Abstract
European gypsy moths (Lymantria dispar dispar) are an invasive species in North America, and are listed by the International Union for the Conservation of Nature as one of the 100 most destructive invasive species worldwide. They have several known viruses, some of which are used as biological control agents. However, there are no detailed descriptions of many entomopathogenic viral infections, including in European gypsy moths, using bright-field microscopy. In this study, 11 European gypsy moth caterpillars were evaluated histologically: 4 were experimentally infected with Lymantria dispar multicapsid nucleopolyhedrovirus (LdMNPV; Baculoviridae); 4 were experimentally infected with Lymantria dispar cytoplasmic polyhedrosis virus (LdCPV; Reoviridae); 3 control animals were uninfected. A complete tissue set was evaluated in all animals from all groups using bright-field microscopy, including epidermis, cuticle, striated muscle, tracheae, foregut, midgut, hindgut, Malpighian tubules, hemocytes, fat body, and nervous system. LdMNPV-infected caterpillars had marked karyomegaly and intranuclear viral inclusions in cells of the epidermis, tracheae, fat body, and hemocytes. LdMNPV-infected caterpillars also had hyperplasia and hypertrophy of epidermal and tracheal epithelial cells. LdCPV-infected caterpillars had numerous granular eosinophilic intracytoplasmic viral inclusions in midgut epithelial cells. Both LdMNPV-infected and LdCPV-infected caterpillars had atrophy of fat body adipocytes; this change was more pronounced in LdCPV-infected caterpillars. This work provides the first detailed descriptions of these viral infections in European gypsy moth caterpillars using bright-field light microscopy and provides images of normal histology from control caterpillars.
Keywords
Entomopathogenic viruses include at least 11 viral families and are named according to the host’s scientific name and the viral family (eg, Lymantria dispar cytoplasmic polyhedrosis virus is named LdCPV). 5 Three families of entomopathogenic viruses can form occlusion bodies (also called inclusion bodies, see below; Fig. 1), including Baculoviridae, Reoviridae, and Poxviridae. Nucleopolyhedroviruses (NPV) are common entomopathogenic viruses from the family Baculoviridae. They have dsDNA, form polyhedral or granular occlusion bodies, and have enveloped, rod-shaped viral particles. Nucleopolyhedroviruses are classified as single NPV or multiple NPV referring to the presence of one (SNPV) or multiple (MNPV) virions per envelope (Fig. 2). 