Abstract
There is an unacknowledged clinical presentation of ovine toxoplasmosis characterized by early abortions and lesions of fetal leukoencephalomalacia. To investigate the pathogenesis of this condition, the extent and distribution of leukomalacia and the variations in the cell populations associated with it were characterized in 32 fetal brains from 2 previously published experimental studies of Toxoplasma gondii infection in pregnant sheep. Immunohistochemical labeling of βAPP allowed for the detection of leukomalacia in 100/110 (91%) studied samples. There was no clear influence of the challenge dose or the area of the brain (frontal lobe, corpus callosum, midbrain, and cerebellum). In tissues with leukomalacia, there was loss of oligodendrocytes and increased number of astrocytes and microglia both in the areas of necrosis but also in the surrounding area. These findings were similar to those described in ovine experimental models (inflammation syndrome and hypoxic models) of periventricular leukomalacia in humans. Thus, a fetal inflammatory syndrome may be involved in the pathogenesis of early abortion in ovine toxoplasmosis. However, further studies are needed to determine the pathogenesis of this clinical presentation because placental thrombosis and resulting hypoxia could also be responsible for the leukomalacia.
Ovine toxoplasmosis caused by the protozoan parasite Toxoplasma gondii is an important reason for reproductive failure in sheep. Sheep become infected through food or water contaminated with oocysts shed in the feces of infected cats, the definitive host. 16 Classically, abortion due to ovine toxoplasmosis has been considered to occur 4 weeks after infection and to be caused by an imbalance in the local immune response in the placenta or due to extensive damage to the placenta and fetus leading to lymphocytic multifocal inflammation and necrosis in the placenta and fetal organs. 8
However, a number of experimental studies have also described a poorly understood clinical presentation of the disease characterized by the occurrence of early abortion shortly after infection (eg, between 1 and 2 weeks after experimental inoculation of sheep), and the histological lesions differ from those classically described, suggesting that these 2 clinical presentations may differ in their pathogenesis. 5,36 In this less understood form of early abortion, lesions are characterized by infarcts in the placentomes associated with thrombosis in the maternal vessels. 9 In the fetus, the brain is the only organ with lesions, which are characterized by leukomalacia denoted by eosinophilic areas of coagulative necrosis and axonal swelling, together with diffuse congestion and scattered hemorrhages in the white matter. 5,9 Histological examination of the placenta is frequently hindered by autolysis, which makes the fetal brain the organ of choice to diagnose this condition. While the pathogenesis of late abortions of ovine toxoplasmosis has been investigated in a number of studies, 6,8,9,41 the mechanisms involved in fetal death during early abortions are yet unknown.
The fetal lesions in early abortion in ovine toxoplasmosis are similar to those described in the human condition known as “cerebral palsy.” 11,13,45 This is a neurological disorder caused by perinatal brain injury that occurs after intrauterine infection during pregnancy or when the fetus suffers hypoxia during the period of brain development. 26 The main lesion in this condition is periventricular leukomalacia (PVL). 1,17,27
Sheep are commonly used as animal models for PVL through intrauterine administration of bacterial lipopolysaccharide (inflammation model) or antenatal hypoxia-ischemia surgery (hypoperfusion model). 13 In the inflammation model specifically, the inflammatory changes and alterations in the white matter in these sheep models have been described as microgliosis, cell death (eg, neurons and astrocytes), and oligodendrocyte loss. 13,23,34,38 Additionally, immunohistochemical labelling of beta amyloid precursor protein (βAPP) has been used as a marker of axonal injury in these studies and has been proposed as a sensitive marker of PVL. 46
The aim of the present work is to better understand the pathogenesis of the early abortions taking place during the acute phase of ovine toxoplasmosis using histopathology and immunohistochemistry to characterize the cell populations and axonal damage in the associated fetal brain lesions. The comparison of these findings with those described in ovine experimental models of PVL as well as in preterm human infants with perinatal brain injury might help to better understand the pathogenesis of early abortions in ovine toxoplasmosis.
