Abstract
Hemorrhagic bowel syndrome (HBS) is a sporadic and fatal disease of predominantly lactating dairy cattle, characterized by segmental hemorrhage and luminal clot formation in the small intestine. Although, Clostridium perfringens and Aspergillus fumigatus have been associated with HBS, the pathogenesis and cause are currently unknown. In this study, 18 naturally occurring cases of HBS (7 necropsied immediately following euthanasia, 11 with 12–48 hour postmortem intervals) were investigated to characterize the pathology and the intestinal microbiome. Hemorrhagic bowel syndrome was characterized by a single small-intestinal, intramucosal hematoma with dissection of the lamina muscularis mucosae. In most cases necropsied immediately after euthanasia (4/7), the intestinal mucosa proximal to the hematoma contained 9 to 14, dispersed, solitary or clustered, erosions or lacerations measuring 4 to 45 mm. In 77% (37/48) of these mucosal lesions, microscopic splitting of the lamina muscularis mucosae comparable to the hematoma was present. These findings suggest the intramucosal hematoma to originate from small mucosal erosions through dissecting hemorrhage within the lamina muscularis mucosae. No invasive fungal growth was observed in any tissue. Bacteriological cultivation and nanopore sequencing showed a polymicrobial population at the hematoma and unaffected intestine, with mostly mild presence of C perfringens at selective culture. Gross and microscopic lesions, as well as the culture and sequencing results, were not in support of involvement of C perfringens or A fumigatus in the pathogenesis of HBS.
Keywords
Hemorrhagic bowel syndrome (HBS) or jejunal hemorrhage syndrome (JHS), is an acute and fatal disease mainly affecting adult dairy cattle, with Brown Swiss being overrepresented in several studies.7,25 Management practices targeting high milk production are suggested as risk factors for this disease. 6 Hemorrhagic bowel syndrome is characterized by segmental luminal clot formation and obstruction in the jejunum. Affected animals initially show signs of small-intestinal obstruction, with subsequent development of abdominal distention, and often the appearance of melena in the feces. 12 This is followed by shock, which if left untreated, results in death within hours or up to 2 days after the first clinical signs.2,12 The disease is difficult to diagnose clinically, and a final diagnosis is often made upon laparotomy or necropsy. 12 Hemorrhagic bowel syndrome was first described in 1991, 4 but the cause and pathogenesis are still unknown. 13 Despite the importance of this emerging syndrome for the dairy industry, only a handful of studies have focused on the cause and pathology of HBS. C perfringens type A was strongly suggested as the causative agent of this disease.8,10,11,19 This is based on its isolation from the intestine of affected animals and because C perfringens is known or associated with acute hemorrhagic enteritis in several species.16,32 In addition, in a single study, another facultative pathogen, the common mold A fumigatus, was suggested as the etiologic pathogen based on the detection of this fungus in several tissues and the blood of animals with HBS.15,31 However, the intestinal microbiome of cattle with HBS has not been documented. As dietary factors and high milk yield are associated with the disease, a change in small-intestinal microbiome might play a role in this syndrome.
Gross and histological descriptions of the lesions are inconsistent in literature. Hemorrhagic bowel syndrome was mostly described as a segmental necrohemorrhagic enteritis with intraluminal blood clot formation.8,10,19 Histologically, prior studies have reported nonspecific acute inflammatory changes in the intestines.2,8,10,19 More recently, it was reported that the hemorrhage originates within the intestinal wall, and the lesion is in fact an intramural hematoma.3,22 In a single study, it was suggested that the hemorrhage starts in the lamina propria secondary to a disturbance of blood or lymphatic flow, 3 while vascular fibrinoid necrosis in the submucosa was proposed in another study. 22 Both studies were hampered by either the limitations of a retrospective study or only the affected intestinal segment was investigated. Although rarely discussed, previous investigations might have been impeded by postmortem change and bacterial overgrowth, which occur rapidly in the intestines, especially in cattle.21,30
Because of the lack of consistent reporting of the pathologic findings in literature, and the lack of knowledge on the pathogenesis and cause, we performed a descriptive study with the objective to characterize the macroscopic and microscopic findings in HBS. In addition, intestinal samples were analyzed for the presence of possible etiological pathogens by bacteriological cultivation and 16S nanopore sequencing.
Materials and Methods
Animals
In this descriptive study, field cases were divided into 2 groups of dairy cattle based on their postmortem intervals at the time of necropsy. Group 1 consisted of 7 animals with minimal to no postmortem autolysis, allowing for optimal histological and bacteriological examination. 21 Lactating cows with a presumptive diagnosis of HBS based on the presence of acute (<3 days) signs of intestinal obstruction, loss of milk production, and bloody feces were included in the study. Animals were referred to the Clinic of Large Animal Internal Medicine of Ghent University for confirmation of the HBS diagnosis. To confirm the diagnosis, a complete clinical examination, abdominal and thoracic ultrasonography, complete blood count (CBC), and serum biochemistry were performed. As a part of routine diagnostics, a blood sample was taken for blood culture. If HBS was highly suspected or the animal was in very poor clinical condition, and the owner gave permission after being informed of the prognosis, the cow was euthanized. Thereafter, a necropsy was immediately performed at the Laboratory of Veterinary Pathology of Ghent University. Formal ethical approval was waived by the chairperson of the ethical committee, based on Belgian and European legislation (EU directive 2010/63/EU), as all tissues were derived postmortem.
Group 2 consisted of 11 animals with HBS that were necropsied within 12 to 48 hours (h) postmortem, which resulted in varying degrees of postmortem autolysis. Necropsies were performed at the Animal Health Service-Flanders (DGZ Flanders) or the Laboratory of Veterinary Pathology of Ghent University.
Necropsy
A complete standard necropsy, with special attention to the gastrointestinal tract, was performed on the animals with minimal postmortem autolysis (group 1). The intestinal hematoma was individually examined and opened with blunt scissors at the mesenterial site. Samples for bacteriology and nanopore sequencing were immediately taken, followed by samples for histology. The entire remaining intestinal tract was carefully opened with a blunt-ended scissors at the mesenteric site; and the mucosa was carefully rinsed with tap water for gross inspection.
Animals necropsied within 12 to 48 h postmortem (group 2) were examined with the focus on the gross morphology of the hematoma, and only samples for histopathology were taken. No samples for sequencing or bacteriological examination were taken because of presumed postmortem microbiome alteration.
