Abstract
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) causes respiratory disease in mink similar to human COVID-19. We characterized the pathological findings in 72 mink from US farms with SARS-CoV-2 outbreaks, localized SARS-CoV-2 and its host cellular receptor angiotensin-converting enzyme 2 (ACE2) in mink respiratory tissues, and evaluated the utility of various test methods and specimens for SARS-CoV-2 detection in necropsy tissues. Of SARS-CoV-2-positive animals found dead, 74% had bronchiolitis and diffuse alveolar damage (DAD). Of euthanized SARS-CoV-2-positive animals, 72% had only mild interstitial pneumonia or minimal nonspecific lung changes (congestion, edema, macrophages); similar findings were seen in SARS-CoV-2-negative animals. Suppurative rhinitis, lymphocytic perivascular inflammation in the lungs, and lymphocytic infiltrates in other tissues were common in both SARS-CoV-2-positive and SARS-CoV-2-negative animals. In formalin-fixed paraffin-embedded (FFPE) upper respiratory tract (URT) specimens, conventional reverse transcription–polymerase chain reaction (cRT-PCR) was more sensitive than in situ hybridization (ISH) or immunohistochemistry (IHC) for detection of SARS-CoV-2. FFPE lung specimens yielded less detection of virus than FFPE URT specimens by all test methods. By IHC and ISH, virus localized extensively to epithelial cells in the nasal turbinates, and prominently within intact epithelium; olfactory mucosa was mostly spared. The SARS-CoV-2 receptor ACE2 was extensively detected by IHC within turbinate epithelium, with decreased detection in lower respiratory tract epithelium and alveolar macrophages. This study expands on the knowledge of the pathology and pathogenesis of natural SARS-CoV-2 infection in mink and supports their further investigation as a potential animal model of SARS-CoV-2 infection in humans.
Keywords
The COVID-19 pandemic has resulted in more than 250 million severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infections and 5 million deaths in humans worldwide, as of 25 November 2021. 72 Sporadic natural infections have also been documented in various animals, including cats, dogs, ferrets, mink, zoo felids, zoo gorillas, and otters, from 30 countries around the world; additional species have shown experimental susceptibility to infection.23,26,27,42,50,61,68 While most animal cases are the result of human-to-animal transmission, only mink have shown both human-to-animal and animal-to-human transmission confirmed by sequencing;35,49,52 spread to other animals on and around mink farms has also been documented. 71 This has raised concern for mink’s potential as a SARS-CoV-2 reservoir and source of viral mutations that may thwart the development of medical countermeasures in the global battle against COVID-19.34,49
Mink farming for pelt production is concentrated in Europe, with more than 27 million pelts produced annually from more than 2000 farms. 20 Between 23 April 2020 and 9 August 2021, SARS-CoV-2 has been detected on 435 mink farms in 12 countries, with devastating effects on portions of the industry, including forced depopulation of the entire farmed mink population (approximately 17 million animals) in Denmark—previously the world’s largest producer of pelts—after detection of a mink-derived SARS-CoV-2 variant in 12 people working on mink farms or living in the surrounding community.35,46 Mink farming in the United States is on a much smaller scale, with smaller individual farms than in Europe, and occurs predominantly in states of the Midwest and West. Between 17 August 2020 and 29 July 2021, 17 US mink farms have confirmed SARS-CoV-2 infections. 69 In the United States, respiratory disease and increased mortality have been seen among mink on most affected farms, and all farms have had associated human cases.12,14,51,70
Although mink are the species with the second-highest number of documented individual SARS-CoV-2 infections, the only nonhuman species for which SARS-CoV-2 infection is consistently associated with significant morbidity and mortality, and the only animal species associated with confirmed zoonotic transmission to humans, descriptions of SARS-CoV-2-associated pathological findings in mink are limited.21,48,52,65 Furthermore, understanding the pathology and pathogenesis of SARS-CoV-2 infection in mink may provide insights into COVID-19 in humans. As with any novel infectious disease, effective animal models are crucial for preventive and therapeutic development efforts. Both mink and ferrets—closely related mustelids which are already commonly used as models of other human respiratory viral infections—have shown susceptibility to experimental SARS-CoV-2 infection, with development of some features similar to human disease in small numbers of animals reported.4,59,64,65 Viral replication in lung, the primary target of SARS-CoV-2 in humans, appears more efficient in mink than in ferrets, and mink may prove valuable animal models for this disease. 65 To this end, we sought to characterize the pathology and localization of SARS-CoV-2 and its host entry receptor angiotensin-converting enzyme (ACE2) in tissues from a large cohort of naturally infected US-farmed mink and to further assess whether disease in mink may recapitulate features of human disease to support the use of this species as a model for COVID-19 research. Furthermore, we compared the utility of various test methods and specimen types for SARS-CoV-2 detection in mink necropsy tissues, which can help improve SARS-CoV-2 mortality surveillance efforts on mink farms.
Materials and Methods
Specimen Collection
All animal work was conducted under the approval of the Centers for Disease Control and Prevention’s Institutional Animal Care and Use Committee’s approval (protocol 3104BARMULX) and with the permission of the US Fish and Wildlife Service, the Utah Division of Wildlife Resources, and the Utah Department of Agriculture and Food. All animals were humanely handled and sampled in strict accordance with the Guide for the Care and Use of Laboratory Animals (Committee for the Update of the Guide for the Care and Use of Laboratory Animals, National Research Council of the National Academies 2011).
Beginning with the first confirmation of a US mink infection on 17 August 2020, the Centers for Disease Control and Prevention (CDC) COVID-19 One Health Working Group deployed multiple field teams during August to December 2020, at the request of the state, to investigate outbreaks of natural SARS-CoV-2 infection in humans and multiple animal species on mink farms in Utah. Necropsy specimens from 72 mink collected during one field team deployment in October 2020 were included in this study. Following necropsy, lung, heart, trachea, liver, kidney, spleen, and intact heads were placed in 10% neutral buffered formalin and transferred to CDC’s Infectious Diseases Pathology Branch for pathological evaluation and SARS-CoV-2 testing. Brain and coronal sections of the rostral nasal tissues and nasal turbinate specimens were subsequently collected from the fixed intact heads. Extraction of nasal turbinates was performed by using rongeurs to open the nasal bone and nasal process of the frontal bone and remove the conchae. Fresh lung and colorectal tissue specimens were also collected and frozen at the time of necropsy and sent to the US Geological Survey National Wildlife Health Center, Madison, WI, for SARS-CoV-2 real-time reverse transcription PCR (RT-rtPCR) assay targeting the N1 and N2 genes.
Histopathology and Immunohistochemistry (IHC)
Formalin-fixed tissues were routinely processed, paraffin embedded, sectioned at 4 µm, and stained with hematoxylin-eosin for histopathologic evaluation. IHC was performed for SARS-CoV-2 antigen detection on upper respiratory and lung tissues for all cases, and on extra-respiratory tissues to evaluate for systemic infection in select cases that showed extensive labeling in the respiratory tract and/or potentially virus-associated pathology in other tissues. Gram stain, Ziehl-Neelsen stain, and/or IHC for bacterial agents were performed in cases with histopathologic findings suggestive of a bacterial etiology. Trichrome stain was performed to highlight fibroblast proliferation/fibrosis in lungs.
Immunohistochemical assays were performed using an immunoalkaline phosphatase detection system and Mach 4 Universal AP Polymer Kit (Biocare Medical, Pacheco, CA) with Permanent Red Chromogen (Cell Marque; MilliporeSigma, St. Louis, MO). Primary antibodies and pretreatments used are listed in Supplemental Table S1. Appropriate positive and negative controls were run in parallel. Double stains were performed using the DoubleStain IHC Kit M&R on human tissue (DAB and AP/Red) (Abcam, Cambridge, MA) as described with modifications listed here. Slides were blocked with BLOXALL Endogenous Blocking Solution, Peroxidase and Alkaline Phosphatase (Vector Laboratories, Burlingame, CA) for 10 minutes and subjected to heat-induced epitope retrieval (HIER) using citrate-based buffer (Biocare Medical, Pacheco, CA). Slides were incubated with goat-anti-olfactory marker protein (OMP) or goat-anti-ACE2 antibodies for 30 minutes, followed by a mouse anti-SARS-CoV-2 spike protein antibody (Genetex, Irvine, CA) mixed with a rabbit anti-goat IgG antibody (catalog ab6697; Abcam) for 30 minutes and Mouse HRP/Rabbit AP polymer or Rabbit HRP/Mouse AP polymer (from Triple Stain IHC Kit: M&M& R on Human tissue [Abcam, Cambridge, MA]), mixed together for 30 minutes. DAB (Abcam, Cambridge, MA) and Permanent Red (CellMarque; MilliporeSigma, St Louis, MO) were applied sequentially for 15 minutes each. Slides were counterstained in Mayers Modified Hematoxylin, blued in Lithium blue and mounted with aqueous mounting media (Polysciences, Warrington, PA).