Materials and Methods
Origin of the Samples
The current study was performed on 128 formalin-fixed paraffin-embedded blocks of 32 fetal brains obtained from previous studies (Table 1). 10,41 Briefly, each infected group (G1–G3) included fetuses from 6 ewes of the Rasa Aragonesa breed. At 90 days of gestation, they were orally inoculated with different doses of oocysts of a Spanish sheep isolate of T. gondii (TgShSp1), diluted in 50 ml of phosphate-buffered saline (Table 1). 41 Samples used as control group (G4), that is, noninfected fetuses with no lesions, came from a different study where control noninfected pregnant ewes of the Churra breed were euthanized at different stages of gestation. 10 The age of the control fetuses (G4) was the same as the early abortions of the infected animals of G1, G2, and G3 (100–112 days of gestation; Table 1). A total of 28 fetuses with early fetal death (between 8 and 11 days postinfection) were included (Table 1). Previous polymerase chain reaction testing of brain samples for T. gondii was negative in all cases. 10,41
Origin of the Fetal Brain Samples Used in the Present Study.
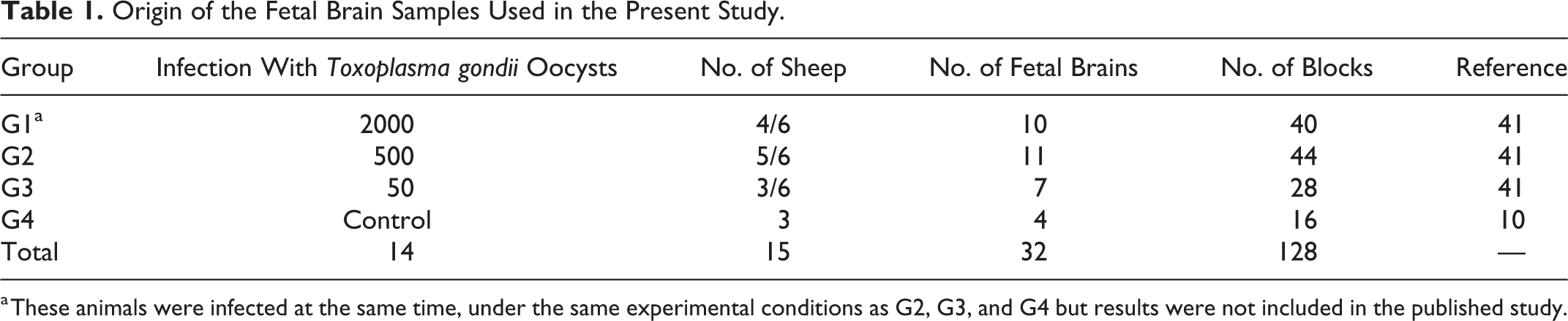
a These animals were infected at the same time, under the same experimental conditions as G2, G3, and G4 but results were not included in the published study.
Histopathology and Immunohistochemistry
In each brain, formalin-fixed paraffin-embedded samples from different areas were studied: frontal lobe (sample labeled as A), corpus callosum (B), midbrain (C), and cerebellum (D). Sections 4 µm thick were routinely prepared and stained with hematoxylin and eosin (HE). Additionally, one section from each paraffin wax block was immunolabeled for βAPP expression. Briefly, the poly-
A section from the corpus callosum (B) block from the 3 best-preserved brain samples of each infected group, as well as the 4 negative control samples, were processed for double immunohistochemical labeling with the purpose of studying the distribution of different populations of cells in relation to the areas of axonal damage (ie, leukomalacia). First, sections were labeled with antibodies against oligodendrocytes, astrocytes, microglia, and macrophages, following the same procedure mentioned above with few modifications (Table 2). Second, the double immunohistochemical labeling was performed in every section already labeled for cellular markers. For this purpose, the ImmPRESS-AP Detection System (Vector Laboratories) was used following manufacturer instructions. Briefly, subsequently to the last wash after DAB development, the slides were blocked with normal horse serum (2.5%) for 20 minutes and incubated overnight at 4°C with the primary monoclonal antibody against βAPP (1:35 000, 22C11, Millipore). After washing, the slides were incubated with ImmPRESS-AP (Alkaline Phosphatase) Polymer Anti-Mouse IgG Reagent for 30 minutes. After washing, the immunoreactivity was revealed using AP substrate solution producing a red stain. Finally, sections were rinsed in tap water and counterstained with Mayer’s hematoxylin for 10 seconds.
Details of the Antibodies and Procedures Used for the Immunohistochemical Examination.
