Abstract
Peritumoral lesions identified during in vivo imaging of feline injection-site sarcoma (FISS) are frequently interpreted as neoplastic. We recently showed that most peritumoral imaging-identified lesions (PTIILs) in FISS are non-neoplastic. In this article, we describe a protocol to target PTIIL for microscopic examination and report on the protocol’s performance. Ten client-owned cats with FISS were prospectively enrolled. A fiducial marker sutured onto the skin, centered on the palpable mass, served as reference point throughout the study. Each FISS and surrounding tissue was imaged in vivo by dual phase computed tomography angiography and multiple magnetic resonance imaging pulse sequences and each PTIIL documented. Subgross measurements obtained during trimming aided localization and identification of PTIIL during microscopy. Histologic findings were categorized by descending clinical relevance: neoplastic, equivocal, non-neoplastic, within normal limits (WNL). Based on in vivo imaging resolution limits, histologic findings were ≥3 mm in at least one dimension and ≥3 mm apart. Surgical margins served as control tissue for PTIILs. Eighty-one of 87 PTIIL were examined histologically; 13 were neoplastic, 16 equivocal, and 28 non-neoplastic; 24 had no identified histologic correlate. Two neoplastic and 10 equivocal findings were located outside of PTIILs but none of them were located in sections of surgical margins. Computation of a simple confusion matrix yielded fair sensitivity (70.4%) and low specificity (59.7%) for prediction of PTIIL by histologic findings. After combining instances of normal microanatomy with non-neoplastic histologic findings, specificity increased (85.1%) and sensitivity decreased (35.8%). The protocol is a blueprint for targeting PTIIL for microscopic examination but may benefit from further refinement.
Keywords
Soft tissue sarcomas are common neoplasms in cats. 15 Soft tissue sarcomas developing in cats at sites of previous injection with vaccines or long-acting therapeutics grow rapidly and invasively and are often more aggressive than spontaneously developing soft tissue sarcomas. 35 Cancer treatment of single mass lesions relies heavily on surgery with the primary goal of complete excision. The invasive growth and deep infiltration of the surrounding tissue by feline injection-site sarcoma (FISS) can render the complete surgical excision challenging and often result in amputation and disfigurement. 9,16,33,36 Surgery may be deemed unfeasible if in vivo imaging identifies extensive tumor infiltration of surrounding tissues. The recurrence rate of FISS ranges from 15% to 20% even when wide (3–5 cm) surgical margins are achieved. 9,23 When surgical margins are deemed complete (free of neoplasia) based on histologic examination, it is undetermined whether recurrence is driven by unidentified residual disease, inability to unequivocally differentiate between neoplastic and reactive fibroblasts, field cancerization, intrinsic characteristics of the neoplastic cells, or other factors. 17,21,27,28,41 FISS outcomes may be improved by adjuvant or neoadjuvant therapies such as pre- or postsurgical radiation, 23 conventional chemotherapy, or treatment with tyrosine-kinase inhibitors, 13,24 with recombinant interleukin-2 immunotherapy holding the biggest promise. 18
We recently have shown that peritumoral lesions identified during in vivo imaging of FISS by computed tomography (CT) and magnetic resonance imaging (MRI) are predominantly non-neoplastic. 31 These results challenge the current practice of interpreting peritumoral imaging-identified lesions (PTIILs) of FISS as extensions of the tumor and, thus, neoplastic invasion. We also demonstrated that neither imaging technique by itself identified all peritumoral lesions in FISS and that neither dual-phase CT angiography nor multiple MRI pulse sequences alone or in combination can accurately predict which FISS-associated PTIIL represents neoplastic infiltration. 31
Ex vivo contraction and potential deformation of surgical samples is an important consideration in the design of a process that allows tracking of in vivo imaging findings to microscopic examination. It has long been suspected that the size of tumor margins changes from in situ to processed histology sections. Only recently have these changes been examined in more detail and rigor. 38,39 For example, lateral tissue margins of tumors were up to 50% smaller in postfixation imaging when compared to gross palpation in vivo. 39 Further margin reduction during processing from fixed tissues to histologic slides may occur. 39 Interestingly, a 54% increase was reported between depth measurements taken after the first incision into the normal feline skin of cadavers and measurements determined by microscopy. 19 However, these depth measurements, ranging from 1.3 mm to 3.9 mm, only included the epidermis and dermis. 19 We have previously shown that tissue shrinkage upon fixation of surgically removed FISS and surrounding tissue largely occurs parallel to the skin surface (x- and y-axis) and only to a small degree perpendicular to the skin surface (z-axis). 39
Our previous work focused on the histologic characterization of PTIIL and was based on the assumption that microscopic examination would capture and accurately represent all in vivo imaging findings. 31 To extend the work reported in prior publications, 31 this study describes the sample processing protocol in detail and provides a systematic investigation of its performance.
Materials and Methods
This study used data obtained from in vivo imaging as previously reported. 31,38,39 Some previously published data from microscopic examination 31 are included for comparison purposes. New data in this study include the detailed description of the trimming protocol, additional histopathology findings in the “non-neoplastic” category and from areas outside of PTIIL including the surgical margins, and data analyses to elucidate the performance of the developed trimming protocol. 31
Definitions
Trimming is used here to describe the process of cutting slices of fixed tissue samples for subsequent processing to paraffin blocks and histologic sections. Trimmed tissue slices were approximately 3 mm thick and in some cases subdivided into smaller segments to fit into processing cassettes (holding space 25 mm × 20 mm × 3 mm). For the purposes of this study, histologic findings are any microscopic features deemed relevant to the study by the study pathologist; more specifically, histopathologic changes or prominent (micro)anatomic structures identified during bright field microscopic examination of routine hematoxylin and eosin (H&E) stained slides. We defined peritumoral imaging identified lesions (PTIIL) as any structures in the tissues surrounding a FISS that were identified by a radiologist with in vivo imaging using CT, MRI, or both. Surgical margin sections are histologic sections generated from tissue slices trimmed to examine the physical margins of the surgical samples. Areas of particular interest to the surgeons were labeled with tissue ink in colors readily identifiable in histology slides; 29 by default, surgical margin sections did not include PTIIL. We defined non-PTIIL as the histologically examined areas outside of identified and labeled PTIIL. More specifically, non-PTIIL comprised surgical margin sections whose longest dimension exceeded 20 mm and any other tissues with histologic findings located in areas outside a 3 mm buffer surrounding the coordinates of PTIIL.