Histological Examination
In group 1 animals, the entire hematoma and samples of the duodenum, 5 distant samples of the jejunum, ileum, colon, cecum, duodenal mesenteric lymph node, rumen, reticulum, omasum, abomasum, lung, heart, liver, kidney, pancreas, spleen, and adrenal gland were collected. Additional samples of the intestine were taken if lesions were present. All samples were fixed in 10% buffered formalin for 24 to 72 hours. The hematoma was entirely fixed for 1 week, which was followed by sampling at selected sites that included the proximal and distal transition with normal mucosa, the mesenteric attachment, central areas with ruptured or intact mucosa, and, if present, smaller partial hematomas at the edge. After fixation, all specimens were routinely processed, cut in 5-µm-thick sections and stained with hematoxylin and eosin (HE). Selected sections were immunohistochemically labeled using antismooth muscle actin antibody (SMA IHC). Antigen retrieval was performed by immersion in citrate-buffered (0.01 M, pH6) distilled water and microwaving. Endogenous peroxidase was blocked with 0.03% hydrogen peroxidase (Dako, Glostrup, Denmark) at room temperature. Sections were incubated for 30 minutes with monoclonal mouse antismooth muscle actin (Dako) as primary antibody. Labeling was visualized with antimouse/EnVision+ System-HRP (Dako). Smooth muscle tissue of the intestinal sections served as positive control. Negative controls were prepared from serial sections in which the primary antibody was omitted and replaced by dilution buffer. In group 2, histological sampling was limited to the rumen, abomasum, duodenum, jejunum, ileum, colon, cecum, liver, kidney, and the entire hematoma. After 1 week of fixation, the hematoma was sampled at the proximal and distal transition, central areas, mesenteric attachment, mesentery, and adjacent mesenteric lymph node.
Bacteriological Examination
To isolate potentially causative bacterial strains, mucosal swabs were taken at 3 sites of the small intestine and stored at 4°C for less than 12 hours. These included intact mucosa at the hematoma and several meters proximal and distal to the hematoma. In the proximal and distal samples, care was taken to avoid segments with bloody content, as much as possible. Swabs were inoculated in duplicate on Columbia agar supplemented with 5% sheep blood (Oxoïd, Hampshire, UK) within 12 hours after sampling, and incubated for 24 hours at 37°C in either aerobic or anaerobic conditions. Second, a selective chromogenic culture medium for C perfringens was used (CHROMagar C perfringens, CHCP), allowing a better qualitative and semiquantitative estimation. 18 Each swab was inoculated on CHCP and anaerobically incubated for 24 hours at 37°C. After incubation of the nonselective plates, 4 to 7 colonies of different morphologies were further purified and identified with an Autoflex III smartbeam MALDI-TOF mass spectrometry, using FlexControl and MBT Compass software (Bruker Daltonics, Bremen, Germany) as described before. 18 The toxinotype of the C perfringens strains was determined by a multiplex polymerase chain reaction (PCR) targeting the genes encoding alpha toxin (cpa), beta toxin (cpb), epsilon toxin (etx), iota toxin (ibp), enterotoxin (cpe), and NetB (netB) as described by Rood et al. 28 If animals were already treated with antimicrobials, bacteriological examination was not performed (2/7 excluded).
Before euthanasia, animals in group 1 were aseptically sampled for blood culture at the jugular vein, followed by inoculation on 3 types of culture medium: BD BACTEC, Plus Aerobic, BD BACTEC, Lytic Anaerobic, and BD BACTEC, Mycosis-IC/F, which were subsequently incubated at 35°C for 120 hours. If growth was present, identification was conducted using MALDI-TOF mass spectrometry.
Nanopore Sequencing
To identify the microbiome composition in the small intestine of HBS cases, intestinal content was collected at the hematoma and the proximal intestine (at the identical location of the mucosal swabs for culture) and were stored at −20°C prior to analysis. In case the intestinal lumen was completely filled with blood at the hematoma site, no content was taken (2/7 excluded). Also, when animals received antimicrobials, no samples were collected (2/7 excluded). After thawing, samples were subjected to DNA isolation using the ZymoBIOMICS DNA Miniprep kit (D4300; Zymo Research) according to manufacturer’s instructions. Quality of resulting DNA was verified on nanodrop, and samples with lowered quality were cleaned using CleanNGS magnetic beads (CNGS-0050; CleanNA). Subsequently, 10 ng DNA was used for targeted 16S rRNA gene sequencing following the 16S barcoding kit (SQK-16S024; ONT). Sequencing was performed on a R9.4.1 flow cell on a GridION sequencer (ONT). Resulting sequencing data were processed in an in-house bioinformatics pipeline allowing accurate 16S amplicon classification to determine relative abundances up to the genus level. This workflow includes conversion of raw data to bases using Guppy v5.0 (ONT), followed by read filtering, trimming, and demultiplexing (qcat v1.1.0 [ONT] and NanoFilt v2.7.1). 9 Cleaned sequencing reads were then used for taxonomic classification using the SILVA 138.1 database and de novo consensus NCBI validation. 26 No samples from animals in group 2 were subjected to sequencing.
Results
Clinical and Anamnestic Findings
All animals (n = 18) were of the Holstein-Friesian breed, aged between 2 and 8 years (mean: 4.1; standard deviation [SD]: 1.6) and originated from different farms in Flanders (Belgium), except for 2 animals that were from the same farm. Time since parturition ranged between 84 and 366 days (mean: 159.2; SD: 90.3). The lactation number ranged between 1 and 6 (mean: 2.6; SD: 1.5), with 3 animals within first lactation. On 3 farms (3/17), at least one other confirmed case of HBS was seen in the past. All cases were collected during 2020 to 2021.