RT-PCR and in Situ Hybridization (ISH)
RNA was extracted from formalin-fixed paraffin-embedded (FFPE) tissue sections and evaluated by conventional RT-PCR (cRT-PCR) for SARS-CoV-2 with primers targeting the nucleocapsid gene of SARS-CoV-2, using the OneStep RT-PCR kit (Qiagen, Valencia, California) and 5 μL of RNA template, as previously described; Sanger sequencing of N-gene PCR amplicons was performed for confirmation. 6 RT-rtPCR for detection of influenza A and B viruses was performed using previously published primers and probes. 15 Quality of RNA was ensured by RT-PCR amplification of reference genes (18S rRNA or β2-microglobulin genes).6,15,63 Upper respiratory tissues (rostral nasal tissues, turbinates, and trachea) were combined for RNA extraction and tested as one specimen (upper respiratory tract [URT]) for RT-PCR assays.
To directly localize SARS-CoV-2 genomic RNA in the FFPE tissues, an ISH assay was performed on all cRT-PCR-positive tissues using RNAscope probes (Advanced Cell Diagnostics, Newark, California) targeting the N-gene of SARS-CoV-2, as previously described. 6
Transmission Electron Microscopy (TEM)
Ultrastructural examination was performed on tissues from 4 animals using 2-mm punch samples removed from FFPE blocks or 4-µm-thick FFPE upper airway and lung tissue sections affixed to glass slides (“on-slide”). 41 Areas of interest for TEM were selected based on corresponding areas that showed positive SARS-CoV-2 immunolabeling. FFPE specimens were deparaffinized in xylene and rehydrated before being embedded in epoxy resin. On-slide sections were removed from slides by immersing the sections in boiling water and removing sections from slides with a razor blade. Areas of interest were cut out and glued onto a blank TEM block prior to being thin sectioned. TEM sections were stained with uranyl acetate and lead citrate and examined on a Tecnai BioTwin electron microscope.
Interpretation of Positive Cases
An animal was classified as “SARS-CoV-2-positive” when SARS-CoV-2 was detected by at least one test method used in this study, including RT-rtPCR on fresh lung or colorectal specimens or IHC, ISH, or cRT-PCR on FFPE upper respiratory tract (rostral nasal tissues, turbinates, and trachea) or lung tissues. An animal was classified as “SARS-CoV-2-negative” if there was no virus detection by any method in this study. Additional epidemiological and laboratory testing data regarding the human and animal SARS-CoV-2 investigations on the Utah mink farms will be published in separate reports.
Results
Clinical Data and Gross Findings
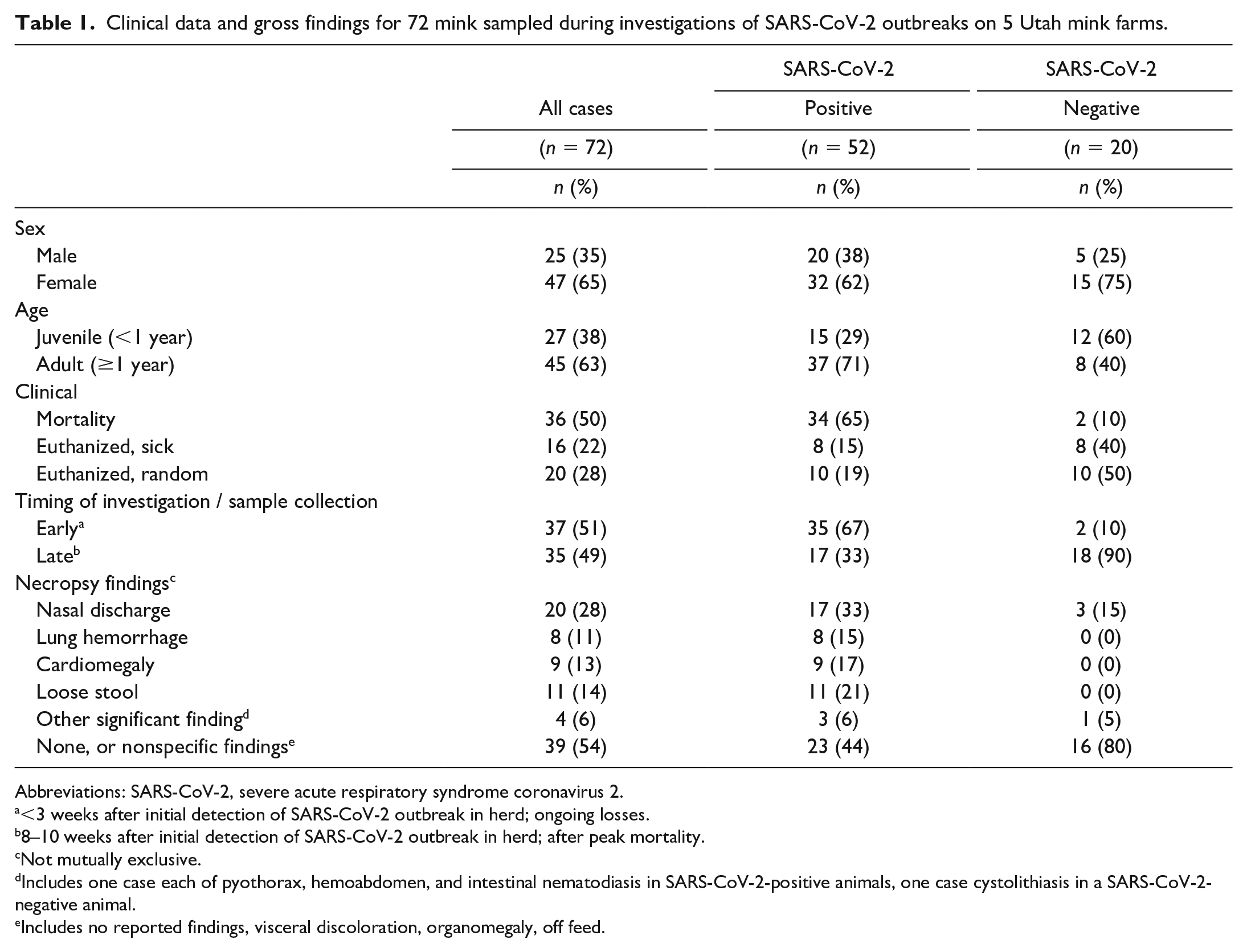
The study evaluated 72 mink from 5 farms in Utah. Available clinical data and gross necropsy findings reported by the field team are summarized in Table 1. There were 47 (65%) females, and 45 (63%) mink were adults (older than 1 year). There were 36 (50%) animals collected for necropsy after being found dead (mortalities) in the mornings during the field investigation, while 16 (22%) were euthanized at the time of necropsy due to respiratory or unspecified illness (euthanized, sick) and 20 (28%) were randomly selected for euthanasia regardless of signs of illness (euthanized, random). Thirty-seven (51%) animals from 3 farms were sampled within 3 weeks following initial detection of the SARS-CoV-2 outbreak in the herds, which were experiencing ongoing mink losses, whereas 35 (49%) animals were from 2 farms that were sampled 8 to 10 weeks following the initial detection of the SARS-CoV-2 outbreaks in the herds and for which the increase in mink losses had subsided at the time of the field investigation.
Clinical data and gross findings for 72 mink sampled during investigations of SARS-CoV-2 outbreaks on 5 Utah mink farms.
Abbreviations: SARS-CoV-2, severe acute respiratory syndrome coronavirus 2.
<3 weeks after initial detection of SARS-CoV-2 outbreak in herd; ongoing losses.
8–10 weeks after initial detection of SARS-CoV-2 outbreak in herd; after peak mortality.
Not mutually exclusive.
Includes one case each of pyothorax, hemoabdomen, and intestinal nematodiasis in SARS-CoV-2-positive animals, one case cystolithiasis in a SARS-CoV-2-negative animal.
Includes no reported findings, visceral discoloration, organomegaly, off feed.