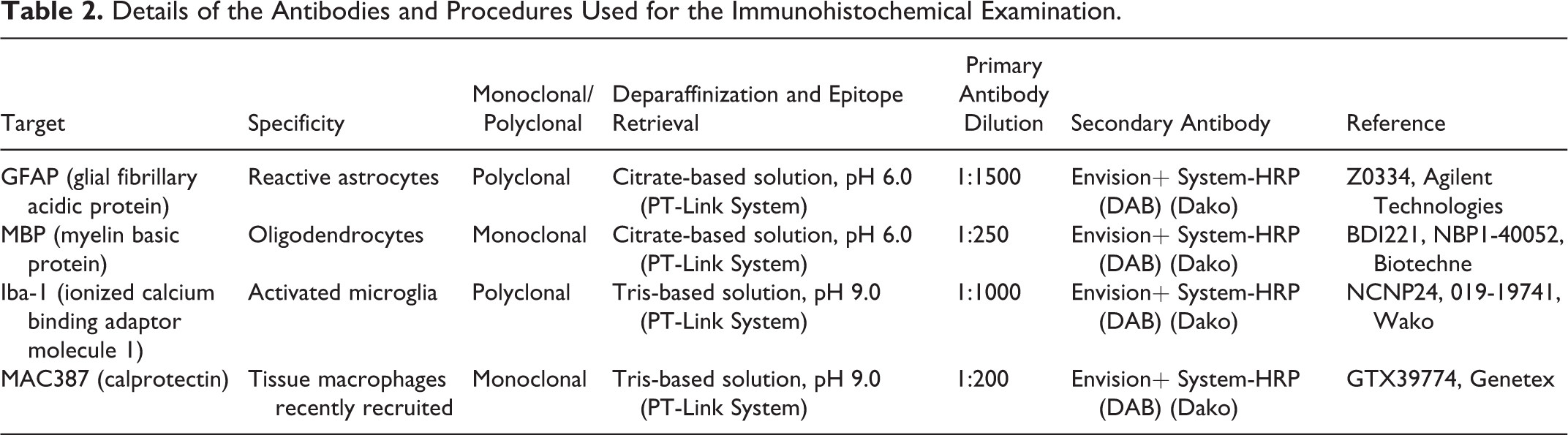
Appropriate species and isotype-matched immunoglobulins were used as a control. These included sections with an isotype control for each primary antibody, and the omission of the primary antibody. As a positive control, sections from tissue samples processed in the same manner as the fetal brains and known to contain the cell population of interest were included with the primary antibodies as positive controls.
Digitally Assisted Quantification of Immunolabeling
In order to measure the area labelled for βAPP expression, a digital analysis method was developed. A digital camera incorporated to a microscope (Leica ICC 50W) was employed to take digital pictures of the lesions. βAPP expression was evaluated in the 4 sections (ie, A, B, C, and D) from each brain. Pictures of all βAPP-labeled regions were taken under a 20× objective with bright-field microscope using standardized illumination and image acquisition settings and were subsequently analyzed. The measurement of the labelled area was carried out with the aid of the software Image J (US National Institutes of Health, Bethesda, MD). In order to calculate the percentage of the section that was labelled with βAPP, the individual measurements of each area labeled positive for βAPP in a given section were added and then divided by the total area of the whole section, also calculated with Image J.
In the 9 double-stained sections (ie, B sections from well-preserved brains), the expressions of GFAP, MBP, Iba-1, and calprotectin were measured in the area of white matter fibers of the internal capsule, between the putamen and caudate nuclei, in which leukomalacia was demonstrated (areas positive labeled for βAPP). Additionally, the same cell populations were studied in the white matter closely related to the lesions but not showing actual leukomalacia. For this, a 25- to 50-µm wide strip of white matter adjacent to the leukomalacia foci but with no evident histological lesion or βAPP labeling was evaluated. Positive labeling was measured on digital pictures through Image J, using the same microscope mentioned above and pictures taken under 20× objective. In addition, GFAP, MBP, Iba-1, and calprotectin expression from the 4 negative control sections were analyzed in the same neuroanatomical region mentioned above. Both the regions with leukomalacia and the adjacent areas were demarcated as regions of interest (ROIs) to carry out the image analysis. In the control brains, a similar demarcation was carried out, selecting the white matter bundles of the internal capsule as ROIs without making a distinction between adjacent or central area (since there was no lesion, no difference could be established). Because individual identification and counting of cells labeled for GFAP, MBP, Iba-1, or calprotectin proved to be difficult due to the presence of long unbranched and branched processes of cytoplasm, especially in those cases with a high number of positively labelled cells, the fractional area labeled positively and referred to as proportional target area (PTA) was evaluated. The PTA is defined as the fractional area labeled, which is a reflection of either the number and/or size of positively labeled cells in the ROIs. 20 For this purpose, between 15 and 20 images per section of white matter with leukomalacia were acquired and a subsequent image analysis was conducted. Briefly, pictures were converted to CMYK color mode, channels were split, and channel K was selected. After color inversion, the positive labeling signal was automatically detected through a thresholding method based on 2-dimensional Renyi’s entropy, and the PTA for GFAP, MBP, Iba1, and calprotectin stained images calculated.