5 Due to high host specificity, easy host-to-host transmission, and high pathogenicity, some NPVs have been developed for microbial control of pest species. 2 The primary environmental impact of microbial control with NPVs is reduction of pest species, though there are occasional negative impacts on populations of nontarget, beneficial insects (especially lepidopterans). Natural NPV epizootics are recorded in many insect species. Vertebrates can facilitate transport of NPVs via consumption and defecation of virus-infected insects. 13 Cypoviruses (CPV) are from the family Reoviridae. 6 They have dsRNA and icosahedral viral particles occluded within polyhedra up to 10 µm in diameter. 1,4 Cypoviruses have a broader host range than NPVs to include dipterans and hymenopterans, and less commonly cause epizootics. Due to their broad host range and only moderate pathogenicity, few CPVs have been developed for biological control. 4,5
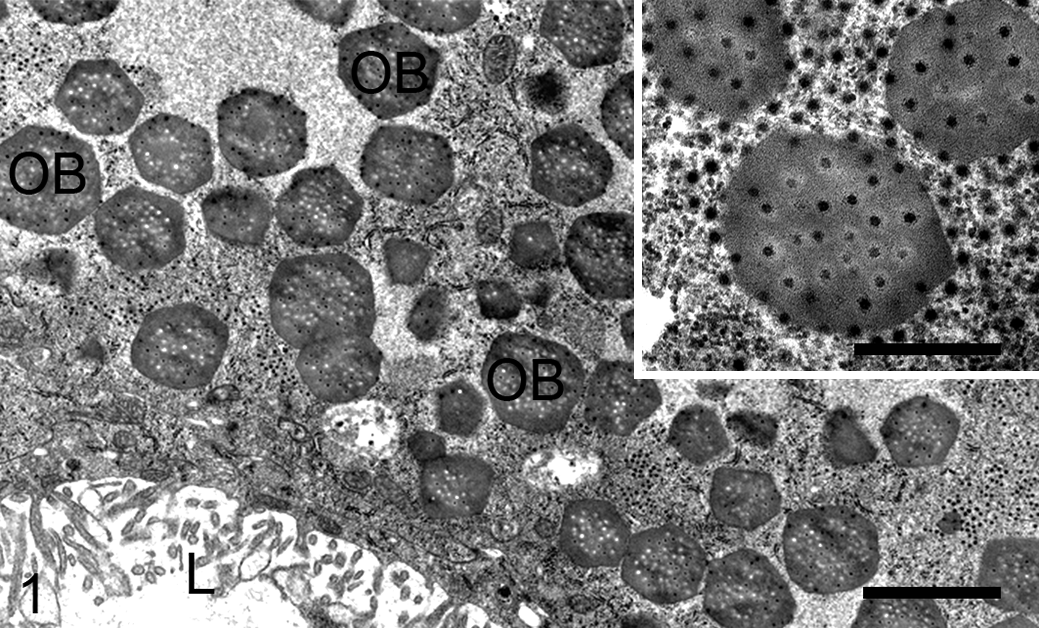
Lymantria dispar cytoplasmic polyhedrosis virus infection, midgut, European gypsy moth caterpillar. Occlusion bodies (OB) fill the cytoplasm of an epithelial cell and contain numerous viral particles (inset). The midgut lumen (L) is lined by microvilli extending from the apical surface of the midgut epithelial cell. Transmission electron microscopy (TEM). Scale bar, 2 µm; inset scale bar, 500 nm.
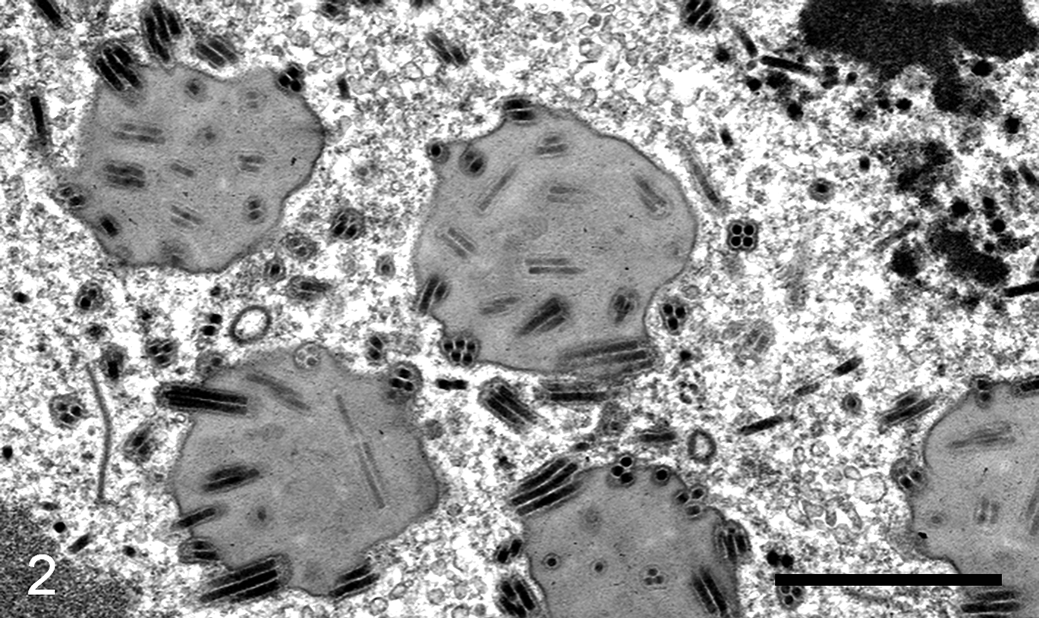
Lymantria dispar multicapsid nucleopolyhedrovirus infection, fat body, European gypsy moth caterpillar. In an adipocyte nucleus, rod-shaped viral particles are present and form multiple nucleocapsids per envelope within each occlusion body in the nucleoplasm. TEM. Scale bar, 1 µm.
While in-depth work has been done on the genome, phylogeny, evolution, transmission, pathogenesis, and ultrastructural features of CPV and NPV, histologic studies using bright field microscopy are lacking. 1,4 As the field of veterinary pathology is constantly expanding and more pathologists are examining invertebrate species, studies detailing histologic lesions of insects using bright field microscopy are needed. 14,16 The objective of this work is to describe the histologic lesions associated with 2 common viral infections (ie, LdMNPV and LdCPV) in European gypsy moth caterpillars (Lymantria dispar dispar). The aim of this work is to create reference material for veterinary pathologists examining insects by bright field light microscopy.