At necropsy, nasal discharge was noted in 20/72 (28%) animals and was the most common gross finding in both SARS-CoV-2-positive and SARS-CoV-2-negative mink. Loose stool, cardiomegaly, and pulmonary hemorrhage were noted in 11 (14%), 9 (13%), and 8 (11%) animals, respectively; all occurred in animals with SARS-CoV-2 infection confirmed in this study. Other sporadic, significant gross findings included one case each of pyothorax, hemoabdomen, and intestinal nematodiasis in animals with SARS-CoV-2 detected, and one case of cystolithiasis in an animal without SARS-CoV-2 detection. In 39/72 (54%) animals, no or mild nonspecific gross findings were reported at necropsy.
Histopathologic Findings in SARS-CoV-2-Infected Mink
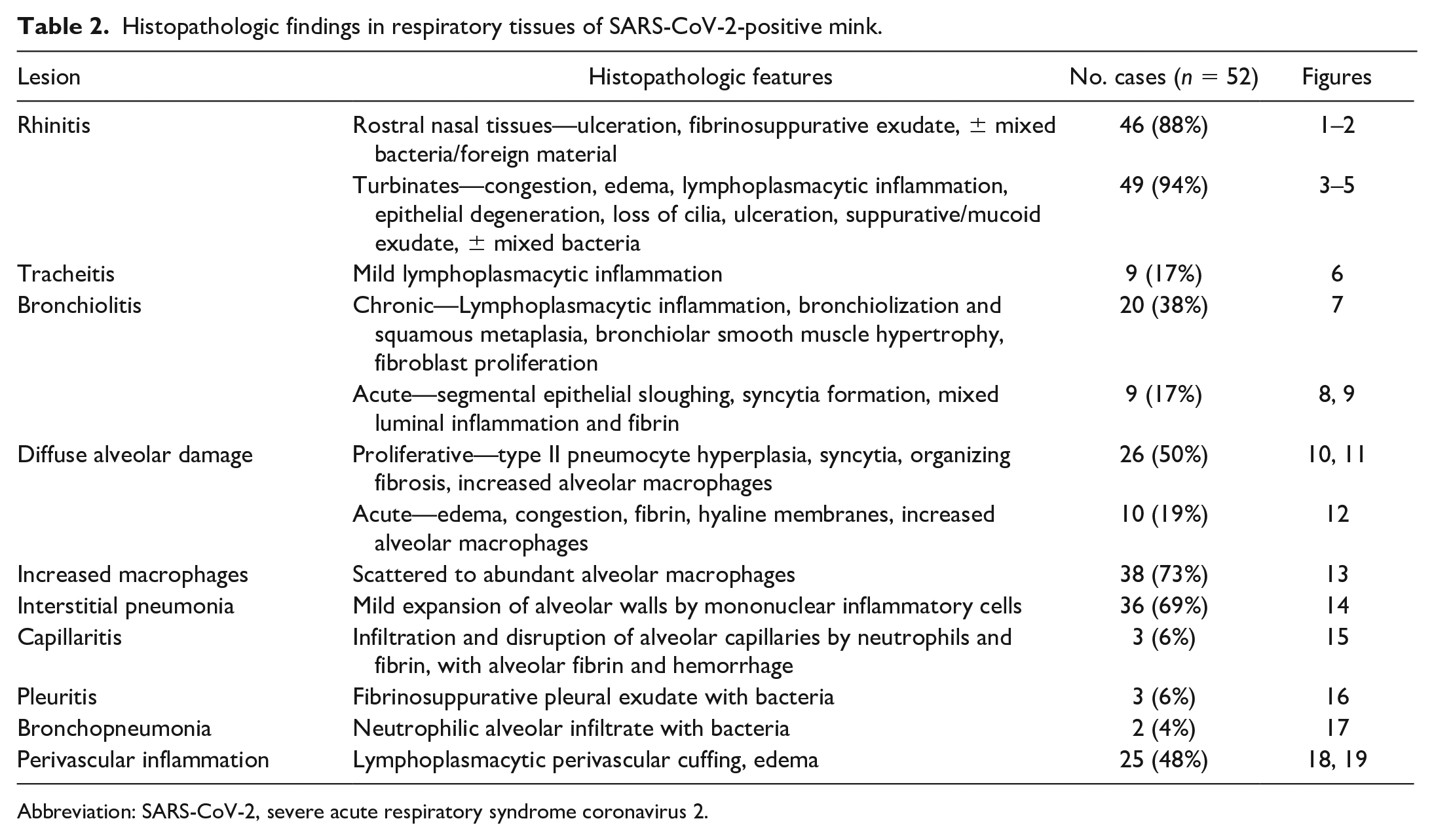
Of 72 mink, SARS-CoV-2 was detected in 52 (72%) by at least one test method (referred to as “SARS-CoV-2-positive”). Histopathologic findings are summarized in Table 2. Findings were most consistently present in nasal tissues, regardless of clinical status. Rhinitis was the most common finding and was seen in 49/52 (94%) SARS-CoV-2-positive animals (Figs. 1, 2). Rostral nasal cavity sections had ulceration of the squamous mucosa, with acute or subacute inflammation and variably dense luminal fibrin and neutrophils (exudate), often accompanied by mixed bacteria (gram-positive cocci and gram-negative bacilli and occasionally filamentous rods) and sometimes plant material. Turbinates had submucosal congestion, edema, and/or predominantly lymphoplasmacytic inflammation, with variable and often subtle epithelial degeneration; overt turbinate epithelial necrosis was present in 6 cases (Figs. 3–5). Luminal suppurative and mucoid exudates were consistently present, often abundant, and sometimes accompanied by mixed bacteria. Tracheitis was uncommon (17%) and, when present, was typically characterized by only mild lymphoplasmacytic submucosal infiltrates (Fig. 6).
Histopathologic findings in respiratory tissues of SARS-CoV-2-positive mink.
Abbreviation: SARS-CoV-2, severe acute respiratory syndrome coronavirus 2.
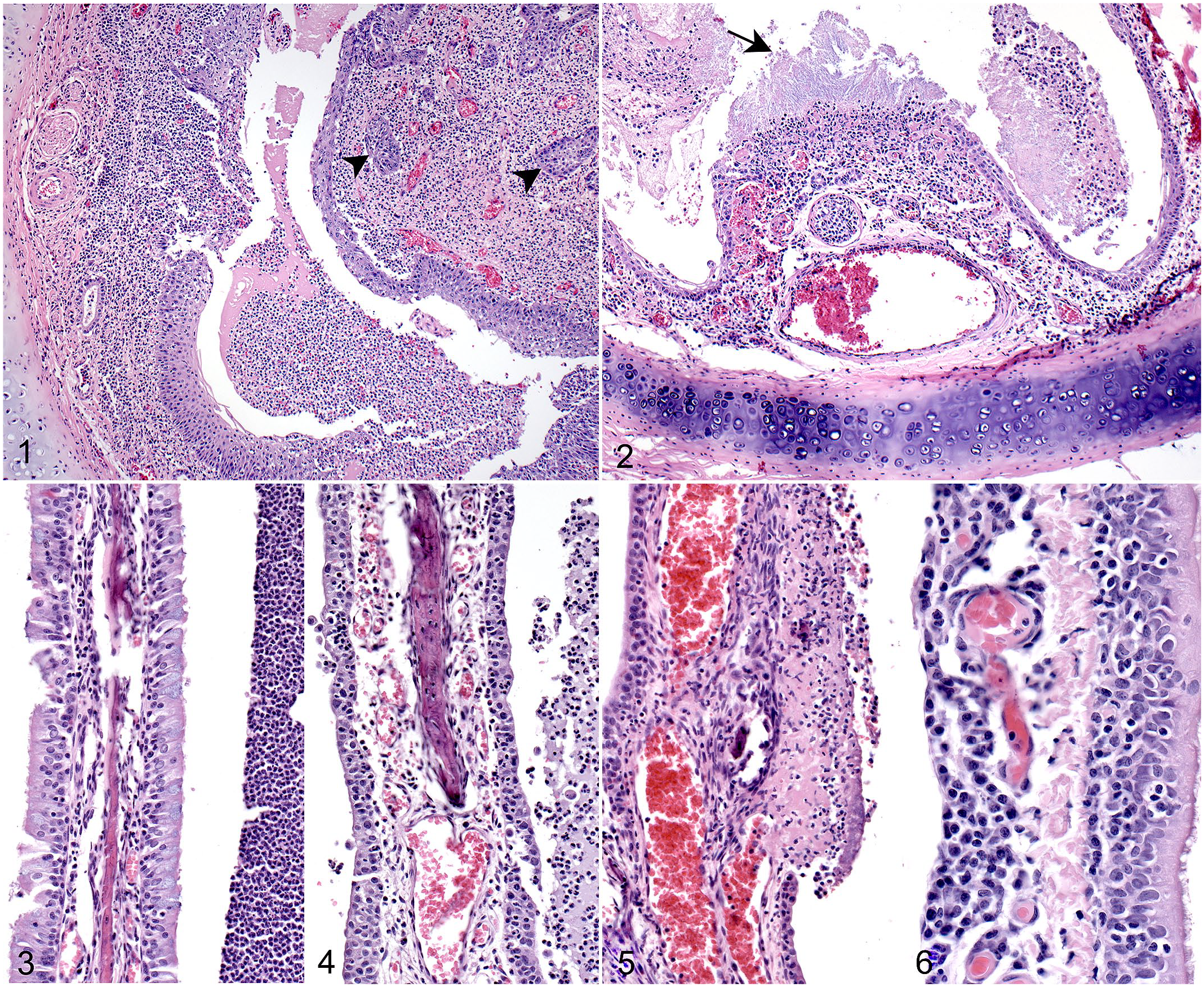
Natural SARS-CoV-2 infection, upper respiratory tract, mink.
