Abstract
Coronavirus disease 2019 (COVID-19), caused by the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), is an emergent, amphixenotic infection that resulted in a pandemic declaration in March 2020. A rapid search for appropriate animal models of this newly emergent viral respiratory disease focused initially on traditional nonhuman primate research species. Nonhuman primate models have previously been shown to be valuable in evaluation of emerging respiratory coronaviruses with pandemic potential (ie, SARS-CoV and Middle East respiratory syndrome coronavirus). In this article, we review the pulmonary histopathologic characteristics and immunohistochemical evaluation of experimental SARS-CoV-2 infection in the rhesus macaque, pigtail macaque, African green monkey, and squirrel monkey. Our results indicate that all evaluated nonhuman primate species developed variably severe histopathologic changes typical of coronavirus respiratory disease characterized by interstitial pneumonia with or without syncytial cell formation, alveolar fibrin, and pulmonary edema that progressed to type II pneumocyte hyperplasia. Lesion distribution was multifocal, frequently subpleural, and often more severe in lower lung lobes. However, squirrel monkeys showed the least severe and least consistent lesions of the evaluated nonhuman primates. Additionally, our results highlight the disparate physical relationship between viral antigen and foci of pulmonary lesions. While classic respiratory coronaviral lesions were observed in the lungs of all nonhuman primates evaluated, none of the primates exhibited severe lesions or evidence of diffuse alveolar damage and therefore are unlikely to represent the severe form of SARS-CoV-2 infection observed in fatal human cases.
On March 11, 2020, the World Health Organization (WHO) declared a pandemic in response to the rapid global spread of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), the viral agent associated with coronavirus disease 2019 (COVID-19). 32 To date, SARS-CoV-2 has infected an estimated 180 million people globally, and COVID-19 has been associated with the death of over 3.8 million individuals. 16 Clinical symptomology consistently reported in humans, regardless of disease severity, includes cough, shortness of breath, and fever. 8 An abundance of autopsy samples since the initiation of the pandemic has led to a pathophysiologic understanding of the terminal phase of disease 3,5,7,8 ; however, autopsy samples have frequently been acquired from patients in which comorbidities, chronic interventional therapy, and prolonged ventilator-assisted respiration have been administered. These interventional strategies do not allow full understanding of the acute pathophysiology of disease. Rare lung biopsy reports exist in which uncomplicated pulmonary disease can be assessed to evaluate the acute phase of disease in humans. 26 The fatal form of severe acute respiratory syndrome (SARS) and COVID-19 is clinically linked to acute respiratory distress syndrome (ARDS). The Berlin definition of ARDS, created in 2011, identifies the main clinical features as acute presentation (occurring within 1 week of a known insult), bilateral lung opacities, hypoxia, and ruling out other pulmonary disease and other clinical causes of pulmonary edema such as heart disease. 19
While ARDS defines the clinical presentation of the most severe form of SARS and COVID-19 in humans, the histopathologic presentation is defined by diffuse alveolar damage (DAD). 2,9,27 Coronavirus disease 2019 has been shown to be histologically indistinct from other causes of DAD. 11 DAD is induced by inflammatory cytokines resulting in increased vascular permeability and epithelial and endothelial cell death. 13 Histologically, this process results in alveolar flooding with protein-rich fluid and organization of polymerized fibrin that entraps degenerate leukocytes and pneumocytes in distinct hyaline membranes. 15 Endothelial damage is attributed to increased TNF-α, interleukin (IL)-1, IL-6, and IL-18 stimulating degranulation of recruited neutrophils with collateral damage to pulmonary endothelium. 15 Interestingly, while DAD is considered to be the pathognomonic change associated with ARDS, approximately half of patients diagnosed with ARDS lack histopathologic changes consistent with DAD. 25 To date, no animal model has consistently exhibited DAD following inoculation with SARS-CoV-2.
Animal models of infectious disease continue to be a pivotal tool used not only to further the understanding of key pathologic mechanisms of disease development but also as crucial platforms for vaccine efficacy, safety testing, and interventional therapy assessment. To date, the broad search for applicable animal models of SARS-CoV-2 infection have focused primarily on the most severe and fatal form of COVID-19. Little is known of the histopathologic changes associated with asymptomatic or mildly symptomatic SARS-CoV-2 infections in humans, and how animal models may mimic this form of disease. Additionally, animal models evaluating the long-term effects of coronavirus-associated pulmonary lesions for any emerging human respiratory coronavirus (SARS-CoV-1 and MERS) are unreported in current literature. The presence of post-acute sequelae of SARS-CoV-2 infection (long COVID-19) suggests this is an area of emerging interest. 23 Nonhuman primates serve a crucial role in both model development and pharmaceutical intervention strategies for infectious disease work as their genomes, proteomes, and physiology are comparable to those of humans. Herein we describe the histopathologic changes associated with inoculation of SARS-CoV-2 in 4 commonly used nonhuman primate species.
