered neutrophils per 40× field; moderate = 50 to 100 lymphocytes and plasma cells per 40× field, 20 to 50 eosinophils per 40× field, or 20 to 50 neutrophils per 40× field; marked = >100 lymphocytes and plasma cells per 40× field, >50 eosinophils per 40× field, or >50 neutrophils per 40× field. 8 For pancreatitis, mild = inflammation affecting <25% of parenchyma, moderate = inflammation affecting 25% to 50% of parenchyma, and marked = inflammation affecting >50% of parenchyma. 9 Cholangitis scores were developed through referencing WSAVA guidelines as well as previous studies7,34 and were defined by the following density of inflammatory cells surrounding bile ducts as previously described: mild = 1 to 2 cell layers, moderate = 3 to 5 cell layers, and marked = >5 cell layers.
Data were retrospectively collected from medical records for each cat, including signalment (age, sex, breed) and significant findings from the postmortem examination (primary lesions identified, cause of death, and the presence of other reported hepatobiliary or gastrointestinal lesions identified on postmortem examination). Additional slides from the postmortem examination sample set were reviewed for confirmation of lesions as warranted. Inflammatory lesions and/or neoplasms affecting stomach or large intestine were noted, but not included in tabulation of small intestinal pathology. Where data were available and review was warranted for individual animals (ie, cases of triaditis), antemortem clinical and clinicopathologic data (including history, clinical presentation, complete blood count, serum biochemistry panel, and diagnostic imaging) were reviewed and correlated to histopathologic lesions. Clinicopathologic data up to 6 months prior to postmortem evaluation were considered in the review. In order to characterize lymphoid aggregates at the MDP, for a subset of 4 individuals (2 cats with no intestinal lesions, 2 cats with small intestinal EATL2), immunohistochemical labeling for CD3 and CD20 was performed on the MDP using commercially available antibodies (Supplemental Table S1). Briefly, automated immunohistochemical staining was performed using the intelliPATH FLX (Biocare Medical LLC, Pacheco, California), and sections were evaluated using the ImPRESS polymer detection kit (Vector Laboratories, Burlingame, California). Positive tissue controls consisted of appropriate feline tissues (lymph node). Negative tissue controls consisted of tissue sections in which the primary antibodies were replaced by phosphate-buffered saline and negative antibody controls consisted of non-immune mouse immunoglobulins (Mouse Super Sensitive Negative Control, BioGenex, Fremont, California).
Associations between paired categorical variables were analyzed using 2-tailed Fisher’s exact probability tests, with a P-value of <.05 being considered significant (VassarStats, Poughkeepsie, New York). Logistic regression was used to examine the relationships between triaditis or individual tissue inflammation and complex MDP anatomy, prominent MDP lymphoid tissue, MDPitis, and the presence of EATL2, with significance set at α = .05; collated data from this analysis is available in Supplemental Table S2. Fisher’s exact test was applied in cases where there was complete separation by a single covariate. Associations between numerical variables (ages of cats with or without triaditis) were analyzed using a Mann-Whitney rank sum test.
Results
Case Demographics
A total of 124 cats were included in the study. Cats ranged from 1 month to 21 years of age (average, 10.13 years; median, 11 years). The age of 3 cats was unknown. There were 68 castrated males, 49 spayed females, 5 unneutered males, and 2 unspayed females. The majority of cats were classified as domestic shorthair, medium hair, or longhair (n = 100). Remaining breeds included Maine coon (n = 4), Himalayan (n = 3), ragdoll (n = 3), Persian (n = 3), Siamese (n = 2), Russian blue (n = 2), Bengal (n = 1), Abyssinian (n = 1), ragamuffin (n = 1), exotic shorthair (n = 1), British shorthair (n = 1), Sphynx (n = 1), and Birman (n = 1).
Major Duodenal Papilla Histomorphology
The majority of examined MDP (n = 104/124, 84%) lacked 2 distinct ductular lumina at the entry point into the duodenum in any examined section (Fig. 1a). Instead, this area comprised a complex network of serpiginous, interweaving ducts that were lined by a simple cuboidal to columnar epithelium. This histologic appearance was interpreted to represent conjoining of the common bile duct and pancreatic duct prior to entry at the MDP, and here after will be referred to as “conjoined anatomy.” Collagenous stroma intercalated between ducts, and several small, isolated bundles of smooth muscle surrounded the entire network of ducts. This musculature was distinct from the duodenal muscularis layer. No distinguishing features were present to differentiate pancreatic from biliary ducts. Uncommonly (n = 20/124, 16%), 2 distinct and separate ducts were identified in one or more examined MDP sections (Fig. 1b).
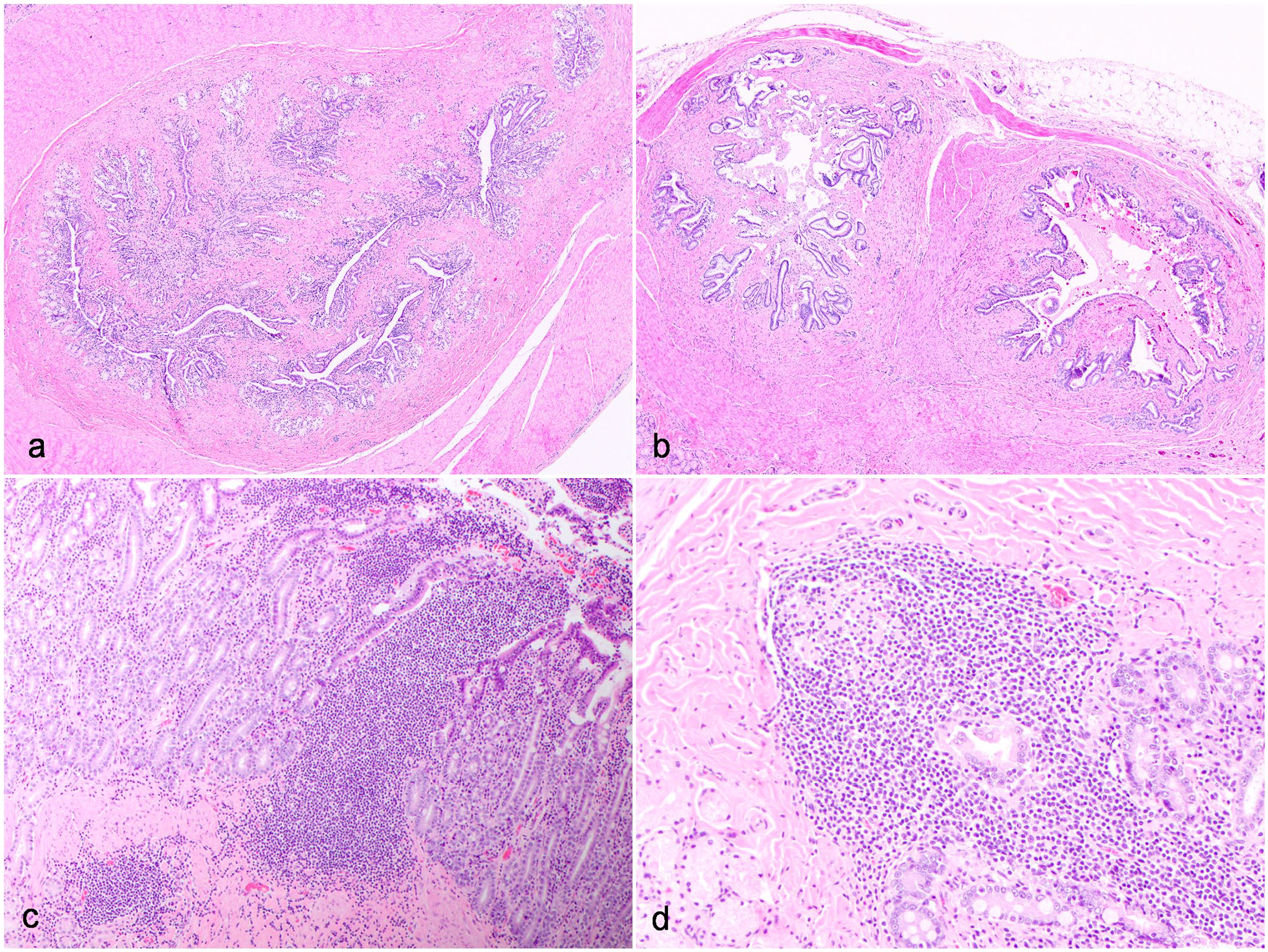
Major duodenal papilla (MDP) histomorphology in domestic cats (Hematoxylin and eosin). (a) Conjoined anatomy of the MDP, domestic cat (case 22). The majority of cats have a complex ductular network at the MDP. (b) Separate biliary and pancreatic ducts at the MDP, domestic cat (case 87). A minority of cats had 2 distinct and separate ducts visualized at the MDP. (c and d) Prominent lymphoid aggregates at the MDP, domestic cat (case 86). Prominent lymphoid aggregates are present near or within the MDP in the majority of cats. (c) In some cases, lymphoid aggregates formed dense sheets without overt organization. (d) In other cases, lymphoid aggregates showed vague follicular organization.
