Abstract
In animals, most cases of systemic amyloidosis are of amyloid A type, and the other types of systemic amyloidoses are rare. This study analyzed systemic amyloidosis in a 15-year-old female Tsushima leopard cat. Amyloid deposits strongly positive for Congo red staining were observed in the arterial walls as well as the interstitium in multiple organs. Mass spectrometry–based proteomic analysis with laser microdissection of amyloid deposits identified epidermal growth factor–containing fibulin-like extracellular matrix protein 1 (EFEMP1) as a prime amyloidogenic protein candidate. Immunohistochemistry showed that the amyloid deposits were positive for the N-terminal region of EFEMP1. From these results, the present case was diagnosed as EFEMP1-derived amyloidosis. It is the first such case in an animal. EFEMP1-derived amyloidosis in humans has recently been reported as a systemic amyloidosis, and it is known as an age-related venous amyloidosis. The present case showed different characteristics from human EFEMP1-derived amyloidosis, including the amyloid deposition sites and the amyloidogenic region of the EFEMP1 protein, suggesting a different pathogenesis between Tsushima leopard cat and human EFEMP1-derived amyloidosis.
Keywords
Amyloidosis is a general term for a disease group characterized by systemic or localized amyloid deposition that results from the misfolding of normal proteins and proteins with genetic and/or enzymatic alterations. Amyloid A (AA) amyloidosis involving serum amyloid A (SAA) protein is the most common systemic amyloidosis in animals. 13 To date, 6 types of systemic amyloidosis, including AA amyloidosis, have been reported in animals. 2 However, systemic amyloidoses other than AA amyloidosis, such as amyloid light-chain amyloidosis and amyloid transthyretin amyloidosis, which are major systemic amyloidoses in humans, are rare in animals. In the Felidae family, AA amyloidosis has been the only reported systemic amyloidosis. 2 AA amyloidosis is well-known and hereditary in Siamese and Abyssinian cats, but sporadic cases in other breeds are not uncommon. 3,7,12 In AA amyloidosis–affected cats, amyloid deposits are most severe in the liver and kidneys, and they often cause liver rupture. 1,5,15,20
The Tsushima leopard cat (Prionailurus bengalensis euptilurus) is an endangered species that inhabits Tsushima Island in Japan. Only brain Aβ amyloidosis has been previously reported in Tsushima leopard cats, 4 and there has been no report of systemic amyloidosis in these cats. This study describes the pathology and molecular findings in systemic amyloidosis in a Tsushima leopard cat in which epidermal growth factor–containing fibulin-like extracellular matrix protein 1 (EFEMP1) was identified as an amyloidogenic protein.
Materials and Methods
Case Information
A 15-year-old captive female Tsushima leopard cat showed clinical signs of anorexia and lethargy for 4 days before it died in a Japanese zoo. To determine the cause of death, necropsy and diagnostic analyses were performed.
For gene sequence analyses, skeletal muscle tissues from two other Tsushima leopard cats were obtained from a wildlife conservation center in Japan. Both cats were wild female individuals that died in a car accident; one was a young adult, and the other was a juvenile. Histopathological analyses showed that both cases were negative for amyloid deposition (data not shown).
Histopathologic and Ultrastructural Analysis
At necropsy, the liver, spleen, heart, lung, kidney, thyroid gland, adrenal gland, stomach, small and large intestine, pancreas, salivary gland, urinary bladder, ovary, uterus, skeletal muscle, cerebrum, cerebellum, medulla oblongata, and spinal cord were collected and fixed in 10% neutral-buffered formalin. Formalin-fixed paraffin-embedded tissues were prepared and cut into 3-µm sections, and stained with hematoxylin and eosin or Congo red. Amyloid deposition was confirmed using Congo red–stained specimens under polarized light.
Formalin-fixed heart tissues were postfixed, embedded in resin, cut into 70-nm sections, and stained as described previously. 14 The stained sections were examined with a JEOL JEM-1400 electron microscope at 80 kV (JEOL, Tokyo, Japan).