Statistical Analysis
The effect of the infection dose as well as the influence of the staining method (HE vs immunochemistry) on the presence of leukomalacia was analyzed by using the Freeman-Halton test, a variation of the Fisher exact test for 2 × 3 contingency tables. The comparison of the percentage of βAPP-positive area between the different groups (G1, G2, and G3), sections (A, B, C, and D), and sections of each group was carried out using the nonparametric Kruskal-Wallis test. In a second step, to analyze pairwise differences between specific groups, a post hoc analysis (pairwise Wilcoxon rank sum test) with Bonferrroni correction was performed for significance.
A nonparametric Mann-Whitney U test was used to compare differences in the PTA of GFAP, MBP, Iba-1, or calprotectin between infected animals (n = 9; areas with leukomalacia and the adjacent area) and noninfected animals (n = 4). In addition, a nonparametric Wilcoxon signed rank test for dependent samples was employed to compare differences in the PTA of each antibody between both areas (with leukomalacia and the adjacent area) in infected animals. P values <.05 were considered to be statistically significant. All statistical analyses were performed with the InStat 6.05 software (GraphPad).
Results
Clinical Observations and Gross Lesions
The clinical consequences of the infection in G2 and G3 have been previously described. 41 In the acute phase of the disease (ie, first 2 weeks after inoculation), all infected ewes showed fever 3 to 4 days after infection, and 12/18 ewes developed early abortions during this phase of the disease (Table 1). None of the collected brains showed macroscopic lesions. Due to the variable degree of autolysis that hampered the complete sampling procedure, 2 sections from one brain in G1 were discarded from the study.
Histopathological Findings
Histological examination showed leukomalacia in 15/28 (54%) studied brains from groups G1 to G3. The leukomalacia was characterized by scarce foci of variable size located mainly at the periventricular area, with coagulative necrosis demarcated by discrete axonal swelling (Fig. 1). The neuropil surrounding these areas sometimes had microvacuolation. Generalized congestion of the white matter was present in most samples. In HE-stained sections, lesions of leukomalacia were detected in 12/38 (32%) of the G1 sections (in 5/10 brains), in 9/44 (20%) of the G2 sections (in 6/11 brains), and in 5/28 (18%) of the G3 sections (in 4/7 brains). There were no statistically significant differences between groups (P > .05; Fisher’s t test).