Six of 7 animals in group 1 demonstrated acute signs of weakness, ileus, milk drop, and bloody feces and/or melena for less than or equal to 24 hours, while 1 animal had clinical signs for 3 days. One animal was euthanized at the farm near the university because of clinical deterioration. The other 6 animals received an extensive clinical exam. All but one animal examined had an increased heart rate (mean: 91 beats per minute; SD: 12.0) and normal respiratory rate (mean: 28 beats per minute; SD: 6.2). Two cows had severe hypothermia (<36°C). The mucosal membranes were pale in 3 animals. Capillary filling times were increased in all but one animal and skin pinch was prolonged in 3 animals. The mental state ranged from slightly apathic to severely lethargic, with 3 animals in a shock state. Abdominal auscultating revealed steelband and slushing sounds and absence of rumen motility in all animals. Two animals were unable to stand upon arrival, making it impossible to execute a proper rectal examination. Ruminal impaction was present in all animals that were rectally palpated, and enlarged small intestines were identified in 2 animals. Ultrasound revealed ileus and dilated intestines, with luminal blood clots in 2 animals. The most significant clinical pathologic findings were high lactate levels (mean: 8.6 mmol/L; SD: 3.5), hyperglycaemia in whole blood (mean: 231 mg/dL; SD: 55.4) and serum (mean: 12.7 mg/dL; SD: 2.9) and hypochloraemia (mean 86.5 mmol/L; SD: 7.8) in all animals. Multiple animals showed severe hypocalcaemia for both ionized (mean 0.82 mmol/L; SD: 0.18) and total calcium (mean 1.3 mmol/L; SD: 0.4). Only one animal had a decreased haematocrit (17%) (mean 28%; SD: 8.4).
Clinical signs of cows in group 2 (n = 11), described by the referring veterinarian and farmer, were similar to those observed in group 1, although 1 animal was found dead without preceding signs. In 6 animals, bloody feces were noted. The interval between death and the time the first clinical signs were noticed was less than 24h in 6 of 7 animals with spontaneous death; while 4 cows were euthanized because of recumbency and a poor prognosis.
Gross Pathology
In all group 1 animals (n = 7), the small intestine contained a single, firm, distended, and coiled intestinal segment, with often severes subserosal hemorrhage (Fig. 1a). This segment was located at different sites of the small intestine from 2 meters distal to the pylorus to 1.9 meters proximal to the ileocecal junction. Overall, distal jejunum was the most affected site (5/7) (Table 1).
Summary of gross and histological findings at the hematoma and small mucosal (early-stage) lesions in cattle with hemorrhagic bowel syndrome (group 1, necropsied immediately after euthanasia).
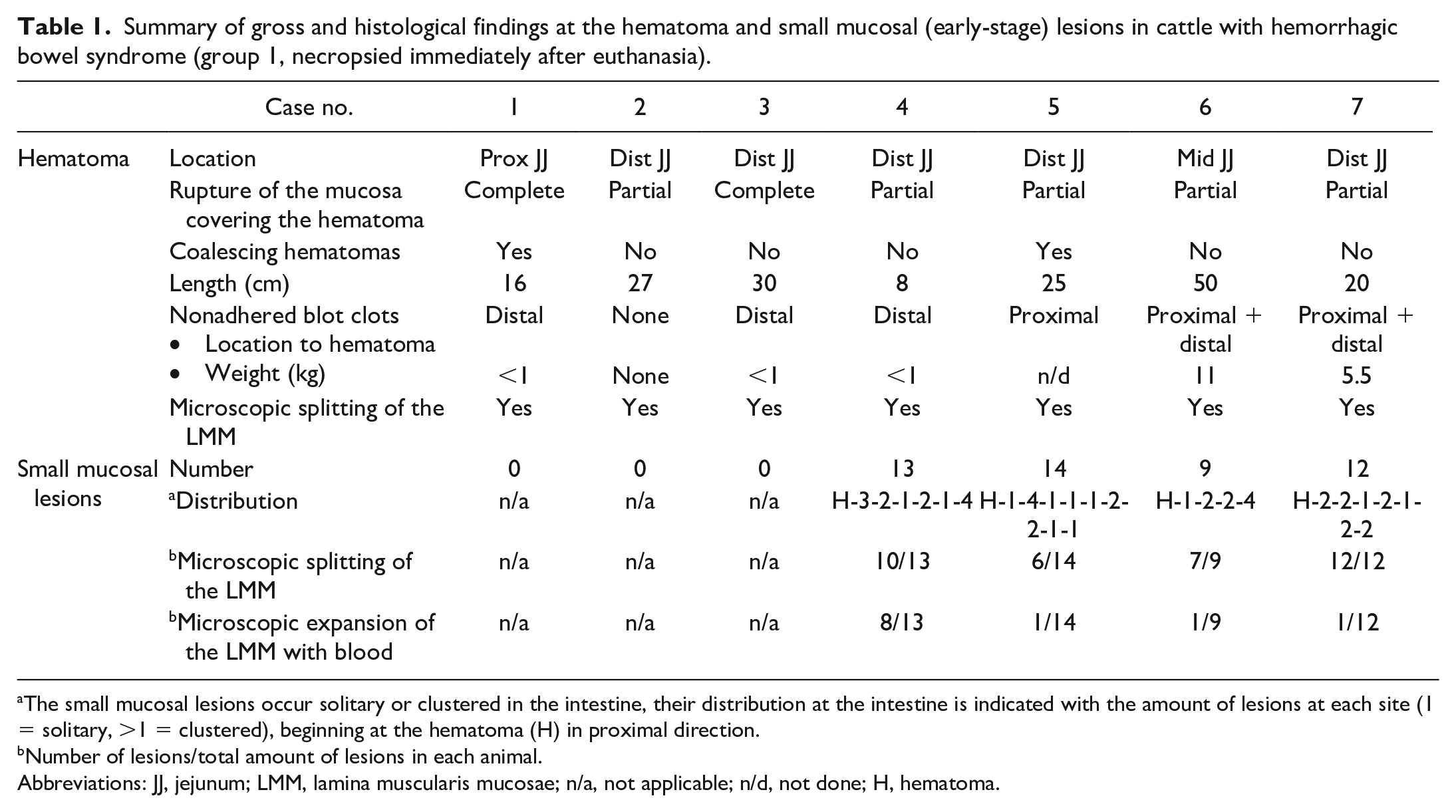
The small mucosal lesions occur solitary or clustered in the intestine, their distribution at the intestine is indicated with the amount of lesions at each site (1 = solitary, >1 = clustered), beginning at the hematoma (H) in proximal direction.
Number of lesions/total amount of lesions in each animal.
Abbreviations: JJ, jejunum; LMM, lamina muscularis mucosae; n/a, not applicable; n/d, not done; H, hematoma.