Materials and Methods
Ethics
All animal studies were conducted under the approval of the Institutional Animal Care and Use Committee of Rocky Mountain Laboratories (National Institutes of Health [NIH]) and carried out by certified staff in an Association for Assessment and Accreditation of Laboratory Animal Care International–accredited facility. Animal care and study guidelines were performed in accordance with the institution’s guidelines for animal use and following the guidelines and basic principles in the NIH Guide for the Care and Use of Laboratory Animals, the Animal Welfare Act, and the US Department of Agriculture and US Public Health Service Policy on Humane Care and Use of Laboratory Animals.
Virus and Biosafety
All SARS-CoV-2 studies were approved by the Institutional Biosafety Committee (IBC) and performed in high biocontainment (BSL3/BSL4) at Rocky Mountain Laboratories (RML), National Institutes of Allergy and Infectious Diseases, National Institutes of Health. All sample processing in high biocontainment and sample removal followed IBC-approved Standard Operating Protocols (SOPs). 10
All nonhuman primates were inoculated with the same SARS-CoV-2 isolate, nCoV-WA1-2020, obtained through the Centers for Disease Control and Prevention and propagated in VeroE6 cells as previously described. 18
Animal Work
All nonhuman primates enrolled in this study were recruited through preexisting experiments that are either published 18,24,30 or publication is pending (squirrel monkey and pigtail macaque). All nonhuman primates were housed in a climate-controlled room with a fixed 12-hour light-dark cycle. Rhesus macaques, African green monkeys, and pigtail macaques were singly housed in adjacent primate cages allowing social interaction and provided with commercial monkey chow, treats, and fruit twice daily with water. Squirrel monkeys were cohoused, provided with commercial monkey chow, treats, and fruit twice daily with water ad libitum. Environmental enrichment was provided with a variety of human interaction, manipulanda, commercial toys, movies, and music. Prior to enrollment, all rhesus and pigtail macaques were tested and found negative for simian immunodeficiency virus, simian retrovirus, simian T-lymphocyte virus, and herpes B virus via serologic analysis. Prior to enrollment, all African green monkeys were tested and found negative for simian immunodeficiency virus, simian T-lymphocyte virus, simian herpesvirus SA-8, and measles virus. Pre-enrollment serologic screening was not performed on squirrel monkeys. A summary table of species, route of inoculation, inoculation dose, age range of enrolled primates, and time point of necropsy is included in Supplemental Table S1.
Eight rhesus macaques were inoculated with 2.6 × 106 TCID50 SARS-CoV-2 and tissues harvested at necropsy at 3 (n = 4) or 21 (n = 4) days post inoculation (DPI) as previously described. 18 An additional cohort of n = 6 macaques were infected and evaluated at 7 DPI identically to those macaques described above as previously described. 30 Deep sedation was achieved via intramuscular administration of ketamine and euthanasia was achieved through intracardiac administration of pentobarbital and phenytoin sodium.
Eight African green monkeys were inoculated with 2.6 × 106 TCID50 SARS-CoV-2 and tissues harvested at necropsy on 3 (n = 4) or 10 (n = 4) DPI as previously described. 24 Deep sedation was achieved via intramuscular administration of tiletamine-zolazepam and euthanasia was achieved through intracardiac administration of pentobarbital and phenytoin sodium.
Six pigtail macaques were inoculated via a combination of intranasal and intratracheal route with a total dose of 2.6 × 106 TCID50 of SARS-CoV-2 diluted in sterile DMEM (Dulbecco’s modified Eagle’s medium) as previously described. 18,24 All pigtail macaques were euthanized, and tissues harvested at necropsy 7 DPI. Deep sedation was achieved via intramuscular administration of tiletamine-zolazepam and euthanasia was achieved through intracardiac administration of pentobarbital and phenytoin sodium.
Four groups of 4 (n = 4/group) squirrel monkeys were inoculated either through the intratracheal (n = 8) or intranasal (n = 8) route with 2.6 × 104 TCID50 SARS-CoV-2 diluted in 1 mL (intratracheal) or 150 µL per naris (intranasal) of sterile DMEM. One group of each inoculation route were euthanized at 4 or 7 DPI and tissues were harvested at necropsy. One squirrel monkey met early euthanasia criteria for disease unrelated to the study and was excluded from the study. Deep sedation was achieved via intramuscular administration of ketamine and euthanasia was achieved through intracardiac injection of pentobarbital and phenytoin sodium.
