Abstract
Stony corals (Scleractinia) are in the Phylum Cnidaria (cnidae referring to various types of stinging cells). They may be solitary or colonial, but all secrete an external, supporting aragonite skeleton. Large, colonial members of this phylum are responsible for the accretion of coral reefs in tropical and subtropical waters that form the foundations of the most biodiverse marine ecosystems. Coral reefs worldwide, but particularly in the Caribbean, are experiencing unprecedented levels of disease, resulting in reef degradation. Most coral diseases remain poorly described and lack clear case definitions, while the etiologies and pathogenesis are even more elusive. This introductory guide is focused on reef-building corals and describes basic gross and microscopic lesions in these corals in order to serve as an invitation to other veterinary pathologists to play a critical role in defining and advancing the field of coral pathology.
The reef-building members of Scleractinia, commonly known as stony corals, are keystone species that create the structural and ecological foundation supporting the high biological diversity found in tropical marine ecosystems. 69 Corals offer a unique paradigm in that the animal provides the foundation of the reef ecosystem building the 3-dimensional infrastructure that allows a multiplicity of habitats providing nurseries, foraging grounds, and shelter for a diversity of invertebrates and vertebrates. In addition, they are themselves a mesocosm composed of a population of modular, usually clonal coral polyps, algal symbionts, and complex microbiomes.
Scleractinian corals are one of the most rapidly declining taxonomic groups of animals on the planet, with 50 species (out of almost 800 currently identified) listed as endangered or critically endangered and an additional 200 classified as vulnerable by the IUCN Red List, 59 24 species are listed as threatened or endangered by the US Endangered Species Act,154,155 and all are protected by Appendix II of the Convention on International Trade in Endangered Species of Wild Fauna and Flora. 30 Significant stressors including climate change,54,63 ocean acidification, 56 coastal development, and land-based pollution159,168 have contributed to the degradation of coral reef habitat and contribute to disease susceptibility.60,94 The rate of current population loss and the inability of existing reefs to overcome repeated disease outbreaks in the face of anthropogenic stressors are of concern. 55 The cause of coral population decline is poorly understood. Climate change and land-based pollution have gained the most attention, with less attention directed toward investigating the tissue and cellular changes of diseases that lead to coral death and are the proximal cause of significant losses. 131 Understanding how physical, chemical, and infectious agents affect corals at the tissue and cellular levels is needed, not only to understand disease processes, but also to potentially identify points of intervention to halt immediate threats to coral. For example, in the late 1980s through the mid-1990s, the western Atlantic lost >80% of Acropora palmata and Acropora cervicornis, branching corals that comprised the 3-dimensional structure of Caribbean reefs, leaving massive and encrusting corals as the dominant type. 10 This loss of rugosity and microhabitats led to an overall reduction in biodiversity within Caribbean reef ecosystems.31,96 Studies suggest that the disease associated with mortality of A. palmata and A. cervicornis in the 1980s and 1990s, white band disease, is caused by a transmissible agent, but the cause has not been identified and there are few reports of microscopic changes46,68,111 associated with the progression of this disease. More recently, Caribbean reefs have undergone marked coral losses due to a newer disease, stony coral tissue loss disease (SCTLD), which is identified by the appearance of (usually) multifocal, rapid tissue loss affecting a consistent pattern of multiple (>24) species, but sparing acroporids.35,115,139,140 Tissue loss disease consistent with this description is now spreading even to more pristine reefs in the wider Caribbean,8,35,41 and has led to local extinction of some coral species. 94 This disease is currently defined by gross and microscopic morphology as well as the assemblage of affected species, with no etiology identified.72,139 It is not known whether this is a more virulent form of earlier tissue loss diseases or white syndromes (eg, white plague) affecting these species or is truly a separate entity(ies). 33
While numerous coral diseases have been documented, the cause of most coral diseases remains unknown. 113 This is, in part, because like many diseases, those in coral are likely multifactorial and complex, and also because many have similar or overlapping gross appearances. 33 The tendencies to diagnose diseases using only field characteristics or to focus on molecular assays of samples taken from an animal immersed in a sea of microorganisms, without direct correlations to lesions, have been common confounding themes of coral disease investigations. 182 This is even more difficult when the described lesions are nonspecific. Confusion in the literature exists due to names and presumed etiologies being assigned absent morphologic evidence to support many conclusions. In this context, the skills of veterinary pathologists and coral histologists have the potential to provide valuable insight to coral disease investigations by characterizing the cellular and tissue-level responses to stress or injury. In vertebrates, these morphological changes are used to deduce insight into pathogenesis and potential etiologies. Systematically applying this process to corals has the potential to create a framework that differentiates unique diseases with similar presentations. In recent years, transdisciplinary efforts have led to considerable improvements in classifying gross lesions and undertaking histology on diseased corals, such as investigation of the role of ciliates in tissue loss,149,171 understanding of growth anomalies, 174 and the discovery of a chimeric parasite with molecular features that indicate fusion of 2 coral species involved in Montipora tissue loss. 179 Such efforts illustrate that the disease specialist has much to offer this field.36,37,72,126
Similar to other invertebrates, histopathological interpretation can be challenging due to limited tissue responses, the need to distinguish between symbiotic and pathogenic microbes, and the lack of well-defined normal histology for many species. The aim of this review is to systematically describe gross and microscopic lesions encountered in corals to facilitate their recognition and standardized nomenclature while emphasizing etiological associations and knowledge gaps (Table 1). We see this as the beginning of a conversation in which pathologists may contribute to defining both normal and abnormal in this colonial animal that depends on an intimate relationship with microorganisms to survive. We also summarize fundamental aspects of physiology and anatomy, sampling, and ancillary diagnostics relevant to coral diagnosticians.
Common microscopic changes seen in corals and correlates.
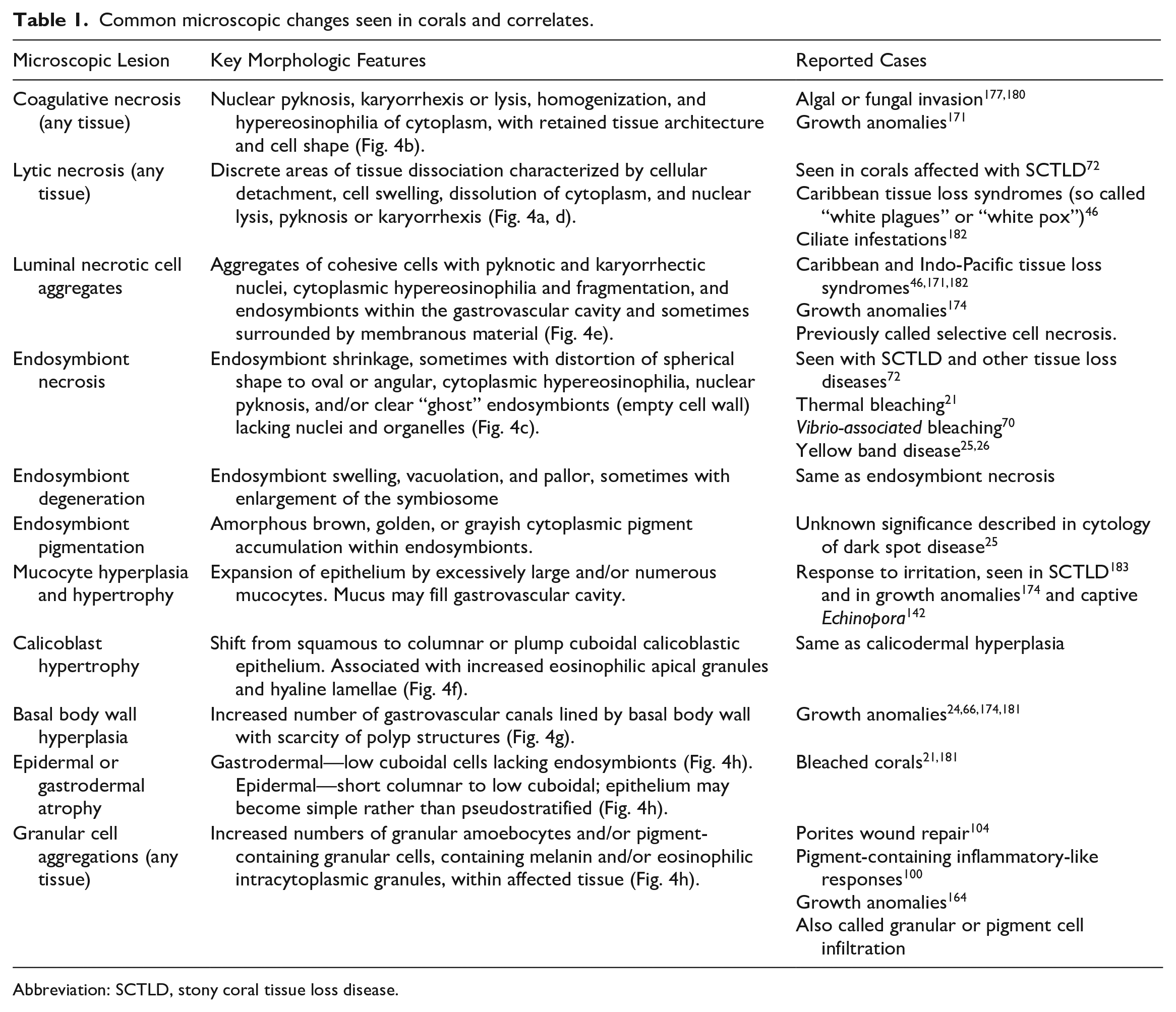
Abbreviation: SCTLD, stony coral tissue loss disease.