Proteomic Analysis
To detect candidate amyloidogenic proteins, laser microdissection followed by liquid chromatography-tandem mass spectrometry (LC/MS/MS) was performed. Congo red–positive amyloid deposits were collected from formalin-fixed paraffin-embedded specimens of the thyroid gland using laser microdissection (PALM MicroBeam; Carl Zeiss AG) and digested with trypsin, then subjected to LC/MS/MS analysis as described previously. 8,9 The resulting MS/MS data were correlated to theoretical fragmentation patterns of tryptic peptide sequences from the NCBI database using Mascot Server (Matrix Science Inc). Statistically significant proteins/peptides were extracted by Mascot’s probability-based scoring algorithm.
Gene Sequencing and Analysis
Total RNA was extracted from the skeletal muscle tissues of the present amyloid-positive case and the 2 amyloid-negative cases described above. 10 Primer were sequences designed with Primer Blast in NCBI based on the mRNA sequences of Felis catus EFEMP1 from the GenBank database (accession: XM_003984015; Supplemental Table S1). One-step RT-PCR (SuperScript, Invitrogen) followed by 5′ and 3′ rapid amplification of cDNA ends were performed as described previously. 10 Nucleotide sequences were determined using the BigDye Terminator v3.1 Cycle Sequencing Kit with an ABI Prism 3130 (Applied Biosystems).
The predicted amino acid sequence of EFEMP1 in Tsushima leopard cats was obtained by translating the open reading frame of the nucleotide sequence of EFEMP1 using EMBOSS Transeq. 16 Multiple sequence alignment of the amino acid sequences of EFEMP1 of Tsushima leopard cat, domestic cat (UniProt accession number: M3VVE6), and human (UniProt accession number: Q12805) was analyzed by Clustal Omega. 18
Immunohistochemistry
Immunohistochemistry (IHC) was performed with the following commercially available polyclonal antibodies: anti-human SAA (PAA885Hu01, Cloud-Clone Corp), anti-human transthyretin (TTR, PAA726Hu01, Cloud-Clone Corp), anti-N-terminal region (antigen: Tyr9-Val196) of human EFEMP1 (EFEMP1-N; PAF422Hu02, Cloud-Clone Corp), anti-C-terminal region (antigen: Cys241-Phe476) of human EFEMP1 (EFEMP1-C, PAF422Hu01, Cloud-Clone Corp), anti-kappa light chain (A80-111A, Bethyl Laboratories), and anti-lambda light chain (RB-334-A0, Thermo Scientific). Horseradish peroxidase–labeled anti-rabbit antibodies (Dako) were used as the secondary antibodies, and positive reactions were visualized by 3,3′-diaminobenzidine tetrahydrochloride staining. For all antibodies, positive reactions in normal tissues (liver, spleen, or skeletal muscle depending on each antibody) of Tsushima leopard cats were demonstrated (data not shown). For the negative controls, the primary antibody was omitted.
Results
Pathologic Findings
At necropsy, the animal weighed 3.5 kg, was in good nutritional condition, and had no abnormal appearance. The liver was mildly enlarged. The thoracic cavity was filled with clear red pleural fluid, and fibrin deposits were observed on the pulmonary pleura. In the heart, the left ventricular wall was severely thickened, and the lumen was narrowed. There was diffuse yellow-white discoloration of the subendocardium. No significant changes were observed in the valve membrane.
Histologically, amyloid deposits were observed in arterial walls and interstitial tissue throughout the body (Fig. 1). Amyloid deposits were especially severe in the interstitium of the atrioventricular valve, intestinal submucosa, and thyroid interstitium (Figs. 2–4). Amyloid was present in blood vessel walls (especially arteries) in liver, spleen, kidney, heart, lung, salivary gland, and skeletal muscle, as well as the interstitium of the liver, epicardium, small and large intestine, salivary gland, urinary bladder, thyroid gland, skeletal muscle, and spinal cord (Supplemental Table S2). All amyloid deposits stained strongly positive with Congo red and showed strong green birefringence under polarized light as amyloid-specific reactions (Figs. 2–4). The yellow-white lesions of the endocardium observed at necropsy were fibrosis and amyloid deposits. In addition to amyloid deposition, myocarditis and myocardial degeneration, pleuropneumonia, and focal necrosis of the pancreas with fat necrosis were also observed.