Histology and Immunohistochemistry
Histopathologic changes for all evaluated species was limited to the lungs and tissue processing, and evaluation was limited to tissues with lesions. For rhesus macaques, pigtail macaques, and African green monkeys, 3 sections of each lung lobe (left and right upper, middle, and lower) were obtained yielding a total of 18 sections per animal for histologic evaluation. One sample of each lung lobe (left and right upper, middle, and lower) was obtained from each squirrel monkey yielding a total of 6 sections per animal for histologic evaluation. All tissues were fixed for a minimum of 7 days in 10% neutral-buffered formalin with 2 changes. Following fixation, tissues were embedded in paraffin, sectioned at 5 μM, and allowed to air dry overnight prior to staining with hematoxylin and eosin. Immunohistochemistry (IHC) on lung sections was performed for rhesus macaques and African green monkeys using a custom-made rabbit antiserum against SARS-CoV-2 N proteinat a 1:1000 dilution as previously described. 18,24 In the pigtail macaque and squirrel monkey, IHC was done using antibody to SARS-CoV/SARS-CoV-2 nucleocapsid (Sino Biological cat#40143-MMO5) at a dilution of 1:1000 as previously described. 20 Positive and negative controls performed as expected. Histopathologic and immunohistochemical analysis was performed by a blinded, board-certified veterinary pathologist.
Results
Rhesus Macaque
At 3 DPI (the earliest evaluated time point), 3 of 4 (75%) evaluated macaques had histopathologic changes consistent with viral interstitial pneumonia (Figs. 1, 2a). Lesions at this time point included mild to moderate expansion of alveolar septa by fibrin and edema, accumulation of macrophages and fewer neutrophils in alveolar spaces, and occasional pooling of proteinaceous fluid and fibrin in more severely affected sections. Minimal bronchiolitis was characterized by rare individual epithelial cell necrosis, attenuation of the adjacent epithelium, and influx of low numbers of neutrophils, macrophages, and occasional eosinophils. When specifically evaluating interstitial pneumonia, 14/24 (58%) of lung lobes had no interstitial pneumonia, 4/24 (16%) had minimal interstitial pneumonia, 2/24 (8%) had mild interstitial pneumonia, and 4/24 (16%) had moderate interstitial pneumonia.
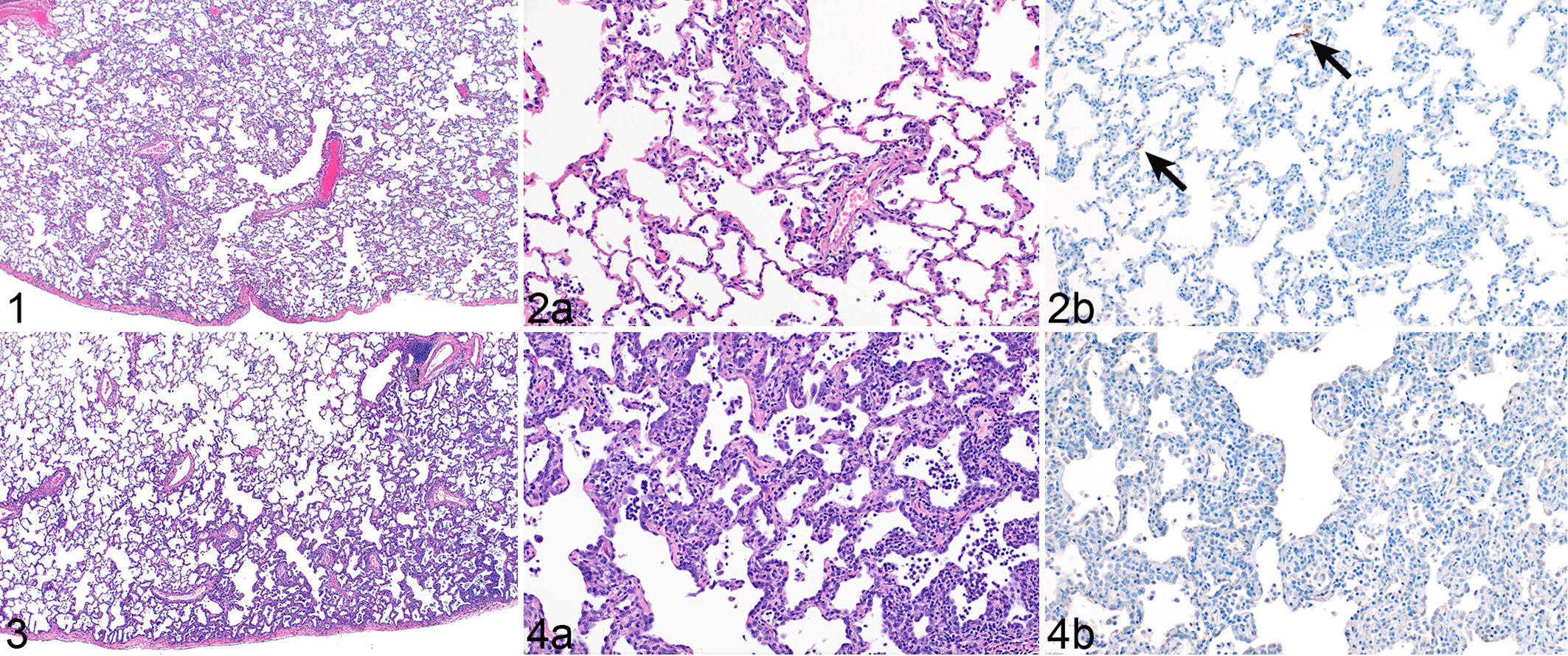
SARS-CoV-2 interstitial pneumonia, lung, rhesus macaque, 3 days post inoculation (DPI).