There are multiple, in-depth publications defining terminology used in insect pathology. 7,15,18,19,20 Some unique terms used when describing entomopathogenic viral infections are given below. After infection and reproduction within the host, certain viruses produce occlusion bodies, which are aggregates of proteins and virions; inclusion bodies (which is used synonymously with occlusion bodies in this article) are analogous to occlusion bodies, but the former is used more commonly in human and veterinary pathology. 7 Occlusion bodies are formed within an infected cell and contain infectious viral particles. 15 This resistant stage persists in the environment to infect additional insect hosts. Polyhedrin is a crystalline protein that surrounds virions to form an occlusion body. Polyhedron (plural: polyhedra) is an occlusion body containing polyhedrin; “polyhedron” is sometimes used interchangeably with “occlusion body.” 4,15 Viruses that form polyhedra in the nucleus are referred to as nucleopolyhedroviruses and those that form polyhedra in the cytoplasm are referred to as cytoplasmic polyhedrosis viruses. 4,15
European gypsy moths are lepidopteran insects that are invasive to North America. This species is listed by the International Union for the Conservation of Nature as one of the 100 most destructive invasive species worldwide. 11 Their infamy is due to a remarkable ability to defoliate large areas of native forests. 3
To conduct these studies, recently hatched L. dispar dispar of the New Jersey Standard Strain were obtained under APHIS (Animal and Plant Health Inspection Service) permit, from a USDA laboratory colony (USDA APHIS, Plant Protection and Quarantine, Buzzards Bay, MA). All subsequent rearing was conducted at 23 °C, 65% relative humidity, and 14:10 light–dark. The larvae were reared on gypsy moth diet (Southland Products Inc) in groups of 10 larvae within plastic cups (6.7 cm diameter × 5.1 cm height) with clay-coated paper lids. LdMNPV was the wild-type Hamden, CT strain registered as Gypchek by the USDA Forest Service. 17 LdCPV was originally isolated from L. dispar dispar in China and was obtained from the Chinese Academy of Forestry, Beijing, China. At 24 to 48 hours after molting to the third instar, larvae were transferred into cups of artificial diet overlaid with 1 ml of 5 × 106 occlusion bodies/ml. Larvae were allowed to feed for 2 days and were then transferred to noninoculated diet, and euthanized 5 to 7 days post inoculation. For controls, the diet was not inoculated with virus.
Caterpillars from 3 treatment groups (4 LdMNPV-infected, 4 LdCPV-infected, and 3 uninfected controls) were euthanized in formalin. Longitudinal and transverse sections of all caterpillars were processed routinely into paraffin, sectioned at 5 µm, and stained with hematoxylin and eosin. The following tissues were examined histologically in all cases and controls: epidermis, cuticle, striated muscle, tracheae, foregut, midgut, hindgut, Malpighian tubules, hemocytes, fat body, and nervous system.
LdMNPV-infected caterpillars had marked karyomegaly and intranuclear inclusions (also called occlusion bodies) in >75% of epidermal epithelial cells, 25% to 75% of tracheal epithelial cells, >75% of fat body adipocytes (also called trophocytes), and 10% to 50% of hemocytes. Nuclei of these cells were swollen up to 50 µm in diameter and frequently had margination of chromatin with numerous, globular, refractile, pale eosinophilic inclusions, and one to multiple, prominent, basophilic nucleoli that were up to 15 µm diameter (Fig. 3; compare to control Fig. 4). Epidermal and tracheal epithelial cells were diffusely enlarged with increased, pale eosinophilic cytoplasm, and were hyperplastic, forming clusters of piled cells up to 10 cell layers thick (Fig. 5; compare to control Fig. 6). Epidermal epithelial cells at the junction with the cuticle were occasionally spindloid (Fig. 7, compare to control Fig. 8). Although fat body adipocytes were enlarged due to karyomegaly, they had reduced cytoplasm (interpreted as atrophy) and a resultant increased nucleus: cytoplasm ratio. Hemocytes with inclusions were enlarged up to 16 µm in diameter and control hemocytes were up to 10 µm in diameter. The cuticle in affected animals subjectively had less frequent layering (ie, epicuticle, exocuticle, endocuticle) and rare foci of melanization; not all regions of the cuticle had discrete layers in either control or affected animals. Cells comprising the striated muscle, Malpighian tubules, foregut, midgut, hindgut, and nervous system were unremarkable in LdMNPV-infected and control animals.