Coral Anatomy
Colonial stony corals are composed of many interconnected polyps with tentacles in multiples of 6 that surround the oral disk, the center of which contains the mouth that transitions to the gastrovascular cavity (Fig. 1-20) through the actinopharynx (Fig. 1-17). Often the tips of the tentacles contain a concentrated number of cnidocytes (Fig. 1-9) (stinging cells) containing a complex stinging organelle unique to this phylum, the cnidocyst or nematocyst, 13 which together form a bulbous structure known as the acrosphere (Fig. 1-16). Along the length of the tentacles are smaller structures composed of cnidocytes called batteries. Cnidocytes can also be found in the cnidoglandular band of the mesenterial filament (Fig. 1-8, 9) and distributed in the epidermis or gastrodermis, depending on coral species. Other species may lack tentacles and deploy cnidocytes on filaments for digestion. 48 The 3 main cnidocyte types seen in corals are the microbasic mastigophore (Fig. 1-9a) that has a hollow tube through which toxin is released into prey when the cnidocyst discharges; the holotrichous isorhiza (Fig. 1-9b) that has a coiled hollow tube covered with short spines along its length and can inject toxin; and the spirocyst (Fig. 1-9c) that has a coiled solid sticky thread to attach to prey. Food and waste transit through the mouth. The polyp sits in a cup in the aragonite exoskeleton called a corallite (Fig. 1-19). Thin calcareous divisions or septae radiate from the walls and floor of this cup toward the center and provide support for the polyp.
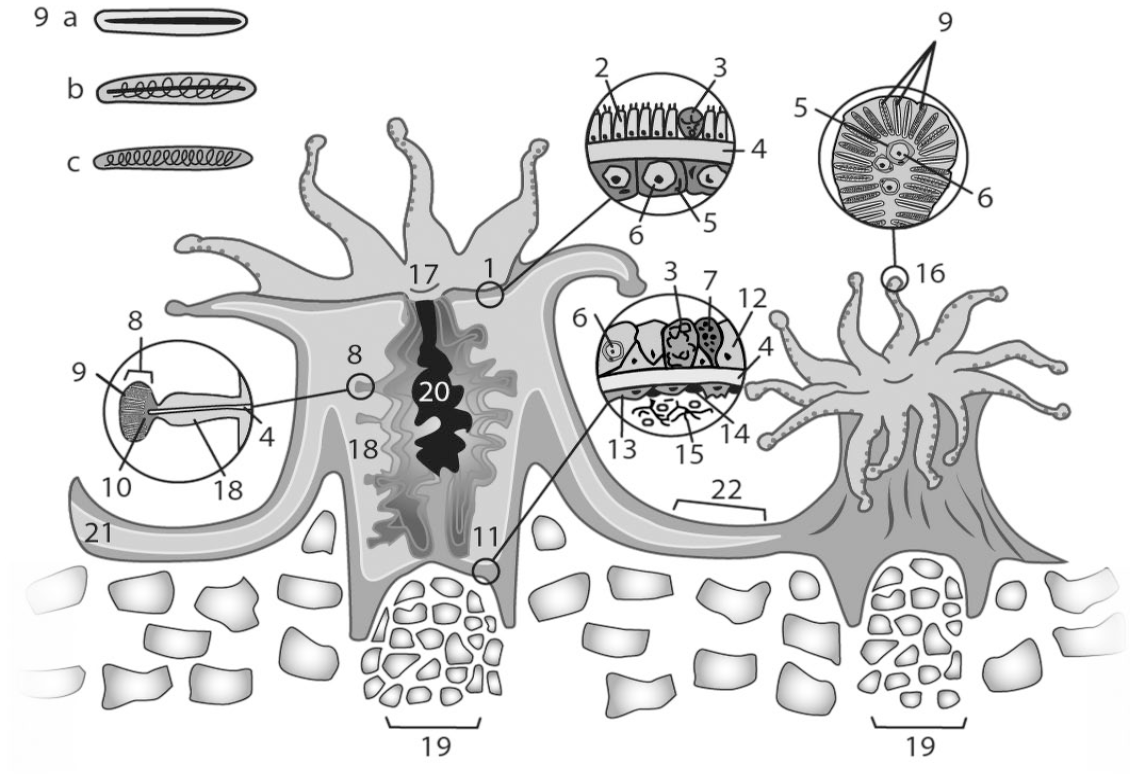
Polyp diagram showing generalized, major anatomic and microscopic features. 1. Surface body wall. 2. Ciliated columnar epithelium. 3. Mucocyte. 4. Mesoglea. 5. Gastrodermal cell. 6. Endosymbiont. 7. Granular cell (often pigment-containing granular cells within gastrodermal and epidermal layers). 8. Cnidoglandular band (along margin of mesenteries). 9. Cnidocyte; 9a. Microbasic mastigophore. 9b. Holotrichous isorhiza. 9c. Spirocyst. 10. Granular gland cell–rich area 11. Basal body wall. 12. Basal gastrodermal cell. 13. Calicoblast. 14. Desmocyte. 15. Endolithic filamentous structures (eg, fungi, algae). 16. Acrosphere. 17. Actinopharynx. 18. Mesenteries. 19. Corallite. 20. Gastrovascular cavity. 21. Gastrovascular canal. 22. Coenenchyme. Source: Kevin Seline, artist.
Coral polyps are diploblastic, possessing 2 cell layers: the epidermis (outer epithelium of adult organisms) or ectoderm (in embryos) facing the environment (open water or skeleton) and the gastrodermis (inner epithelium in the adult) or endoderm (in the embryo) facing the gastrovascular space. 109 The space between these layers is filled by a mostly acellular connective tissue layer, the mesoglea (Fig. 1-4), which provides some additional structural support, and together these comprise the surface body wall (Fig. 1-1). Round to oval cells with a clear cytoplasm (agranular amoebocytes) can occasionally be seen within the mesoglea. Smooth muscle bands called myonemes, which are extensions of epitheliomuscular cells within the epidermis, interdigitate into mesoglea, in effect anchoring the epitheliomuscular cells to allow for contraction of the polyp or other movements. 16
The tissue connecting polyps is known as coenenchyme (Fig. 1-22) and is composed of 2 sets of tissue layers, each consisting of epithelium, mesoglea, and gastrodermis, that lines the gastrovascular canal (Fig. 1-21). Tissue partitions, the mesenteries (Fig. 1-18), extend from the mesoglea of the body wall into the gastrovascular cavity and are lined on either side by gastrodermis. The mesenteries increase gastrodermal surface area and can provide additional support. Mesenteries may also contain ova and/or spermaries in reproductive polyps. The free margins of the mesenteries (facing the gastrovascular space) are modified into thickened structures referred to as mesenterial filaments (Fig. 1-8). The margins form cnidoglandular bands (Fig. 1-8), covered by a layer of thin columnar ciliated epithelial cells, granular gland cells (Fig. 1-10), mucocytes, and cnidocytes, which are used to attack zooplankton captured by the tentacles and brought to the gastrovascular cavity for consumption. 109 Immediately adjacent to the margins, at the base of cnidoglandular bands and attached to the mesoglea, are nutrient-absorbing gastrodermal cells that form paired lobes in some species. In many species of corals, these filaments are actively extruded through the mouth of the polyp to attack adjoining corals in the never-ending competition for space in the marine environment. 29
The thickness of the living coral tissue can range from millimeters to a few centimeters, depending on species of coral. The surface epidermis is in contact with seawater and consists of simple or pseudostratified columnar epithelial cells, which are often ciliated to help move mucus or sediment. The epidermis may also contain mucocytes, cnidocytes, neurons, sensory cells, pigment-containing granular cells, granular amoebocytes, and epitheliomuscular cells.
Cells containing cytoplasmic granules may be found in every coral tissue. Presence and numbers of granular cells within coral tissues vary with species of coral, and while there is observable variation in granular cell morphology within and between coral species, most have not been characterized by function. It may not be possible to differentiate all of these with routine histopathology, and many are yet to be clearly identified by type. Terminology in the literature can be confusing, and granular cells have been called granular amoebocytes, granular pigment cells, melanin-containing granular cells, or eosinophilic amoebocytes depending on appearance.103,104,143 Some broad distinctions can be made between granular gland cells (discussed further with the gastrodermis), granular amoebocytes (discussed further under coral immunity), and pigment-containing granular cells. Pigment-containing granular cells are most common in the surface body wall epidermis and gastrodermis. They are characterized by fine cytoplasmic granules that may contain melanin and/or fluorescent pigments, and stain variably with hematoxylin and eosin (HE). Fluorescent pigments in corals have been shown to be photoprotective, and these can be found both in granules within cells and free within intracellular spaces; 137 however, the amount of fluorescent proteins/pigments may diminish with temperature stress. 133
The basal body wall (Fig. 1-11) is lined internally by gastrodermal epithelial cells (Fig. 1-12) interspersed with granular cells (Fig. 1-7) and mucocytes (Fig. 1-3). The calicodermis (outer epithelium of the basal body wall) is composed of calicoblasts (Fig. 1-11). These are modified epithelial cells that form the coral skeleton through a complex interaction between organic matrix, composed of collagens, laminins, and fibronectin, and calcium-binding proteins such as coral acid–rich proteins (CARPs). 15 Seven CARPs have been characterized; these are high in aspartic and glutamic acids, which interact with carbonic anhydrases and cadherin-like calcium-binding proteins (eg, von Willebrand factor proteins), to precipitate aragonite into the skeleton. 92 This exoskeleton accreting over time forms the massive support structures that we recognize as the reef. Desmocytes (Fig. 1-14) are differentiated epithelial cells distributed among calicoblasts that provide focal points of attachment to the aragonite skeleton. The entire exoskeleton is known as the corallum and develops into different growth forms. 109 Endolithic filamentous organisms (Fig. 1-15) are often seen in the superficial layers in small crevasses and spaces created by the skeletal deposition process and may also be found in deeper layers.