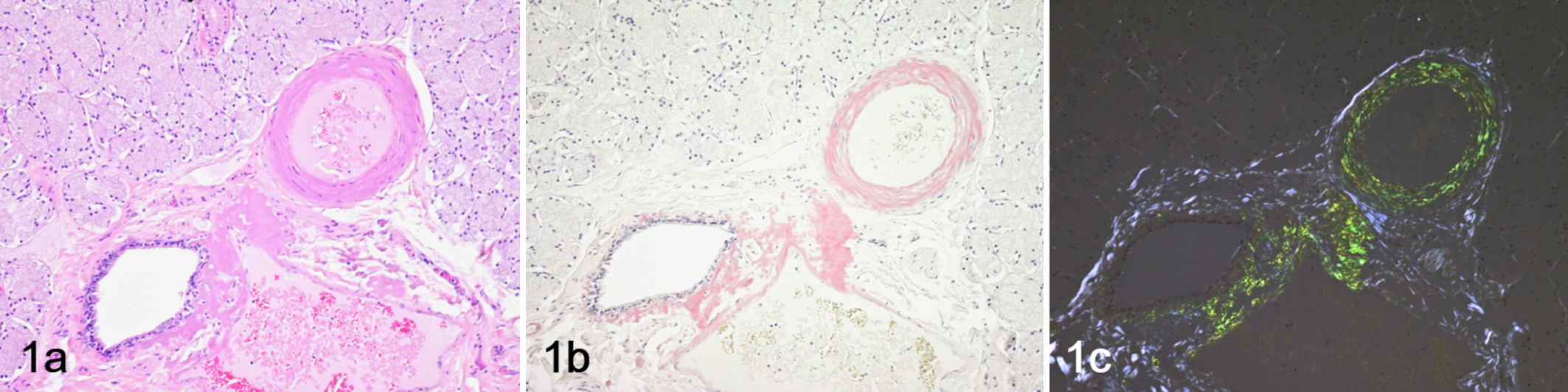
Amyloidosis, salivary gland, Tsushima leopard cat. (a) Hematoxylin and eosin stain. (b) Congo red stain. (c) Congo red stain under polarized light. Amyloid deposits are observed in the arteries and surrounding stroma.
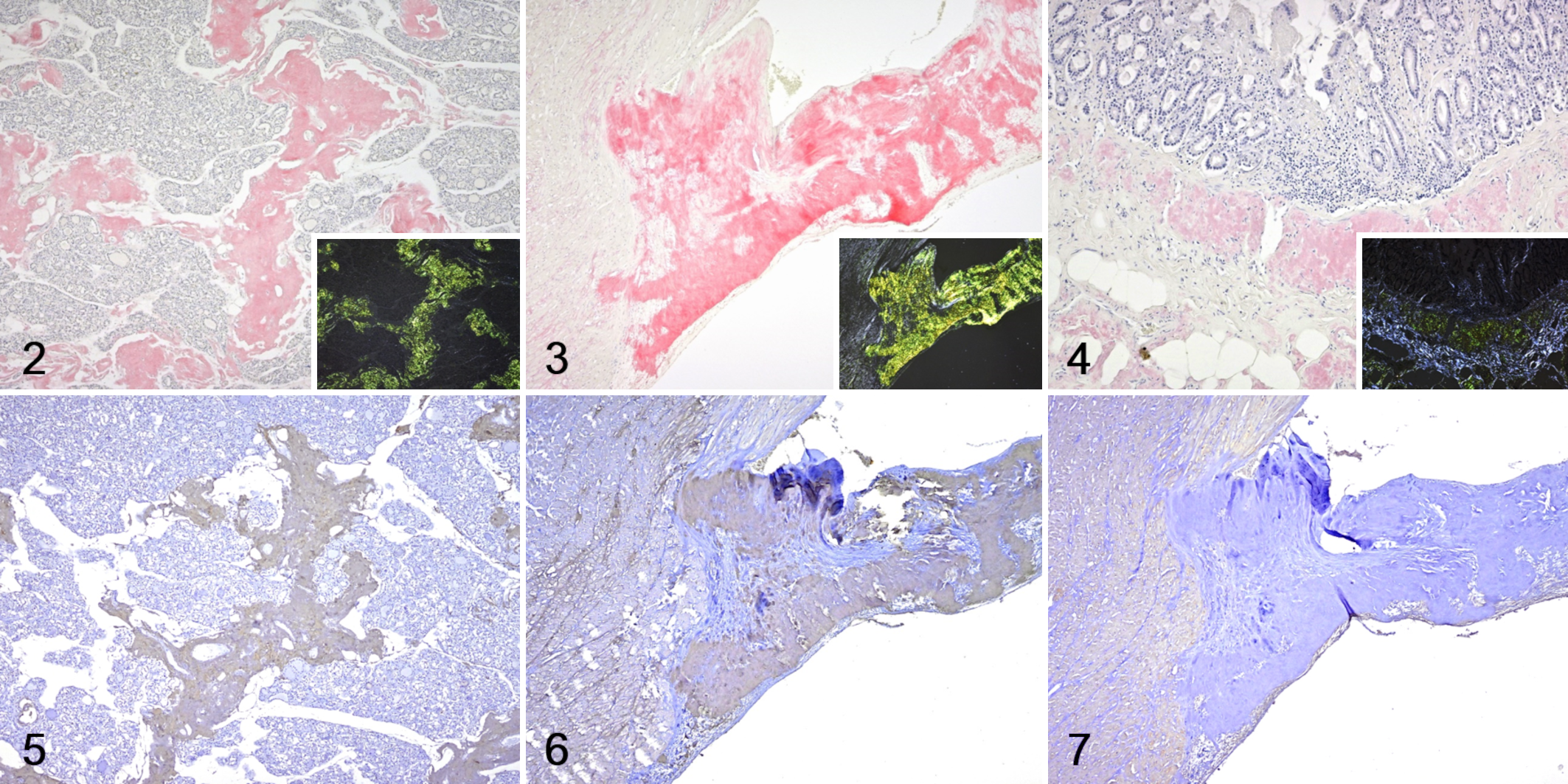
Amyloidosis, Tsushima leopard cat.
Ultrastructurally, straight, unbranched, solid, 12-nm-diameter fibrils were observed in the amyloid deposits. The fibrils were densely deposited like hay bales (Fig. 8).

Amyloidosis, heart, Tsushima leopard cat. The amyloid deposits contain straight and unbranched microfibrils of 12 nm in diameter that are arranged in dense bundles. Transmission electron microscopy. Bar = 200 nm.
Proteomic Analysis
Table 1 shows the proteins detected by laser microdissection followed by LC/MS/MS in the amyloid deposits of the thyroid gland. The amyloid signature proteins vitronectin, apolipoprotein E, clusterin, and apolipoprotein AI were detected as the 1st, 6th, 11th, and 13th most abundant proteins based on Mascot score, respectively. 12 These proteins also showed high scores on the exponentially modified protein abundance index (emPAI), which is a quantitative indicator in Mascot analysis. Collagen, serum albumin, thyroglobulin, and complement C3 were also detected, but they were considered to be non-amyloid proteins derived from the serum or the tissue component of the thyroid gland. However, EFEMP1, which is unlikely to be derived from the thyroid gland or serum, was also detected at a high level, and it also had a high emPAI score.
The proteins identified from the microdissected amyloid fragments by LC/MS/MS and Mascot analysis.