In more than half of the examined MDP and surrounding duodenum (n = 63/124, 51%), one or more prominent submucosal lymphoid aggregates were present. Lymphoid aggregates were present either within the submucosa of the MDP itself, within the duodenum submucosa and/or lamina propria at the entry point of the MDP, or within the duodenum submucosa and/or Brunner’s glands immediately adjacent to the MDP. Lymphoid aggregates either were comprised of dense well-demarcated sheets of small lymphocytes or formed follicle-like aggregates with variably prominent antigen-related polarity (Fig. 1c–d). Immunohistochemical labeling for CD3 and CD20 on a subset of 4 cats revealed that the majority of lymphocytes in these aggregates were CD20-positive, with peripheral cuffs of CD3-positive cells, consistent with lymphoid follicle organization (Fig. 2). Lymphoid aggregates with follicle-like organization was present in all cats examined, including those with MDPitis/duodenitis and/or duodenal enteropathy-associated T-cell lymphoma (EATL2; Fig. 2).
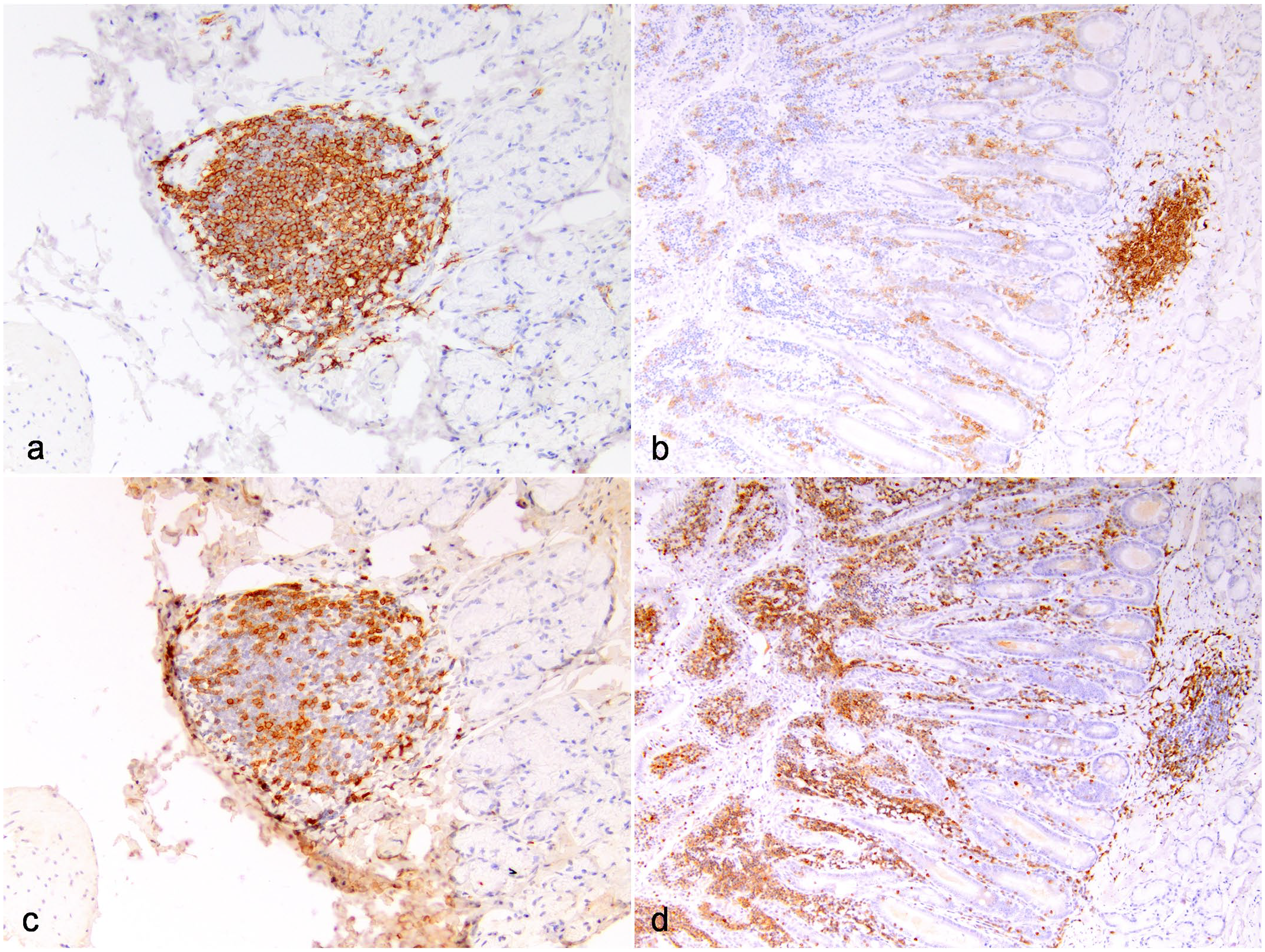
Lymphocyte aggregates at the feline major duodenal papilla (MDP) represent lymphoid tissue. Lymphocyte aggregates were consistently comprised of a central aggregate of CD20-positive lymphocytes representing follicular formation (a and b, immunohistochemistry, CD20) with peripheral cuffs of CD3-positive lymphocytes (c and d, immunohistochemistry, CD3). (a) and (c) Lymphoid tissue at the MDP in a cat with histologically unremarkable small intestine, domestic cat, case 105. (b) and (d) Lymphoid tissue at the MDP in a cat with enteropathy-associated T-cell lymphoma type 2 (EATL2) in the duodenum arising on a background of chronic lymphoplasmacytic enteritis, domestic cat, case 108.