The gastrodermis lines the gastrovascular cavity and canals and is composed of columnar to cuboidal gastrodermal cells interspersed with variable numbers of cnidocytes, granular amoebocytes, pigment-containing granular cells, granular gland cells, and mucocytes.
Granular gland cells are found in groups within cnidoglandular bands, within the actinopharynx, and individually within the gastrodermis.109,125,162,178 These are a secretory epithelial cell with granules or vesicles that are often larger than the ones found in the granular amoebocytes that are described aggregated at sites of injury. In the stony coral Stylophora they have been shown to contain enzymes (eg, chymotrypsin, lysosomes) speculated to be involved in assimilation of food.123,167,178 Granules are variably sized and stain bright red to pink with HE. 123
Gastrodermal epithelial cells (sometimes termed gastrodermal supporting cells or symbiotic endoderm cells) often have a cytoplasmic vacuole, the symbiosome, that contains symbiotic, single-celled dinoflagellates of the family Symbiodiniaceae, known as endosymbionts (formerly zooxanthellae). Other intracellular organisms may be found within coral cells, but the term endosymbiont in corals refers to Symbiodiniaceae. These are most dense within the gastrodermis of the surface body wall but may also be found in other gastrodermal regions including mesenterial filaments and basal body wall gastrodermis. The taxonomy of Symbiodiniaceae has been recently reorganized, and genera found in corals include Symbiodinium (formerly known as clade A), Breviolum (formerly clade B of Symbiodinium), Cladocopium (clade C), Durusdinium (clade D), Effrenium (clade E), Fugacium (clade F), and Gerakladium (clade G). 71 One colony may host multiple species, but there are typical associations between given coral and endosymbiont species. Endosymbiont genus, species, and subclade can affect the coral’s resistance to thermal stress, and may vary with environmental conditions because the endosymbionts are variably resistant to coral stress. 118 Coral cells act as hosts to endosymbionts, providing nutrients in the form of nitrogenous compounds, while the endosymbiont provides sugars and lipids to the coral in the form of photosynthates. Endosymbionts provide much of the color to the polyp and also mediate deposition of the aragonite skeleton through incompletely characterized biochemical processes.78,91 Endosymbionts also play a critical role in coral health, and loss of these organisms often leads to a phenomenon called bleaching, leaving translucent tissues over a white skeleton. This occurs most commonly due to elevated water temperatures,7,21,39,57,73,75,163 but may also occur due to other stressors, including oxidative damage from UV radiation, cold water, ocean acidification, toxic insults (eg, pesticides or cyanide), and bacterial infection. 112 The mechanism of endosymbiont loss is not completely understood, but involves destruction of the endosymbionts in at least some cases,22,73 and the process of recolonization is not known. If tissues are not recolonized by endosymbionts, this can lead to death of the coral colony.
Coral tissues adopt 2 basic skeletal architectural plans: perforate and imperforate. In imperforate corals, coenenchyme (common tissue) connecting polyps contains gastrovascular canals that connect to adjacent polyps’ gastrovascular cavities only along the surface of the skeleton (as in Figs. 1, 2a). In perforate corals, numerous canals lined by coral tissue connect to adjacent polyps’ gastrovascular cavities at multiple levels and course throughout the exoskeleton (Fig. 2b). Histologically, this results in a more complex appearance for perforate corals with numerous cross, longitudinal, and oblique sections of small channels embedded within the skeleton and interconnecting the surrounding polyps.
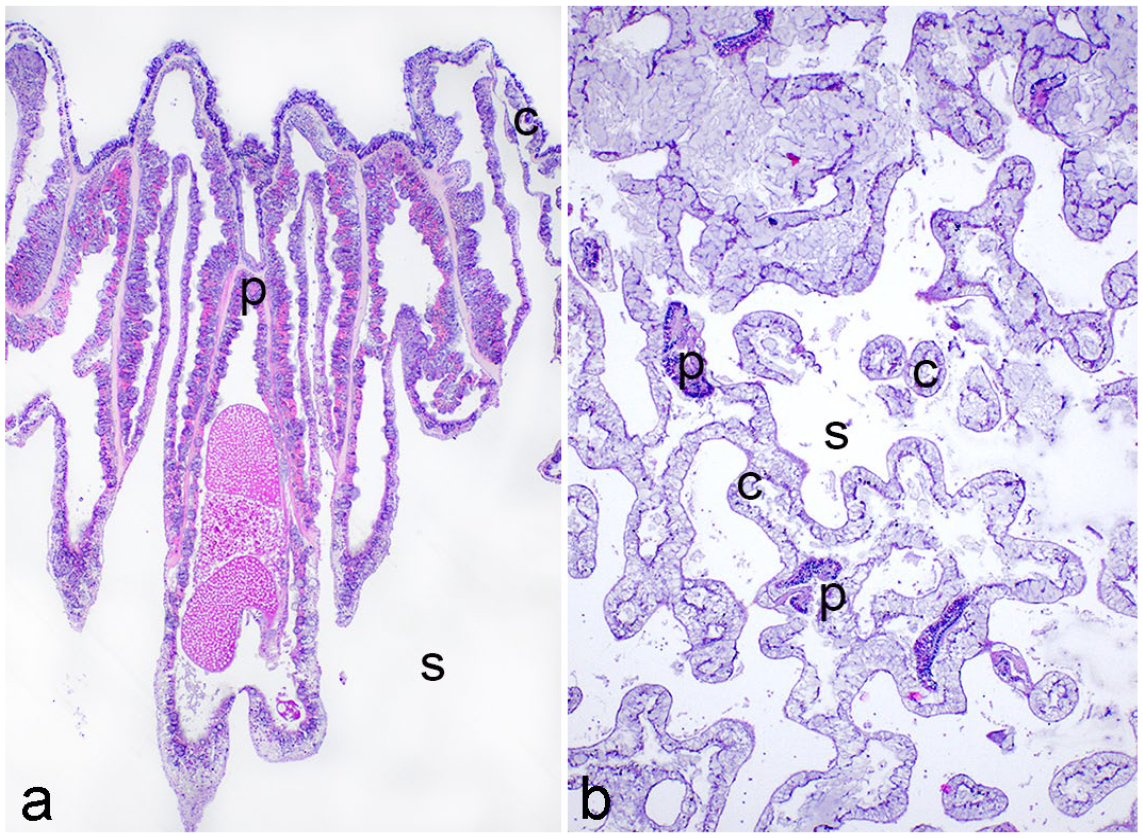
Imperforate and perforate coral growth forms. Hematoxylin and eosin. a. Orbicella annularis. Imperforate coral with coenenchyme (c) connecting polyps (p) and gastrovascular canals restricted to skeletal (s) surface. b. Porites astreoides. Perforate coral with coenenchyme network composed of gastrodermis-lined gastrovascular canals (c) running through skeleton (s) and connecting polyps (p) at multiple levels along the gastrovascular cavities.
Rather than a single animal, corals are best described as a meta-organism: an array of symbiotic organisms including the cnidarian animal, the dinoflagellate algae (family Symbiodiniaceae), bacteria, archaea, fungi, protists, and viruses with a biodiversity that is equal to or greater than any other known system. 17 Holobiont is the term used to describe this collective group of organisms, while microbiome describes the symbiotic microbiota and their surrounding environment including interspecific and host interactions, viruses, and abiotic environmental conditions (eg, temperature).14,151 Alterations in the microbiome are implicated in disease susceptibility and the term “pathobiont” has been used to describe disease resulting from disruption of the microbiome.144,160
Immunity in Corals
Corals lack an acquired immune system but possess many of the same intracellular inflammatory signaling pathways and receptors seen in innate immune responses of other animals, including lipoxygenases, cyclooxygenases, 82 nuclear factor kappa B (NF-κB), pattern recognition receptors (PRRs), integrins, Toll-like receptors (TLR), C-type lectins, and a complement-like system C3.104,153 They also possess a group of cells termed amoebocytes, which are pleomorphic cells that are thought to function as the main cells of an innate immune system. 104 These are not well characterized histologically and studies to demonstrate directed movement are lacking, but amoebocytes in the gorgonian Swiftia have phagocytotic activity, 97 and it seems likely that hard corals possess phagocytic and mobile amoebocytes as well. The healing process varies by species. Healing documented in Porites cylindrica consists of 4 stages (plug formation, immune cell infiltration, proliferation, and maturation) that are common to many other animals, while in Montipora capitata, plug formation and inflammatory cell infiltration were not observed.103,104
Current thinking places coral amoebocytes in 2 broad categories: granular amoebocytes (or pigment-containing granular amoebocytes) and agranular amoebocytes, both of which have been observed in locally increased numbers at sites of injury in at least some species, and are presumed to have migrated, as this change occurs rapidly after injury and mitotic figures were not noted. 104 Granular amoebocytes may contain dark pigment on HE that stains positive with Fontana-Masson and may contain fluorescent proteins. 10
Many coral species have the appearance of multiple morphologically distinct granular cells but whether all of these are different cell types with distinct roles is not known, and it is not always apparent whether groups of granular cells within tissue are a response to injury or a normal finding. Comparison with healthy examples of the same species is invaluable in distinguishing normal from abnormal.