Cases
For this descriptive, cross-sectional, method-comparison study, 10 cats were enrolled between March 2012 and March 2015 with the approval of the Institutional Animal Care and Use Committee (Oregon State University) and the owners’ consent, as previously reported. 31,38 Briefly, inclusion criteria were that cats presented to the Lois Bates Acheson Veterinary Teaching Hospital of the Carlson College of Veterinary Medicine at Oregon State University (OSU-VTH) for surgical treatment of a cytologically or histologically diagnosed FISS. Cats that had received neoadjuvant therapy or prior surgical treatment of the FISS were excluded from the study; thus, all cases were treatment-naïve. All enrolled cats received a single anesthetic episode, following protocols determined by the OSU-VTH Anesthesia Service, during which imaging and wide surgical excision were performed as described elsewhere. 31 A fiducial marker was sutured over the center of the palpable mass after induction of general anesthesia, followed by in vivo imaging of the tumor region. 31,37 –39 The marker remained in place during surgical treatment and throughout all further examinations and handling of the surgical specimen and served as reference point for all subsequent PTIIL-related documentation. 31
Imaging
Imaging of the mass, surrounding tissues, and regional lymph nodes was performed as previously described using dual-phase CT angiography and MRI. CT angiographic studies were conducted with a 64-detector helical CT scanner (Toshiba Aquilion 64) pre- and post-IV iodinated contrast medium (Iopamidol 300 mg I/mL IV; except in case 1) administration. 31 In brief, MRI was performed using a 1-Tesla scanner (GE Signa Horizon) and including the following sequences: transverse, dorsal, and sagittal STIR (Short Tau Inversion Recovery—TR 3000–3400 ms, TE 54–57 ms; TI 100 ms]) and transverse pre- and post-IV Gadolinium contrast medium administration (Magnevist, Gadopentetate Dimeglumine) T1-FS (T1 fat-saturated—TR 400–566 ms; TE 15 ms). The images from each modality were examined by 1 of 2 board-certified radiologists at time of surgical treatment. 31 The FISS mass was defined as a round, oval, or lobular soft tissue structure with a well-defined outer margin. Soft tissue structures in contact with the mass or in tissue surrounding the mass and with imaging characteristics similar to the mass but beyond the margins of the mass or different from that of the mass were defined as PTIIL. 31 Each PTIIL was assigned a unique identifier (eg, CTA1, CTA2, CTA3, etc; or MRI1, MRI2, MRI3, etc), and the location of each PTIIL was recorded using distance (in mm) from the fiducial marker (x- and y-coordinates) and depth from the skin surface (z-coordinates). For each FISS, all PTIIL were recorded in a schematic drawing and classified as reported previously. 31
Surgery
Surgery was performed by a board-certified small animal surgeon with the fiducial marker left in place as previously reported. 31,39 In brief, after surgical preparation including routine aseptic treatment with 4% chlorohexidine scrub, the marker was covered with an antimicrobial incision drape (Ioban, 3M). The palpable tumor margins and 4 cm to 5 cm circumferential lateral surgical margins were outlined using a sterile surgical pen. The tumor and associated tissues were removed “en bloc” with a minimum of 2 deep fascial planes with curative-intent. 33 In cases of skin and soft tissue excision, samples were fixed to cardboard backing with at least 4 sutures to avoid shifting of tissue planes during transport and fixation (Suppl. Fig. S1). In cases requiring hemipelvectomy, a compartmental excision approach was used while still obtaining 4 cm to 5 cm circumferential lateral surgical margins and a minimum of 2 tissue planes deep. 2 The resultant surgical samples included 3 resections of the lateral flank musculature, 2 of caudal hemipelvectomies, and single samples of complete hemipelvectomy, hind limb amputation via coxofemoral disarticulation, forequarter amputation, partial scapulectomy, and excision of the dorsal neck musculature including vertebral spinous process ostectomy (Table 1).
Signalment, Surgical Approach, Number of Peritumoral Imaging-Identified Lesions (PTIILs) Using Computed Tomography and Magnetic Resonance Imaging, and Histologic Findings From Areas Outside of PTIIL, From 10 Cats With Injection-Site Sarcomas.a
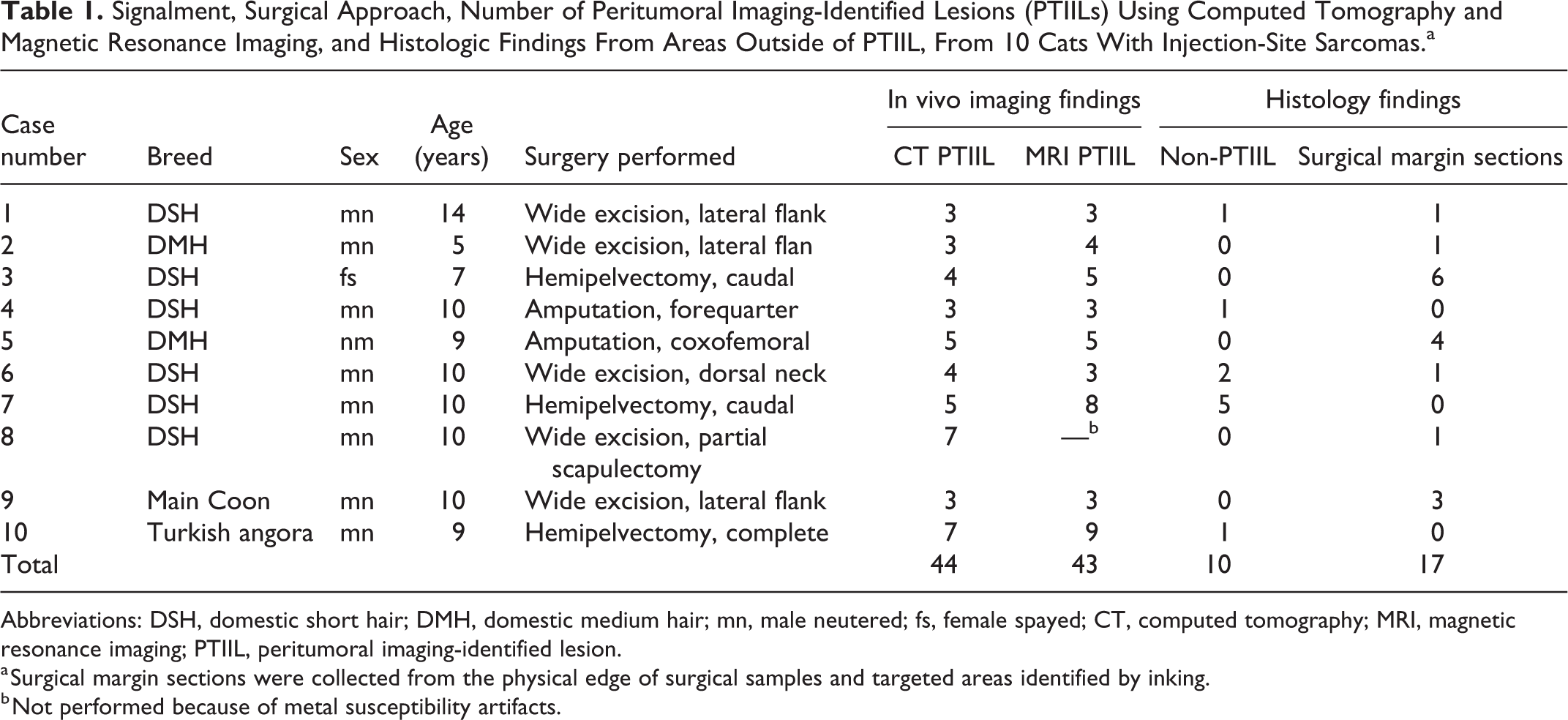
Abbreviations: DSH, domestic short hair; DMH, domestic medium hair; mn, male neutered; fs, female spayed; CT, computed tomography; MRI, magnetic resonance imaging; PTIIL, peritumoral imaging-identified lesion.
a Surgical margin sections were collected from the physical edge of surgical samples and targeted areas identified by inking.
b Not performed because of metal susceptibility artifacts.
Imaging Data Recording and Interdisciplinary Communication
Following the surgery, tissue dye was applied to surgical margins by 1 of 2 surgeons to aid with orientation of the sample in space (cranial and caudal, deep and either dorsal and ventral, or left and right) and to identify any areas of concern for complete surgical excision (Suppl. Fig. S1). The perimeter of the palpable mass was labeled by the surgeons by applying a yellow line (roughly circular) on the skin surface (Suppl. Fig. S1). 31,38,39 Then, the PTIIL recorded by radiologists were marked on the surgical sample: orange for CT-identified and black for MRI-identified PTIIL. 31 When a PTIIL was seen in the same location with both imaging modalities, the path was drawn on the skin in one color and a dot was placed at the peripheral boundary of the PTIIL in the other color (Suppl. Fig. S2). 31 Radiologists also recorded findings from in vivo imaging in schematic drawings (Suppl. Fig. S3).