Major Duodenal Papilla Lesions
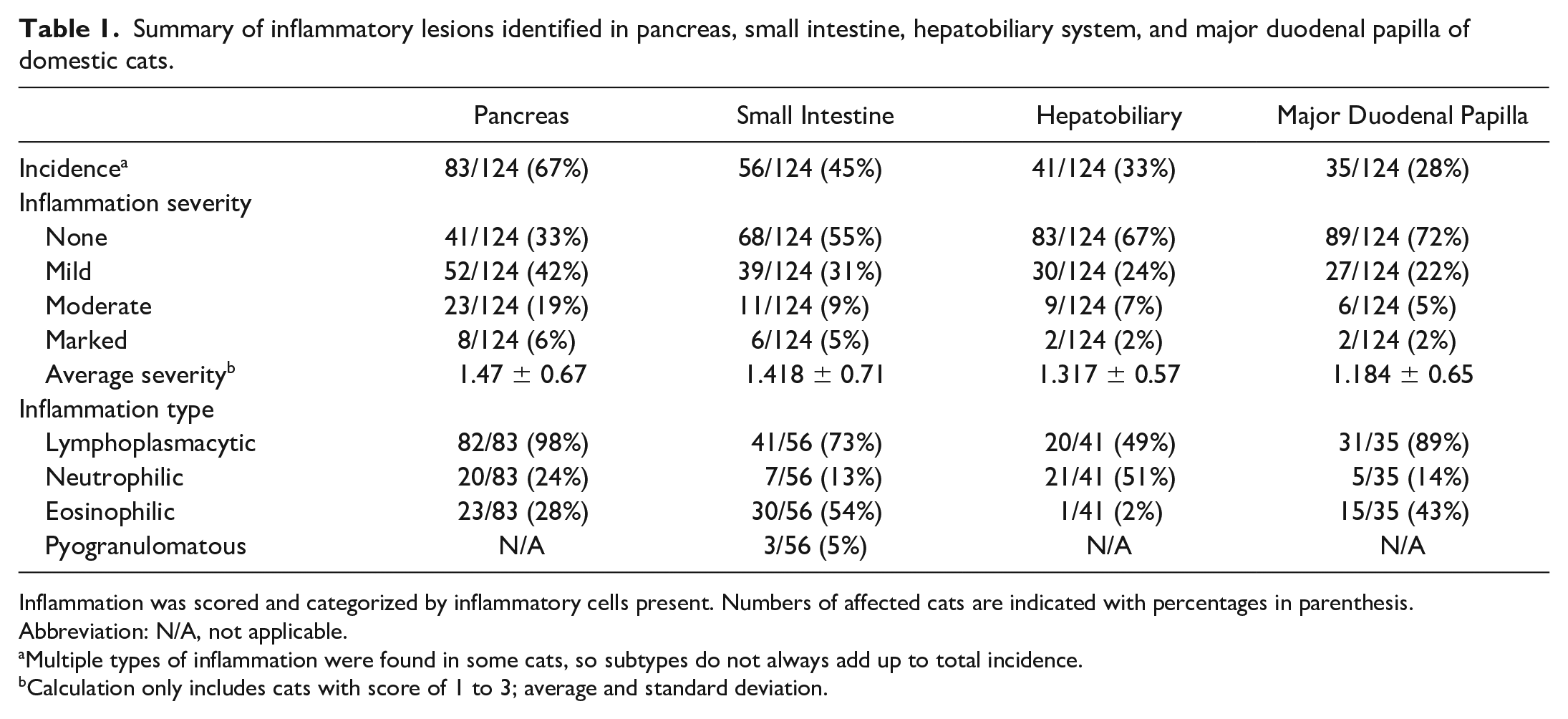
A total of 43 of 124 (34.7%) cats had a lesion at the MDP. These lesions were categorized as inflammatory (MDPitis), neoplastic, and non-inflammatory/non-neoplastic (Supplemental Figure S2). Major duodenal papillitis was the most common lesion identified at the MDP (n = 35/43 cats with MDP lesions, 81%, Supplemental Figure S2) and was mild in the majority of cats (Table 1). Lymphoplasmacytic inflammation was most common, followed by eosinophilic and neutrophilic inflammation (Table 1).
Summary of inflammatory lesions identified in pancreas, small intestine, hepatobiliary system, and major duodenal papilla of domestic cats.
Inflammation was scored and categorized by inflammatory cells present. Numbers of affected cats are indicated with percentages in parenthesis.
Abbreviation: N/A, not applicable.
Multiple types of inflammation were found in some cats, so subtypes do not always add up to total incidence.
Calculation only includes cats with score of 1 to 3; average and standard deviation.
Non-inflammatory/non-neoplastic lesions were the next most common lesions (n = 11/43, 26%) and included duct dilation, periductular fibrosis, epithelial injury (ie, erosion, ulceration, hyperplasia, or attenuation), subjective duct enlargement, hemorrhage, and luminal pancreatolithiasis (Fig. 3). The majority of these lesions were seen in conjunction with MDPitis (Supplemental Figure S2). Among cats with concurrent inflammation and non-inflammatory/non-neoplastic lesions, 4 of 7 cats had moderate to marked MDPitis.
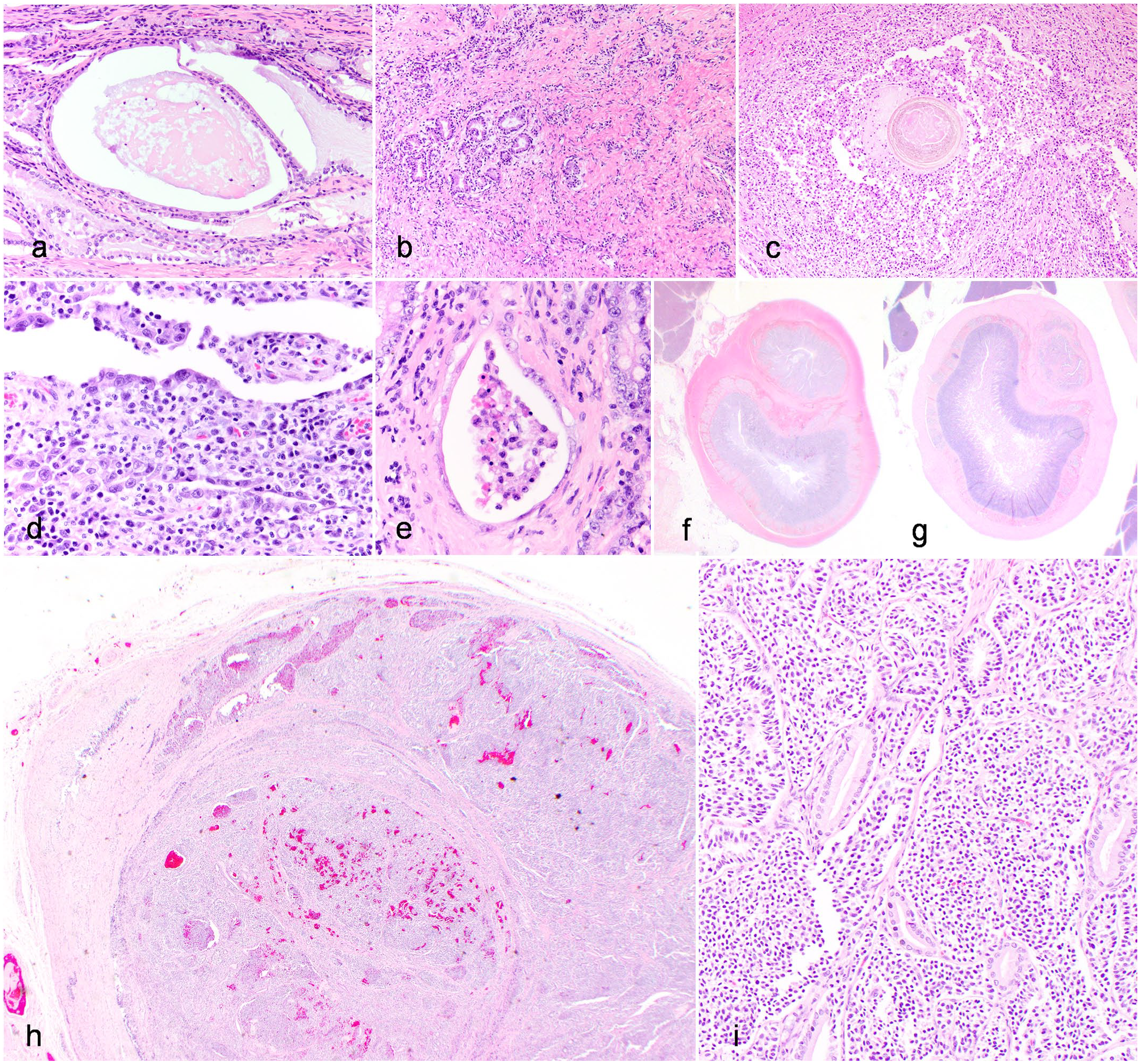
Lesions of the major duodenal papilla (MDP) in domestic cats (Hematoxylin and eosin). (a–f) Non-inflammatory/non-neoplastic lesions seen at the MDP in cats. (a) Dilation of ducts in the MDP, domestic cat, case 44. Dilation is often accompanied by concurrent luminal mucin and/or proteinaceous fluid. (b) Fibrosis in the MDP, domestic cat, case 82. Increased collagenous stroma is present between ducts, with multifocal loss of ducts. (c) Pancreatolithiasis at the MDP, domestic cat, case 31. A spherical concretion of material is present in a duct at the MDP with concurrent ulceration and suppurative inflammation. Similar concretions were identified in adjacent pancreatic ducts (not pictured). (d) Epithelial injury of the MDP, domestic cat, case 111. There is erosion to ulceration of the mucosa, with concurrent mixed inflammation in the underlying lamina propria. (e) Crypt abscesses-like lesion of the MDP, domestic cat, case 58. There is distension of a crypt at the conjoining of the duodenum and MDP and filling of the lumen with necrotic cellular debris. (f) Enlargement of the MDP, domestic cat, case 94. The MDP is enlarged with a diameter approximately half the size of the duodenum. (g) Normal size of MDP, domestic cat, case 118. In contrast to image (f), the MDP diameter is approximately one-fourth the diameter of the duodenum. (h and i) Carcinoid adjacent to the MDP, domestic cat, case 76. (h) An expansile and infiltrative mass fills and obliterates normal bile duct architecture. (i) The neoplasm comprises packets and nests of polygonal neoplastic cells supported by a fine fibrovascular stroma, consistent with a neuroendocrine neoplasm (carcinoid).