Transcriptomics has confirmed the existence of 2 main types of immune cells in the stony coral Stylophora pistillata. One of the cell types expresses interleukin-1 receptor orthologs and secretes proteins including perforins, prosaposins, and lipopolysaccharide-binding proteins, and the other expresses antimicrobial apextrin C-terminal proteins 80 and tyrosinase. Both express nuclear factor of activated T cells and interferon regulatory factor orthologs that are similar to those involved in the vertebrate innate immune response, 78 but these phenotypes have not been linked to any histologically identified cell types, so it is not known how this corresponds to observable cellular morphologies.
The skeleton itself forms a physical barrier and houses an endolithic community composed of coral-associated fungi, microalgae, and other microbes that are part of the holobiont; the role of these in coral immunity is largely unexplored, but they may help the coral colony recover from bleaching. 107 Calicoblasts also express antimicrobial compounds, as well as genes with possible dual-immune and biomineralization functions, such as cathepsin L and peroxidasin. 79
While coral do not have the adaptive immunity or inflammatory cell types seen in vertebrates, the holobiont itself may adapt in ways that protect the coral colony from disease. For example, after an outbreak of Vibrio-associated bleaching in Oculina patagonica, surviving individuals showed resistance to further Vibrio-associated bleaching. Disease could be produced in resistant colonies by inoculation with Vibrio only if the colony was pretreated with antibiotics. In addition, Vibrio growth could be inhibited by compounds produced by 2 bacterial strains identified in resistant coral fragments prior to antibiotic treatment, suggesting a loss of the protective microbial community that conferred immunity to previously pathogenic bacteria. 90 Disruption of the microbiome may predispose to, or even cause, disease18,83,160 as it provides many functions that support the coral colony, including exchange of metabolites and nutrient production, nitrogen fixing, elaboration of antimicrobial peptides, and competition with potential pathogens. 157 Combining histopathology with paired microbiological analyses of microbiome composition and diversity may help in clarifying the role of dysbiosis in coral diseases.
Gross Lesions
Corals have a tissue response repertoire composed of a restricted set of gross changes limited to 3 broad categories: tissue loss, discoloration, or growth anomaly.44,172
Tissue Loss
Tissue loss is a commonly seen gross lesion on corals and can be explained or unexplained. 130 Explained tissue loss are those lesions where a clear cause can be determined grossly; examples include mechanical injury caused by boat anchor scrapes, corallivorous fish, 134 snails, 122 fireworms (Fig. 3c), 89 or sea stars. 114 Some corallivorous fish can leave characteristic patterns of tissue loss in corals; for instance, parrot fish remove both coral tissues and underlying skeleton revealing characteristic scrape marks. 1 Blennies suck tissues off in a circular pattern leaving a pock-marked appearance. 134 Butterflyfish will suck out individual polyps leaving empty calices. 58 Further examples are illustrated by Bruckner and Bruckner (2016). 23 Ultimately, the distinction between tissue loss due to predation versus other causes relies on in situ observations of predators or their characteristic patterns, because predation cannot be definitively diagnosed via histopathology. Recognizing predation requires keen field observation skills as predators may be cryptic, well-camouflaged, or inactive during daytime. Unexplained tissue loss includes all other tissue loss lesions for which no clear cause is evident.

Gross lesions in coral. a. Meandrina meandrites. Acute tissue loss showing white, exposed skeleton (asterisk) and adjacent brown, intact tissue. b. Meandrina meandrites. Subacute tissue loss with a band of exposed white skeleton where recent tissue loss has occurred (arrows), and greenish turf algae overgrowth on area of older tissue loss (asterisk). c. Fireworm (Hermodice carunculata) predation on Diploria labyrinthiformis showing a fireworm (arrow) leaving a trail of bare skeleton (asterisk). Note similarity of lesion to nonpredatory tissue loss disease. d. Montastraea cavernosa. Pigmented band disease showing an advancing band of dark pigment due to a microbial mat at tissue loss margin (arrows) leaving behind bare skeleton (asterisk). e. Acropora humilis with a dark speckled band representing ciliate aggregations (inset) at the tissue loss margin (circled). f. Bleached Orbicella annularis colony with white skeleton showing through translucent tissue (asterisk). Polyp tissue is present including oral disk and tentacles (inset). g. Lavender discoloration of bleached Siderastrea siderea (asterisk) adjacent to normally colored reddish-brown tissue. h. O. annularis with yellow discoloration (arrows) along the border of tissue loss areas (asterisks). i. Exophytic growth anomaly (arrow) on Colpophyllia natans with enlarged, distorted rows of tissue.
Chronicity of tissue loss can be further characterized as acute, in which bare white coral skeleton is revealed (Fig. 3a), or subacute in which intact tissues are separated from algae-covered skeleton by a band of bare intact skeleton suggestive of a more slowly progressing process (Fig. 3b). 172 This temporal pattern can be inferred based on the fact that bare coral skeleton, like other bare benthic substrata, are rapidly colonized (within a few days) by fast-growing turf algae. 67 In some cases, tissue loss lesions can be bordered by a band of discoloration; examples include polymicrobial mats (eg, cyanobacteria) 135 (Fig. 3d), ciliate infections 28 (Fig. 3e), and other protozoal or metazoan invaders that may be primary or secondary contributors to tissue loss.
Discoloration: Bleaching and Pigmentation
Lesions of discoloration are characterized by intact tissue with abnormal tissue color and can be subcategorized into bleaching or pigmentation. The color changes with bleaching are due to loss of intracellular symbionts that often impart the full, normal color of coral tissues.
Bleaching
Bleaching is apparent by tissue pallor progressing to complete translucency, making the underlying white skeleton increasingly visible through translucent, intact coral tissue (Fig. 3f). Some bleached corals may simply become pale or discolored. An example is Siderastrea siderea that turns pale blue or lavender (Fig. 3g), presumably due to bacterial or native intracellular pigments within tissues that are uncovered subsequent to loss of endosymbionts.137,138 Bleaching can be diffuse, focal, or multifocal. In cases where lesions are white, bleaching can be distinguished from tissue loss by careful examination to ensure presence of intact polyps (Fig. 3f). Focal bleaching also tends to have faded and poorly circumscribed borders relative to the sharp borders of acute tissue loss. Mechanisms of bleaching are discussed with the associated microscopic lesions.
Pigmentation
Lesions of pigmentation differ from bleaching due to the gain of color not normally observed in the species under examination. Coral coloration is typically more strongly influenced by the brownish photosynthetic pigments of its endosymbionts rather than the pigments of the coral cells; however, coral tissues do contain fluorescent proteins that can impart their own colors. 38 Yet, apart from loss of Symbiodiniaceae causing marked or total coral color loss (bleaching), there are few described instances where endosymbiont abnormalities result in pigmented lesions in corals. For example, in Stephanocoenia intersepta and Siderastrea siderea with dark spot disease (also referred to as dark spot syndrome), grossly observed areas of dark brown–pigmented coral tissues have endosymbionts with dark cytoplasmic pigment seen cytologically. 25 These lesions seldom recover and may lose tissue slowly. The dark brown pigment has not been characterized, and the cause is unknown but is speculated to be a stress response. One study found an organism closely related to Rhytisma, a fungal genus associated with tar spot on maple tree leaves, within dark spot–affected areas of S. intersepta using polymerase chain reaction (PCR). This was speculated to be causing color change in endosymbiont chloroplasts via a similar mechanism as the terrestrial plant pathogen, 145 but histology was not performed so it is unknown whether the endosymbionts in these samples were showing the pigmented changes previously described.
Some coral species with dark-colored pigment lesions, similarly and confusingly given a field diagnosis of dark spot disease, have endolithic hypermycosis which is discussed further below, whereas others are uninvestigated and may represent entirely different processes.