Histopathologic findings in the lungs differed between animals that were found dead (mortalities) and those that were euthanized. Of 34 mortalities, 19 (56%) had multifocal to extensive, proliferative, and/or necrotizing and inflammatory bronchiolar lesions. Proliferative lesions were more common and included lymphoplasmacytic peribronchiolar infiltrates and bronchiolar epithelial hyperplasia with bronchiolization of surrounding alveoli, characterized by extension of cuboidal epithelium beyond the bronchiole (peribronchiolar metaplasia; Fig. 7). Squamous metaplasia, bronchiolar smooth muscle hypertrophy, and fibroblast proliferation commonly accompanied bronchiolization. More acute lesions were characterized by focal or segmental bronchiolar epithelial necrosis, with epithelial attenuation or sloughing, syncytia formation, and luminal fibrin with mixed inflammatory cells (Figs. 8, 9).
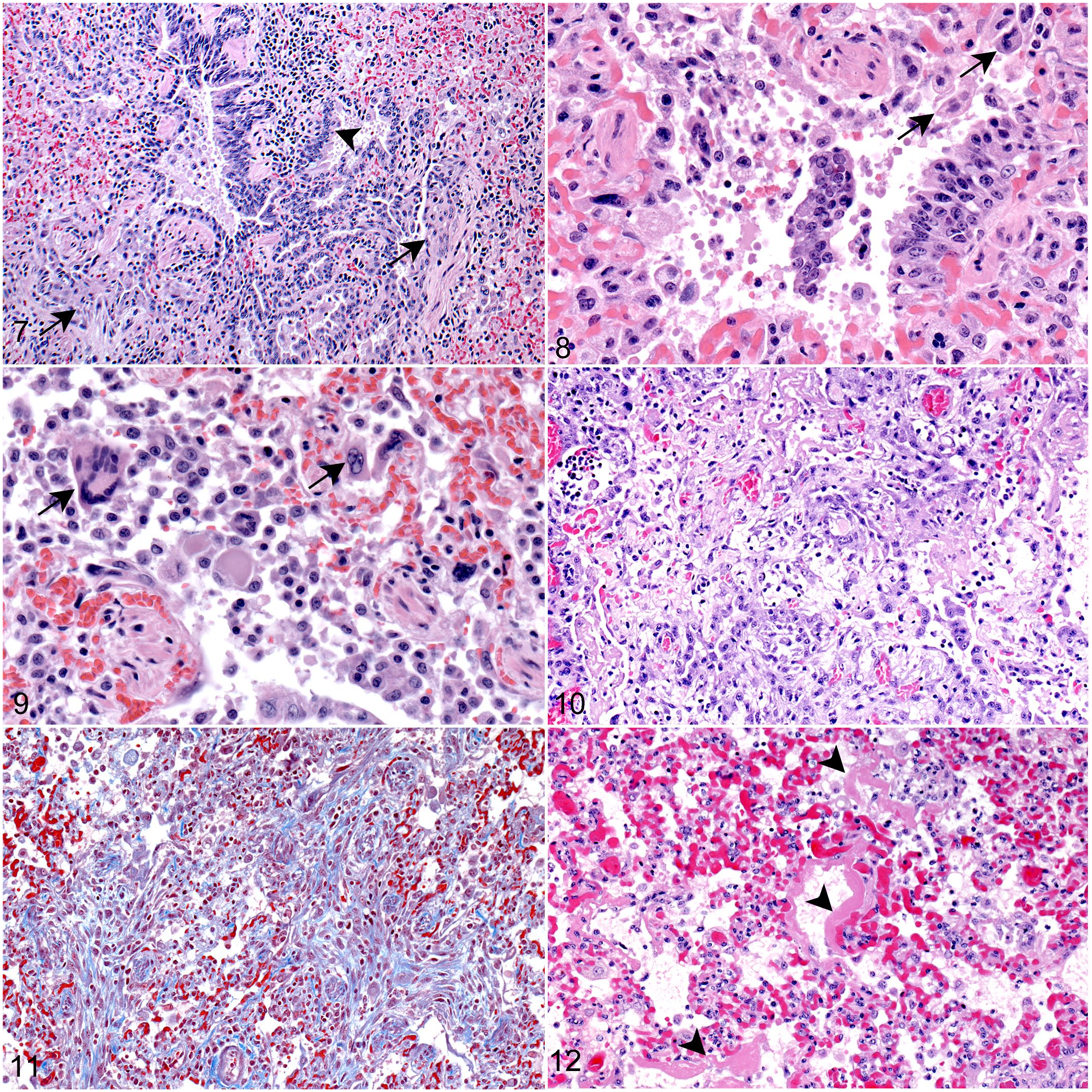
Natural SARS-CoV-2 infection, lung, mink.
The proliferative/organizing phase of diffuse alveolar damage (DAD) was present in 21/34 (62%) mortalities, including 15 with bronchiolar lesions. DAD was patchy, and the extent varied both within and among the different lung sections examined for each animal; some lung sections had no or focal DAD while other sections from the same animal had multifocal or diffuse changes. Features included type II pneumocyte hyperplasia and reactive hypertrophy with scattered syncytia; viral inclusions were not seen (Fig. 10). Foci of fibroblast proliferation accompanied DAD in 6 cases (Fig. 11). Of 21 animals with proliferative DAD, 10 also had similarly patchy evidence of acute alveolar injury, with congestion, edema, and alveolar fibrin, in addition to desquamation of pneumocytes and the presence of hyaline membranes (Fig. 12). Increased, and sometimes abundant, alveolar macrophages and scattered mixed alveolar inflammatory infiltrates were also common (Fig. 13). Twelve mortalities with DAD also had interstitial pneumonia with mild thickening of alveolar walls by mononuclear or mixed inflammatory cells; one had neutrophilic interstitial inflammation with disruption of alveolar walls and hemorrhage (capillaritis). One mortality with DAD also had suppurative pleuritis with Escherichia coli and Pasteurella detected by IHC, and one each had neutrophilic bronchopneumonia or features of sepsis, with no bacterial cause identified.