Neoplasia was found either directly affecting the MDP (n = 4) or immediately adjacent to the MDP (n = 2). These included 3 cats with EATL2, 1 cat with large cell gastrointestinal lymphoma, 1 cat with a carcinoid arising from the common bile duct adjacent to the MDP (Fig. 3), and 1 cat with an adenoma arising from the proximal duodenum adjacent to the MDP.
Characterization and Incidence of Pancreatitis, Cholangitis, Enteritis, and Triaditis
Inflammation of the pancreas (83/124, 67%), small intestine (56/124, 45%), and biliary system (41/124, 33%) were commonly observed in this population of cats (Table 1). Lymphoplasmacytic followed by concurrent eosinophilic infiltrates were the most common inflammatory cells identified within the pancreas and small intestine. Neutrophilic inflammation was present in nearly a quarter of pancreatitis cases (Table 1). Lymphocytic cholangitis and neutrophilic cholangitis were found at similar incidences in cats (Table 1). Concurrent inflammation in 2 or more of the examined tissues was also common, and triaditis was diagnosed in 19 of 124 (15%) cats (Supplemental Figure S3, Table 2). When investigating relationships between pancreatitis, cholangitis, and enteritis, there was a significant association between the presence of cholangitis (collectively including both neutrophilic and lymphocytic variants) and enteritis (P < .05).
Summary of inflammatory findings in cats with triaditis.
Of the 124 cats prospectively evaluated, 19 cats had concurrent inflammation in the pancreas, small intestine, and hepatobiliary system (“triaditis”). Inflammation was scored and categorized by inflammatory cells present. Numbers of affected cats are indicated with percentages in parenthesis.
Abbreviation: N/A, not applicable.
Multiple types of inflammation were found in some cats, so subtypes do not always add up to total incidence.
Association Between Anatomy of the Major Duodenal Papilla, Lesions of the Major Duodenal Papilla, Other Examined Tissues, and Triaditis
Conjoined anatomy of the MDP was associated with the presence of enteritis (P = .024) and triaditis (P = .041, Supplemental Figure S4), but not pancreatitis, cholangitis, or MDPitis. No cases of triaditis were observed among cats with separation of the common bile duct (CBD) and pancreatic duct (Supplemental Figure S4).
MDPitis was frequently present in conjunction with cholangitis, pancreatitis, or enteritis (91%, 32/35). The incidence of pancreatitis, enteritis, and cholangitis were higher in cats with concurrent MDPitis (Supplemental Figure S5), with a significant association found between the presence of enteritis and MDPitis (P = .012). Furthermore, there was significant association (P < .001) between MDPitis and triaditis (Supplemental Figure S6): 12 of 19 (63%) cats with triaditis had concurrent MDPitis, while only 23 of 105 (22%) cats without triaditis had concurrent MDPitis. Furthermore, cats with MDPitis were 6.64 times more likely to have concurrent triaditis (Supplemental Table S2). Examples of the most common inflammatory patterns seen in the tissues of cats with concurrent triaditis and MDPitis are shown in Figs. 4–6.
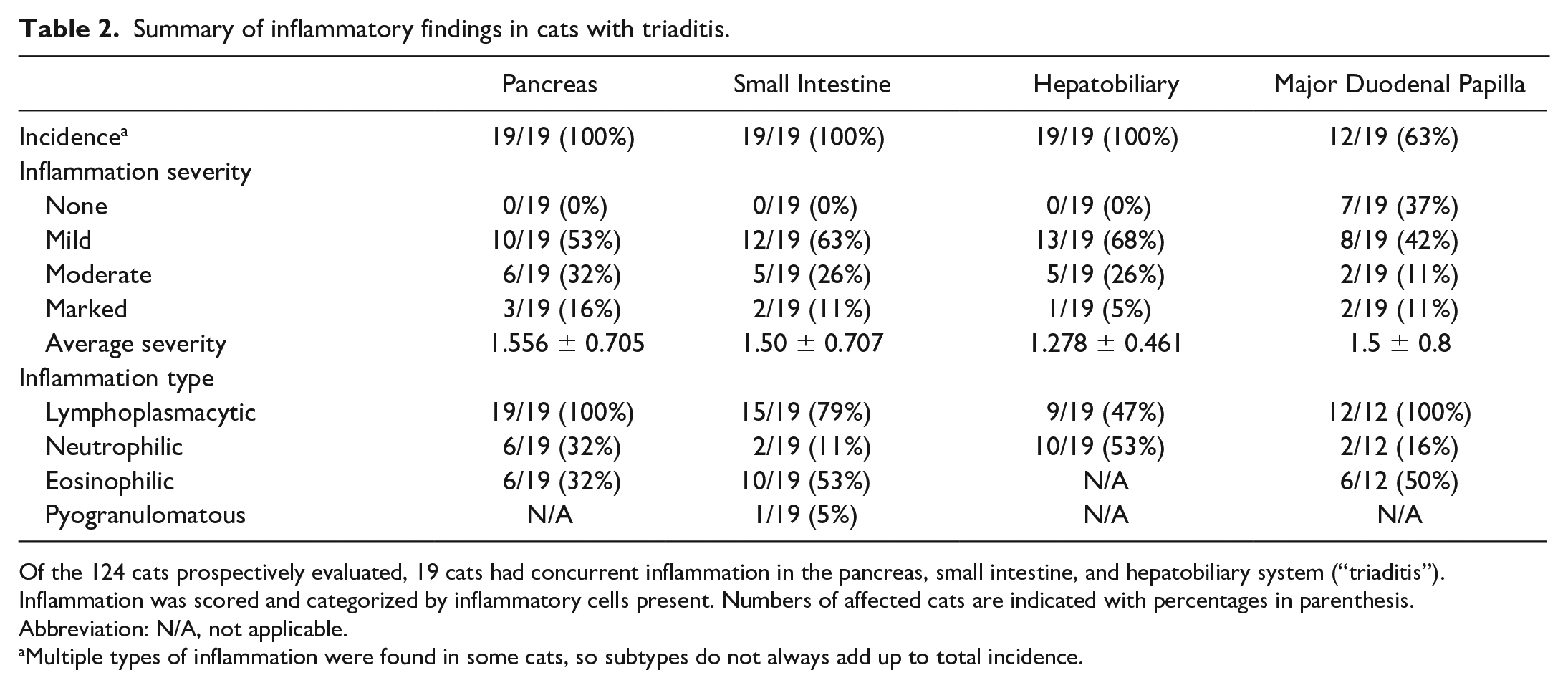
Suppurative pancreatitis, neutrophilic cholangitis, and chronic enteritis with intra-ampullary pancreatolithiasis in a domestic cat (Hematoxylin and eosin). Domestic cat, case 31. This cat had a luminal concretion within the major duodenal papilla (MDP) and adjacent pancreatic ducts (see Fig. 1c). (a) Pancreas. There is disruption of the pancreatic parenchyma by degenerate neutrophils and fibrin (suppurative pancreatitis), which extends into adjacent surrounding adipose (steatitis). (b) Liver. Neutrophils cuff and infiltrate bile ducts (neutrophilic cholangitis), which are increased in number (ductular reaction) and further surrounded by concentric spindloid cells and collagenous stroma (fibrosis, extrahepatic obstruction). (c) MDP. Ducts within the MDP are filled with degenerate neutrophils and fibrin, and the lamina propria is expanded by neutrophils, lymphocytes, and plasma cells. (d) Jejunum. The lamina propria of the villi is mildly to moderately expanded by lymphocytes and plasma cells (mild to moderate chronic lymphoplasmacytic enteritis).