Abbreviation: emPAI, exponentially modified protein abundance index.
Gene Sequencing
Amplification of the Felis catus EFEMP1 gene by PCR (polymerase chain reaction) generated the predicted amino acid sequences shown in Supplemental Figure S1 for the 1 amyloid-positive case and the 2 amyloid-negative Tsushima leopard cats. No differences in the open reading frame of the EFEMP1 gene or in the translated amino acid sequence were detected between the amyloid-positive and amyloid-negative cases. The amino acid sequence of EFEMP1 of Tsushima leopard cats showed high identity to that of the domestic cat (F. catus). Although the whole amino acid sequence of EFEMP1 of Tsushima leopard cats showed relatively high identity to human EFEMP1, a region with low homology was seen in a part of the N-terminal region.
Immunohistochemistry
Immunohistochemistry revealed that the amyloid deposits were negative for SAA, TTR, kappa light chain, lambda light chain, and EFEMP1-C (Fig. 7). In contrast, the amyloid deposits showed clearly positive immunolabeling for EFEMP1-N (Figs. 5, 6).
Discussion
AA amyloidosis in cats is characterized by amyloid deposition mainly in the hepatic perisinusoidal space, renal glomeruli, and intestinal lamina propria. 1,5,7,15,20,22 Amyloid deposits in the present case, however, were observed mainly in arterial walls throughout the body. By IHC, the amyloid deposits were negative for SAA, TTR, and the kappa and lambda light chains. IHC also revealed that the amyloid deposits were positive for EFEMP1. These results indicate that the present case represents systemic amyloidosis from an amyloid precursor protein (EFEMP1) other than SAA.