Sample Trimming and Tissue Processing
In accordance with standard operating procedures of the Oregon Veterinary Diagnostic Laboratory, at least one representative section from each mass was trimmed and processed after 24 to 48 hours of fixation in formalin to ensure optimal preservation for diagnostic and archival purposes. Whenever possible, these tissues slices included at least 5 mm of the clinically identified tumor area (from within the yellow circle). The otherwise-intact surgical sample was fixed for 1 week. In case of amputated legs and hemipelvectomies, samples were re-imaged by CT immediately prior to trimming, as described elsewhere, 39 and imaging findings were reviewed and updated as needed by the primary radiologist on the case. The sample trimming for diagnostic purposes was performed using recommended standardized techniques. 20
Trimming and processing of fixed surgical samples for study purposes followed the same general guidelines and were conducted as detailed below and illustrated in Figure 1. The location and orientation of trimmed tissue slices were included in the trimming notes generated by the study pathologist, which included a gray scale print of photographs of the surgical sample and the surgical field after placement of the fiducial marker and prior to surgery (see Suppl. Fig. S4). One board-certified anatomic veterinary pathologist (CVL; study pathologist) performed all trimming for the study with exception of the surgical margin sections for 3 cases (cats 3, 4, and 5). For these cases, 1 of 3 board-certified anatomic veterinary pathologists trimmed a representative slab of the mass and the inked surgical margins for diagnostic purposes. This initial trimming left the fiducial marker and areas marked for PTIIL intact. The study pathologist confirmed the suitability of surgical margin sections trimmed for diagnostic purposes for inclusion in the study.
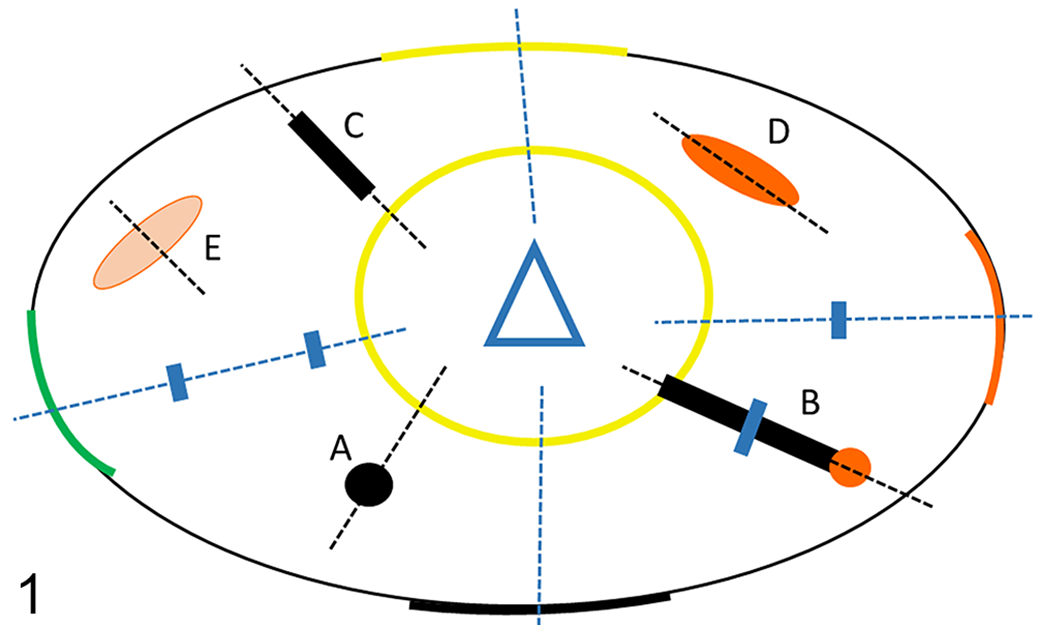
Schematic depicting cuts placed during trimming of feline injection-site sarcomas (FISS). Hatched blue lines indicate surgical margin sections. Small blue rectangles indicate subdivision of surgical margin sections to fit tissues into cassette chambers. Black hatched lines indicate placement of cuts to trim peritumoral imaging-identified lesions (PTIIL) for histologic examination. (A) Black dot. Discontinuous small spherical PTIIL on magnetic resonance imaging (MRI). (B) Black line ending in an orange dot. Continuous linear PTIIL on MRI and computed tomography (CT) perpendicular to sample margin. (C) Black line. Discontinuous linear PTIIL on MRI perpendicular to sample margin. (D) Orange ellipse. Discontinuous linear PTIIL (satellite) on CT not perpendicular to sample margin. (E) Pale orange ellipse. Deep (>30 mm from skin surface) discontinuous linear PTIIL (satellite) on CT not perpendicular to sample margin. Black ellipse: Physical margin of the surgical sample. Colored segments (yellow, orange, green, and black): Orientation of sample in space and inked margins of interest to surgeon. Yellow circle: Palpable tumor margin. Blue triangle: Fiducial marker.
For tissue processing, the cut surface of the tissue slice most relevant to the study was placed face down into the cassette, to be oriented face-up in the paraffin blocks by histology personnel. Tissue slices too large to fit into a single cassette were divided into multiple segments to fit into cassette chambers; care was taken to avoid division at reference points or prominent anatomic landmarks. Tissue slices in cassettes were left in formalin overnight and routinely processed to histologic sections in accordance with established standard operating procedures. From each paraffin block, a 3- to 5-µm section was obtained, stained with hematoxylin and eosin, and coverslipped. An example of the set of histology slides generated in trimming of the tumor, the surgical margins, and PTIIL is shown in Supplemental Figure S5.
Identification of PTIIL
Photographs taken before surgery and after inking of margins and marking of PTIIL served as reference and for further documentation during trimming (Suppl. Fig. S3). Individual orange and black markings were identified and labeled according to the reference numbers assigned during in vivo imaging (CT1, CT2, etc; MR1, MR2, etc). For complex surgical samples (amputated limbs and hemipelvectomies), the pathologist met with the radiologist to ensure that findings from in vivo imaging recorded in schematic drawings and notes were interpreted correctly. A ruler was used to measure the distance from the fiducial marker to the boundaries of each orange and black marking (closest to marker = central; furthest from marker = peripheral). When applicable, additional measurements were taken from the markings to anatomic landmarks such as fascial planes, bone, or boundaries of muscle bellies in any relevant 3-dimensional plane.
Trimming of sample margins and microchip sites
Trimming was performed to generate tissue slices oriented perpendicular to skin surface (vertical) whenever possible. Radial cuts (by default vertical) were oriented perpendicular to the physical (ie, lateral) sample margin (Fig. 1; Suppl. Fig. S4).
Sample margins
Margin cuts targeted areas inked by the surgeons. Trimming avoided the sutures of the affixed fiducial marker to leave the reference point intact until the completion of the study. Tissue slices spanned from the fiducial marker or the tumor bulk within the yellow circle to the inked sample margins and contained at least 15 mm of tissue extending beyond the palpable mass. Where inked sample margins included bone, the bone was removed from the sample with an approximately 5 mm rim of soft tissue attached, then it was decalcified in formic acid-EDTA, and a representative slice was trimmed perpendicular to the inked margin and processed.
Microchip
Two surgical samples (cases 6 and 8) contained a microchip. A slice containing the microchip and surrounding tissue was trimmed to cassette size, centered on the chip, and the chip was removed before tissue processing.
Trimming of PTIIL
A schematic representation and examples of the location and orientation of trimmed slices are provided in Figure 1 and Supplemental Figure S4, respectively. Radial cuts extended from the tumor bulk (from within the yellow circle) to 5 mm past the peripheral border of PTIIL markings. The shape of the PTIIL markings (which were either dots or lines) guided the trimming as follows.
For dots, the cut was placed through the center of the dot (Fig. 1A). For lines, the cut followed the long axis in the center of the line. For lines ending in a dot (to indicate overlapping linear CT and MRI findings), the cut extended through the center of the dot (Fig. 1B).
PTIIL that were discontinuous from the tumor (satellite PTIIL) were indicated by markings not connected to the tumor bulk (yellow circle). These were bisected along their long axis and perpendicular to the skin surface, irrespective of their orientation to the tumor or sample margin (Fig. 1C, D). PTIIL that were at least 30 mm deep to the skin surface (deep PTIIL) were elongate or linear, and they were trimmed perpendicular to their long axis to maximize likelihood that PTIIL were represented in tissue slices (Fig. 1E). Multiple reference points (eg, skin surface, bone, and fiducial marker) were used to locate deep PTIIL. Incisions to generate tissues slices were placed perpendicular to the skin surface whenever possible.