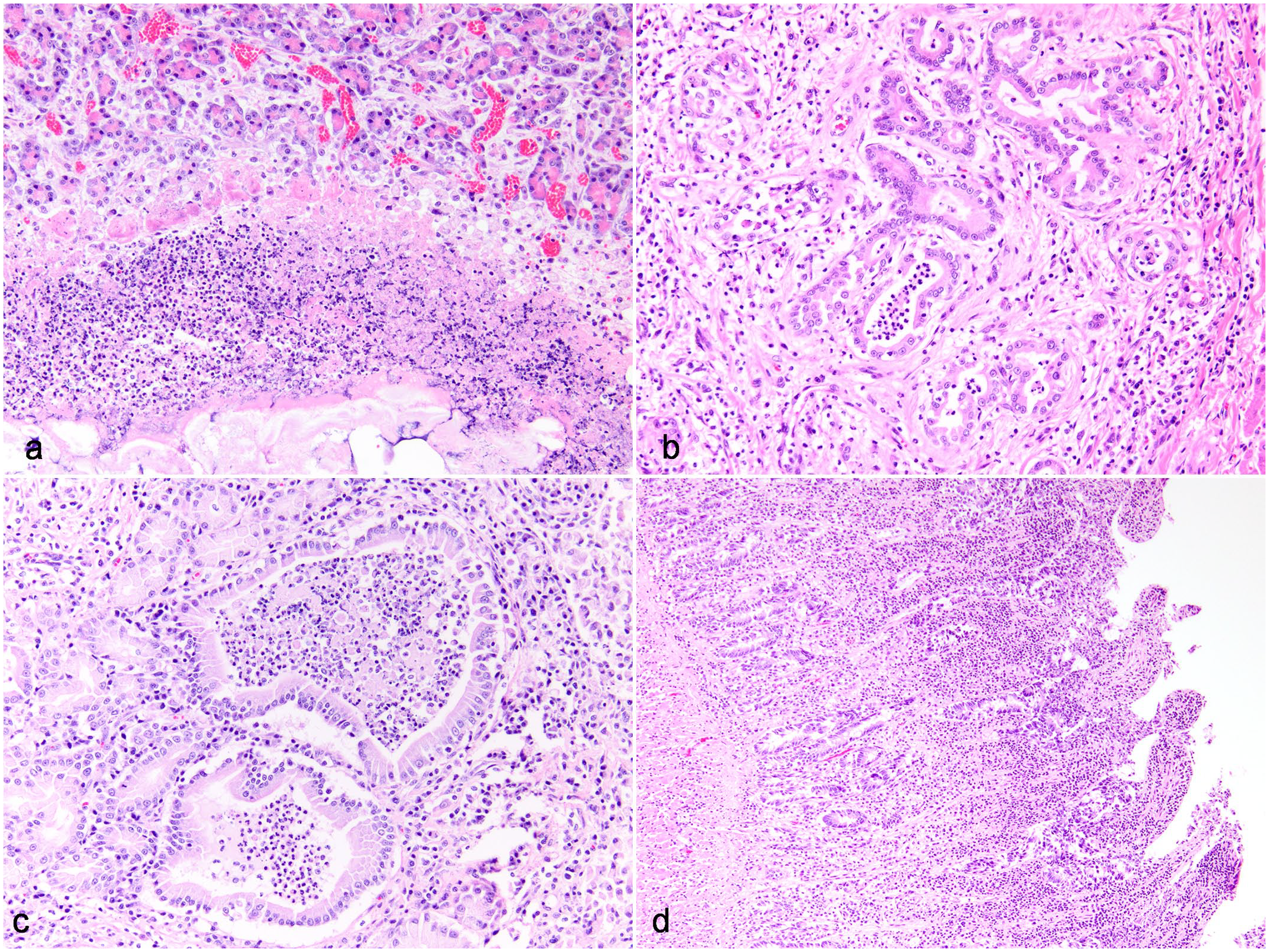
Chronic pancreatitis, cholangitis, and enteritis associated with marked extrahepatic biliary dilation in a domestic cat (Hematoxylin and eosin). Domestic cat, case 82. Grossly, this cat had significant pancreatic atrophy and fibrosis with secondary severe dilation of the extrahepatic biliary tree. (a) Pancreas. There is multifocal loss and atrophy of acini with replacement of tissue by abundant collagenous stroma with intermixed lymphocytes and plasma cells (marked chronic lymphoplasmacytic pancreatitis). (b) Liver. There is neutrophilic cholangitis with peribiliary concentric fibrosis, consistent with extra-hepatic obstruction, interpreted to be secondary to pancreatic fibrosis. (c) Major duodenal papilla (MDP). Similar inflammation and fibrosis as seen in the pancreas was present in the MDP, with concurrent dilation of multiple ducts. (d) Jejunum. There is multifocal blunting and fusion of villi with expansion of the lamina propria by lymphocytes and plasma cells (chronic lymphoplasmacytic enteritis).