EFEMP1 is member of the fibulin family of proteins, and it has an epidermal growth factor–like module and a fibulin module. 11 EFEMP1 is an amyloidogenic protein that has been reported in recent years; 3 papers have reported it in humans thus far. 6,19,21 This study is the first report of EFEMP1-derived amyloidosis in an animal. However, the present case had some characteristics that differed from the human cases. According to previous reports, 6,19,21 human EFEMP1-derived amyloidosis has the following characteristics: weakly positive Congo red staining and weak or no birefringence under polarized light; amyloid deposits are often localized only to the intestinal lamina propria; as for systemic deposition, amyloid fibrils are deposited in the venous walls, but not in the artery, and deposition is not seen in the heart; and TEM analysis reveals sparse fine fibers. These characteristics of human EFEMP1-derived amyloidosis differ significantly from those of the present case.
In this study, the amyloid deposits were strongly positive for EFEMP1-N, but negative for EFEMP1-C. On the other hand, in human EFEMP1-derived amyloidosis, amyloid fibrils contained the C-terminal region of EFEMP1. 19 In human AA amyloidosis, AA amyloid is derived from C-terminally truncated SAA. Interestingly, the phenotype of amyloid deposition pattern changes depending on the truncation site of SAA. SAA(1-76), which is the most typical AA protein, is characterized by amyloid deposits in the glomerulus, whereas SAA (2-45) and SAA (2-85) cause amyloid deposits in the renal medulla and vessel walls instead of glomeruli. 17 It is possible that the difference in the pathological features between humans and Tsushima leopard cats resulted from the difference in the amyloidogenic region. In the amino acid sequences of Tsushima leopard cat and human EFEMP1, there is a region with low homology in a part of the N-terminal sequence. Therefore, the difference in the lesions between humans and Tsushima leopard cats may be due to low homology in the N-terminal region of EFEMP1.
In this study, no differences in the amino acids of EFEMP1 were seen between the amyloid-positive case and the amyloid-negative cases, so amino acid mutations do not appear to have been an amyloidogenic factor. In human EFEMP1-derived amyloidosis, aging has been suggested to be a risk factor. 19 Therefore, it is possible that EFEMP1-derived amyloidosis in Tsushima leopard cats is a geriatric disease. Furthermore, EFEMP1 is highly conserved in the Felidae family, so it is possible that similar disorders may occur in other feline species. The present case did not show clinical signs that may have resulted from the amyloid deposits despite severe amyloid deposits in systemic organs. The incidence of EFEMP1-derived amyloidosis is high in elderly humans, but it is rarely diagnosed since it is asymptomatic. 6,19 Therefore, like in humans, EFEMP1-derived amyloidosis may also be overlooked in members of the Felidae family.
In conclusion, we report EFEMP1-derived amyloidosis in a Tsushima leopard cat as a non-AA systemic amyloidosis. It is interesting that the pathology of EFEMP1-derived amyloidosis differed between humans and the Tsushima leopard cat even though it is caused by the same amyloidogenic protein. Comparative pathological studies widely targeting animals are useful for understanding or predicting human diseases. This study contributes to better understanding of the relationship between the amino acid sequence-dependent amyloidogenicity and pathogenesis.
Supplemental Material
Supplemental Material, sj-pdf-1-vet-10.1177_03009858211048650 - Systemic amyloidosis derived from EFEMP1 in a captive Tsushima leopard cat
Supplemental Material, sj-pdf-1-vet-10.1177_03009858211048650 for Systemic amyloidosis derived from EFEMP1 in a captive Tsushima leopard cat by Shinya Miyazaki, Yuki Kobayashi, Fuyuki Kametani, Kyoko Kobayashi, Susumu Iwaide, Tokuma Yanai and Tomoaki Murakami in Veterinary Pathology
Footnotes
Acknowledgements
We are greatly indebted to Prof Satoshi Nakaba (Tokyo University of Agriculture and Technology) for technical assistance with the TEM analyses. We also greatly appreciate the helpful information from Prof Takuya Itou (Nihon University).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was partially supported by JSPS KAKENHI (Grant Number 20K15660) and the TUAT president’s discretionary funding to support the launch of next-generation research projects.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