Endogenous pigments of corals vary widely by species, many having roles in photoprotection or improving light availability for photosynthesis. Increased cellular expression or deposition of melanin, 100 carotenoids, 76 and fluorescent pigments101,137 have all been documented in pigmented lesions of corals. In some cases, bleached corals display vibrant color. This can be induced by brief temperature or nutrient stress that causes loss of pigmented endosymbionts, revealing green fluorescent protein-like pigments produced by the coral cells. 19 Extracellular organisms that associate with or infiltrate coral tissues, including fungi, cyanobacteria, helminths, and ciliate protists, or that colonize their skeleton, such as overgrowth of endolithic fungi, algae, and cyanobacteria, may contain their own variety of pigments20,128,177 or induce pigment production by coral cells.101,119,145 Together, these pigment sources provide a wide range of possibilities for the color observed in pigmented lesions in corals. For example, pink pigmentation can be observed in Porites compressa with parasitic infection and is attributed to Porites epidermal production of a red fluorescent protein. 101 Melanin-like pigment production in response to heat stress or injury in stony corals is part of an innate immune pathway involved in antimicrobial and encapsulation processes. 103 In many instances, the pigment(s) responsible for a pigmented lesion have not been elucidated. Pigmented lesions may develop areas of tissue loss (Fig. 3h), 25 remain static, or occur in areas where the colony’s contour is depressed, suggesting comparatively slower growth of affected polyps. 37
Growth Anomalies
Growth anomalies present as a mass lesion, with considerable morphological diversity, including, but not limited to, annular, bosselated, crateriform, exophytic, fimbriate, nodular, ruminate, and vermiform variations (Fig. 3i).174,175 They may show a growth pattern that compresses, encroaches on, or effaces adjacent corallites, and may have accelerated or reduced growth compared to surrounding tissue. Growth anomalies may also be bleached or pigmented. Corals may have focal cellular proliferations or various tissue deposits that encapsulate invading organisms, such as crustaceans that similarly present as nodules.2,176 Therefore, examination of growth anomalies should include attempts to identify potentially embedded organisms. Otherwise, 2 divergent lesion themes appear to underlie most growth anomalies: polyp and corallite gigantism, and a variety of basal body wall proliferations, both of which also produce distorted skeletal support.36,110,123,174 Soft tissue growths, lacking underlying skeletal malformations, have also been observed but these findings are not published (Esther Peters, oral communication).
Considerations for Histological Sampling and Processing
Corals present unique sampling and processing challenges due to their dense exoskeleton and delicate overlying tissue layers. Briefly, samples should measure at least 2-cm long in branched stony or soft corals such as finger corals or sea fans, or 2-cm-diameter cores that are deeper than the polyp in the case of massive corals like brain coral. Sampling of lesions should strive to capture the transition between normal and abnormal tissue. Two centimeters is the minimum size because tissues at the edges of the collection site where the fragment is severed are invariably damaged, complicating microscopic interpretation. When sampling a coral colony with a lesion, it is standard practice to sample a lesion fragment and a paired fragment from the same colony with no evident gross lesion to provide a comparative basis to interpret microscopic pathology. If permitting allows, a sample from an unaffected colony on the same reef is also beneficial for comparison, as apparently normal tissue from diseased colonies may have microscopic lesions. Guides113,169 exist with more detailed instructions, and sampling should be done in such a way as to prevent potential contamination of the unaffected site, other corals, or introduction of novel pathogens,52,99 notably from dive gear, sampling tools, and ballast decontamination between sites. 62 Coral fragments should be placed in a labeled individual tube or bag filled with seawater on collection, held in seawater for no longer than a single dive, and fixed as soon as possible after collection to minimize artifacts due to the effects of transport and storage of live polyps. 169 Seawater-buffered zinc formalin is the most commonly used fixative, though neutral-buffered formalin in seawater can provide adequate fixation if the former is not available. 116 Bouin’s fixative may also be used but fixation should be limited to a few hours or the tissue becomes brittle. 116 Subsamples may be placed in fixative for transmission electron microscopy (TEM), and formulations for TEM fixative are published in the work by Price and Peters. 116 Fixation in freshly prepared paraformaldehyde in phosphate-buffered saline (PBS) may be considered for parallel microbiome DNA analysis and histology, 50 though simultaneous adjacent samples flash frozen or preserved in DNA- or RNA-preserving buffers is preferable when possible.
The calcareous exoskeleton of stony corals means that for paraffin sections tissues must be decalcified prior to embedding. Decalcification can disrupt histologic architecture and may cause endolithic organisms to lose their spatial association with coral tissues or be lost altogether; therefore, careful processing is essential to histologic interpretation. Methyl methacrylate thin sections as for bone or teeth can be prepared if the exoskeleton is of primary interest, with detailed instructions in the work by Price and Peters. 116
Since samples of wild coral collected for histology are traumatically excised from the colony and typically stored in a plastic bag during the collection dive for a period prior to fixation, a significant margin of tissue is rendered unreadable, and reaction to irritation such as increased mucus production may occur artifactually. Thus, cell injury and traumatic tissue death may occur as an artifact of collection and should be interpreted with caution and in light of the gross lesions. Surgical ink can be used to distinguish tissue loss margins from the traumatized edges of a biopsy. Studies using atraumatic methods of collection such as immediate whole fixing of small aquarium coral may be needed to differentiate artifacts from lesions.
Microscopic Lesions
While basic pathology principles still apply, invertebrates present some unique scenarios which are difficult to interpret through extrapolation from vertebrates. While progress has been made in describing coral histology and histopathology, there is a need for further experimental work to better understand coral responses to injury, and how this relates to specific diseases. 182 There have been efforts to elucidate both normal histology and histological changes in coral disease, with published studies of many diseases including SCTLD, 72 white syndromes of Acropora, 171 growth anomalies, 164 and wound repair 129 (Table 1), but corals and their diseases are diverse and much remains to be described. As well as a relative lack of descriptive normal histology of many species for comparison, there is some confusion regarding the terminology for histologic changes. Defining microscopic changes in corals is an evolving process. Here, we present current recommended nomenclature for some commonly seen histologic lesions expanded and collated from the Coral Disease and Health Workshop, 44 Diseases of Coral, 109 and recent publications.72,143,164,172 These represent an invitation for pathologists to join ongoing conversations to clarify the histology and histopathology of reef-building corals.
Coral Cell Injury and Death
Cell swelling
Generalized swelling of the cells is an early alteration in reversible injury that has been widely described in corals5,39,61,72,167,175 and seems to be a primary response to stress.1,4 In particular, vacuolation and swelling are commonly observed in degenerating Symbiodiniaceae.25,72 Gastrodermal endosymbiont host cell swelling and enlargement of the symbiosome is seen along with degeneration of endosymbionts. 72 Vacuolation within mucocytes, gastrodermal cells, and epidermis is also commonly seen in multiple diseases involving tissue loss. 44 Differentiating cell swelling from post-sampling artifact and mucus production triggered by the trauma of collection can be problematic with light microscopy, so parallel TEM could be beneficial in confirming ultrastructural features of cell injury while also searching for submicroscopic causative agents.
Necrosis
Necrosis is commonly seen in coral diseases that lead to tissue loss. Necrosis in corals follows the same general pattern as other animals manifesting as cytoplasmic hypereosinophilia, pyknosis, and karyorrhexis. However, not all morphological forms of necrosis observed in corals conform to conventional classifications used in mammals and the mechanisms involved may differ from those found in animals possessing a vascular system. The patterns of necrosis described in coral cells include coagulative and lytic necrosis within epithelia, as well as luminal necrotic cell aggregates.72,175 As we come to better understand coral diseases, distinctive morphology may correspond to differential pathogenesis. We define these common forms of necrosis to encourage consistency in their diagnosis. In some cases, necrosis cannot be further classified with confidence, for example, when cells have been sloughed leaving bare mesoglea (Fig. 4c).