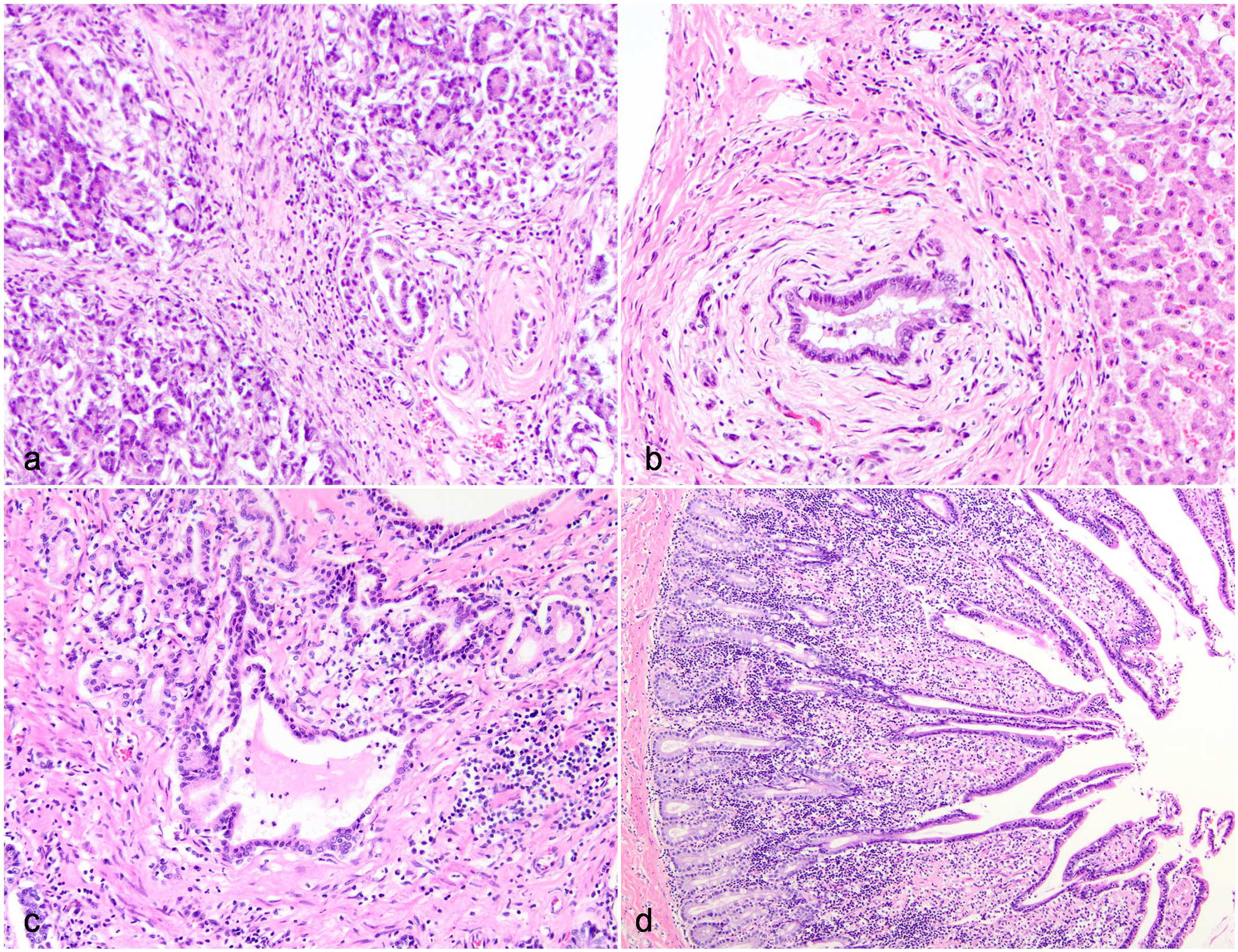
Mild chronic lymphoplasmacytic pancreatitis, cholangitis, major duodenal papillitis (MDPitis), and enteritis with emergence of small cell enteropathy-type lymphoma (EATL2) in a domestic cat (Hematoxylin and eosin). Domestic cat, case 22. The majority of cats with inflammation in examined organs exhibited mild, chronic lymphoplasmacytic inflammation in multiple organs, as evidenced in this case in all examined organs. (a) Pancreas. Lymphocytes and plasma cells expand the interstitium between acini (mild chronic lymphoplasmacytic pancreatitis). (b) Liver. Lymphocytes cuff bile ducts, with occasional infiltration into bile duct epithelium and mild ductular reaction (mild chronic lymphocytic cholangitis). (c) Major duodenal papilla. Lymphocytes and plasma cells expand the lamina propria between ducts (mild MDPitis). (d) Jejunum. There is multifocal fusion of villi and expansion of the villus and deeper lamina propria with lymphocytes, plasma cells, and fewer eosinophils (mild chronic lymphoplasmacytic and eosinophilic enteritis). Furthermore, there is evidence of transition from enteritis (top inset) to an EATL2 with transmural infiltration of small lymphocytes (arrowheads), intra-epithelial lymphocyte nests, and plaques (bottom inset).
Cats with more severe MDP inflammation or additional non-inflammatory MDP lesions tended to have concurrent triaditis. Specifically, half of the cats with moderate-marked MDPitis had concurrent triaditis (n = 4/8, 50%). Of these 4 cats, 3 had additional non-inflammatory/non-neoplastic MDP lesions, including pancreatolithiasis and MDP enlargement, fibrosis, or dilation. Furthermore, 3 of the 6 cats with neoplasia at or near the MDP had triaditis (Fig. 3h, i, Supplemental Figure S7).
MDP lymphoid aggregates were associated with enteritis (P = .04) and MDPitis (P = .04), but were not associated with pancreatitis, cholangitis, or triaditis.
Association Between Histologic Evidence of Triaditis and Antemortem Clinical and Clinicopathologic Data
Cats with triaditis were significantly older (median 15 years, range = 5–19 years, n = 19) than those without triaditis (median 10 years, range = 1 month–21 years, n = 102) (P < .001). Among cats with triaditis, 9 of 19 (47%) presented for evaluation of clinical signs attributable to gastrointestinal or hepatobiliary disease; the remainder presented for a variety of other reasons (vehicular trauma, septicemia, respiratory disease, neoplasia outside the gastrointestinal/hepatobiliary system, or euthanasia only; Supplemental Table S3). Antemortem clinicopathologic data (complete blood count and serum biochemistry panel) was available for 15 of 19 cats with triaditis; an additional cat also had a venous blood gas that included hematocrit (Supplemental Table S3). Of these cats, the most common abnormalities on complete blood count were anemia (13/16, 81%) or neutrophilia (12/15, 80%). Total bilirubin was increased in 8 of 15 (53%) cats. Increases in alkaline phosphatase, alanine transferase, and gamma-glutamyl transferase were uncommon, each being identified in 6 or fewer cats (<40% of cats with triaditis; Supplemental Table S3).
Incidence of Neoplasia in Examined Tissues and Association of EATL2 With Triaditis
Neoplasia was identified in the liver, pancreas, small intestine, or MDP of 37 of 124 cats (30%). Neoplasia was most common in the small intestine, followed by the hepatobiliary system, pancreas, and MDP (Table 3). The spread of neoplasia in one or more of the examined tissues was seen in 18 cats and spread to 3 of the examined tissues was seen in 7 cats. Only one cat had neoplasia (large cell lymphoma) in all 4 examined tissues.
Summary of neoplastic lesions identified in pancreas, small intestine, hepatobiliary system, and major duodenal papilla of 124 domestic cats.

Abbreviation: EATL2, enteropathy-associated T-cell lymphoma type 2.
Multiple types of neoplasia were found in some cats, so specific tumors do not always add up to total incidence of neoplasia (37/124 cats).
One carcinoid arose within the common bile duct, while one adenoma arose from pylorus/proximal duodenum; both of these were immediately adjacent to the major duodenal papilla.
Lymphoma was the most common category of neoplasia identified (28/37 cats, 78%, Supplemental Figure S8, Table 3), and among this group, EATL2 was the most common (n = 16/28, 57%). Among the 16 cats with EATL2, spread to at least one other examined tissue was identified in 6 of 16 cats (37.5%; Supplemental Figure S8), the pancreas being the most commonly affected organ (n = 4/16 cats, 25%). Involvement of 3 of the examined tissues was less common (n = 2, Fig. 7), and no cats had involvement of all 4 tissues.