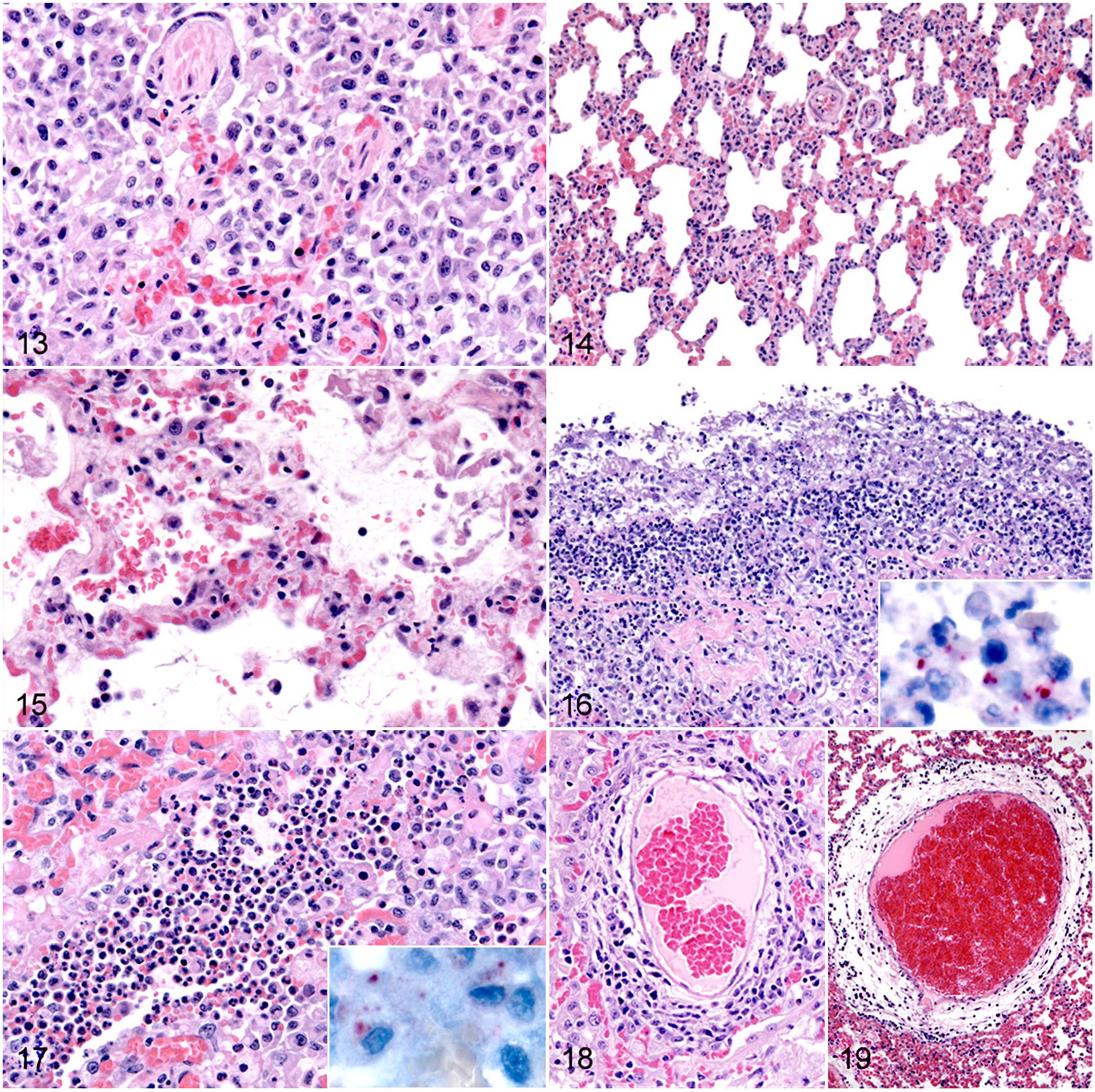
Natural SARS-CoV-2 infection, lung, mink.
The examined lung sections had no histological lesions of DAD in 13/34 (38%) mortalities. Of these, 10 had mild interstitial pneumonia, 2 of which had a neutrophilic component and evidence of capillaritis (Figs. 14, 15). Two animals had suppurative pleuritis with mixed bacteria identified (one also with sepsis), and one had bacterial bronchopneumonia (Figs. 16, 17). Of 34 mortalities, 25 (74%) had perivascular cuffing by variable and often dense, lymphocytic infiltrates, frequently with perivascular edema (Figs. 18, 19); 17 were cases with DAD, and 8 had no DAD. Perivascular inflammation involved small, medium, and large caliber vessels and both arteries and veins, with small to medium-sized veins most notably affected; thrombi were not observed.
Of 52 SARS-CoV-2-positive animals, 18 (35%) had been euthanized (8 with signs of illness and 10 by random selection). Rhinitis similar to that seen in mortalities was consistently present in both euthanized groups. Findings in the lung of euthanized animals included congestion, edema, and increased number of alveolar macrophages. Of 18 euthanized animals, 14 (78%; 9 randomly selected and 5 sick) also had interstitial pneumonia and perivascular lymphocytic inflammation and edema. One was an animal that was agonal prior to euthanasia and also had bronchiolization, organizing DAD with squamous metaplasia, and fibroblast foci. Four other euthanized animals had evidence of focal type II pneumocyte hyperplasia; DAD or bronchiolar changes were otherwise not seen in euthanized animals that were positive for SARS-CoV-2.
Extrapulmonary findings in SARS-CoV-2-positive animals included hepatic lipidosis in 25/34 (74%) mortalities, 4/8 (50%) sick euthanized animals, and 1/10 (10%) randomly euthanized random animals; lymphoplasmacytic infiltrates in the kidneys in 6/34 (18%) mortalities, 4/8 (50%) sick euthanized animals, and 3/10 (30%) randomly euthanized animals; and mild mononuclear inflammation in the heart in 7/34 (21%) mortalities, and one each in sick euthanized and randomly euthanized animals.
SARS-CoV-2 Detection and Localization in Upper Respiratory Tract and Lung
Table 3 summarizes the detection of SARS-CoV-2 in various specimen types by each of the test methods used, according to age and clinical status of the animals. RT-rtPCR on fresh lung and colorectal tissues yielded more SARS-CoV-2-positive specimens in mortalities than in euthanized animals, with detection in 25/32 (78%) adult mortalities and 2/2 (100%) juvenile mortalities on at least one of the 2 fresh specimen types. For one adult, only the colorectal specimen was positive, while both juvenile mortalities had detection only in the colorectal specimen. Also notably, fresh colorectal RT-rtPCR was the only assay by which SARS-CoV-2 was detected in juvenile mortalities. RT-rtPCR performed on fresh lung and colorectal tissues was less sensitive for SARS-CoV-2 detection in euthanized animals than in mortalities.
SARS-CoV-2 detection in various necropsy specimen types from US-farmed mink.
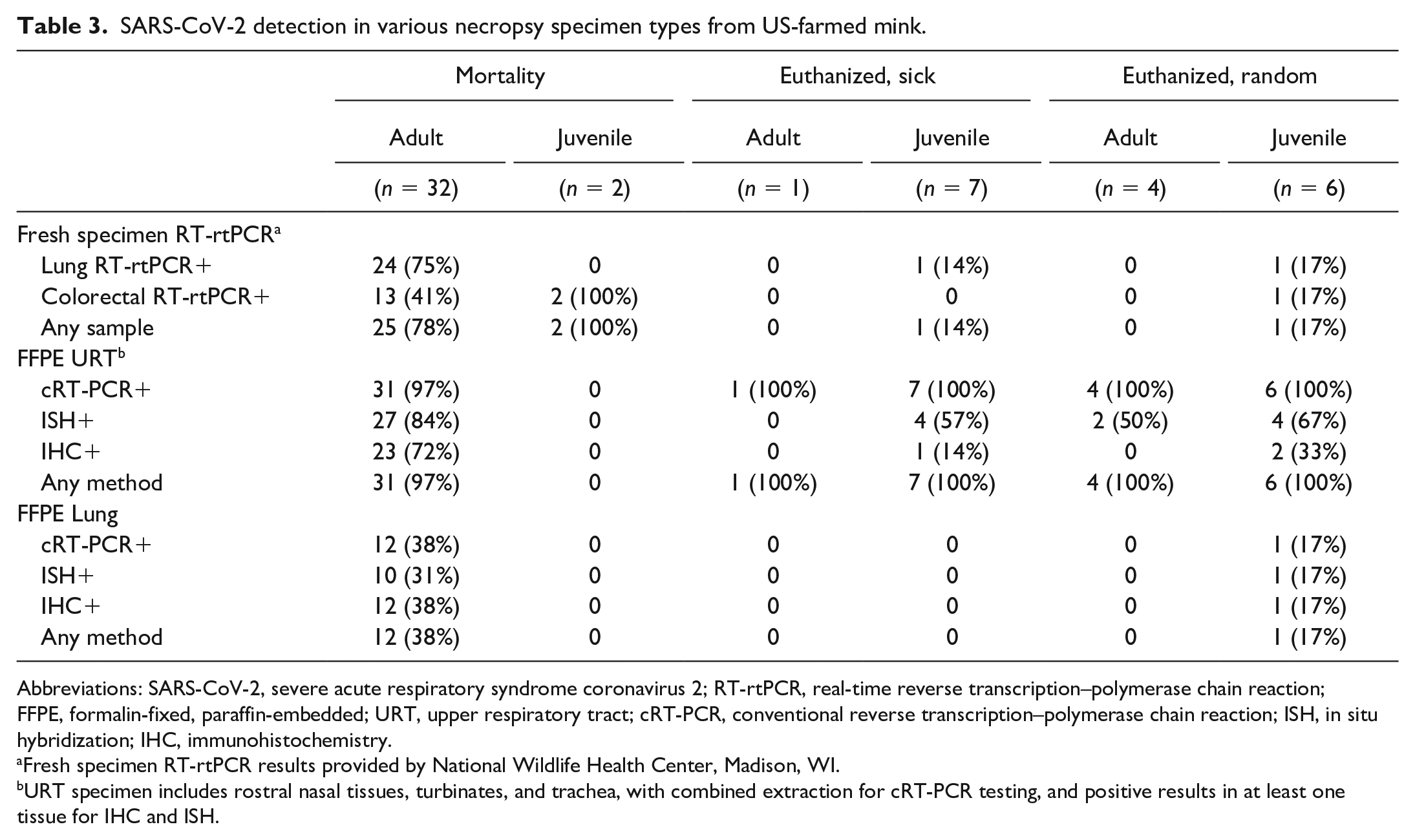
Abbreviations: SARS-CoV-2, severe acute respiratory syndrome coronavirus 2; RT-rtPCR, real-time reverse transcription–polymerase chain reaction; FFPE, formalin-fixed, paraffin-embedded; URT, upper respiratory tract; cRT-PCR, conventional reverse transcription–polymerase chain reaction; ISH, in situ hybridization; IHC, immunohistochemistry.