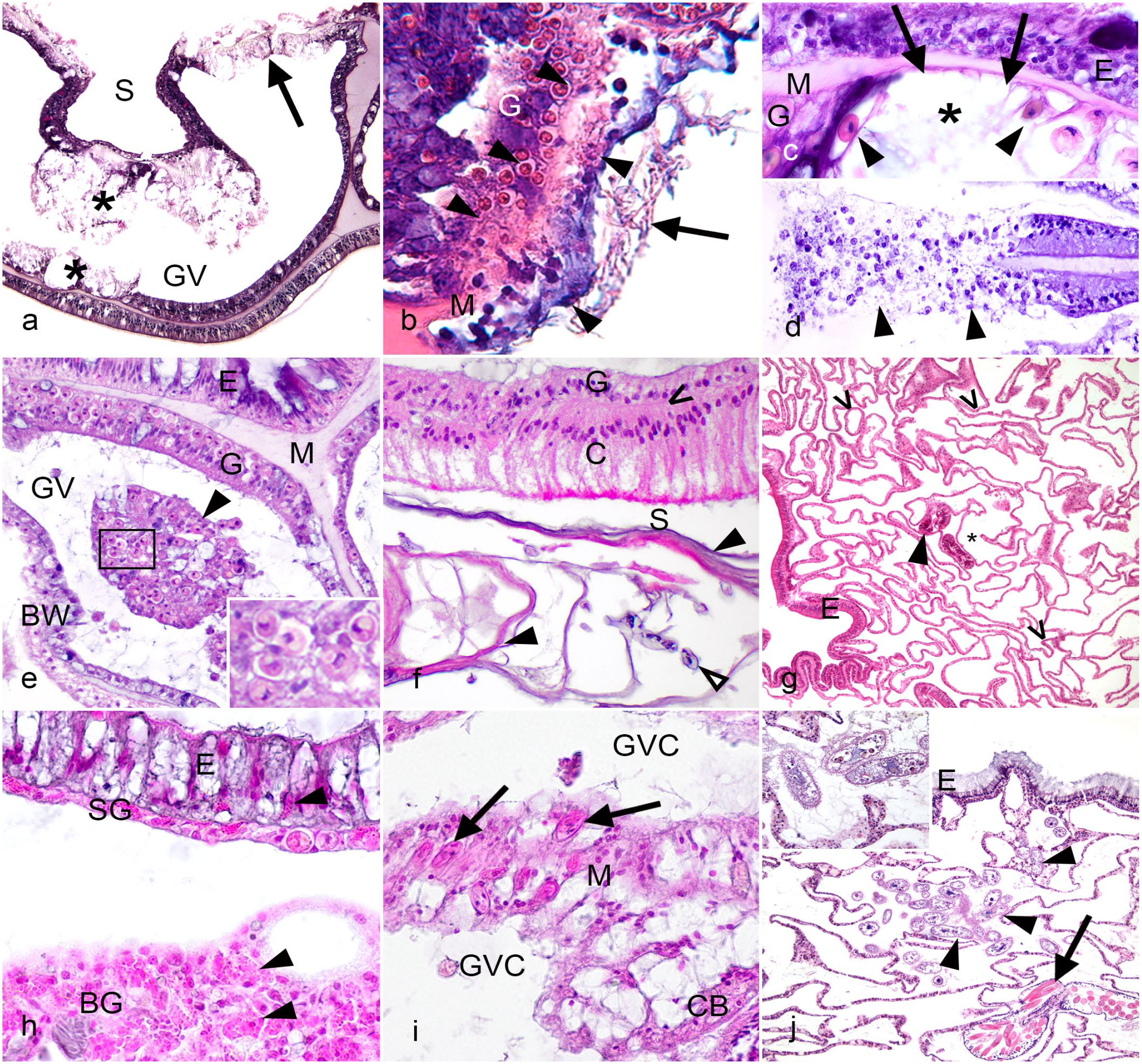
Microscopic lesions in coral. Hematoxylin and eosin. a. Montastraea cavernosa. Lytic necrosis of mucocytes within the gastrodermis (asterisks) indicated by dissociation and dissolution of mucocytes, and lytic necrosis extending through the body wall affecting gastrodermis and calicodermis (arrow), indicated by cell swelling and tissue dissociation. GV indicates gastrovascular space, S indicates skeletal space. b. Orbicella faveolata. Basal body wall with coagulative necrosis (arrowheads) characterized by homogenization and hypereosinophilia of cytoplasm and lysis of nuclei with preservation of tissue architecture. Endolithic hyphal growth is present within the skeleton along the calicodermis (arrow). G indicates gastrodermis of the basal body wall, M indicates mesoglea; note loss of definition within the necrotic area. c. Montastraea faveolata. Surface body wall with loss of gastrodermal tissue possibly due to lytic necrosis (asterisk). Note denuded (arrows) mesoglea (M) and flattened, aspherical endosymbionts (arrowheads). G indicates remaining gastrodermis, E indicates surface epidermis. d. Orbicella annularis. Lytic necrosis of gastrodermis (arrowheads) in a mesenterial filament as indicated by the dissociation and dissolution of gastrodermal cells, cellular debris, and loss of mesoglea. e. Diploria labyrinthiformis. Gastrovascular canal (GV) containing luminal necrotic cell aggregates (arrowhead). Note presence of endosymbionts (rectangle, inset). E indicates surface epidermis, G gastrodermis of the surface body wall, M mesoglea, BW basal body wall. f. Orbicella faveolata. Basal body wall with calicoblast hyperplasia and calicoblast hypertrophy. Large, columnar calicoblasts with closely apposed nuclei and bright pink apical granules (C) separated from gastrodermis (G) by a thin layer of mesoglea (caret). Hyaline lamellae (closed arrowheads) are present within the skeleton (S) along the external margin of the calicodermis, and around endolithic organisms (open arrowhead). g. Growth anomaly of Acropora abrotanoides with basal body wall hypertrophy as indicated by numerous disorganized gastrovascular canals (carets), and relative scarcity of polyp structures, such as cnidoglandular bands (arrowhead). E indicates epidermis. Skeleton and gastrovascular cavity can be difficult to distinguish after decalcification, but the area containing cnidoglandular band is gastrovascular space, and the area on the opposite side of the body wall from cnidoglandular band can be identified as skeleton (asterisk). h. Agaricia agaricites. Atrophy of the surface body wall as indicated by low cuboidal surface gastrodermis (SG), short columnar epidermis (E) which appears dominated by mucocytes. Note the scarcity of endosymbionts within the surface gastrodermis indicating bleaching, and granular cells (granular amoebocytes and/or pigment-containing granular cells) (arrowheads) within epidermis and gastrodermis. BG indicates basal gastrodermis. i. Orbicella faveolata. Apicomplexans of unknown significance (arrows) within mesenterial filament (M) at the base of cnidoglandular band (CB). GVC indicates gastrovascular cavity. j. Acropora palmata. Presumptive pathogenic scuticociliates (arrowheads) within gastrovascular cavity. E indicates surface epidermis, arrow indicates cnidoglandular band with numerous large, eosinophilic cnidocytes. Inset shows magnification of ciliates containing endosymbionts in stages of digestion, inferred to be ingested with coral tissue.
Coagulative necrosis, as in vertebrates, maintains tissue architecture and cell shape while intracellular details are lost, nuclei are pyknotic, karyorrhectic or lysed, and cytoplasm is homogenized and eosinophilic (Fig. 4b). This is observed in coral diseases involving tissue loss and growth anomalies, but it is not known whether it is associated with local hypoxia, as is usually the case in vertebrates, microbial toxins, or endogenous processes.72, 126 In contrast, lytic necrosis is characterized by dissociation of tissues associated with cellular detachment, cell swelling, cytoplasmic dissolution, and nuclear lysis, pyknosis or karyorrhexis, leaving behind cellular debris or denuded mesoglea (Fig. 4a), or in some instances, extending through mesoglea, and disrupting the full thickness of the body wall or mesentery (Fig. 4a, d). Because coral tissues are so delicate, comprising 2 cell layers, comparatively less differentiation, and relatively simple tissue organization, many forms of injury could arguably result in lytic necrosis, but in vertebrates, metabolic, toxicologic, and viral causes of injury are often suspected. While morphological loss of architecture is conceptually similar to liquefactive necrosis observed in some vertebrate lesions due to release of enzymes and free radicals from neutrophils, this process is not seen in corals and different terminology is used.
Lytic necrosis may be observed in different cell types and tissues, affecting the mucocytes within the gastrodermis in SCTLD (Fig. 4a). In these corals, distinct areas of swelling and vacuolation putatively reflect accumulation of mucus within affected tissues that culminates in lysis and tissue fragmentation with disruption and pallor of the mesoglea, 72 and mucocyte membrane lysis can be seen in TEM images of affected tissue. 183
Aggregates of cohesive necrotic cells and endosymbionts within the lumen of the gastrovascular canals or cavity may be seen in corals with grossly visible tissue loss or within growth anomalies (Fig. 4e). This has been referred to as selective cell necrosis45,169,170,176 but the term luminal necrotic cell aggregates is preferred. In Acropora, luminal necrotic cell aggregates at a tissue loss margin may appear to be surrounded by cells or membranous material (Miller et al, in Supplemental Fig. 2G). 88 The adjacent gastrodermis is often intact, and it is not always evident where the debris originated though it has been inferred to be gastrodermal cells by the accompanying free endosymbionts (Fig. 4e inset).
Endosymbiont degeneration and necrosis
The endosymbionts should be evaluated carefully for evidence of injury and degeneration. Normal coral dinoflagellate endosymbionts are round, with a small square nucleus composed of permanently condensed chromosomes (dinokaryon) and contain a pyrenoid which appears as an eosinophilic cytoplasmic granule surrounded by clear margin. 16 This is a starch storage organelle that may be lost in states of stress or disease of the endosymbiont, or with poor fixation. Staining with a periodic-acid Schiff reagent procedure can help assess this organelle. Degenerate endosymbionts are vacuolated and swollen with pale cytoplasm and are sometimes accompanied by enlargement of the symbiosome. Necrotic endosymbionts are shrunken, becoming ovoid or angular (Fig. 4c), and have hypereosinophilic cytoplasm and nuclear pyknosis or karyorrhexis, or have nearly clear cytoplasm and nuclei with only the cell wall visible, resembling empty “ghosts”. 72 Cytological examination of endosymbionts can facilitate identification of degenerative changes. For example, phase contrast microscopy on cytological preparations of unstained endosymbionts from Diploastrea heliopora with bleached foci attributed to yellow band disease were pale, vacuolated, and deplete of organelle structures. 26
In the process of bleaching, endosymbionts may be removed through autophagy more often than endosymbiont release.39,103,163 Autophagy is a highly conserved eukaryotic mechanism that recycles cytoplasmic material through an orderly process as part of normal cell maintenance and tissue remodeling as well as during conditions of stress or starvation.42,84 In corals, a specific and selective autophagic process termed symbiophagy is characterized by the transformation of the symbiosome to a digestive organelle resulting in consumption/degradation of the endosymbionts in response to stress events. 39
Overall endosymbiont density can be estimated histologically, but may be quantified using a hemocytometer by homogenizing a standardized volume of tissue in seawater, 143 or as part of metagenomic analysis. 152
Tissue Repair
Little is known of the histologic progression of wound repair in corals. Grossly, wound repair in corals is influenced by lesion size and shape, 166 colony size, 11 temperature, 74 and health status of the coral colony. 40 The cellular processes of wound repair may vary between genera of corals. In Montipora, wound repair involves development of pluripotent cell layers and re-epithelization that arises from the calicodermis (Work and Aeby in Figure 2). 173 In Pocillopora, wound repair involves anastomoses of mesoglea with formation of new gastrovascular canals that are later epithelialized (Rodriguez-Villalobos et al in Figure 2). 129 Porites is the only genus where a visible recruitment of cellular response characterized by infiltrates of melanin-containing pigmented amoebocytes has been observed (Palmer et al in Figure 6a–d). 104
Alterations of Growth and Cellular Adaptation to Injury
Hyperplasia and hypertrophy
Mucocyte hypertrophy and increased mucus production may occur as a response to irritation or disease.124,142,183 It has been described in SCTLD,72,183 as a response to toxic exposure, 124 and in blister-like growth anomalies of captive Echinopora, 142 and is characterized by an increase in mucocyte size, often with mucus filling gastrovascular spaces. Mucocytes appear to dominate the gastrodermis and/or epidermis, and may be increased in number as well as size (hyperplasia), which can produce a pseudostratified appearance with increased epithelial depth. Mucocyte hypertrophy and hyperplasia can be seen concurrently, and it may not always be possible to distinguish whether mucocytes are increased in number or simply enlarged.