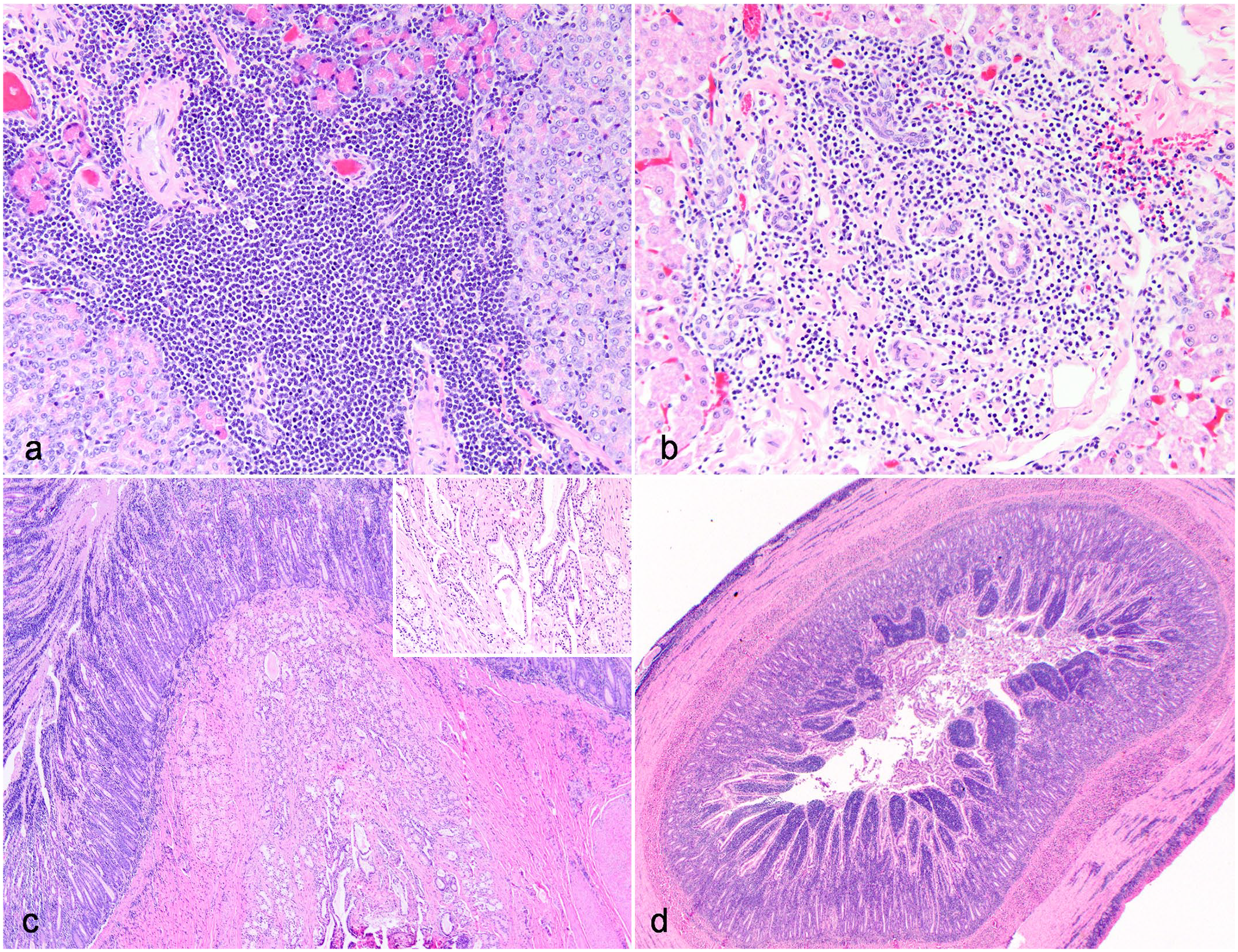
Enteropathy-associated T-cell lymphoma type 2 (EATL2) within the small intestine, pancreas, and liver of a domestic cat (Hematoxylin and eosin). Domestic cat, case 38. EATL2 was the most common neoplasm identified in cats. (a) Pancreas. Sheets of small neoplastic lymphocytes efface acini and the interstitium. (b) Liver. Sheets of small neoplastic lymphocytes expand the stroma surrounding the biliary tree. Lack of biliary infiltration and accompanying plasma cells supports a diagnosis of lymphoma over lymphocytic cholangitis. (c) Duodenum and major duodenal papilla (MDP). Sheets of small lymphocytes expand and efface the lamina propria of the duodenum, but the mucosa and lamina propria of the MDP is spared (inset). (d) Jejunum. Small neoplastic lymphocytes expand and efface villi and extend transmurally through the bowel wall with expansion of the serosa.
There was a significant association (P = .02) between the presence of triaditis and EATL2 (Supplemental Figure S9): 7 of 19 (37%) cats with triaditis had concurrent EATL2 (Supplemental Figure S7), while 9 of 105 (9%) cats without triaditis had concurrent EATL2. In addition, there was a significant association between MDPitis and EATL2 (P = .005): 8 of 35 (23%) cats with MDPitis had concurrent EATL2, while 8 of 89 (9%) cats without MDPitis had concurrent EATL2 (Supplemental Figure S9). There was no significant association between EATL2 and pancreatitis (n = 12/83, 14%), EATL2 and cholangitis (n = 9/41, 22%), or EATL2 and enteritis (n = 11/56, 20%).
In contrast to cats with EATL2, involvement of multiple examined tissues was more common in cats with large cell lymphoma (9/13, 69%). Involvement of 4 and 3 examined tissues was noted in 1 and 4 cats, respectively. Also, in contrast to cats with EATL2, only 1 cat had concurrent triaditis and large cell lymphoma in the examined tissues.
Discussion
The current findings represent the first large-scale prospective study investigating the role of the MDP in the pathogenesis of pancreatic, hepatobiliary, and enteric inflammation and neoplasia in cats. Feline triaditis has long been attributed to a merging of the common bile and pancreatic ducts prior to entry into the duodenum at the MDP, thereby providing a direct path of communication between the organs. This conjoined anatomy, similar to the ampulla of Vater in humans, 4 was previously only supported by limited data in cats.2,32 Our findings in the present study confirm that merger of the common bile and pancreatic duct proximal to entry into the duodenal wall is present in a majority, but not all cats. We found that the MDP is histologically elaborate and comprised of a tangle of ducts surrounded by small, isolated bundles of smooth muscle surrounding the periphery of the MDP. Consistent with previous morphometric studies of the MDP musculature in cats, 32 the common bile duct and pancreatic duct lacked anatomic evidence (ie, distinct, separate muscle bundles) for separate sphincteric control. It has been shown that bile flow into the duodenum in cats is reliant upon a pressure gradient and that bile is not released into the duodenum if duodenal pressures exceed that of the bile duct. 3 Collectively, the anatomy and physiology of this site may predispose cats to retrograde reflux of bile acids, pancreatic enzymes, intestinal bacteria, and intestinal contents in the setting of altered intestinal motility, inflammation, obstruction, and dysbiosis, ultimately leading to inflammation in all 3 tissues. This theory is supported by the association seen between conjoined anatomy of the common bile duct and pancreatic duct and triaditis in this study.
It is of additional interest that half of the examined cats had prominent lymphocyte aggregates present at or immediately adjacent to the MDP within the duodenum. These aggregates were confirmed to be tertiary lymphoid structures via immunohistochemistry. The presence of this tissue, at the very least, suggests that this is an anatomic site for which constant immune surveillance and antigen sampling is warranted. This also implies that these foci, which presumably have associated overlying M cells, may be entry points for opportunistic bacterial colonization and infection, which could contribute to development of diseases such as ascending bacterial cholangitis. Future studies would benefit from further immunohistologic characterization of lymphoid follicles and the overlying presumptive M cells, as well as further characterization of the bacterial flora (eg, culture, 16S sequencing, in situ hybridization) present at this location.