Measurements and data recording
For each trimmed tissue slice, the study pathologist drew a rough schematic to depict anatomic characteristics such as tumor boundaries, tissue planes, tissue types, and large vessels (Suppl. Fig. S4). The following measurements were recorded: overall length and height of the tissue slice; distances from the central edge of the tissue slice to the central and peripheral boundaries of each marking with the ruler aligned with the skin surface (eg, “to end of black line” or “yellow line to orange dot”); distances from the central edge of the tissue slice to the point(s) of subdivision into multiple segments to fit cassette chambers; or other relevant landmarks as described above. Cassette labels with the case-specific laboratory accession number and a running number for each cassette were recorded on the trimming schematic (Suppl. Fig. S4) and printed on histology slides (Suppl. Fig. S5).
Microanatomy and Histopathology
All histology slides were read by the study pathologist using a Nikon Eclipse E400 bright-field microscope. After tissue landmarks observed during trimming were aligned with microanatomic features present in histology slides, the location of all findings recorded up to that point were drawn onto the glass slides with fine-tipped pens using a ruler to aid with orientation (Suppl. Fig. S5). This included the boundaries of the palpable tumor bulk (yellow circle) and all in vivo imaging findings (orange and black markings on the skin surface corresponding the x- and y-coordinates; depth range corresponding to z-coordinates). The location of any deviation from normal microanatomy was marked with felt pen on the histology slides including the depth range (closest and furthest point from the skin surface). Their distance to the skin surface (z-coordinates) was measured digitally using a Nikon Digital Sight DS Fi1; 14 Mpixel camera, and the NIS-BR Elements imaging software package (v21.1; www.nikonmetrology.com).
The results from the microscopic examination were recorded in a Microsoft Office Professional Plus 2016 Excel spreadsheet by cross-referencing the PTIIL labels from MRI or CT, their qualitative imaging finding(s), and their depth range (z-coordinates). Each slide that did not correspond to a PTIIL identified by MRI or CT was designated “Histology” and listed on a separate row (Suppl. Table S1).
All deviations from normal microanatomy were assigned to 1 of 3 broad categories as previously reported. 31 Clearly identifiable atypical spindle-shaped to stellate cells in streams or bundles were categorized as “neoplastic.” Edematous and/or well-vascularized connective tissue with individual or small groups of plump fibroblasts were categorized as “equivocal.” All other histologic findings deviating from normal microanatomy were categorized as “non-neoplastic.” If no deviation from normal microanatomy was observed, the result was recorded as “within normal limits” (WNL).
Data Analysis
Given the limited number of observations, we combined findings from both imaging modalities for further analyses. In order to account for the differences in lower resolution limitation of the 2 imaging modalities (3 mm for MRI and 0.5 mm for CT), we decided to set a minimum size threshold for histologic findings. Histologic findings had to exceed the MRI resolution limit (3 mm) and a distance of at least the resolution limit from each other (3 mm) to be included in results and statistical analysis. We took a similarly conservative approach to identifying discrepancies between in vivo imaging and histologic findings by adding a 3 mm buffer zone to PTIIL in all recorded dimensions. We defined a histologic finding of a non-PTIIL as any histologic finding that fulfilled the size and distance parameters established above and that was outside the 3 mm buffer zone of the nearest PTIIL. Each histology slide generated to examine surgical margins was considered a separate location when the length of the tissue section (as measured parallel to the skin surface) was at least 20 mm. Histologic findings in surgical margin sections meeting this requirement were also designated as histologic findings of a non-PTIIL.
Multiple unique histologic findings present within the coordinates of the same PTIIL or non-PTIIL and histologic findings less than 3 mm apart were collapsed into a single data point for statistical data analyses and designated, in descending order, as neoplastic, equivocal, non-neoplastic, or WNL; within the “non-neoplastic” category in descending order as inflammation, vascular changes, increased collagen, or myositis.
Data were recorded in Microsoft Office Professional Plus 2016 Excel. Python 3.8.5 and the libraries Matplotlib 3.3.0, Seaborn 0.10.1, pandas 1.0.5, and numpy 1.19.1 were used to manipulate the data and generate pairwise plots for data visualization and examination for trends (not shown). To evaluate the performance of the protocol, a simple confusion matrix with 2 binary variables (PTIIL yes/no and histologic finding yes/no) was set up, and parameters of diagnostic accuracy were calculated. Results from in vivo imaging were used as the reference (true); results from histologic examination as prediction. As such, we defined PTIIL with any histologic findings (any histology category other than WNL) as true positives; surgical margins (by definition non-PTIIL) without histologic findings as true negatives; PTIIL without histologic findings as false negatives; and areas outside of PTIIL with histologic findings as false positives. Microsoft Office Professional Plus 2016 Excel was used for simple data processing (primary statistics) and assembly of simple confusion matrices. Parameters of accuracy were calculated using an online calculator (https://onlineconfusionmatrix.com/). Further statistical analyses were not performed because the size of the data set and number of variables (limited degrees of freedom).
Results
Cases
The 10 client-owned cats with FISS were prospectively enrolled into the study (Table 1). The breeds were domestic shorthair (n = 6), domestic medium hair (n = 2), Maine Coon (n = 1), and Turkish angora (n = 1). The cats had the following signalment: median age 9.8 years (range 5.3–14 years), median body weight 5.5 kg (range 3.8–7.8 kg); 1 spayed female, 9 neutered males.
Imaging
As previously reported, 31 PTIIL were identified in all 10 cases (range 3–12; average 8.7; median 7). Of 87 PTIIL total, 18 were identified by MRI only, 19 by CTA only, and 25 identified by both modalities (total of 62 unique lesions totaling 87 observations or PTIIL). Three cats had metal susceptibility artifacts on MRI (2 microchips in the periscapular tissues, 1 metallic projectile in the rear limb). Artifacts were focal in 2 cats (cats 3 and 6) and did not interfere with diagnostics and evaluation for PTIIL, but it was severe in cat 8, precluding the use of MRI to evaluate PTIIL in this case. Imaging characteristics were classified as described in detail elsewhere. 31
Histology
A total of 159 histology slides were generated. Examples of specific findings are provided in Figures 2 to 7. Histologic findings other than neoplastic (Figs. 2, 3) or equivocal (Fig. 4) included increased or abnormally distributed collagen; vascular changes such as increased vascularity, edema, and hemorrhage (Figs. 5, 6), and presence of extravascular leukocytes (Fig. 7). Eosinophilic myositis was present in one case (cat 3).