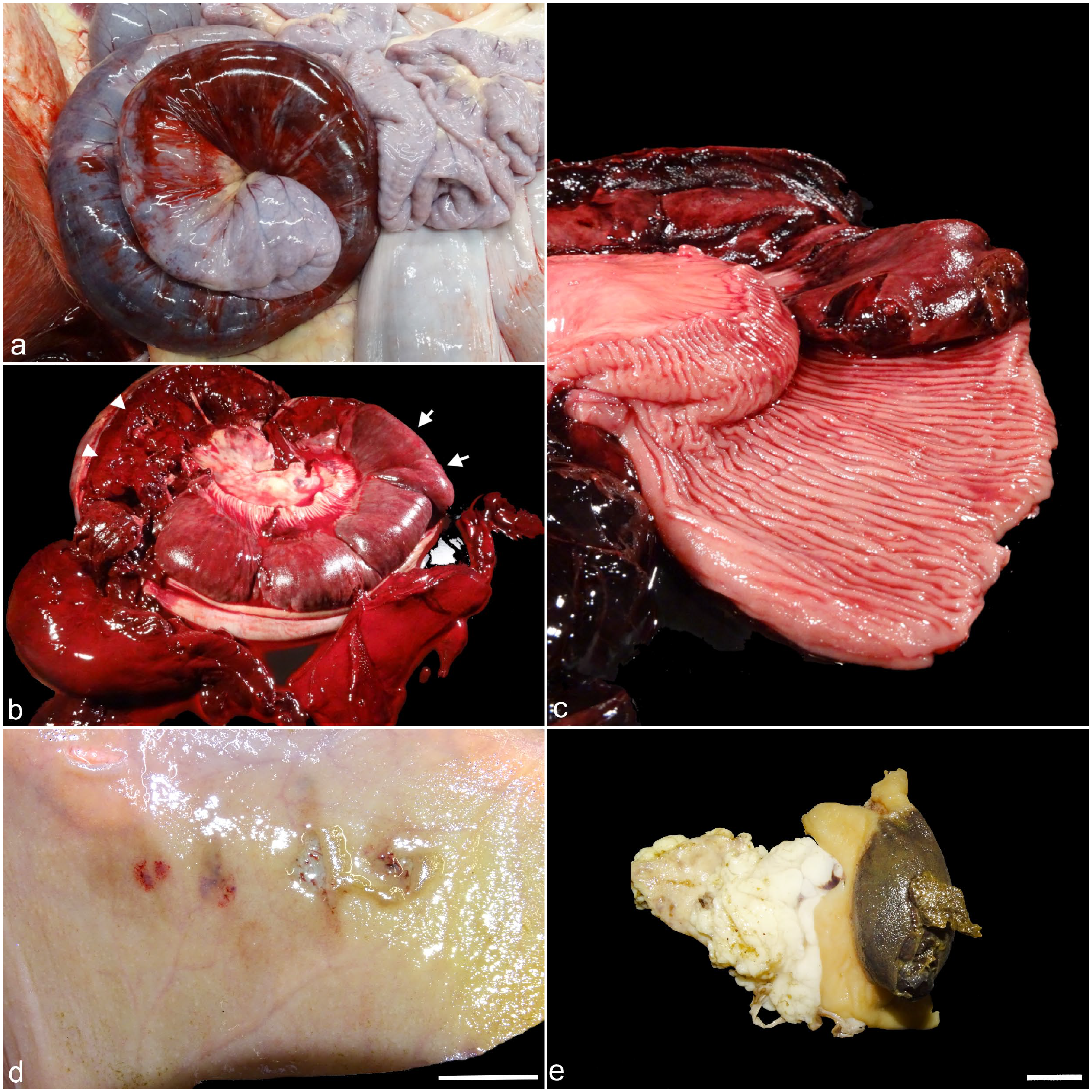
Hemorrhagic bowel syndrome, small intestine, bovine. Serosal and mucosal side of the intestinal hematoma (a-c) and small mucosal (early-stage) lesions (d-e). (a
In 5 of 7 animals, the mucosa covering the hematoma was partially ruptured exposing the submucosal-adhered coagulum (Fig. 1b). In the other 2 cases, the hematoma consisted of blood clots adhering to the intestinal wall, covered with only remnants of necrotic mucosa. In a minority of animals (2/7), the hematoma was composed of multiple often variably sized coalescing hematomas. In all cases, there was an abrupt transition from normal to elevated mucosa with apparent submucosal blood collection (hematoma) (Fig. 1c). The hematoma had an average length of 25.1 cm (range: 7.9 – 50 cm; SD: 13.3) (Table 1).
In all cases, a large nonadhered variably sized blood clot was present in the intestinal lumen because of mucosal rupture at the hematoma (Fig. 1b). In 4/7 animals, the intraluminal blood clot of the hematoma extended 15–25 cm distal to the hematoma and weighed less than 1 kg; while in 3 cases, clots extended more extensively distal and proximal to the hematoma. In these 3 cases, clots were up to 5 meters proximal to the hematoma, and weighed several kilograms (up to 11 kg) (Table 1).
In 4 animals, a total of 48 multifocal small mucosal erosions proximal to the hematoma were found. The number of lesions ranged from 9–14 in each individual animal, 73% clustered with 2 to 4 within a few centimeterss of each other (Table 1). These lesions tended to occur more closer together proximal to the hematoma. Grossly, they were characterized by discrete mucosal erosions or lacerations of 4 to 45 mm in diameter, often with detachment of the mucosal edges and/or mild hemorrhage (Figs. 1d, 2d, g). One solitary lesion, measuring 45 cm in length and located 30 cm proximal to the hematoma, had an oval vesicular appearance with apparent submucosal blood accumulation (Fig. 1e).