In addition to documenting an association between anatomical arrangement of the MDP and triaditis, we directly investigated the possibility that lesions at the MDP could predispose to triaditis in cats. Humans and non-human primates are predisposed to a variety of mechanical, inflammatory, and neoplastic conditions at the MDP that further predispose the development of biliary and pancreatic inflammation and neoplasia.18,25,31,33 Given their similar anatomy, we suspect that cats may also suffer from similar conditions with related sequelae, but reports in the literature are few and limited to antemortem observations. 13 In this study, over a quarter of necropsied cats had histologic evidence of MDPitis, and cats with MDPitis were 6.64 times more likely to have concurrent triaditis. Moderate to marked MDPitis was uncommon but frequently associated with additional pathology at the MDP, such as choledocholithiasis, MDP dilation, MDP enlargement, and erosion or ulceration. We suspect that in these more severe cases, significant injury to the MDP predisposed to extrahepatic biliary obstruction and subsequent cholangitis, as well as pancreatic duct obstruction and/or reflux of bile acids into the pancreas, contributing to pancreatitis. For example, in the current study, a single cat with triaditis (case 31) had microscopic evidence of pancreatolithiasis at the level of the MDP with concurrent suppurative MDPitis, cholangitis, and pancreatitis; this diagnosis was only reached with postmortem histologic evaluation of the MDP. While this lith may not have been detectable via antemortem imaging (ie, ultrasound), we suspect that the enlargement of the MDP would have been seen ultrasonographically. To this point, we have personal experience (JLG) treating cats with ultrasound-diagnosed transient MDP or common bile duct liths, sludge, or debris, and similar lesions have been documented in recent studies in cats. 5 We suspect that such lesions lead to pancreatitis due to reflux of bile acids and/or bacteria into the pancreas. This phenomenon is a suspected cause of recurrent pancreatitis in cats, 5 but remains unproven; however, the current case also suggests this pathogenesis. Ultimately, we suspect that these conditions may be underdiagnosed in cats for a variety of reasons, including a lack of specific antemortem clinical signs and clinicopathologic findings, insensitivity of imaging techniques to detect MDP lesions or ductal debris, intermittent passage of liths/debris, and rarity of MDP evaluations on postmortem examinations. Therefore, we recommend that the MDP should be collected routinely on postmortem examinations in cats for which hepatobiliary, pancreatic, or intestinal disease is suspected, and recommend applying the methods used for histologic evaluation detailed in this study (ie, trimming of serial sections, application of definitions of inflammation).
Neoplasia was identified in at least one of the 4 examined tissues in over a quarter of cats in this study. Among that population, just under half of the examined cats had neoplastic involvement of at least 2 examined tissues. The spread of neoplasia between these organs may be facilitated by a variety of mechanisms, including direct spread due to the close proximity of the organs or spread via lymphatics. Given the prominent lymphoid tissue present at the MDP in the majority of cats, it is possible that this tissue may serve as a unique “portal” to facilitate lymphoid invasion and regional and/or disseminated metastasis.
Lymphoma was the most common neoplasm identified in this cohort, and among cats with lymphoma, EATL2 was the most common. There was a significant association between the incidence of triaditis and EATL2 as well as the incidence of MDPitis and EATL2. It is conceptually established that chronic inflammation predisposes cats to the development of neoplasia, including injection site sarcoma and trauma-induced ocular sarcoma.29,38 In humans, chronic inflammatory bowel disorders (eg, celiac disease) have been associated with the development of small T-cell enteropathy lymphoma,10,14,15,22,27,39 and a similar process has been postulated for cats with idiopathic chronic enteritis.1,6,11,14,15,17 Furthermore, changes in enteric bacterial populations have been documented in cats with chronic lymphoplasmacytic enteritis, EATL2, and large cell lymphoma,14 –16,21 suggesting that alterations in gut microbiome (ie, dysbiosis) may play a role in the pathogenesis of the progression from idiopathic chronic enteritis to EATL2. Given the suspected relationship between enteritis and EATL2 in cats, as well as the significant association seen between triaditis, MDPitis, and EATL2 in this study, further studies investigating relationships between these inflammatory lesions and development of EATL2 are warranted. While additional work is needed to investigate the relationships between these conditions, these findings have significant clinical implications; namely, cats with triaditis or MDPitis may be at risk of EATL2 and should be monitored accordingly.
A few observations were made in this study tangential to its primary aims. First, it should be noted that the proportion of cats with pancreatitis (67%), cholangitis (33%), and enteritis (45%), as well as the types and severity of inflammation present, are similar to those reported in previous studies.6,7,9,12,36 These data suggest that these disease entities are relatively common in cats, and based on the limited antemortem data from cats with triaditis in this study, may be present at a sub-clinical level. Second, the clinical presentation of cats with triaditis was highly variable; less than half had clinical signs or history attributable to triaditis, and no consistent clinicopathologic changes were identified in these cats as a group. Notably, interpretation of the significance of the few clinicopathologic changes present (eg, hyperbilirubinemia in just over half of cats with triaditis) is limited by the lack of evaluation of these parameters in cats without triaditis. Finally, cats with triaditis were significantly older than those without triaditis. While the reason for this cannot be definitively defined from this study, this may be due to the tendency for triaditis to be a disease of chronicity; this is supported by the over-representation of chronic lymphoplasmacytic inflammation seen in the majority of organs in triaditis cases in this study. Other factors more common in aged patients, such as immunosenescence and the impact of systemic comorbidities, may also contribute.
This study has several limitations. First and most importantly, while descriptive histologic evaluation of MDP greatly aids in our understanding of the anatomy of the feline MDP, this type of evaluation is limited to few static postmortem “snapshots” of the area and is unable to assess dynamic movements in order to understand sphincter contraction and interactions with duodenal musculature. To this point, given that only 2 to 4 sections were evaluated for each cat, it is possible that more overt “separation” of the ducts at the MDP, or more significant intercalating sphincter musculature, was missed in section. For example, while we identified 2 discrete ducts at the duodenum entry point in only a subset of cases (<20% of cats), we suspect that this low percentage was due to limitations of tissue orientation and sectioning, and that more cats may have this visualized in sections that captured the ducts more proximally in the duodenum. Notably, previous detailed studies of this site2,32 identified extension of duodenal musculature between ducts prior to merging at the ampulla, and similar septal musculature is present in humans. 4 Future studies detailing anatomy of the feline MDP would benefit from advanced histologic imaging techniques on postmortem specimens and advanced imaging (ie, computed tomography) on live patients that would allow for 3D visualization of the MDP. 28 Second, a lack of consistent clinical history, clinicopathologic data, or antemortem diagnostic imaging precluded correlation between histologic findings and clinical features of the disease. Future studies would benefit from prospectively identifying triaditis suspects antemortem, monitoring them during the course of clinical disease, and performing postmortem histologic evaluation of pertinent tissues, including the MDP, if applicable. Third, for ease of analysis, we grouped different types of inflammation together when assessing correlations between inflammation in different tissues. However, this is likely an oversimplification, as the pathogenesis behind different types of inflammation in tissues (eg, neutrophilic vs lymphocytic cholangitis) is likely different. 34 This approach may have resulted in overlooking more subtle correlations between different inflammatory patterns in tissues. Finally, beyond EATL2 and the one case of intravascular lymphoma, large cell lymphomas were not further subtyped, limiting further assessment and conclusions regarding more specific types of large cell lymphomas and associations with triaditis.
In conclusion, in this study, we performed the first large-scale histomorphologic characterization of the feline MDP to date and have identified unique features about this site that may contribute to triaditis pathogenesis. In turn, we have identified several features of feline triaditis that were previously unknown, including associations with MDPitis, conjoined anatomy, and EATL2. While the pathogenesis of these relationships is currently unclear, these data underscore the importance of thorough clinical workup in cats suspected to have triaditis, including evaluation of the MDP.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858231189450 – Supplemental material for Histologic characterization of the major duodenal papilla and association with concurrent biliary, pancreatic, and intestinal pathology in cats
Supplemental material, sj-pdf-1-vet-10.1177_03009858231189450 for Histologic characterization of the major duodenal papilla and association with concurrent biliary, pancreatic, and intestinal pathology in cats by Megan E. Schreeg, John M. Cullen, James Robertson and Jody L. Gookin in Veterinary Pathology
Footnotes
Acknowledgements
We acknowledge the expertise and contribution of the staff of Ms Laura Miller and the North Carolina State University College of Veterinary Medicine Histology Laboratory. We thank the numerous North Carolina State University College of Veterinary Medicine pathology faculty, residents, staff, and students that aided in tissue collection; without this team effort this study would not have been possible.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