At 7 DPI, pulmonary lesions consistent with viral interstitial pneumonia was noted in 5 of 6 (83%) of evaluated macaques. The interstitial pneumonia was multifocal, primarily limited to subpleural regions, and most frequent in lower lung lobes (Figs. 3, 4a). Upper lung lobes rarely contained any histologic changes consistent with viral pneumonia. Pulmonary edema and syncytial cells were noted in all affected macaques. Lesions frequently involved less than 20% of any evaluated lung section. When specifically evaluating interstitial pneumonia, 26/36 (64%) of lung lobes evaluated had no interstitial pneumonia, 5/36 (14%) had minimal interstitial pneumonia, 2/36 (5%) had mild interstitial pneumonia, and 6/36 (16%) had moderate interstitial pneumonia. Rare SARS-CoV-2 immunoreactivity, primarily in type I and II pneumocytes as well as pulmonary macrophages, was noted at 3 and 7 DPI. Immunoreactivity was multifocal, random, and rarely intralesional (Figs. 2b, 4b).
SARS-CoV-2 interstitial pneumonia, lung, African green monkey, 3 days post inoculation (DPI).
By 21 DPI, minimal pulmonary lesions were noted in all evaluated macaques. At this time point, when evaluating specifically for interstitial pneumonia, none of the 24 lung lobes exhibited interstitial pneumonia. Foci of pathology were characterized by minimal to mild septal fibrosis, type II pneumocyte hyperplasia, and rare aggregates of alveolar macrophages admixed with lymphocytes surrounding blood vessels. Immunolabeling for SARS-CoV-2 was not detected at 21 DPI.
African Green Monkey
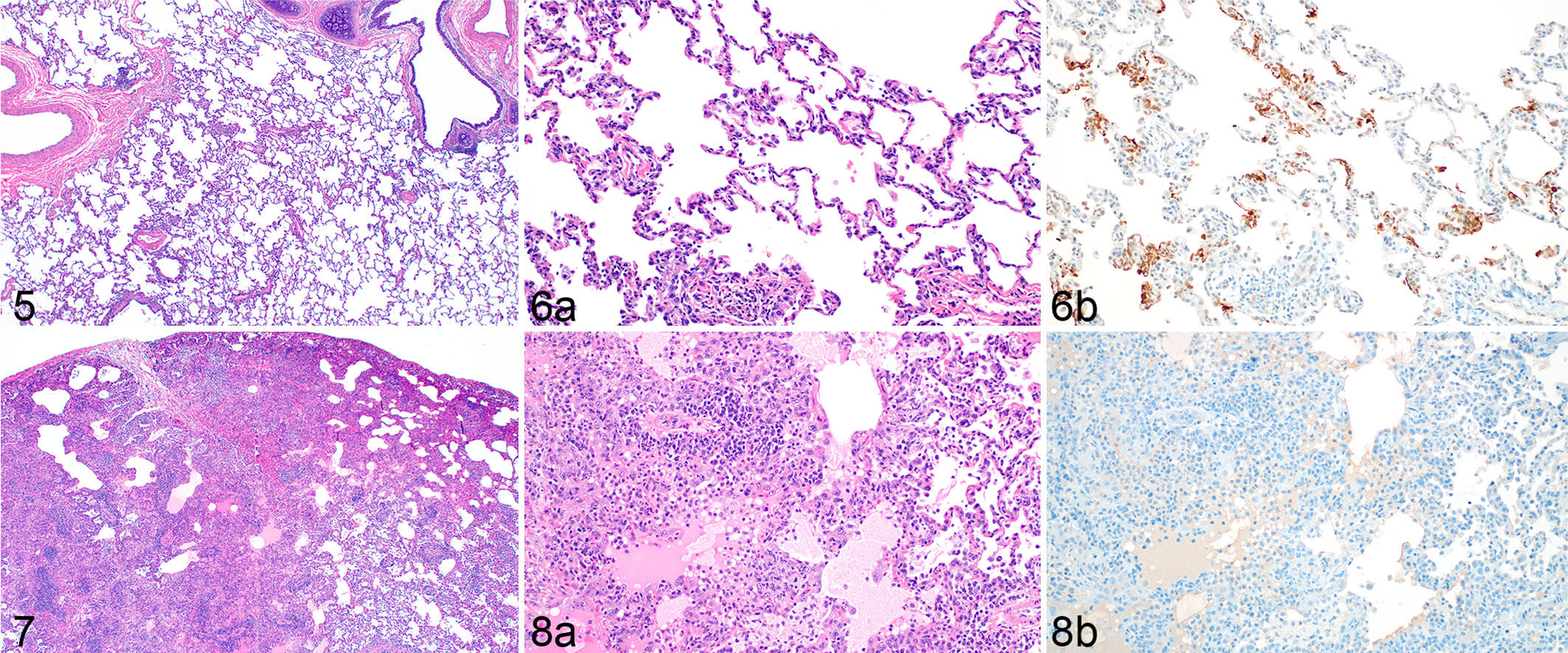
Pulmonary pathologic findings were limited to minimally to moderately thickened alveolar septa and increased inflammatory cellularity in all 4 AGMs at 3 DPI (Figs. 5, 6a). At this time point, none of the 24 evaluated lung lobes reached diagnostic criteria of interstitial pneumonia. At 10 DPI, lesions consistent with viral interstitial pneumonia was observed in 2 of 4 AGMs. Features of interstitial pneumonia in these cases included septa thickened by fibrin and edema, infiltrating leukocytes, type II pneumocyte hyperplasia, increased alveolar macrophages, and variable amounts of alveolar edema and fibrin accumulation (Figs. 7, 8a). When specifically evaluating interstitial pneumonia, 17/24 (58%) of lung lobes had no interstitial pneumonia, 4/24 (16%) had minimal interstitial pneumonia, 2/24 (8%) had mild interstitial pneumonia, and 1/24 (4%) had moderate interstitial pneumonia. In rare cases, a high magnification field of pneumonia appeared subjectively severe, but only a small fraction of the evaluated section was affected (Figs. 7, 8a). Thus, most lung tissue remained normal or minimally affected and normal lung was sometimes juxtaposed to a focus of interstitial pneumonia.