Fresh specimen RT-rtPCR results provided by National Wildlife Health Center, Madison, WI.
URT specimen includes rostral nasal tissues, turbinates, and trachea, with combined extraction for cRT-PCR testing, and positive results in at least one tissue for IHC and ISH.
For FFPE specimens, SARS-CoV-2 was detected most frequently for all animal groups by cRT-PCR testing of URT specimens (rostral nasal tissues turbinates, trachea; combined). Thirty-one of 32 (97%) adult mortalities, and all SARS-CoV-2-positive euthanized animals, had virus detection by FFPE URT cRT-PCR. ISH and IHC were less sensitive than cRT-PCR for virus detection in FFPE URT across all groups. SARS-CoV-2 was detected less frequently in FFPE lung specimens than URT specimens for all groups. Only 12/32 (38%) adult mortalities had virus detection by at least one test method from FFPE lung, and among all other groups, only one randomly euthanized juvenile had virus detection in FFPE lung. Unlike for FFPE URT specimens, virus was detected with the same frequency by cRT-PCR and IHC performed on FFPE lung specimens from adult mortalities.
SARS-CoV-2 distribution and cellular localization within respiratory tissues was evaluated by IHC, ISH, and TEM. IHC and ISH labeling of viral antigens and RNA, respectively, consistently had a very patchy distribution in both URT and lung, with the most overall labeling in nasal turbinates. In the rostral nasal tissues, viral antigens localized primarily within desquamated epithelial cells and associated exudate; rare labeling of intact, superficial squamous cells was seen (Fig. 20). In turbinates, antigen labeling was also found in degenerate and sloughed epithelium and luminal exudate; however, extensive labeling of intact turbinate respiratory epithelium was also commonly present (Fig. 21). IHC labeling of tracheal ciliated epithelium and glands was rare. In lung, occasional labeling of bronchiolar epithelium was seen, but labeling was primarily within scattered intra-alveolar cells interpreted as macrophages and pneumocytes (Figs. 22, 23); there was also rare labeling of extracellular fibrin or hyaline membrane material in cases with DAD. ISH labeling was seen in the same overall locations and cell types as IHC labeling (Figs. 24, 25). IHC and ISH were also performed in extra-respiratory tissues (liver, heart, kidney, spleen, and brain) for 6 animals; all extra-respiratory tissues were negative by IHC and ISH.
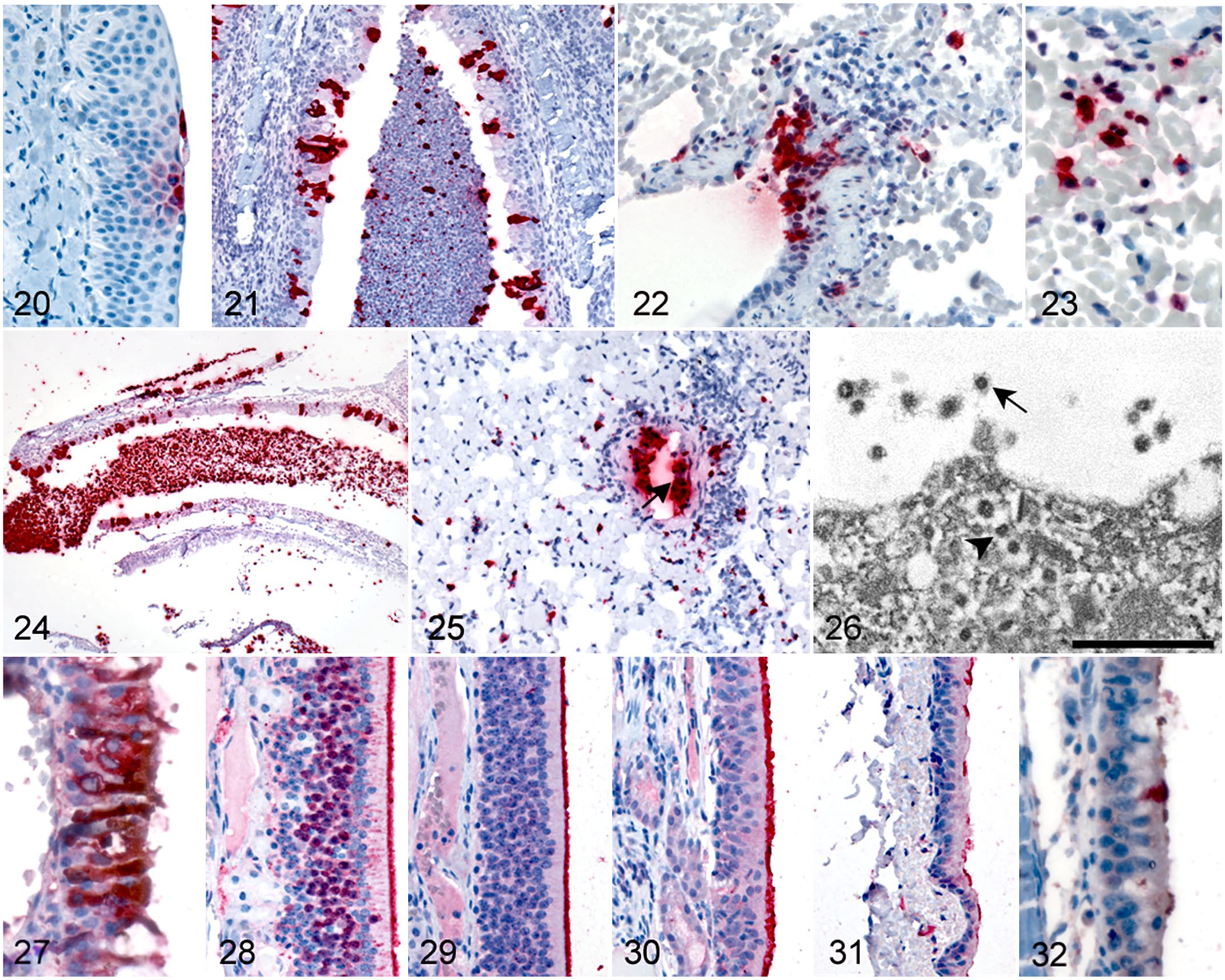
SARS-CoV-2, mink.
Ultrastructural evaluation by TEM was performed on FFPE upper respiratory and lung specimens from 4 cases that had extensive detection of SARS-CoV-2 by IHC. Rare clusters of viral particles were found in 3 of the 4 cases. Virions were present within membrane-bound vacuoles in the cytoplasm of tracheal and bronchiolar epithelium and type I pneumocytes, and extracellular virus was also associated with degenerating epithelium in nasal turbinate and trachea (Fig. 26). Viral particles were 65 nm in diameter and had morphology consistent with the family Coronaviridae, although viral and tissue ultrastructure were deteriorated due to extensive tissue processing. Characteristic coronaviral surface spikes were observed on extracellular viral particles, appearing as a faint fringe lining the virus surface. Cross sections through the viral nucleocapsids were apparent on both intracellular and extracellular viral particles.
On evaluation of turbinate sections, there appeared to be general sparing of olfactory epithelium by SARS-CoV-2 immunolabeling. IHC for OMP was performed in the 26 cases that had turbinate SARS-CoV-2 immunolabeling to aid in distinguishing olfactory from respiratory epithelium and determine whether SARS-CoV-2 infection of this specialized epithelium occurs in mink. Overall, there was correlation of strong OMP immunolabeling with olfactory mucosal morphology and a notable lack of SARS-CoV-2 labeling in these same areas. In only 3 cases (all adult mortalities), there were focal, very small regions of attenuated epithelium with both OMP and SARS-CoV-2 labeling seen in serially stained sections. Double labeling with OMP and SARS-CoV-2 confirmed colocalization in these areas (Fig. 27).