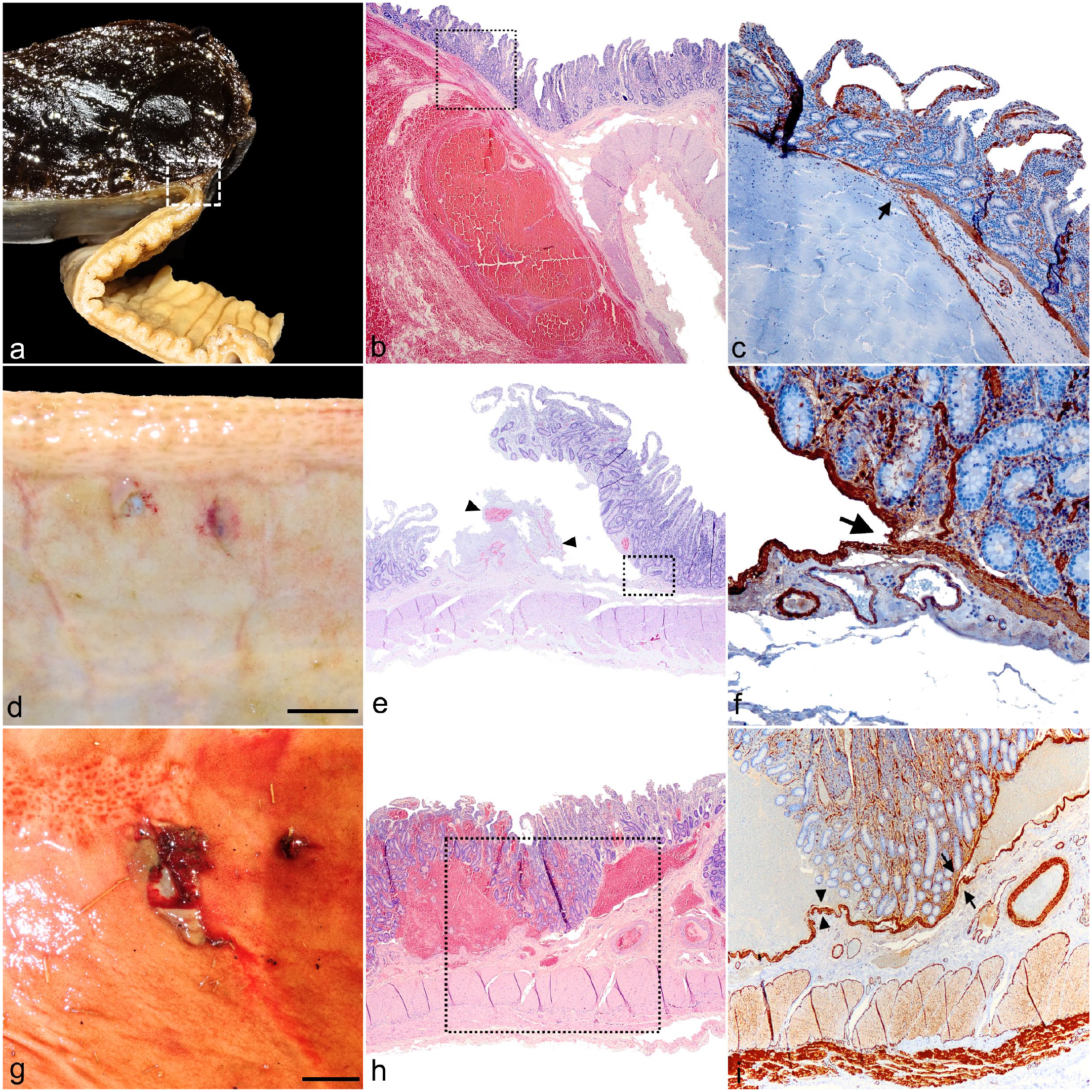
Hemorrhagic bowel syndrome, small intestine, bovine. Intestinal hematoma compared grossly and histologically to the small mucosal (early-stage) lesions. (a-c) Hematoma, at the transition with normal intestine. (a) There is an abrupt transition from normal to elevated mucosa due to intramucosal hematoma formation. Formalin fixed. (b) Section indicated in (a) with a square. Abrupt intramural hemorrhage with splitting and deviation of the mucosa and submucosa/muscularis in almost an 180° angle. Hematoxylin and eosin (HE). (c) Section indicated in (b) with a square. There is abrupt splitting of the lamina muscularis mucosae (LMM) (arrow). Smooth muscle actin immunohistochemistry (SMA IHC). (d-f) Small mucosal (early-stage) lesions. Most lesions show mild to no hemorrhage. (d) Grossly, there are 2 discrete lesions with ulceration and mucosal hemorrhage. Bar, 1cm. (e) Mucosal laceration with detachment and formation of a mucosal flap. Arrowheads show mild hemorrhage and fibrin deposition underneath. HE. (f) Section indicated in (e) with a square. There is abrupt splitting of the LMM (arrow). SMA IHC. (g-i) Small mucosal (early-stage) lesions. Some lesions have a more hemorrhagic appearance, with similar microscopic features as the hematoma. (g) There is a focal erosion with mucosal detachment and hemorrhage. Bar, 1cm. (h) Two expanding hemorrhages within the basal mucosa elevating the propria. HE. (i) Section indicated in (h) with a square. The hemorrhage is above the LMM (arrow heads) as well as (comparable with the hematoma) dissecting within the LMM (arrows). SMA IHC.
In all cases, the intestine was obstructed because of the hematoma. Proximal to the hematoma, the intestine was dilated and contained brown-green liquid admixed with a variable amount of blood and blood clots. Distal to the hematoma, the intestine was contracted and contained mostly no blood, although segmental presence of small blood clots admixed with some liquid blood occurred. The attaching mesentery and associated lymph nodes, at and proximal to the hematoma, were edematous (6/7), and had dilated mesenteric lymphatic vessels. The colon and cecum contained dark tarry mucous admixed with fresh blood. The rumen was filled with fibrous content and the abomasum often contained fluid with undigested silage (4/7). In 4 animals, there was generalized pallor of the skeletal muscles (indicating severe blood loss).
In group 2 animals(n = 11), a single hematoma was located at varying sites in the jejunum in 10 animals, and in the duodenum (pars ascendens) in 1 animal. In 6 cows, the hematoma was identified on the mucosal side, with 4 animals displaying apparent submucosal hemorrhage. The hematoma measured on average 40 cm (SD: 16.58). In most cases, the jejunum contained a large amount of coagulated and noncoagulated blood. The luminal blood clots were weighted in 4 cases, weighing 5.2 to 6.55 kg. In addition, 3 cases displayed generalized muscular pallor (3/11). The rumen was filled with an increased fibrous fraction.