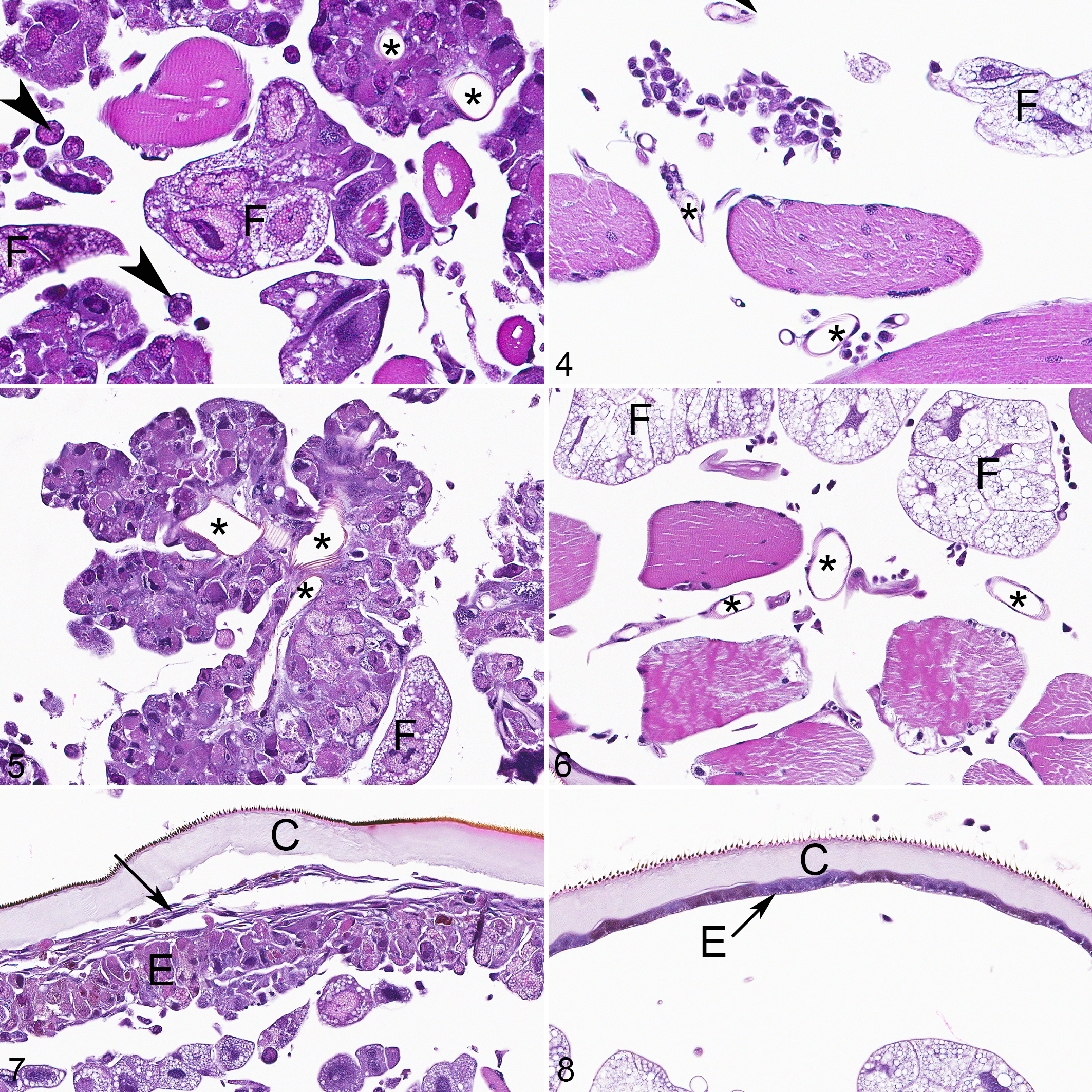
European gypsy moth caterpillars. Arrowheads, hemocytes; asterisks, tracheae lumina; C, cuticle; E, epidermis; F, fat body. Hematoxylin and eosin (HE).