In some sections, nodular aggregates at terminal airways resembled obstructive bronchiolitis or acute fibrinous organizing pneumonia. Additionally, multinucleate giant cells and epithelial syncytial cells were present in variable numbers. Lesion prevalence and distribution were not uniform and diffuse as one would expect in a typical interstitial pneumonia. Terminal bronchioles were multifocally affected, but at 10 DPI, foci of interstitial pneumonia were most often subpleural. At 3 DPI, there were rare foci of immunoreactivity primarily in type I and II pneumocytes as well as pulmonary macrophages (Fig 6b). Exceedingly rare immunoreactivity was noted at 10 DPI, and antigen was frequently not noted immediately adjacent to (Fig. 8b) but not within (not shown) foci of histopathologic change.
Pigtail Macaque
A single time point of 7 DPI was evaluated for pulmonary lesions in the pigtail macaque model. This time point was selected because lesions were maximal at 7 DPI in the rhesus macaques. Interstitial pneumonia consistent with a viral etiology was observed in at least one lung lobe of all six evaluated pigtail macaques ranging in severity from minimal to moderate. In all macaques, the most severe lesions were in the lower lung lobes and were most prevalent in subpleural spaces (Fig. 9). In foci of interstitial pneumonia, septa were extensively thickened by edema, fibrin, and low to moderate numbers of infiltrating leukocytes. Adjacent alveolar spaces contained abundant proteinaceous fluid or a mixture of degenerate and nondegenerate leukocytes (primarily macrophages with fewer lymphocytes and neutrophils) admixed with abundant fibrin and karyorrhectic cellular debris that occasionally formed distinct hyaline membranes (Fig. 10a). Affected alveoli were extensively lined by prominent type II pneumocytes occasionally exhibiting syncytial cell formation or bizarre mitotic figures. When specifically evaluating interstitial pneumonia, 18/36 (50%) of lung lobes had no interstitial pneumonia, 6/36 (16%) had minimal lesions, 6/36 (16%) had mild lesions, and 6/36 (16%) had moderate lesions. There was rare immunoreactivity, primarily in type I and II pneumocytes as well as pulmonary macrophages, occasionally within foci of histopathologic change but most frequently adjacent to regions of interstitial pneumonia (Fig. 10b).