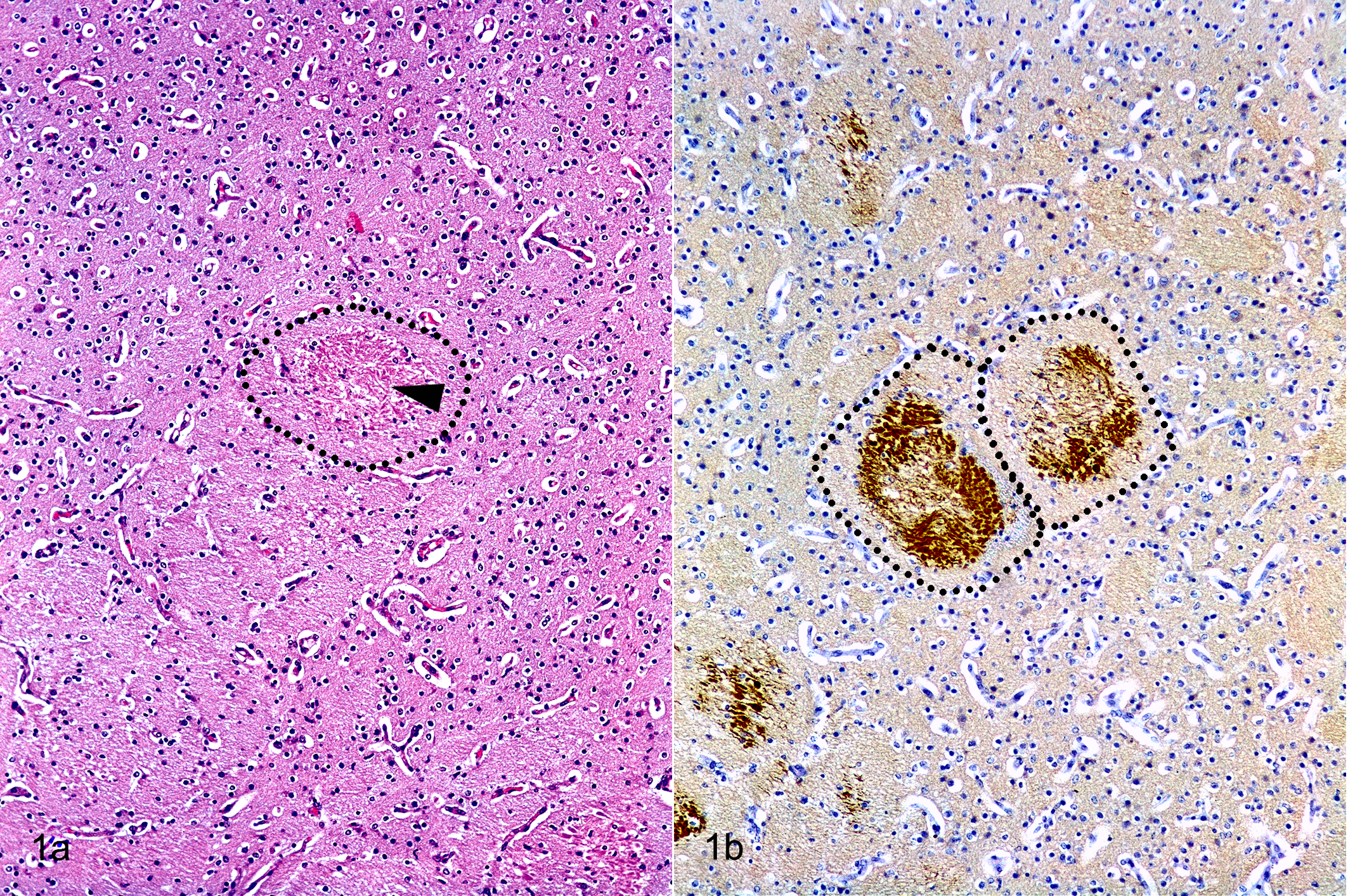
Leukomalacia, brain, fetal sheep. a) Leukomalacia is denoted by a small focus of coagulative necrosis (arrowhead) within the bundle of white matter (dotted line) of the internal capsule, between the putamen and caudate nuclei. Hematoxylin and eosin. b) In the same tissue sample as “a,” there is immunolabeling for βAPP in 2 areas of leukomalacia within the bundles of white matter (dotted line).
Immunohistochemistry
Immunolabeling for βAPP expression showed clear and well-demarcated positive labelling in those sections where leukomalacia was evident in HE-stained sections, but also in a many of the remaining sections, where the coagulative necrosis in the white matter was not clearly identified in HE-stained sections (Fig. 1). Positive labeling for βAPP expression was detected in 31/38 (82%) of the G1 sections (in 9/10 brains), in 43/44 (98%) of the G2 sections (in 11/11 brains), and in 26/28 (93%) of the G3 sections (in 7/7 brains). All brains had at least one section that was positive, except for one brain of the G1 group, which only showed generalized congestion of the white matter. The percentage of βAPP-labeled area varied from 0.1% to 34%. When comparing the extension of the lesion, there were no statistical differences between G1, G2, and G3 (Fig. 2; χ2 = 0.38, degrees of freedom [df] = 2, P = .82) or between the 4 different levels evaluated, that is, A, B, C, and D (χ2 = 5.64, df = 3, P = .13), both when the different groups were pooled (Fig. 2) or analyzed individually (G1: χ2 = 1.12, df = 3, P = .77; G2: χ2 = 5.20, df = 3, P = .15; G3: χ2 = 1.34, df = 3, P = .71; Fig. 3). Although not significant, the average percentage of positive immunoreaction in section D (2.31%; cerebellum) was always lower than in section A (6.18%; frontal lobe), section B (4.23%; corpus callosum), and section C (4.36%; midbrain) in all groups (Fig. 3).