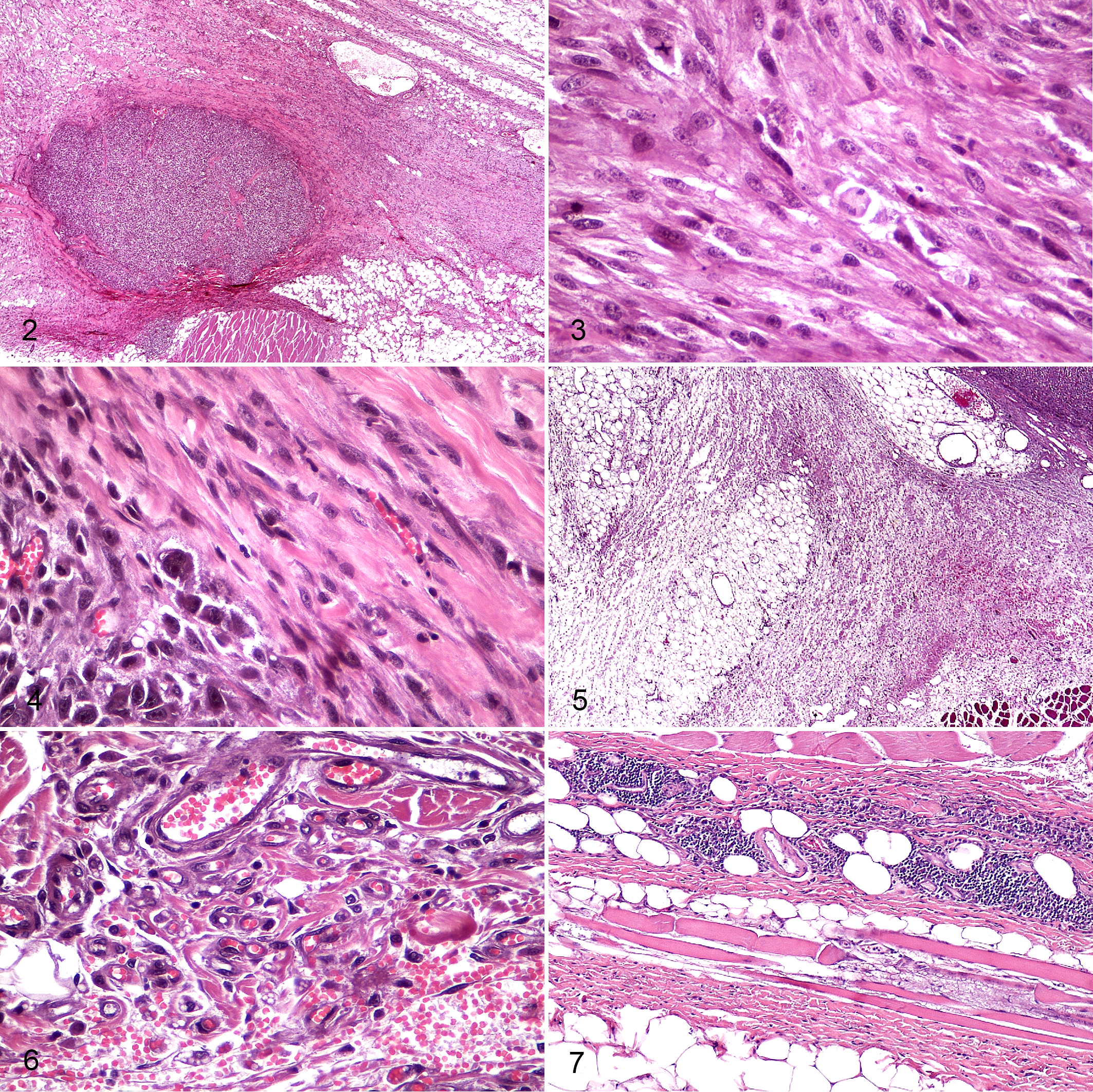
Feline injection-site sarcoma (FISS), skin, cat. Hematoxylin and eosin. The panel shows representative examples of histologic findings in peritumoral imaging-identified lesions (PTIILs).
Results from the microscopic examination are summarized in Table 2. For 6 of the 87 PTIIL, no histology slides were generated (Suppl. Table S1), either due to inconsistencies in data recording, miscommunication or corrections made to PTIIL coordinates by the radiologist after trimming of the sample, alone or in combination. These PTIIL were excluded from further analyses and graphic representations of the results. For 5 PTIIL, multiple histology slides were generated (n = 17; median 2.5) and yielded a total of 9 histologic findings (3 for 1 PTIIL, 2 each for 2 PTIIL, 1 for 1 PTIIL). For data representation and statistical analyses, multiple findings were collapsed to one data point for each PTIIL (neoplastic, equivocal, or non-neoplastic) as described below.
Histopathologic Findings in Areas Identified as Peritumoral Imaging-Identified Lesions (PTIIL) Related to Feline Injection-Site Sarcomas (FISS).
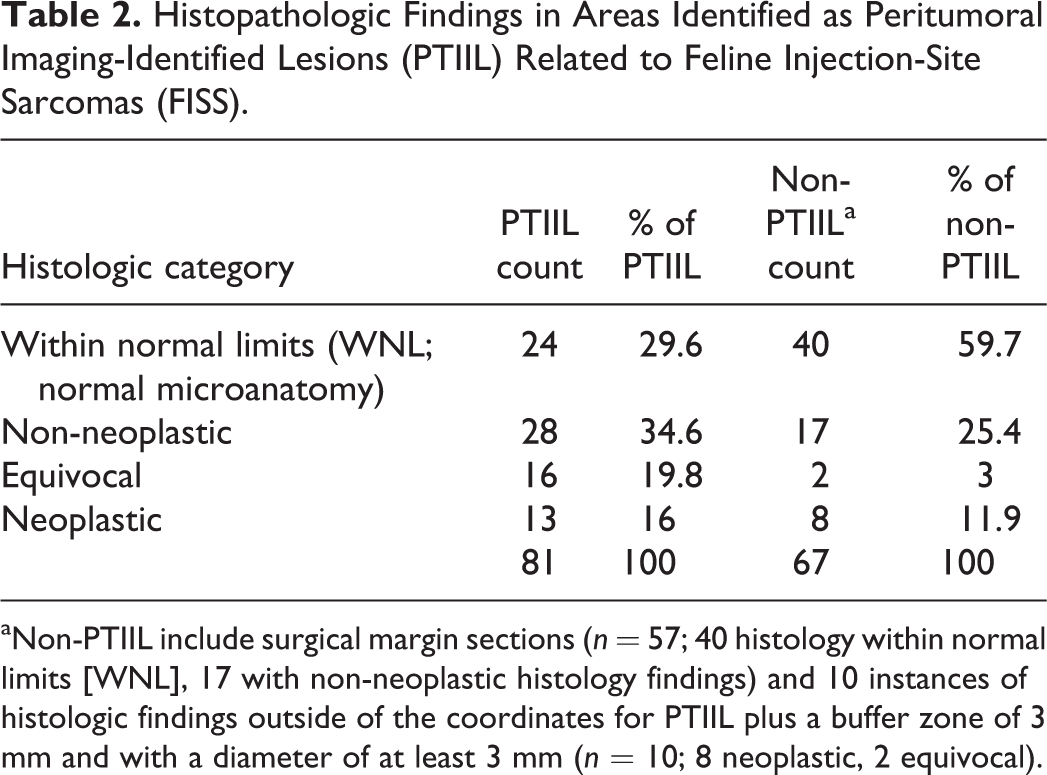
aNon-PTIIL include surgical margin sections (n = 57; 40 histology within normal limits [WNL], 17 with non-neoplastic histology findings) and 10 instances of histologic findings outside of the coordinates for PTIIL plus a buffer zone of 3 mm and with a diameter of at least 3 mm (n = 10; 8 neoplastic, 2 equivocal).
Trimming of surgical margin sections yielded 57 slides (Suppl. Table S1); all reached or exceeded the minimum of 2 cm in the longest dimension; thus, each was included as an individual data point. There were 17 histologic findings in surgical margin sections, all non-neoplastic, and each in a separate slide; all were included as “non-PTIIL with histologic findings” (Table 3, Fig. 8). Thirty-nine additional histologic findings were identified in tissues outside the coordinates recorded for PTIIL (neoplastic n = 8; equivocal n = 3; non-neoplastic n = 28 [inflammation n = 6; increased collagen n = 8; edema n = 3; hemorrhage n = 2; eosinophilic myositis n = 2; large vessel n = 7]). Application of the 3 mm buffer zone around PTIIL and the minimum size requirement of 3 mm eliminated 20 findings, leaving 19 (neoplastic n = 8; equivocal n = 2; non-neoplastic n = 9 [inflammation n = 3; increased collagen n = 1; edema n = 1; hemorrhage n = 1; myositis n = 1; vessel n = 2]). After collapsing multiple histologic findings for a given location and reporting the clinically most relevant category, 10 findings remained (8 neoplastic; 2 equivocal; Suppl. Table S1; Table 2). These 10 data points were included in “non-PTIIL with histologic findings” (Table 3, Fig. 8).