Detection of the SARS-CoV-2 Host Cell Entry Receptor ACE2 in Mink Respiratory Tract
IHC for the SARS-CoV-2 host cell entry receptor ACE2 was performed in URT and lung tissues from 3 animals (2 SARS-CoV-2-positive and 1 SARS-CoV-2-negative) to assess for the presence and distribution of this known viral receptor in mink. ACE2 immunolabeling localized within the apical cytoplasm of the epithelium in both olfactory and respiratory mucosa (Figs. 28–31); labeling was extensive in turbinate and tracheal epithelium and was patchy to rare in bronchiolar epithelium. Membranous labeling of alveolar macrophages was also seen; alveolar epithelial labeling was not observed in the examined sections. Double labeling with ACE2 and SARS-CoV-2 revealed colocalization primarily within respiratory epithelium of the nasal turbinates (Fig. 32).
Findings in Mink Without SARS-CoV-2 Detection
Of 72 mink examined, 20 (28%) did not have SARS-CoV-2 detected by any method in this study (referred to as “SARS-CoV-2-negative”). Rhinitis was also common (18/20; 90%) in these animals. Perivascular inflammation and edema was seen in the lung in 15/20 (75%) of SARS-CoV-2-negative animals, similar to many SARS-CoV-2-infected animals. Mild interstitial pneumonia was present in 8/20 (40%); none had bronchiolar lesions or DAD. Only 2 SARS-CoV-2-negative animals were mortalities; one had severe cystolithiasis identified at necropsy and the other had only mild periportal inflammation of the liver. Of 8 SARS-CoV-2-negative animals euthanized with signs of illness, 7 had lymphoplasmacytic inflammation in the kidney, 4/7 also in liver, and 3/7 also in heart; one animal had hepatic lipidosis. Of 10 SARS-CoV-2-negative animals euthanized randomly, 6 had lymphoplasmacytic inflammation in the kidney, 5/6 also in liver, and 2/6 also in heart.
Discussion
We characterized the pathological findings and localization of SARS-CoV-2 and its host cell entry receptor ACE2 in naturally infected US-farmed mink. Our findings expand upon previous reports of SARS-CoV-2-associated pathology in European-farmed and experimentally infected mink.48,52,65 In our study, adult mink were more often and more severely affected than juveniles, and there was no apparent sex predilection. Nearly half of all SARS-CoV-2-infected animals had no reported clinical or significant gross necropsy findings. Nasal discharge and rhinitis were the most consistent gross and histopathologic findings in infected animals, but also occurred in SARS-CoV-2-negative animals. The presence of lung lesions was variable in SARS-CoV-2-positive animals. Mild interstitial pneumonia was common, and often the only potentially virus-attributable finding in lungs of euthanized animals, but was also present in some SARS-CoV-2-negative animals. The lesions attributed to SARS-CoV-2 infection in mortalities included acute or chronic bronchiolitis and DAD. Acute bronchiolitis, DAD, and interstitial pneumonia have been described in other small cohorts of naturally and experimentally infected mink;48,52,65 however, the proliferative bronchiolar changes found in this study are heretofore undescribed in mink but are occasionally seen in human COVID-19 and have been reported in experimentally infected nonhuman primates.3,5,66
While the disease course in individual mink from this study is not known, the overall clinicopathologic findings align with features of COVID-19 in humans. In both species, the clinical spectrum ranges from asymptomatic infection to respiratory disease and death. DAD is a consistent and characteristic finding in human COVID-19 autopsies and was seen in more than half of mink mortalities in this study.41,54 In other animal models of SARS-CoV-2 infection, interstitial inflammation is common but DAD is only variably reported. Of the most commonly utilized animal model species, DAD has been achieved in transgenic mice expressing human ACE2 receptor, some Syrian hamsters, and few African green monkeys, whereas murine models using mouse-adapted virus, and most hamsters, ferrets, and macaques develop only mild disease without prominent DAD.7,8,37,73,74 On the contrary, the authors have also commonly seen human cases with only minimal findings of pulmonary congestion and edema that test cRT-PCR positive on FFPE (unpublished data), as also occurred in some mink herein. These findings support the use of mink in animal model development, especially with respect to recapitulating DAD in fatal human COVID-19, and also have important implications for SARS-CoV-2 surveillance on mink farms; that is, even asymptomatic animals or those without prominent viral lesions in necropsy tissues may harbor virus and be potential sources of exposure to other animals and humans on the farm.
cRT-PCR on FFPE URT specimens had the highest sensitivity for SARS-CoV-2 detection for all mink groups, with detection in all except one SARS-CoV-2-positive animal. ISH and IHC were less sensitive methods for virus detection in URT, as expected based on the known performance characteristics of these assays in our laboratory. FFPE lung specimens were significantly less useful overall than FFPE URT specimens for detection by cRT-PCR. Surprisingly, IHC was as sensitive as cRT-PCR in FFPE lung of adult mortalities with SARS-CoV-2 infection. ISH and IHC labeling were most abundant in nasal turbinates, and there was a patchy distribution of labeling in both URT and lung, sometimes with only one small focus of labeling among multiple tissue fragments. This has also been our consistent observation in human autopsy tissues (unpublished observation). These findings highlight the value of collecting URT necropsy specimens, which is not always routinely done—especially for nasal turbinates—and emphasize the need for testing multiple tissue specimens from each animal to improve diagnostic yield. The presence of more labeling in URT than lung by IHC and ISH also corroborates the finding of higher viral loads by quantitative RT-PCR in nasal turbinates compared with lung of experimentally SARS-CoV-2-infected hamsters, further suggesting a potential reason for high transmissibility of SARS-CoV-2 compared with other viruses that localize primarily to the lung. 13 SARS-CoV-2 was not detected by IHC or ISH outside the respiratory tract. RT-rtPCR testing of fresh lung was less useful than cRT-PCR testing of FFPE URT samples, and more useful than FFPE lung samples, for virus detection in adult mortalities with SARS-CoV-2 infection, and was overall not useful for detection in juvenile mortalities or in euthanized animals. RT-rtPCR testing of fresh colorectal samples was positive in both juvenile mortalities but in less than half of adult mortalities and was not useful for virus detection in euthanized animals.
A striking and common finding in these mink was the presence of marked lymphocytic perivascular inflammation and edema, which was present in 75% of adult mortalities with SARS-CoV-2 infection, for some of which this was the only significant lung lesion. Severe lymphocytic perivascular inflammation and vasculitis were also noted in experimentally infected mink, and vascular involvement by inflammation and thrombosis is commonly described in human SARS-CoV-2 infection and accounts for some severe outcomes of COVID-19 (ie, stroke).9,28,32,53,65 Capillaritis was seen in 3 SARS-CoV-2-positive mink in this study and is also found in some human COVID-19 autopsies.40,45 Attributing perivascular inflammation to SARS-CoV-2 infection in these animals is not straightforward, due to the possibility of Aleutian disease virus (ADV) infection, a common disease of farmed mink, and with histopathologic features of perivascular lymphoplasmacytic inflammation in various tissues in chronically infected animals.1,55 In fact, 75% of SARS-CoV-2-negative animals also had perivascular inflammation in the lung, further raising consideration for ADV. ADV serology results using the ADV antibody test (Fur Care LLC, Medford, Wisconsin) performed on blood specimens obtained at the time of necropsy were available for 31 euthanized animals from 2 farms; 8 were positive, of which 6 had perivascular inflammation in the lung, including the only 2 ADV-positive that were also SARS-CoV-2-positive. Of 23 euthanized animals that were ADV-seronegative, 19 had perivascular inflammation in the lung, including 12 that were SARS-CoV-2-positive. Furthermore, lymphoplasmacytic inflammation sometimes occurred in extrapulmonary tissues—kidney, liver, and occasionally heart—in both ADV-positive and negative animals. ADV serology was not available for mortalities, but 7 mortalities with SARS-CoV-2 detection had perivascular inflammation only in the lung and not in other tissues, suggesting that pulmonary perivascular inflammation without lymphoplasmacytic infiltrates in other organs could be a distinguishing feature of SARS-CoV-2. Further investigation into this potential association is clearly needed; in the meantime, identification of perivascular inflammation in mink lung warrants consideration of both SARS-CoV-2 and ADV in the diagnostic workup.