Calicoblast hyperplasia is inferred by the close positioning of nuclei reflecting the increased cell density of the tissue. Calicoblast hyperplasia is often accompanied by calicoblast hypertrophy (Fig. 4f), where calicoblasts are enlarged (columnar or plump cuboidal rather than squamous), sometimes with vacuolated cytoplasm or prominent apical cytoplasmic eosinophilic granules. Calicoblast hypertrophy may also occur independently of calicoblast hyperplasia. Calicoblast hypertrophy and hyperplasia have been described in growth anomalies,66,126,181 and in tissue loss lesions that continue to enlarge after initiation by snail predation injury in Porites. 122 Hyaline lamellae are associated with both calicoblast hyperplasia and hypertrophy. These are composed of CARPs and appear as one or more eosinophilic, laminar structures parallel to the calicodermis (Fig. 4f), 122 often in association with fungal or algal hyphal growth in the adjacent skeleton. 92
Metaplasia has been described in the calicodermis as a feature of wound healing in Montipora capitata, 173 in which calicodermis transitions to an epithelial morphology resembling gastrodermis (gastrodermal metaplasia) or epidermis (epidermal metaplasia). In epidermal metaplasia of the calicodermis, calicoblasts become cuboidal to ciliated columnar rather than squamous and are interspersed with specialized cells typical of epidermis (eg, mucocytes, or cnidocytes). The presence of features not found in calicodermis and absence of those typical of calicodermis, such as desmocytes and apical CARP granules, distinguishes metaplasia of the calicodermis from calicodermal hyperplasia and hypertrophy. Similar transitions among tissues have been observed in one type of growth anomaly and were suspected to represent metaplasia. 126
Growth anomalies and basal body wall hyperplasia
Microscopic lesions with various morphologies comprising gigantism or deformation of skeletal structure overlaid by tissues that may be abnormally pigmented or bleached are observed in the lesions grossly known as growth anomalies. These have been described in Acroporidae, 164 Fungiidae, 164 Faviidae, 126 Poritidae,36,143 Pavonidae, 45 and Pocilloporidae.36,143 The cause and pathogenesis of growth anomalies in corals remains unclear, and there is debate as to whether they are a neoplastic, hyperplastic, or some other process. Microscopic examination of growth anomalies in Montipora24,184 and Acropora164,174 from the Pacific Ocean and faviid corals from the Caribbean Sea 126 revealed increased number of gastrovascular canals surrounded by skeleton and lined by well-differentiated basal body wall (calicodermis, mesoglea, and gastrodermis) with reduced to absent polyp structure. In contrast, some others from the Caribbean with similar gross and histologic morphology have been described as calicodermal neoplasia, specifically calicoblastic epithelioma (Fig. 3g).110,184 The criteria to assign cellular processes to neoplasia have been well described in other animals 51 and could likely be applied to corals to resolve this issue, but to date, clear evidence of neoplasia in corals, as defined for vertebrates, is lacking. 34 However, Tarin 150 states that the calicoblastic epithelioma of Acropora palmata 110 is an example of progressive neoplasia based on histopathological criteria.
Atrophy
Tissue atrophy in corals is like that seen in other animals and is manifested by a reduction in the volume of cells and the tissues they comprise. Gastrodermal and epidermal atrophy are best known in bleached corals and the cellular loss of the symbiont. Gastrodermal atrophy associated with bleached corals is due in part to the loss of symbionts, which changes the shape of the cell from tall cuboidal or columnar to low cuboidal. In epidermal atrophy, the epidermis is short columnar to cuboidal rather than tall columnar and may be simple rather than pseudostratified epithelium. 181 In some cases, these tissues appear dominated by mucocytes53,175 due to the relative lack of epithelial cell mass. In these tissues, atrophy can be deduced and differentiated from hypoplasia based on observing cell or tissue size reductions in adult polyps which have grossly reached full size. Remaining endosymbionts may show degeneration or necrosis.22,47,53 One possible mechanism is that loss of symbionts may lead to “starvation” of the coral host pursuant to loss of essential nutrients provided by algal symbionts.53,175 Further studies are needed to confirm this. In some cases, prolonged bleaching is associated with reduction in polyp depth, 86 size and thickness of cnidoglandular bands, and overall polyp size (Dennis and Peters, personal observations).
Pigment changes of coral tissue
Pigmentation
Microscopically, grossly recognized pigmentary changes can reflect organisms that infiltrate coral tissues, changes in endosymbionts, or pigments produced by coral cells. As previously discussed (see “Gross Lesions”), examples of pigmented organisms that may colonize coral tissue include some colored-band diseases where ciliates or cyanobacteria are responsible for the color change,20,177 or the darkening seen in some corals with endolithic hypermycosis. 177 Histological identification of these organisms is addressed in a later section (see “Microbial associations”).
Apart from endosymbiont degeneration seen with bleaching (discussed below), pigmentary changes in Symbiodiniaceae may not be histologically obvious and identification of abnormal endosymbionts may be facilitated by their cytological or electron microscopic examination.25,26 Using phase contrast microscopy of unstained endosymbionts, those from dark spot disease-affected tissue of Stephanocoenia intersepta were observed to be dark. 25
There are few convincing instances where endogenous pigment production was the basis for gross pigment change and was histologically evident. In Porites spp, aggregations of granular amoebocytes containing melanin may be the basis for color change.101,102 The red fluorescent protein producing the gross pink pigmentation seen in Porites trematodiasis was not histologically apparent, 27 but was later demonstrated diffusely throughout epidermis near encysted trematodes using confocal microscopy on cryo-sections. 102 Such instances where the basis for pigmentation is not histologically evident demonstrate the importance of correlating cytological and histological assessments with other techniques that characterize pigments, such as confocal or ramen microscopy, spectrophotometry, or mass spectrometry, in order to improve our collective understanding of the pathological basis for pigmented lesions in corals.
Bleaching
Coral bleaching appears histologically as a marked reduction or absence of endosymbionts within the gastrodermis of the surface body wall, which may occur due to physiologic stressors (eg, temperature changes) or infectious diseases (eg, yellow band disease). Gross bleaching may not be evident until more than 50% of the endosymbionts have been lost. 22 Endosymbionts may have features of degeneration and necrosis, as described above,22,72 and increased numbers of dividing endosymbionts may be observed during thermal bleaching. In some cases, the endosymbionts are released into the gastrovascular spaces 21 and may be free floating with cellular debris. Bleaching may be accompanied by atrophy of coral tissues. 175
Bleaching due to pathogenic microorganisms can be observed in some Vibrio infections,25,26,70,90 and in these cases, the mitotic rate of endosymbionts is decreased. 25 It is also seen in some lesions of SCTLD either as an early lesion or at the margin of tissue loss, where deformation and lysis of endosymbionts is a common finding (Fig. 4c), 72 and anisometric (flexuous rod shaped) viral-like particles have been observed within some affected endosymbionts using transmission electron microscopy, although their significance is uncertain. 183
Granular amoebocyte aggregation
It can be difficult to classify lesions and infer causes and chronicity using the same criteria as in vertebrates, but some general conclusions can be drawn. Granular amoebocytes are more visible and prominent in some taxa, and may be observed in locally increased numbers in areas of injury or inflammation. 104 Their presence can be helpful in distinguishing antemortem processes from postmortem artifact, but they may be inapparent in some antemortem disease processes.
Granular amoebocyte aggregates associated with injury or disease may be seen in coral tissues, with significant variations between species in their numbers, appearance, and distribution. These are most prominent in octocorals (eg, sea fans) where granular amoebocytes are increased in the coenenchyme of corals with various infections 12 and with heat stress. 93 In stony corals, they have been described as previously mentioned in the healing process of P. cylindrica, where two morphologically distinct populations are observed at the lesion edge early in the healing process: one with cytoplasmic melanin granules and one with eosinophilic granules. 104 Increased numbers of granular amoebocytes can be observed in diseased areas (Fig. 4g), but more research is needed to demonstrate cellular mobility and whether directed chemotaxis is occurring.
Microbial associations within coral tissue
Microbial associations in corals occur within tissues and within the skeleton (endolithic organisms) and may or may not be associated with gross or microscopic lesions.