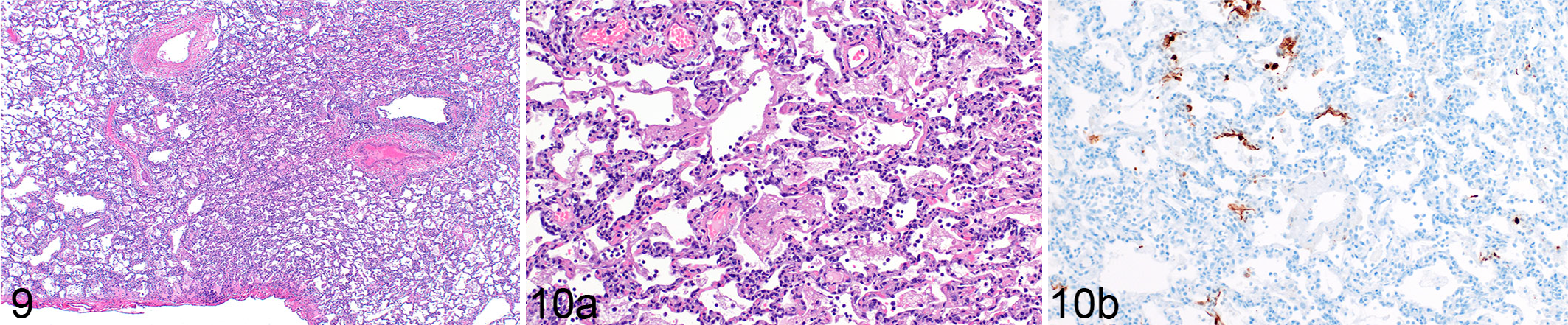
SARS-CoV-2 interstitial pneumonia, lung, pigtail macaque, 7 days post inoculation.
Squirrel Monkey
Pulmonary lesions consistent with viral interstitial pneumonia was observed in 2 of 7 squirrel monkeys at 4 DPI and 6 of 8 squirrel monkeys at 7 DPI. Early in the disease course (4 DPI), minimal to mild interstitial pneumonia characterized by alveolar edema, fibrin and leukocyte accumulation, type II pneumocyte hyperplasia, and rare foci of alveolar wall thickening by edema were observed (Figs. 11, 12a). Lesion severity, while still limited, increased at 7 DPI with mild to moderate interstitial pneumonia noted with the addition of low to moderate numbers of multinucleate giant cells and fewer epithelial syncytial cells (Suppl. Fig. S1) as well as increased numbers of hyperplastic type II pneumocytes with infrequent bizarre or irregular mitotic figures (Figs. 13, 14a). When specifically evaluating interstitial pneumonia, 23/28 (82%) of lung lobes had no interstitial pneumonia, 3/28 (11%) had minimal lesions, and 2/28 (7%) had mild lesions at 4 DPI. Similarly, 5/28 (18%) of lung lobes had minimal interstitial pneumonia, 3/28 (11%) had mild interstitial pneumonia, and 20/28 (71%) had no interstitial pneumonia at 7 DPI. Spatially, at both 4 and 7 DPI, lesions were multifocal to focally extensive and focused on terminal bronchioles primarily in subpleural locations. Histopathologic lesion severity did not correlate to route of inoculation. Exceedingly rare immunoreactivity (one focal area in one squirrel monkey) was noted in type I pneumocytes (data not shown). Regions of histopathologic change showed no SARS-CoV-2 immunoreactivity at either 4 or 7 DPI (Figs. 12b, 14b).
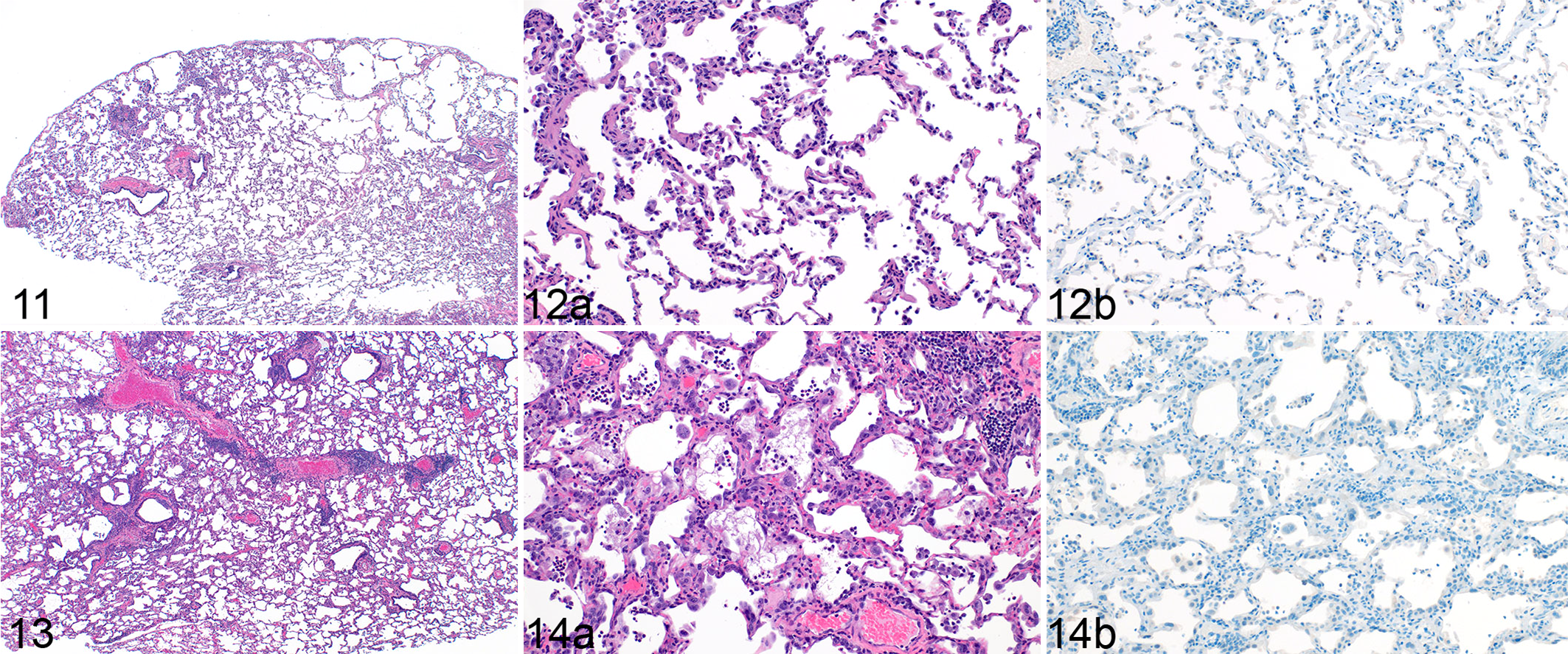
SARS-CoV-2 interstitial pneumonia, lung, squirrel monkey, 4 days post inoculation (DPI).
Discussion
Establishment of an appropriate animal model of viral disease requires first that the selected species is susceptible to infection (ie, has the appropriate viral receptor expressed in the appropriate location for host-viral interaction), and secondly that the species is permissive to viral infection. The host cellular receptor for the SARS-CoV-2 spike protein has previously been identified as the angiotensin I converting enzyme 2 (ACE-2) receptor cell surface protein. 14 Interestingly, this protein is widely expressed throughout the body of mammals, including humans, yet limited lesions were observed outside of the respiratory tract for most natural infections. The ACE-2 receptor gene is highly conserved across nonhuman primate species, 17 but the permissiveness to SARS-CoV-2 among these species was unknown. In part, this is why 4 species of nonhuman primates were evaluated at our facility. While the squirrel monkey ACE-2 receptor gene is very similar to that of humans, 17 it appears that the squirrel monkey is not as permissive to SARS-CoV-2 as Old World nonhuman primates, and does not appear to be a suitable animal model for COVID-19 research.