As with ADV, other coinfections could confound interpretation of SARS-CoV-2 pathology. In addition, DAD is characteristic of, but not specific for, SARS-CoV-2 infection, and particularly other viral causes must be considered because mild interstitial pneumonia was present in both SARS-CoV-2-positive and SARS-CoV-2-negative animals. Mink are susceptible to influenza A viruses, which are commonly associated with DAD; 2 we tested FFPE lung from all animals with DAD by RT-rtPCR assay for influenza A and B viruses, and all were negative. Canine distemper virus (CDV) also infects mink, producing interstitial pneumonia and syncytia with prominent viral inclusions in multiple tissues. 47 Viral inclusions were not present in this study, and furthermore, all mink farms had CDV vaccination practices in place that had been shown highly effective in preventing outbreaks. 60 Bacterial coinfections occur in some human COVID-19 mortalities, but often in the setting of extended hospitalization.33,41 Of 34 (18%) mink mortalities with SARS-CoV-2 detection, 6 (18%) also had histopathologic features of bacterial coinfection, including suppurative pleuritis, bronchopneumonia, and suspected sepsis. Mixed infection was common; bacterial agents detected included Streptococcus spp., Pasteurella spp., E. coli, and Pseudomonas aeruginosa by IHC, and E. coli and Pasteurella canis by culture swab taken at the time of necropsy for one case with pyothorax. No SARS-CoV-2-negative animals had histopathologic evidence of a bacterial disease in the lung. Colonies of mixed bacteria, sometimes associated with foreign material, were present within the suppurative nasal exudate in some animals. These were confined to the airway luminal exudate, interpreted as contaminants, and were not further characterized except for Pseudomonas IHC testing that was negative.
The cardiac findings in these animals are notable. The field team reported subjective cardiomegaly in 13% of SARS-CoV-2-positive animals, and several animals in the study, including both SARS-CoV-2-positive and negative animals, had very mild mononuclear inflammation with rare cardiomyocyte necrosis. Heart is the organ with the second most frequently identified lesions in human COVID-19, including these same findings.9,11,24,38,56,62 Furthermore, cardiac inflammation is a key histopathologic feature of multisystemic inflammatory syndrome, a postinfectious process associated with late-onset cardiac and other complications, occurring most frequently in children.17,18,43 The mechanism of virus-associated cardiac inflammation and injury may be multifactorial and due to a combination of direct viral infection and a generalized hyperinflammatory response (cytokine storm).19,67 Potential cardiac manifestations of, or contributors to, SARS-CoV-2 disease in mink warrant special attention in the further development of mink models of COVID-19, with careful consideration of other potential causes of myocardial inflammation in mink, such as ADV.
SARS-CoV-2 utilizes the ACE2 receptor for spike protein binding and host cell entry in humans. 30 It has been shown that ACE2 receptor in mink and other animals is structurally similar to human ACE2 receptor and also facilitates SARS-CoV-2 spike protein binding.16,25,29,39,58 ACE2 receptor is expressed extensively in ciliated epithelium of the human respiratory tract and we showed extensive immunolabeling in ciliated epithelial cells in the upper respiratory tract of mink, and lesser immunolabeling in bronchioles, pneumocytes, and alveolar macrophages. 36 This progressive decrease in ACE2 expression from upper to lower respiratory tract paralleled the distribution of SARS-CoV-2 immunolabeling, suggesting that concentration of viral load in URT could be directly related to its higher levels of ACE2 expression.
Interestingly, extensive ACE2 labeling was seen in olfactory epithelium—similar to what has been shown in human tissue. 22 In humans, loss of smell is often associated with COVID-19 and can sometimes be the earliest or only symptom experienced. 57 The pathogenesis is not fully understood but may involve direct viral infection of olfactory sensory neurons or olfactory nuclei in the brain, and SARS-CoV-2 virus detection in olfactory mucosa is reported in humans and experimentally infected hamsters.10,44,75 Despite extensive ACE2 labeling, the vast majority of olfactory epithelium was spared of SARS-CoV-2 IHC and ISH labeling. SARS-CoV-2 labeling was also not observed in brain sections that included olfactory bulb. It is unknown whether SARS-CoV-2-infected mink experience anosmia, but our findings suggest that if they do, direct viral infection of olfactory mucosa or olfactory bulb may not be the primary mechanism involved, although ACE2 receptor is present in the mucosa. Spike protein binding to ACE2 receptors represents only the first step in viral entry of host cells; therefore, co-receptors or other cellular components needed to complete viral entry or replication may be absent or minimally expressed in mink olfactory epithelium. 31
Limitations of this study are primarily related to the paucity and vagueness of clinical information available. The full spectrum and distribution of clinical signs for the examined animals was not thoroughly characterized and the specific date of infection for individual animals was not known. The 3 farms from which mink mortalities were received were experiencing ongoing losses due to SARS-CoV-2, whereas the 2 farms on which animals were euthanized were several weeks past the peaks of their outbreaks and had returned to baseline mortality rates. Some animals from these latter farms that tested negative for SARS-CoV-2 may have been in a late recovery phase of infection, beyond the period of viral detection by the assays used. Clinical disease course for individual animals was not known, making it impossible to fully characterize the time course of pathological changes in these animals; this will be an important next step in assessing the utility of mink as models of human COVID-19.
In conclusion, we showed that natural SARS-CoV-2 infection in US-farmed mink is associated with a range of clinicopathologic findings, from asymptomatic infection to death. Suppurative rhinitis and mild interstitial pneumonia were present in a majority of SARS-CoV-2-positive animals but also in SARS-CoV-2-negative animals, and bronchiolitis and DAD were almost exclusively in animals found dead. Bacterial coinfections were also identified. Some SARS-CoV-2-positive animals had only minimal, nonspecific lung findings, indicating that typical viral lesions should not be a requirement for consideration of SARS-CoV-2 infection in mink mortality investigations. Lymphocytic perivascular inflammation was common, and cardiac inflammation was rare, in both SARS-CoV-2-positive and negative animals, and these findings warrant further exploration as possible SARS-CoV-2-associated lesions. We showed that FFPE respiratory tissues are useful for SARS-CoV-2 diagnosis, with viral detection in URT specimens more commonly than lung specimens by all diagnostic methods used (cRT-PCR, ISH, and IHC). Virus labeling by IHC and ISH corroborated this finding, with more extensive labeling in URT than lung. Virus distribution in tissues was patchy, and multiple tissue specimens from each animal should be tested to maximize virus detection when using FFPE diagnostic samples. We demonstrated extensive expression of ACE2 receptor in olfactory and respiratory epithelium, with decreasing detection along the respiratory tract and into the lung. These findings further support ACE2 as the SARS-CoV-2 host cell entry receptor in mink. Overall, this study expands on the previous, limited reports of the pathological findings of SARS-CoV-2 infection in mink, demonstrates similarities to some aspects of human COVID-19, and supports the use of mink models for investigating SARS-CoV-2 pathogenesis and development of medical countermeasures.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858221079665 – Supplemental material for Histopathology and localization of SARS-CoV-2 and its host cell entry receptor ACE2 in tissues from naturally infected US-farmed mink (Neovison vison)
Supplemental material, sj-pdf-1-vet-10.1177_03009858221079665 for Histopathology and localization of SARS-CoV-2 and its host cell entry receptor ACE2 in tissues from naturally infected US-farmed mink (Neovison vison) by Jana M. Ritter, Tais M. Wilson, Joy M. Gary, Josilene N. Seixas, Roosecelis B. Martines, Julu Bhatnagar, Brigid C. Bollweg, Elizabeth Lee, Lindsey Estetter, Luciana Silva-Flannery, Hannah A. Bullock, Jonathan S. Towner, Caitlin M. Cossaboom, Natalie M. Wendling, Brian R. Amman, Robert R. Harvey, Dean Taylor, Hannah Rettler, Casey Barton Behravesh and Sherif R. Zaki in Veterinary Pathology
Footnotes
Acknowledgements
We thank the mink producers and the Utah Department of Health and Utah Department of Agriculture and Food for their participation in these investigations. We thank the US Geological Survey National Wildlife Center, Madison, WI, for performing the fresh tissue real-time reverse transcription–polymerase chain reaction (RT-rtPCR) testing, and the USDA National Veterinary Services Laboratory (NVSL) for performing bacterial culture. We thank Fur Care LLC, Medford, WI, for donation of the Aleutian Disease Virus test kits. We thank Mitesh Patel, Monica Peabody, Rhonda Cole, and Amy Morris from the Centers for Disease Control and Prevention’s (CDC) Infectious Diseases Pathology Branch Operations and Histology teams for accessioning and processing of tissue specimens.
Supplemental material for this article is available online.
Disclaimer
The findings and conclusions herein are those of the authors and do not necessarily represent the official position of the Centers for Disease Control and Prevention (CDC). The mention of company names or products does not constitute endorsement by the CDC.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