Histopathology
Intramucosal hematomas in all group 1 animals had similar histological characteristics. In all cases, both at the proximal and distal transition of the hematoma with the normal intestinal wall, a dissecting hemorrhage with abrupt splitting of mucosa and submucosa was evident (Fig. 2b). The splitting occurred at the level of the lamina muscularis mucosae (LMM), with the top layer of smooth muscle cells lifting off with the mucosa, and the basal layer still adhered to the submucosa, which was clearly demonstrated with SMA IHC (Fig. 2c). The mucosa overlying the hematoma at the transition zone displayed variable necrosis with shortening of the villi, epithelial sloughing, and dilated lacteals containing red blood cells. The mucosa covering the hematoma (if still present), showed a severe stretched appearance and necrosis. Microcolonies of bacteria with different morphologies were often present on the luminal surface of the degenerated mucosa and blood clot. In all examined sites of the hematoma (central area, transition area, and mesenteric attachment) and the mesentery, diffuse moderate to severe acute inflammation was observed in all intestinal layers. This was characterized by edema, dilated lymphatics, neutrophilic influx, and hypertrophied endothelium. Submucosal blood vessels were considered within normal limits, although very rarely mild hyalinization of a submucosal arteriole was present at heavily inflamed sections. In addition, there was often hemosiderophages and proteinaceous edema, neutrophils, and red blood cells to the adjacent jejunal lymph node.
The small mucosal lesions grossly described above, demonstrated uniform microscopical characteristics. In all lesions, there was a mucosal laceration or erosion which transitions into a mucosal flap detaching from its submucosa (Fig. 2e). This separation of the mucosa from its underlying submucosa occurred in most cases within the LMM with splitting of the 2 layers of smooth muscle cells (34/48) (Fig. 2f, i) and, in fewer cases, in between the propria and LMM (13/48) (Fig. 2i). Only in 3 lesions in 1 animal, the mucosal laceration penetrated the superficial submucosa. Hemorrhage and fibrin formation in between the 2 separating layers was mild to moderate in most lesions (29/48) and rarely absent (7/48). In 11 lesions, there was expansion of the LMM through dissecting hemorrhage between the 2 layers of smooth muscle (Fig. 2h, i). Often there was mixed bacterial proliferation and migration of vegetable material under the detached mucosa. No vascular abnormalities nor significant mucosal inflammation were present in any lesions.
In all animals of group 1, the intestine proximal to the hematoma showed mild to moderate dilated lacteals. Frequently, subepithelial hemosiderophages were present in the villus tips. In animals of group 2 of which extra-intestinal tissues were sampled for histology (n = 8), centrilobular hepatic necrosis was present in 2 cases. No significant lesions were present in other tissues of animals from group 1 and 2.
In group 2, the hematoma was histologically evaluated in 7 animals. In 3 cows, dissecting intramucosal hemorrhage with splitting at the level of the LMM was evident. In the other 4 animals, the splitting of the LMM could not be assessed due to loss of mucosa or postmortem autolysis. In 1 animal, multifocal pigmented fungal hyphae associated with vegetable material within the blood clot of the hematoma were observed. The vasculature of the intestinal submucosa showed no lesions, although rarely mild hyalinisation of a submucosal arteriole was present.
Bacteriological Examination
Bacterial culture was performed in 5 group 1 animals; the other 2 were treated with antimicrobials and were not cultured. Nonselective aerobic and anaerobic culture of the mucosa-associated bacteria at the location of the hematoma yielded abundant growth of Escherichia coli and fewer colonies of Proteus mirabilis, Escherichia fergusoni, Paeniclostridium sordelli, Bacteroides spp, and C perfringens. At the hematoma, no C perfringens was isolated on CHCP culture medium in 2/5 animals; 3/5 animals showed few to abundant growth (Fig. 3). In the 3 cases where C perfringens was isolated on CHCP, 1 to 4 C perfringens isolates were genotyped. All isolates show only the presence of cpa, indicating type A. Blood culture was performed in 4 animals of group 1, and no growth was present in any animal in all 3 culture vials.
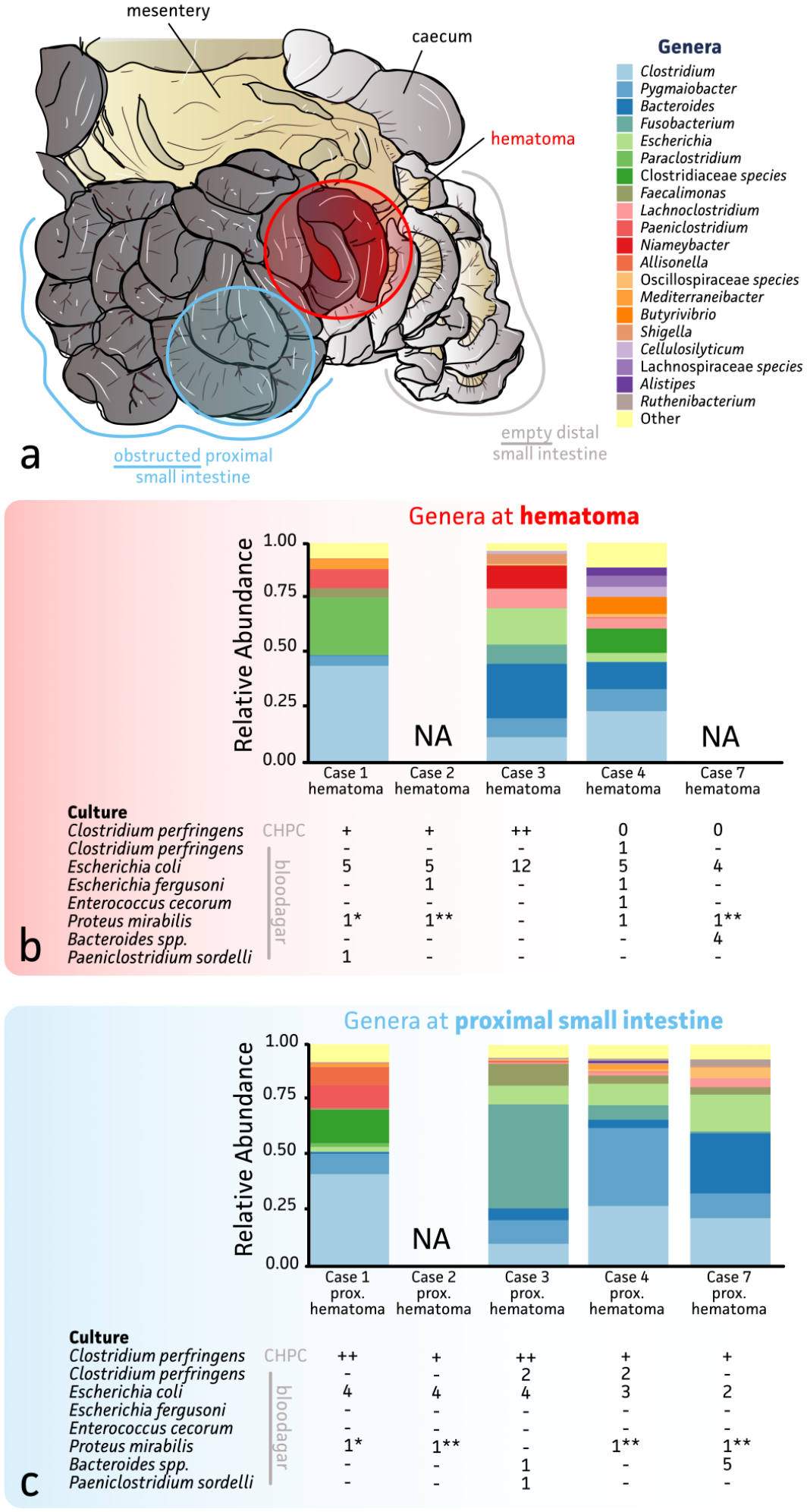
Overview of bacteriology and 16S nanopore sequencing of hemorrhagic bowel syndrome (HBS) cases: (a) illustration of the intestinal tract of a cow with HBS. The location of sampling is indicated. (b) and (c) Overview of the results of full 16S rRNA gene sequencing and bacterial culture of the hematoma (b) and intestine proximal to the hematoma (c). The relative abundance of bacterial genera identified in different samples are demonstrated in the bar chart, only top 20 genera are shown. The amount of growth of C Perfringens at selective culture (CHCP) is estimated (0,+,++), with 0 no growth, + colonies covering 0–33% of the streaked area, ++ colonies covering more than 33% of the streaked area. The bacterial species and the number of colonies identified for each species on nonselective aerobic and anaerobic culture are given. 1*and 1**, anaerobic and aerobic plate, respectively, overgrown by Proteus.; NA, not available.