Microbes found in normal tissues
Corals possess a rich microbiome, sometimes including single-celled eukaryotes and small multicellular organisms of uncertain significance. These may be commensal or unrelated to disease, and others may be opportunistic feeders on diseased coral rather than primary pathogens. In addition to Symbiodiniaceae, a variety of organisms have been documented in apparently normal tissues. For instance, trichodinid ciliates may be seen with no association to a lesion, and some corals have aggregates of bacteria in tissues thought to also be symbiotic and found predominantly in Acropora, Porites, and Pocillopora. 170 Apicomplexa have been seen in some coral species on histology (Fig. 4i)72,156 and by using molecular tools. 65 While these organisms are not associated with pathology, their role in coral health is unclear. 179 Finally, the coral skeleton is known to harbor numerous and diverse endolithic organisms such as algae 141 and fungi, 49 which are often present with no reaction or may cause calicoblast hypertrophy and increased CARP production when abundant. Other microorganisms have been found to cause localized cellular responses within coral tissues that are not grossly visible lesions, such as suspect gregarine cysts in hypertrophied calicoblasts of Porites spp with increased number of CARP-filled granules. 108
Microbes associated with disease
Microorganisms visible on routine staining have also been associated with lesions. One of the earliest characterized coral diseases was black band associated with invasive overgrowth of cyanobacteria and sulfur-metabolizing bacteria. 127 In some cases, endolithic organisms can overgrow the skeleton and lead to abnormal pigmentation, tissue invasion and necrosis 177 (Fig. 4b), and calicoblastic hypertrophy. In other instances, particularly during bleaching, loss of tissue pigmentation can lead to increased light penetration and overgrowth of endolithic algae with potential invasion into coral tissue. 43 Although not strictly a microorganism, corals can also become parasitic and invade other corals. 179
Ciliates are a prevalent microorganism in diseased coral tissue,20,182 but controlled inoculation experiments are lacking and therefore their true role in disease pathogenesis remains under investigation. Ciliates are reported to be absent in healthy coral tissues by microscopy and molecular techniques,146 –148 while a diverse community of ciliates has been associated with a variety of coral diseases, particularly those with tissue loss as a primary feature.20,147,149,171,182 In diseased coral tissues, 3 types of relationships with ciliates are possible: (1) those feeding on bacteria colonizing necrotic tissue and the bare skeleton; (2) predators of other protozoans, attracted to other ciliates colonizing the necrotic tissues of the lesion margin; (3) histophagous (tissue-eating) species that contain ingested coral tissue as evidenced by coral endosymbionts visible within the ciliate, and may play a role in disease pathogenesis. 149 Many ciliates are therefore likely to be secondary invaders (bacterivores, protozoavores) but 2 types of ciliates are described in diseased tissues: folliculinid ciliates (eg, Halofolliculina sp) and scuticociliates (eg, Philaster sp). 81 Histologically, folliculinid trophonts are oval with a macronucleus and found individually or in groups or chains confined within ovoid- to tube-shaped lorica. They are often associated with epidermis or within clear space where denuded skeleton was decalcified. Folliculinids can penetrate exposed coral skeleton and have been associated with skeletal eroding band, a disease characterized by tissue loss and skeletal erosion with a dark, speckled margin composed of the folliculinid loricas, 32 but histologic evidence for tissue invasion is lacking.98,121 Scuticociliates can be found invading into intact coral tissues in advance of the lesion margin and containing ingested endosymbionts 146 (Fig. 4j), suggesting a primary role in tissue damage,121,149 though they may function as part of a consortium with bacterial infection rather than as a sole pathogen.147,148 Scuticociliate trophonts are typically found within the polyp, are oval to pyriform with hyperchromatic macronucleus, and contain cytoplasmic vacuoles with ingested endosymbionts and cellular debris. They also have peripheral rows of cilia best seen in cytological preparations. 146
Ancillary Diagnostic Methods
Ancillary diagnostic methods are less well developed for corals than for most vertebrates. Histochemical stains such as Gomori’s methenamine silver and Giemsa can be useful to highlight fungi and bacteria. Bacterial culture is of questionable utility for identifying pathogens in an animal that lacks any normally sterile tissues, but culture along with molecular techniques such as 16S ribosomal sequencing have been used to identify shifts in the microbiome associated with disease.87,132 Development of useful diagnostic biomarkers from these data is challenging as the individual members of the microbiome can vary widely between coral species and location as well as within healthy and diseased populations, but increases in beta diversity and virulence factors within the microbiome have been observed to correlate with stress and diseased states.152,185 Because shifts in microbiome integrity can potentially cause diseases in coral, going forward it seems important to integrate the search for potential microbiomarkers of disease with histopathology, in an effort to link cellular changes and pathology to changes in the microbiome.
Commonly used tools to assess coral disease are various forms of deep sequencing to target viruses 158 or bacteria. 105 This is most informative when paired with histology, including immunohistochemistry and in situ hybridization, to support their association with lesions at the microscopic level. 180 Given that corals are nonsterile systems, interpreting next-generation sequencing such as viral or bacterial metagenomics is difficult in the absence of other confirmatory tools to localize the agent to the lesion at the microscopic level.
Few examples are available in the literature for immunohistochemical stains in corals, mainly due to a lack of appropriate reagents. Purevel et al 117 immunized mice with proteins extracted from the skeleton of Stylophora that were then used to tag the calicodermis of corals using immunohistochemistry. Cryptochromes (photoreceptor proteins involved in coordinating mass spawning) in Acropora were immunolocalized in ectodermal tissues using antibodies generated from immunizing mice with artificial peptides. 77 Plant-derived lectins have been used to localize different cell types and structures, including granular gland cells, cnidocytes, mesoglea, and gonadal tissues in coral tissues by differential staining. 178 Neither lectin cytochemistry nor immunochemistry have been used to elucidate pathogenesis or etiology of disease in corals; however, TUNEL (terminal deoxynucleotidyl transferase dUTP nick end labeling) staining has been used to investigate the role of programmed cell death in cases of Indo-Pacific white syndrome (tissue loss),5,6 thermal stress, 106 and SCTLD.85,136
Fluorescence in situ hybridization has been extensively used in corals to localize cell-associated microbial aggregates in a variety of coral species.4,3,161 This tool has been used to investigate the role of bacteria in acroporid white syndrome, finding no association between bacteria and lesions, 6 but suffers from difficulties in interpretation because of the propensity for many coral tissue structures to autofluoresce. 3
Scanning electron microscopy has been extensively used to examine fungal interactions by visualizing hyphal morphology, and swellings at the intersections where hyphae penetrate the skeleton, 120 and to observe the presence of thin, haphazardly placed skeletal trabeculae in growth anomalies. 9 Transmission electron microscopy has been used to detect viruses in corals, 165 examine endolithic fungal associations in corals, 120 and to investigate the pathogenesis of SCTLD. 183
Future Opportunities
In vertebrates and many better-studied invertebrates, veterinary diagnosticians routinely use a deductive approach for interpreting gross and histological lesions to determine the most likely causes of diseases. In corals, the descriptive framework that informs this deductive approach is in development, and pathogenesis for many lesions is still speculative and based on extrapolations from other taxa. 180 In order to achieve this level of understanding of corals, we need more histology of both natural and experimental disease, as well as of healthy coral samples from a variety of environments (wild, aquaria). An improved understanding of gross and microscopic lesions will also emerge as these findings are paired with transcriptomics and metabolomics to elucidate and inform the metabolic and biochemical processes that underlie observed cellular changes. Finally, there is a critical need to develop standardized animal models for coral disease.
Establishment of corals as an experimental animal, with known genotypes and standardized husbandry, would help to bring consistency to in vivo laboratory experiments designed to elucidate the pathogenesis of the many diseases threatening coral and allow for sampling throughout the disease progression. The anemone Aiptasia has been suggested as an easier-to-raise model organism that may help explicate the cellular and molecular mechanisms involved in the unique symbiosis of corals and Symbiodiniaceae. 95 Sustainable coral cell lines have been established 64 and their utility in disease investigation is beginning to be explored. Many of these questions could be addressed by veterinary pathologists, a discipline poised to contribute to this agenda.
Behavior as an indicator for disease in corals is also a largely unexplored area. Prior to the manifestation of the gross lesions, coral may show clinical signs of illness including extruding mesenteries, retracting or protruding polyps at the wrong time of day, or decreased to absent extrusion of feeding tentacles suggesting decreased feeding. Public aquaria provide a unique opportunity to make careful behavioral assessments, document the development and progression of gross lesions, and collect appropriate samples to correlate the histological changes associated with gross lesions over the time course of lesion development, something that can be rarely done in the field. These institutions also offer the opportunity to better characterize variations in coral anatomy and cellular biology that might occur under various husbandry regimes and offer potential avenues to develop standardized animal models for coral disease.
While much work remains to be done, corals offer a unique opportunity to the pathologist to expand the understanding of an ecologically important animal. Establishing consistent descriptive terms and, where possible, developing morphologic case definitions of disease through a pathology lens is an important step in accurate diagnosis of coral diseases, as a prelude to development of biomarkers and other specific diagnostic tests. This is crucial information to help guide management plans to mitigate disease and anthropogenic stressors on coral reefs, and work toward their long-term preservation.
Footnotes
Acknowledgements
The authors would like to thank the members of the Histopathology subteam of the Florida Disease Advisory Committee (DAC) for expert assistance and professional input.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