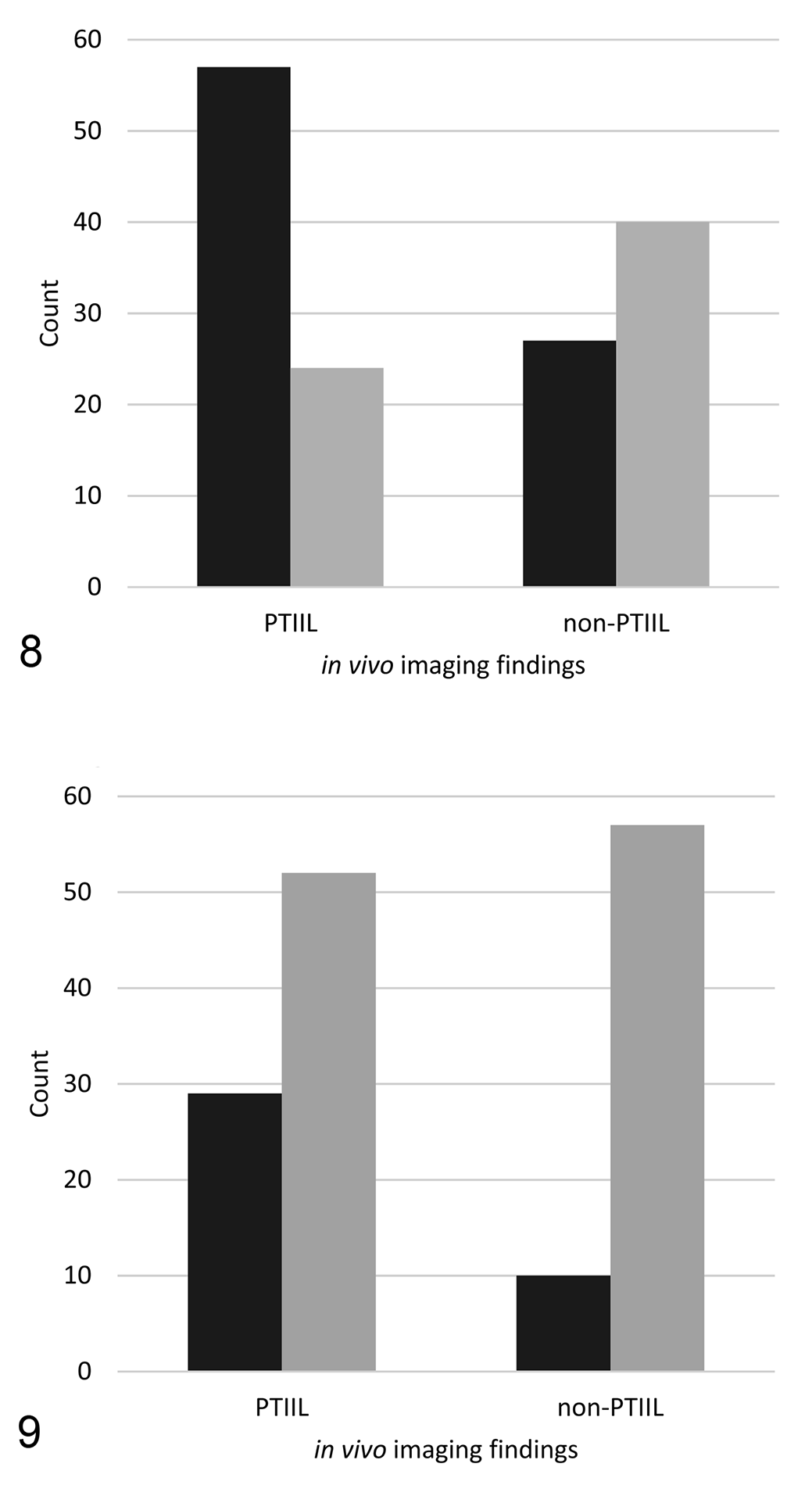
Results from histopathologic examination of peritumoral imaging-identified lesions (PTIILs) and surgical margin sections of feline injection site sarcomas (FISS). Non-PTIIL include surgical margin sections, as well as histologic findings from areas outside the 3 mm buffer zone (z-coordinates) surrounding a PTIIL and with a diameter of at least 3 mm in at least one dimension.
Histologic Lesions in Tissues Surrounding Feline Injection-Site Sarcomas in Areas With and Without Peritumoral Imaging-Identified Lesions (PTIILs).
a Total observations n = 148.
bNon-PTIIL include surgical margin sections (n = 57; 40 histology within normal limits [WNL], 17 with non-neoplastic histology findings) and 10 instances of histologic findings outside of the coordinates for PTIIL plus a buffer zone of 3 mm and with a diameter of at least 3 mm (n = 10; 8 neoplastic, 2 equivocal).
Data Analysis
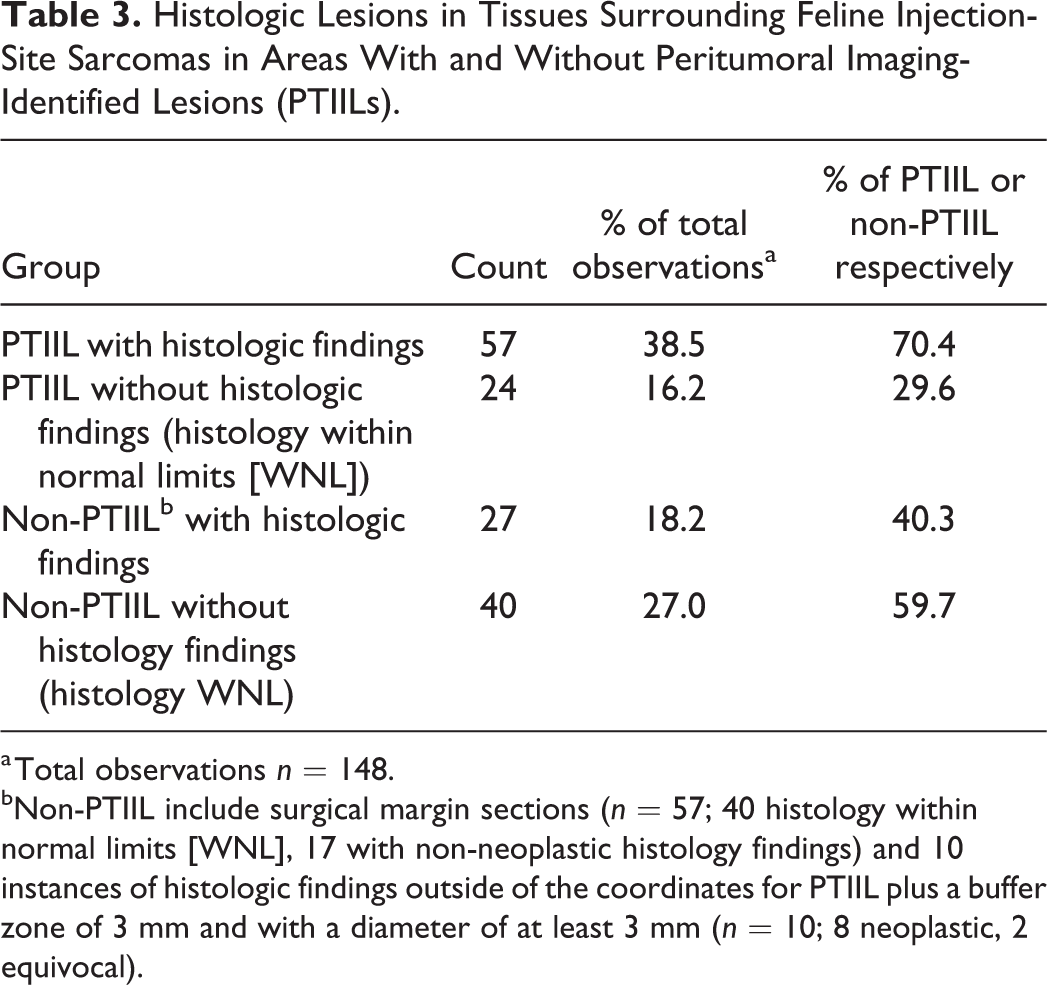
All 148 data points were included in the computation of the simple confusion matrices. The initial confusion matrix compared histologic findings of WNL to all other histologic findings combined (Fig. 8). The sensitivity of histologic findings for predicting in vivo imaging findings was 70.4%, specificity 59.7%, positive predictive value 67.9%, and negative predictive value 62.5%, with an accuracy of 65.5%, F1 score of 0.69, Matthews correlation coefficient of 0.3, and diagnostic odds ratio (DOR) of 3.5. When histologic findings in the “non-neoplastic” category were combined with histologic findings in the “WNL” category (Fig. 9), the results changed to a sensitivity of 35.8%, specificity of 85.1%, positive predictive value of 74.4%, and negative predictive value of 52.3%, with an accuracy of 0.41.9%, F1 score 0.48, Matthews correlation coefficient of 0.24, and a DOR of 3.2.