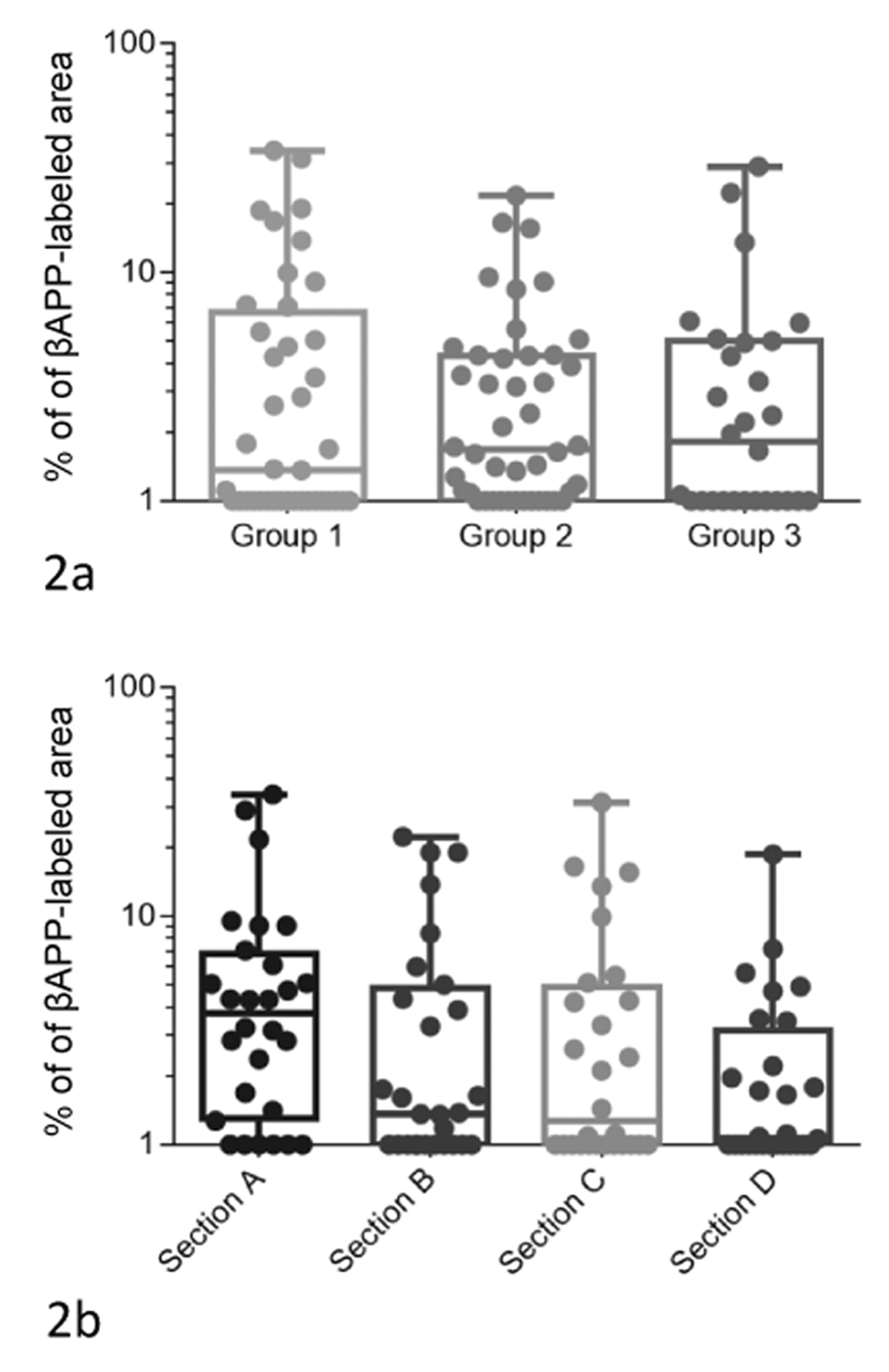
Extent of leukomalacia. The area of fetal brain tissue affected by leukomalacia was determined as a percentage of the total area, based on image analysis. (a) The data are based on analysis of fetal brain from ewes infected with 2000 (G1), 500 (Group 2), and 50 (Group 3) Toxoplasma gondii oocysts. (b) Different neuroanatomic locations: frontal lobe (section A), corpus callosum (section B), midbrain (section C), and cerebellum (section D). There were no significant differences among groups or among anatomic locations.