LdCPV-infected caterpillars had hypertrophy and numerous intracytoplasmic inclusions of midgut epithelial cells. Cytoplasmic inclusions were numerous (ie, dozens per cell), approximately 1 to 2 µm in diameter, roughly spherical, brightly eosinophilic, and distributed throughout the cytoplasm but predominantly in the apical region (Fig. 9; compare to control Fig. 10). Height of midgut epithelial cells in the LdCPV-infected animals ranged from 65 to 230 µm, and in the control animals ranged from 20 to 100 µm. Fat bodies had markedly reduced cytoplasm, causing reduced size of individual adipocytes (Fig. 11; compare to control Fig. 12). Cells comprising the epidermis, cuticle, striated muscle, tracheae, foregut, hindgut, Malpighian tubules, hemocytes, and nervous system were unremarkable in LdCPV-infected and control animals.
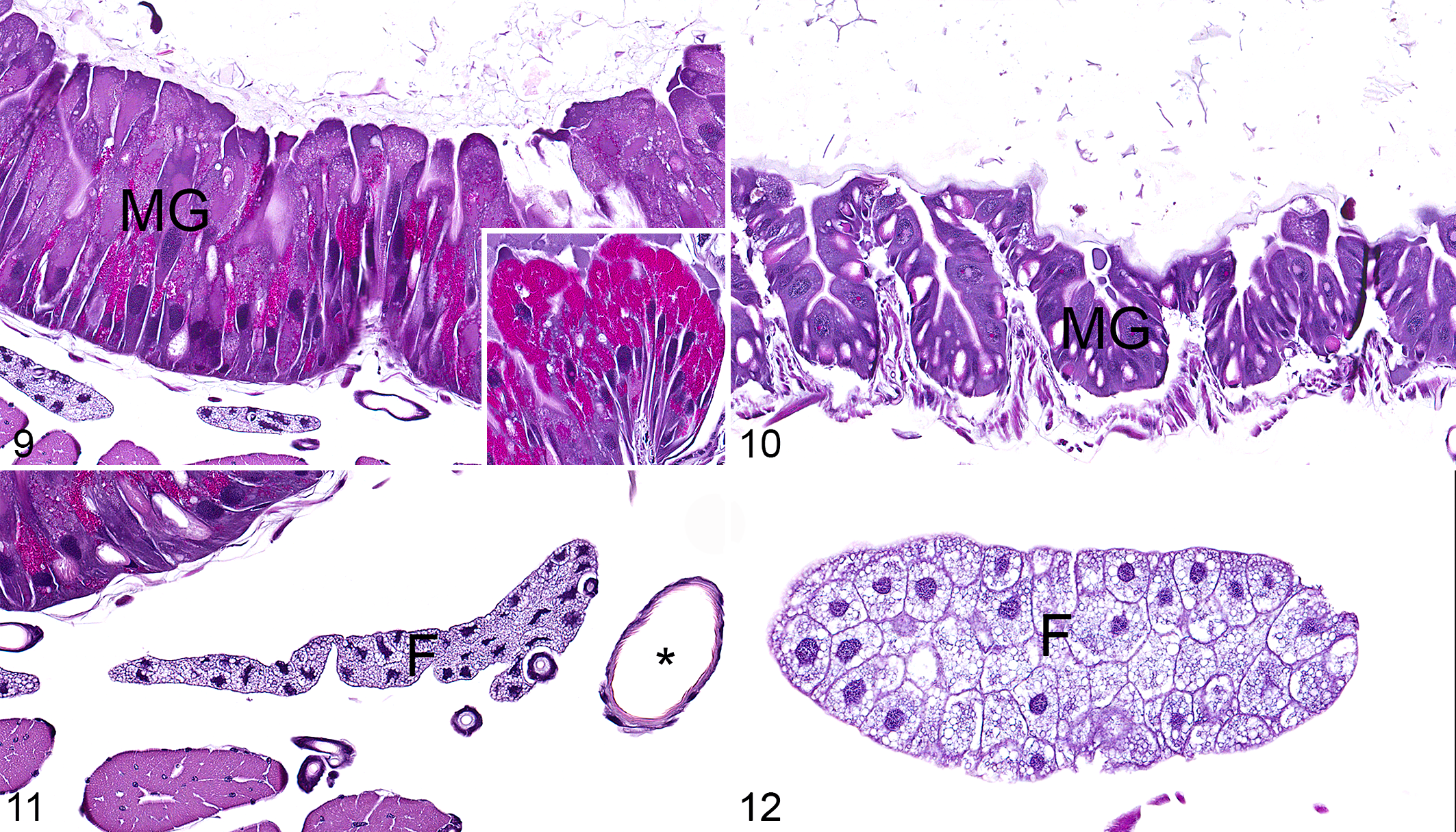
European gypsy moth caterpillars. Asterisk, tracheal lumen; F, fat body; MG, midgut epithelium. Hematoxylin and eosin (HE).