Mucosal swabs of the intestine proximal and distal to the hematoma demonstrated abundant growth of E coli and smaller numbers of C perfringens, P mirabilis, and Bacteroides spp. on nonselective culture. Intestinal samples distal to the hematoma had far less growth compared to the intestine proximal to the hematoma. No growth on any culture medium was present in 2/5 cases at this site. With CHCP anaerobic culture, the proximal small intestine only few colonies were grown in 3/5 animals, while 2 animals showed abundant C perfringens at this site (Fig. 3). The small intestine distal to the hematoma yielded mild (2/5) or no growth of C perfringens (3/5) at CHCP. In 3 animals, aerobic or anaerobic plate(s) of hematoma and/or proximal intestine were excluded because colonies were overgrown with Proteus preventing purification.
Nanopore Sequencing
A total of 8 intestinal digesta samples (from 4 cows in group 1) were subjected to full-length 16S rRNA gene nanopore sequencing, allowing for the determination of the relative bacterial abundance up to the genus level. While most samples showed sufficient sequencing depth (average 222 519 ± 93 773 reads), samples of case 2 did not result in sufficient sequencing output, which is thought to be due to its low biomass and the presence of blood-related inhibitors in the sample. The main genera identified were Clostridium, Bacteroides, and Escherichia. Pygmaiobacter and Fusobacterium were also present in all samples (Fig. 3).
Discussion
The main aim of this study was to describe the pathology and gain better insights in the pathogenesis and cause of HBS. To achieve this, and in contrast to previous work, our study aimed to closely examine the complete intestine immediately after euthanasia to safeguard intestinal microarchitecture and microbiome.
In all cases, a solitary and intramural hematoma was present in the small intestine, as was described by others.3,22 Grossly, the mucosa covering the hematoma was partially or completely necrotic and lacerated, causing severe intraluminal hemorrhage and clot formation. Histologically, this hematoma was characterized by blood collection between the 2 layers of smooth muscle tissue of the LMM. This was consistently found in all animals where histological determination was possible (10/18). The hemorrhage originates within the mucosa itself and is in fact an intramucosal hematoma with hemorrhaging between the 2 layers of the LMM. These results are in contrast to previous studies that located the hemorrhage in the submucosa 23 or lamina propria. 3 Although, Adaska et al 3 noted the splitting of the LMM. Intestinal intramural hematomas are rarely described in horses24,33,34 and dogs.17,23,27 Suggested cause in the described cases comprised iatrogenic rectal trauma, 33 chronic ulceration, 33 foreign body perforation, 23 and chronic pancreatitis. 17 In contrast to HBS, the hemorrhage was located at the submucosa or tunica muscularis and severe intraluminal hemorrhage was not found in these cases. Comparable with HBS, obstruction was present in these species. In humans, intramural hematomas are mainly a rare complication of anticoagulant therapy or blunt abdominal trauma.1,36 Furthermore, the intramural hemorrhage is also located in the submucosa in humans. 1 In cattle with HBS, there is no indication that coagulopathy is present, as problematic hemostasis during or after surgery is not reported. 25 Moreover, 2 animals in this study showed normal coagulation times (data not shown). Furthermore, abdominal blunt trauma or traumatic lesions have been never related with HBS. To the best of the authors knowledge, no comparable hematoma with hemorrhage within the LMM is reported in human or veterinary literature.
The main novelty of this study is the identification of multifocal small mucosal erosions or lacerations in the small intestine. Comparable with the hematoma, most of these lesions showed a dissection of the 2 layers of LMM, with 11/48 lesions exhibiting expansion of the split LMM by blood. Therefore, we find it is reasonable to conclude that these are early-stage lesions might progress to a hematoma. Furthermore, the presence of these multiple small lesions might explain why postoperative recurrence is common.2,3 Second, this might also explain why the hematoma consisted out of multiple coalescing hematomas in some cases (n = 2), as clustered lesions might develop into a hematoma. It is not clear why these lesions were only located proximal to the hematoma. Since they were present in only 4 animals with the hematoma located at mainly the distal jejunum, leaving only a small portion of intestine distal to it, we cannot exclude these lesions do not occur distally.