A secondary consideration for animal model development is disease severity. The nonhuman primates reported herein exhibited mild to occasionally moderate histopathologic changes consistent with viral pneumonia. To date, no nonhuman primate model evaluated at our facility has exhibited severe clinical disease consistent with ARDS, or the clinical signs of moderate to severe COVID-19 in humans. Additionally, there were no histopathologic changes consistent with DAD to support modeling of severe disease. Severe disease in terms of clinical signs and lesions was inconsistently observed throughout studies conducted at multiple facilities to date. A single report citing severe pulmonary lesions in 2 of 4 African green monkeys has been published. 4 While this report attributed terminal disease in these 2 African green monkeys to ARDS, it is important to note that the clinical criteria established in the Berlin definition of ARDS were not noted in the publication and the diagnosis was primarily based on histologic evidence of DAD. In contrast, African green monkeys at our institution as well as those reported at other institutions showed, at most, moderate clinical and histologic evidence of pulmonary disease. 24,31
The utility of nonhuman primates in SARS-CoV-2 research may be dependent on the type of prevention or therapeutic modality in question. Nonhuman primates serve as a good model in vaccine development as large, consecutive blood volumes may be taken sequentially to evaluate antibody titers and T cell responses over time. The size of the nonhuman primate also allows additional sampling modalities to be performed, such as bronchoalveolar lavage, oral fluid collection, and nasal washes to assess mucosal immunoglobulin. Additionally, changes in viral titers in bronchoalveolar lavage and lung tissue have been shown to correlate well with histopathologic scoring in nonhuman primate models of SARS-CoV-2 infection, even in the absence of moderate or severe histologic disease. 21,28,30 When considering treatments for severe disease phenotypes observed in the lethal form of COVID-19 in humans, nonhuman primate models may not be the most appropriate animal model as the species evaluated thus far do not exhibit the prolonged clinical progression of disease. Additionally, some features of pulmonary disease noted in human autopsies that may be contributory to mortality have not been observed in nonhuman primate species. For example, an unexpected clinical presentation, thromboembolic pulmonary disease, has been reported in retrospective autopsy reports in human literature throughout the pandemic. In one report, as many as 7/12 (58%) patients had postmortem computed tomography lesions or histologically observable thromboembolism that was clinically silent. 29 While the mechanism by which thromboembolism occurs in COVID-19 patients has not been established, several mechanisms have been postulated. Considering Virchow’s triad, 2 principal mechanisms of coagulation are considered: induction of a hypercoagulable state and direct endothelial damage. Endothelialitis associated with COVID-19 has been reported in humans, 1 and similar histopathologic changes have been observed in the Syrian hamster model. 22 It is plausible that endothelial dysregulation may occur during active SARS-CoV-2 infection, or localized cytokine storms within the infected lung incite a localized hypercoagulable state. However, histopathologic changes consistent with either of these scenarios have not been observed in the nonhuman primate models studied at our facility.