This study details histologic lesions of LdMNPV and LdCPV infections in gypsy moths, and provides control histology. LdMNPV is spread via ingestion, infects midgut epithelial cells, then enters the respiratory system, and uses tracheae to spread to other tissues throughout the body. 2 The key diagnostic feature of LdMNPV was granular, refractile, intranuclear inclusions with marked karyomegaly in multiple cell types. Histologic lesions of infection were widely disseminated throughout the epidermal epithelial cells and fat body adipocytes with fewer inclusions in tracheae epithelial cells and hemocytes. Interestingly, lesions were not observed within the midgut, which is the reported initial site of infection of LdMNPV. It is suspected that the midgut phase of infection is transient, and these animals were euthanized after virus left the midgut and was disseminated in other organs. LdMNPV has many virulence factors, including chitinase. 4 The caterpillars’ cuticle, which is composed primarily of chitin, had decreased layering, which may have been effects of both chitinase and infection causing dysfunction of epidermal cells. The terminal phase of LdMNPV infection is liquefaction of the host secondary to viral chitinase and proteinase.
The key diagnostic feature of LdCPV was granular, brightly eosinophilic, intracytoplasmic inclusions and hypertrophy in midgut epithelial cells. Histologic lesions of LdCPV were limited to midgut epithelial cells and fat body adipocytes, which were markedly reduced in size, interpreted as atrophy. Hypoplasia of the fat body was considered less likely given the short time frame between inoculation and euthanasia, and that animals did not undergo metamorphosis during the experiment. Generally, LdCPV infections are localized to the midgut in most species, and cause decreased growth and development of the host. 12 The midgut is the site of food digestion and absorption in insects. Unlike the foregut and the hindgut, the midgut lacks a cuticular barrier between ingesta and epithelium, allowing for nutrient absorption. 9 If the midgut is compromised (eg, by viral infection), this may lead to decreased nutrient absorption, and clinical sequelae of slowed growth/development. The function of the fat body is analogous to the vertebrate liver and adipose tissue. Insect fat body cells (also called adipocytes or trophocytes) metabolize a wide array of compounds, produce proteins, and store abundant lipid and glycogen, the latter two of which comprise approximately half of the fat body mass in a healthy insect. 10 Fat body atrophy in European gypsy moths caterpillars was speculatively considered the histologic correlate for reduced nutritional status secondary to viral infection and compromise of midgut epithelial cells.
The importance of histologic controls in invertebrate studies cannot be overstated. Invertebrates have unique anatomy and immune responses compared to vertebrates. For example, the effector cell of the insect immune system, the hemocyte, can phagocytose pathogens, similar to a macrophage, or form capsules and nodules, which are analogous to granuloma formation in vertebrates. These immune responses can be associated with melanization. 8 While encapsulation of infectious agents by hemocytes and formation of nodules of inflammation are relatively easy to identify for even a novice pathologist, subtler lesions may be missed without the use of control tissues. In this case, the lesions of cypovirus may have been difficult to appreciate without control specimens. The fat bodies of the affected animals appeared normal histologically with the exception of their reduced size. Additionally, the viral inclusions in the midgut epithelial cells are not typical of inclusions seen in vertebrate viral infections, and may have been mistaken as digestive granules without control tissues.
Footnotes
Authors’ Note
The views expressed in this article are those of the authors and do not reflect the official policy of the Department of the Army/Navy/Air Force, Department of Defense, or US government. The identification of specific products or scientific instrumentation does not constitute endorsement or implied endorsement on the part of the author, Department of Defense, or any component agency.
Acknowledgements
The authors wish to thank Adina Cummings Tasker and Efrain Perez-Rosario of the electron microscopy department at the Joint Pathology Center (JPC) for specimen preparation. Thank you also to the JPC technical and administrative support staff, including Ann Brown, Andrea Cherilus, Kenenya Gathers, Jessica Lewis, SFC Rafael Lozano, Steven Mcnair, Warren McNeil, and Stacey Tamer. Thank you to John Podwaite for detailed advice on viral infection.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