Results in our study do not support the hypothesis of C perfringens or A fumigatus as etiological pathogen. Several previous reports associated C perfringens type A (ß2 positive) with HBS, as it can be isolated from the intestine of cows with the disease.8,10,19 Production of major lethal toxins and presence of C perfringens was associated with HBS compared to controls with left displaced abomasum. 11 However, there was no statistical difference regarding the occurrence of C perfringens and its major toxins in the intestinal content of HBS cows, compared to non-HBS bloody content and nonbloody content. 3 Also, inoculation in the intestine or abomasum of C perfringens type A + ß2 could not reproduce the disease. 14 In this study, histology of the early-stage lesions did not show any significant inflammation or necrosis, nor the presence of rod-shaped bacteria at the mucosal surface which could be expected with clostridial infection.32,35 A fumigatus has also been associated with HBS in one study. 15 It was hypothesized the fungus causes an intestinal infection through invasion of the intestinal tissue resulting in the observed hemorrhage in HBS.13,15 The main diagnostic criterion of infection with Aspergillus spp. (aspergillosis) is microscopic demonstration of fungal infiltrative growth in the tissue. 29 In our study, only 1 cow had mild growth of pigmented fungal hyphae within the hematoma associated with alimentary vegetal material in the blood clot, but no tissue invasion. Mycotic blood culture was negative in all animals, indicating local nor hematogenous mycotic infection does not play a role in the pathogenesis. Furthermore, to the best of the author’s knowledge, such a detachment at the LMM, as is present at these early-stage mucosal lesions and hematoma, is not a described feature of any kind of infectious intestinal pathology. Although, inflammation and mucosal necrosis at the hematoma was present, we believe it to be a secondary change due to ischemia and tissue damage created by the hematoma, rather than being a form hemorrhagic enteritis, as was suggested by others.10,19
Nonselective and selective bacterial culture of mucosal swabs yielded almost no to respectively little growth of C perfringens at the hematoma and proximal small intestine. Nonselective culture yielded a polybacterial population at the hematoma and unaffected intestine, which was primarily composed of E coli. Full 16S rRNA gene long-read sequencing of the intestinal contents showed a variable microbiome with a comparable composition regarding the bacterial families previously reported in normal dairy cattle. 20 However, at the lumen Clostridium spp. were found to be abundant, in contrast to the results from the bacterial culture of the mucosa. Interpretation of these results is only possible to a limited extent since no unaffected controls were included. However, it is arguable what an adequate control animal would be, as in animals with HBS, intestinal stasis of ingesta and blood is present for greater than 12 hours, most likely changing the microbiome compared to the initial stage of the disease. In addition, the mucosal to luminal microbiome were compared, which is known to be different. 37 Although based on our examinations, we were not able to identify a clear cause for the mucosal early-stage lesions, our results might indicate that HBS is not caused by a bacterial infection. In fact, because of the lack of inflammation or degenerative changes and consistent presence of mucosal laceration at these lesions, it cannot be excluded that trauma to the mucosa plays a role.
Adaska et al 3 suggested an abnormal lymphatic or vascular function might play a primary role in the pathogenesis. This was based on the presence of dilated villus lacteals with proteinaceous edema or blood and submucosal vasculopathy. In our study, dilation of the villus lacteals with edema was also frequently observed at the hematoma and the proximal intestine. In addition, the jejunal lymph nodes of the preobstructed intestine and at the hematoma were swollen due to edema with frequent grossly dilated lymphatic vessels in the mesentery. This was considered a secondary phenomenon due to the intestinal obstruction. This obstruction causes a severe weight increase due to ingesta accumulation, which causes tension to the mesentery. Most likely, this tension hampers the venous and lymphatic return. Both Adaska et al 3 and Owaki et al 22 suggested the observed vasculitis of the submucosal blood vessels to be a primary lesion. However, in our study, very rarely vascular hyalinization was observed in the hematoma, and if present, it was considered secondary to the inflammatory response rather than a primary vasculopathy.
We hypothesize that the formation of a hematoma occurs through propagation of a dissecting hemorrhage within the LMM. At the LMM, arterioles are perforating from the submucosa to the propria where they branch into subepithelial capillary beds. 5 Detachment in between the 2 layers of LMM, would induce rupture of these penetrating arterioles, causing hemorrhage. Propelled by the arteriolar blood pressure, blood might find its way in direction of the least resistance, in between the 2 still attached layers of smooth muscle of the LMM at the edges, causing further splitting of the LMM. In this way, a vicious circle of splitting of the LMM and arteriolar rupture, would generate propagation of this undermining hematoma. Further research is required to confirm this hypothesis.
This study is subject to several limitations. First, there are only a limited number of cows in group 1. This was a consequence of the nonspecific clinical signs and rapid deterioration, as well as the sporadic occurrence of HBS in Flanders. Second, bacteriological cultivation and sequencing was performed in only 5 animals because 2 animals in group 1 were treated with antimicrobials. Third, the small mucosal lesions described in 4 animals of group 1 were seen consistently since they were first noticed. It might be possible lesions were present in the other 3 animals of group 1, but were not recognized since they can be easily overlooked or dismissed as artifact. Fourth, no negative controls were used. Finally, although samples were freshly collected after euthanasia, bacterial overgrowth might already be present in the living animal, since there is stasis of intestinal content and blood over a period of most likely 12 hours or more. As a consequence, a possible etiologic bacterium might be overgrown by other bacteria or lost at the moment of sampling.
In conclusion, this study shows that HBS is characterized by an intramucosal dissecting hematoma. In several animals, there is co-occurrence with multifocal mucosal lesions at the small intestine, which share very similar histological characteristics with the main hematoma. This strongly suggests these lesions are early-stage lesions of the hematoma. Although, this study reveals possible early-stage lesions of HBS, the cause or mechanism of injury remains unknown. The findings of this study do not support any role of C perfringens or fungi in the pathogenesis of HBS.
Footnotes
Acknowledgements
The authors thank the technical staff of the involved laboratories and the collaborating farmers and veterinarians.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by Veepeiler (Animal Health Service-Flanders; project: 562).