Discussion
The Developed Protocol Systematically Examined the Histologic Appearance of In Vivo Imaging Findings
A systematic comparison of results from in vivo imaging to those from histologic examination (histopathology) is infrequently reported in the literature. More specifically, detailed protocols describing the tracking and preparing of surgical tumor samples for the histologic examination of in vivo imaging findings are not available to date. 31,34,42 Our previous publications aimed to address this gap but left important aspects untouched. 31,38,39 Here, we describe our trimming protocol in sufficient detail to allow comparative studies correlating in vivo imaging with histologic findings. In designing our trimming protocol, we ensured current standard of care by including routine examination of the tumor mass and the surgical margins. 20
Our goal was to track all PTIIL throughout the processing of FISS surgical samples, and we strived to optimize communication between surgeons, radiologists, and pathologist. To that end, we included the imaging of complex surgical samples (ie, limb amputations and hemipelvectomies with numerous PTIIL or PTIIL deep [>30 mm] in tissues) postfixation in an effort to minimize the potential effects of tissue shrinkage in general and contraction of long muscle specifically. Furthermore, the most complex samples were trimmed by the pathologist in the presence of the primary radiologist to allow for immediate consultation and the clarification of PTIIL records and tissue markings. Clearly, these additional steps and the time-consuming nature of marking up surgical samples and keeping detailed records for each PTIIL are impractical for a routine diagnostic setting. However, we are confident that, in a research environment, the protocol can be followed and the entire process reproduced.
Tissues Surrounding FISS Including PTIIL and Surgical Margin Sections Have a Wide Range of Histologic Findings
Histologic findings in tissues surrounding FISS matched those previously reported in the tumor margins of vaccine-associated sarcomas in cats or FISS. 6,15,22,40 Neoplastic cells continuous or discontinuous with the primary tumor; proliferating mesenchymal cells not unequivocally distinguishable from neoplastic cells; infiltrates of lymphocytes, plasma cells, and macrophages (with or without gray cytoplasmic deposits interpreted as aluminum); reactive fibroblasts; and perilesional scarring alone or in combination have been described previously. 6,15,22,40 In addition, we observed and recorded vascular changes such as edema, hemorrhage, and increased vascularity. Our understanding of the clinical significance of non-neoplastic changes in peritumoral tissues is continually evolving, and some processes previously viewed as a tissue response to the presence of a neoplasm are now interpreted as signs of potential field cancerization and cancer progression. 17,21,27,28
To investigate our protocol’s performance, a systematic examination of histologic observations away from PTIIL was required. All findings were initially recorded; however, the data set was simplified for purposes of primary statistical analyses and data presentation. We assigned histologic findings to discrete categories following the previously reported schema, to facilitate comparison, 31 with addition of a fourth category for normal microanatomy (no histologic finding or WNL). We condensed data when multiple histologic changes were present in a given PTIIL (location) and only reported one histologic finding: the gravest and most relevant to the clinical evaluation of surgical tumor excision. It is possible that these data manipulations introduced bias, skewed results, or both. Both the data manipulations and the additional data points explain any discrepancies between the results of this report and our previously published study based on the same patient material and imaging findings. 31
Targeting PTIIL for Microscopic Examination Mostly Yielded Non-Neoplastic Histologic Findings or Normal Histology
The majority of equivocal (16 of 18) and neoplastic (13 of 21) histologic findings correctly predicted the presence of in vivo imaging findings. However, only 50% of PTIIL (29 of 57), whose histology deviated from normal microanatomy, had neoplastic or equivocal histologic findings. Clearly, caution has to be executed when interpreting PTIIL in the planning of tumor excisions, especially when more extensive surgical approaches are considered. 7,9,16,25
Of 81 PTIIL examined histologically, 29.6% yielded normal microanatomy (no histologic finding; n = 24), and 34.6% showed non-neoplastic histologic findings. The simplest explanation for these false negatives is that histologic lesions representing PTIIL were missed during tissue processing. Alternatively, normal structures such as lymphatics or regional vasculature may have been (mis)interpreted as PTIIL during in vivo imaging; however, prominent vessels were present in only a small number of histologic sections. Perhaps some PTIIL identified by CT or MRI represented changes in blood flow or vascular permeability, subtle differences in tissue texture, or other biological phenomena for which a histologic correlate could not be identified. 1,5,26,44
Conversely, the physical size or biologic nature of at least some of the non-neoplastic findings may have precluded detection during in vivo imaging. This is especially true for histologic changes not associated with altered perfusion or for changes whose tissue density is similar to that of surrounding tissues. 1 Some changes may have been obscured by other (micro)anatomic units or imaging artefacts in the vicinity. 14,44 In 3 cats, metal artefact was present in MRI images, which was localized and limited in 2 cases but precluded analysis of the MRI study in one case.
The identification of neoplastic or equivocal histologic findings in roughly 15% of non-PTIIL (false positives in this context) is highly relevant, both from a diagnostic and clinical standpoint. In a routine setting with less extensive histologic examination than performed in this study, these lesions could have been missed. Such failures to detect infiltrative neoplastic growth in vivo and ex vivo could provide an explanation for recurrence of those FISS reported to be excised with clean margins. 9,12,16,30
Overall Performance of the Protocol Is Good
We investigated the performance of our protocol overall from 2 perspectives. In our initial analysis, we assumed that all deviations from normal microanatomy that exceeded the size threshold would have been identifiable by the in vivo imaging modalities employed here. In a second analysis, we assumed that all PTIIL would represent only neoplastic and equivocal histologic findings, and therefore we combined non-neoplastic changes with findings of normal microanatomy (histology WNL). 16,35,36 In either statistical context, we made the assumption that the prevalence of PTIIL and histologic findings in this study agrees with that in FISS in general. We decided to include the calculation of the diagnostic odds ratio (DOR) to facilitate comparisons with the performance of protocols that may be developed by others. The DOR is a measure of overall test performance that is not negatively impacted by disparate/highly variable counts within a given confusion matrix, in contrast to other measures, most notably accuracy. 10
In the first analysis, the protocol had a moderate sensitivity (70.4%) and positive predictive value (any histologic finding deviating from normal microanatomy is a true positive or an actual PTIIL; 67.9%) but a mediocre specificity (59.7%) and negative predictive value (histology WNL is a true negative or an actual non-PTIIL; 62.5%). After combining histology WNL with non-neoplastic histologic findings, and considering only neoplastic and equivocal histopathology as noteworthy histologic findings, the protocol had a markedly improved specificity (85.1%) and positive predictive value (74.4%) suggesting that neoplastic and equivocal findings were better at predicting (more likely to represent) PTIIL. It is important to note that these results do not provide evidence that histologically equivocal lesions really are neoplastic, only that PTIIL correspond to both neoplastic and equivocal histologic lesions. In contrast, the negative predictive value was reduced (52.3%) and sensitivity was poor (35.8%), consistent with the fact that many non-neoplastic histologic findings correctly predicted (are representing) PTIIL, in the statistical sense within the context of the study (whereby PTIIL were used as reference and histologic findings as prediction). While overinterpreting PTIIL as truly or potentially neoplastic may negatively affect surgical planning leading to unnecessarily aggressive approaches and recommendation against surgical treatment, missing potentially neoplastic lesions is any clinician’s and diagnostician’s nightmare. In the context of our study this means that false positives are less concerning (and lower specificity is more acceptable) than false negatives (and lower sensitivity is less acceptable).