Interstitial lung disease secondary to an aerosolized agent such as a virus is classically thought to cause disseminated pulmonary disease. Our studies for each of the nonhuman primate species have demonstrated that the majority of pulmonary tissue did not develop lesions, and foci of severe pneumonia were only in a fraction of the tissue sections evaluated. While these rare foci may exhibit severe lesions at high magnification, they remained such a minor portion of the total pulmonary parenchyma that we did not consider the overall lesion to be severe. Additional ante-mortem, high-resolution imaging may more precisely quantify the percentage of the lung affected to allow more accurate interpretation of the histopathologic data. Sampling sites were standardized in all studies to provide a representative histologic picture of the spectrum of disease in each nonhuman primate species; however; due in part to the limitations of sampling size and multifocal lesion distribution of this disease, histopathology remains a poorly sensitive method of evaluating lesion distribution for SARS-CoV-2 infection. Additionally, immunohistochemistry appears to be poorly sensitive for detection of SARS-CoV-2 antigen in both nonhuman primates and human autopsy samples when only foci of gross or histologic lesions are evaluated. Antigen was almost exclusively detected in pneumocytes either peripheral to or distant from focal lesions. This phenomenon has been noted in a nonhuman primate model of MERS-CoV. 6 The near-complete lack of viral antigen within the pulmonary lesions remains poorly understood.
Development of a lethal animal model mimicking severe COVID-19 in humans has yet to be accomplished. Of the nonhuman primate species evaluated at this institution, the pigtail macaque showed the most widely distributed and most consistent lesions observed on histopathology (Table 1). Pigtail macaques have a known TRIM5 mutation, which has been previously hypothesized to make them more susceptible to human immunodeficiency virus type 1 infection relative to all other Old World monkeys. 12 It has yet to be elucidated if TRIM5 plays a role in the pathogenesis of SARS-CoV-2 infection. Full genotyping of nonhuman primates enrolled in further studies may assist in identifying genetic markers that render species more or less susceptible to disease, and therefore, more appropriate for disease modeling. Experimentally inoculated nonhuman primates may mimic asymptomatic or mildly symptomatic disease in humans; however, to date, nonhuman primates remain a poor model for severe SARS-CoV-2 infection in humans. Histopathology does not appear to be a superior readout for interpretation of vaccine and therapeutic evaluations in the nonhuman primate models of SARS-CoV-2 due to the variability of lesion distribution and severity within experimentally inoculated animals in each species. The recommendation of which nonhuman primate species is a superior model should be based on virology data such as viral burden in tissue and immunologic response, as well as further work on genetic factors that contribute to significant disease.
Histologic lesions in 4 nonhuman primate species inoculated with SARS-CoV-2.

a Total enrollment does equate to number of animals within cohort in which lesion is observed; −, not observed; +/−, observed in few primates; +, minimal; ++, mild; +++, moderate.
Supplemental Material
Supplemental Material, sj-pdf-1-vet-10.1177_03009858211067468 - Histologic pulmonary lesions of SARS-CoV-2 in 4 nonhuman primate species: An institutional comparative review
Supplemental Material, sj-pdf-1-vet-10.1177_03009858211067468 for Histologic pulmonary lesions of SARS-CoV-2 in 4 nonhuman primate species: An institutional comparative review by Chad S. Clancy, Carl Shaia, Vincent Munster, Emmie de Wit, David Hawman, Atsushi Okumura, Heinz Feldmann, Greg Saturday and Dana Scott in Veterinary Pathology
Footnotes
Acknowledgements
The authors are grateful for the significant contributions made by Rebecca Rosenke, Tina Thomas, and Dan Long in production of the histology/immunohistochemistry slides, and the animal caretaker staff at Rocky Mountain Laboratories. The authors would also like to sincerely thank the scientific staff of the Laboratory of Virology for their scientific contributions and animal work conducted over the course of the nonhuman primate studies.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Intramural Research Program (NIAID, NIH).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