A Range of Factors May Have Contributed to the Discrepancies Between Histologic Findings and In Vivo Imaging Findings
Sample complexity
Complete surgical excision of FISS in the advanced stage of clinical disease often requires limb amputations or more aggressive approaches. 35 Limb amputation and hemipelvectomies comprised 50% of the surgical samples of this study, perhaps a reflection of our surgical service operating as tertiary referral center combined with changes seen in topographic distribution of FISS over time. 43 As anticipated, complex surgical samples had more PTIIL and included the PTIIL with the most complex 3-dimensional architecture. The complexity of some of the surgical samples made the accurate tracking of in vivo imaging findings difficult and, thus, may have negatively impacted information transfer from radiologists to the pathologist. These challenges may have been responsible for the failure to capture 6 PTIIL during trimming and may have contributed to the inaccurate prediction of imaging findings by some histologic findings. For example, the case with the most discrepancies between in vivo imaging findings and histologic findings was a hemipelvectomy (case 5). Increased distance from the skin surface to PTIIL may have also contributed to discrepancies between in vivo imaging and histologic findings. Most of the neoplastic and equivocal histologic findings outside of PTIIL coordinates were identified in deep tissues; 7 of 10 were located more than 10 mm deep to the skin surface. Given the limited data set, it is not clear what if any connections may exist between these discrepancies and the specific trimming approach we took to capture deep PTIIL for histologic examination (by trimming perpendicular to rather than along the long axis of deep PTIIL). Differences in orientation between the imaging scan planes and the trimming planes have the potential to contribute to false positive or false negative predictions. 1
Tissue shrinkage
The degree of shrinkage of peritumoral tissues varies among tissue types, with skeletal muscle shrinking the most and adipose tissue the least. 38,39 Surgical samples other than amputations were sutured to cardboard backing in at least 4 sites. 39 Nonetheless, some shifting along or between tissue planes may have occurred. We made efforts to reduce the impact of tissue shrinkage in lateral/radial planes by trimming beyond the PTIIL markings on surgical specimens in both directions (closer to the tumor and further to the periphery) and by heavily relying on z-coordinates (depth range) to identify discrepancies between in vivo and histologic findings. Despite our efforts, a contribution of tissue shrinkage to false positives, false negatives, or both and the overall performance of the protocol cannot be ruled out. 1,38,39
Differences between imaging modalities
CT and MRI are based on different physical properties, X-rays versus proton response to magnetism, respectively. 3,11 In some studies, MRI fat attenuation sequences, such as STIR, have resulted in superior differentiation of tumor from surrounding normal tissue as compared with CT. 4,8,44 However, other studies have shown that the performance of CT and MRI for staging of musculoskeletal neoplasms was similar. 32 The combination of improved spatial resolution and use of arterial and venous phase postcontrast CT images would have facilitated identification of the vasculature and related changes by CT compared to MRI. Dual-phase CT angiogram images were obtained at 0.5 mm and reconstructed at 2 mm slice thickness, whereas MRI slices were acquired and reconstructed at 3 mm thickness. The larger area of patient averaged per MRI image versus CT image may explain some of the discrepancies between CT and MRI imaging findings. Furthermore, small histologic changes, whose size was at the lower resolution limit of the in vivo imaging modalities, may have been missed by one or both modalities. 8,31,38,39 Furthermore, the pathologist was blinded to the physical characteristics of the in vivo imaging findings (PTIIL category). 31 It is conceivable that knowledge of these characteristics at time of trimming could lead to improved protocol performance. Unfortunately, the sample size of this study was too small to compare the performance of our protocol for one imaging modality versus the other.
Communication
Effective communication and careful tracking of data points, especially at transition points from one investigator to another, are key to the success of any study and seemed particularly important in this study, owing to the 3-dimensionality of the samples and PTIIL and, in some cases, the complexity of the samples and PTIIL. We attempted to mitigate miscommunication by closely working together at the 2 critical transition points: the marking of PTIIL and inking of areas of interest on the surgical samples (surgeons and radiologists), and the interpretation of the records from in vivo imaging findings (radiologists and pathologist). Individual differences in the execution of the in vivo imaging, surgery, or both and the communication among investigators would undoubtedly impact the outcome of our study and performance of our protocol. Consequently, limiting the number of investigators involved may have improved consistency but may have introduced bias across the entire study.
Study Limitations
The primary limitations of this study are the small sample size of 87 PTIIL in 10 cats, the complexity of some of the surgical samples, and limited number of investigators involved in sample processing and analysis (2 surgeons, 2 radiologists, and 1 pathologist). Additional studies with larger, less heterogeneous sample sets and involvement of multiple investigators at each step of the process will be required to validate the protocol presented herein.
Conclusions
The developed protocol facilitates the targeting of PTIIL for microscopic examination through utilization of a permanent reference point affixed to surgical samples, detailed recording of findings, and frequent interdisciplinary communication. The histologic findings within and outside of PTIIL of the examined series of FISS are consistent with the histopathologic changes in surgical margins of FISS reported in the literature. Importantly, using the developed protocol we confirmed that most PTIIL of FISS correspond to non-neoplastic histologic findings and revealed that some peritumoral histologic findings may not be identified during in vivo imaging of FISS by CT or MRI. Depending on diagnostic and clinical needs, the protocol can be adjusted to emphasize sensitivity or specificity.
Supplemental Material
Supplemental Material, sj-pdf-1-vet-10.1177_03009858211012949 - Targeting Peritumoral Lesions Identified by Computed Tomography and Magnetic Resonance Imaging in Feline Injection-Site Sarcomas for Microscopic Examination
Supplemental Material, sj-pdf-1-vet-10.1177_03009858211012949 for Targeting Peritumoral Lesions Identified by Computed Tomography and Magnetic Resonance Imaging in Feline Injection-Site Sarcomas for Microscopic Examination by Christiane V. Löhr, Susanne M. Stieger-Vanegas, Jesse L. Terry, Milan Milovancev and Jan Medlock in Veterinary Pathology
Supplemental Material
Supplemental Material, sj-xlsx-1-vet-10.1177_03009858211012949 - Targeting Peritumoral Lesions Identified by Computed Tomography and Magnetic Resonance Imaging in Feline Injection-Site Sarcomas for Microscopic Examination
Supplemental Material, sj-xlsx-1-vet-10.1177_03009858211012949 for Targeting Peritumoral Lesions Identified by Computed Tomography and Magnetic Resonance Imaging in Feline Injection-Site Sarcomas for Microscopic Examination by Christiane V. Löhr, Susanne M. Stieger-Vanegas, Jesse L. Terry, Milan Milovancev and Jan Medlock in Veterinary Pathology
Footnotes
Acknowledgements
We thank the anatomic pathologist on diagnostic service for generating routine diagnostic sections of the surgical sample from cat 3 (Sean Spagnoli), cat 4 (Howard Gelberg), and cat 5 (Robert Bildfell); radiologist Sarah Nemanic for invaluable contributions to in vivo imaging, and the personnel of the histology laboratory at the Oregon Veterinary Diagnostic Laboratory for technical support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported, in part, by internal funding mechanisms (Department of Clinical Sciences and Department of Biomedical Sciences, Carlson College of Veterinary Medicine, Oregon State University).
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