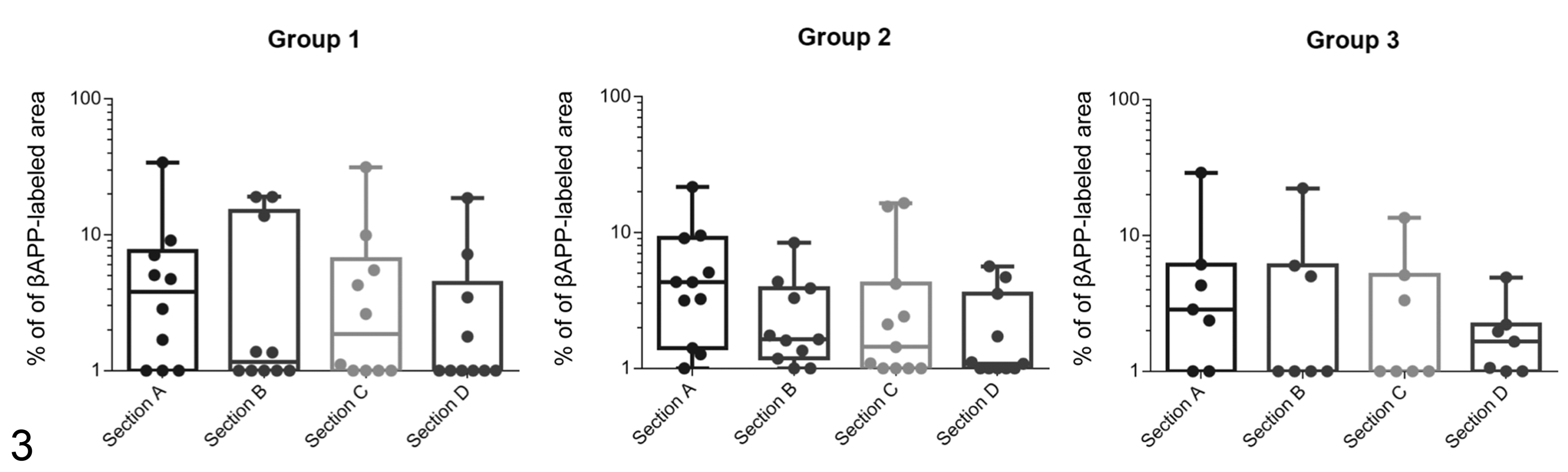
Extent of βAPP labelling. The area of fetal brain tissue with immunolabeling for βAPP was determined as a percentage of the total area. (a) The data are based on analysis of fetal brain from ewes infected with 2000 (G1), 500 (Group 2), and 50 (Group 3) Toxoplasma gondii oocysts. (b) Different neuroanatomic locations: frontal lobe (section A), corpus callosum (section B), midbrain (section C), and cerebellum (section D). There were no significant differences among groups or among anatomic locations.
GFAP-, MBP- and Iba1-positive cells were found in the white matter of all sections of fetal brains studied, either from infected sheep or noninfected controls. In the areas of leukomalacia, cells positively immunolabeled for Iba-1 appeared as highly branched cells intermingled with swollen axons in the area of leukomalacia (Figs. 4–9). However, there were no Iba-1-positive cells in the white matter at the internal capsule, or just low numbers at their periphery, in the control animals (Figs. 4, 5). Similarly, GFAP-positive cells were found in the areas of leukomalacia, in between spheroids and necrotic axons (Fig. 6) while these cells were less numerous in the same region of the nonaffected white matter from control animals (Fig. 7). On the other hand, there was a clear decrease in the number of MBP-positive cells in the areas of leukomalacia (Fig. 8) when compared to the white matter in the same region from nonaffected control animals, where few rounded cells but numerous cytoplasmic branches were labelled (Fig. 9). Finally, there was no labelling for calprotectin in any of the studied sections.
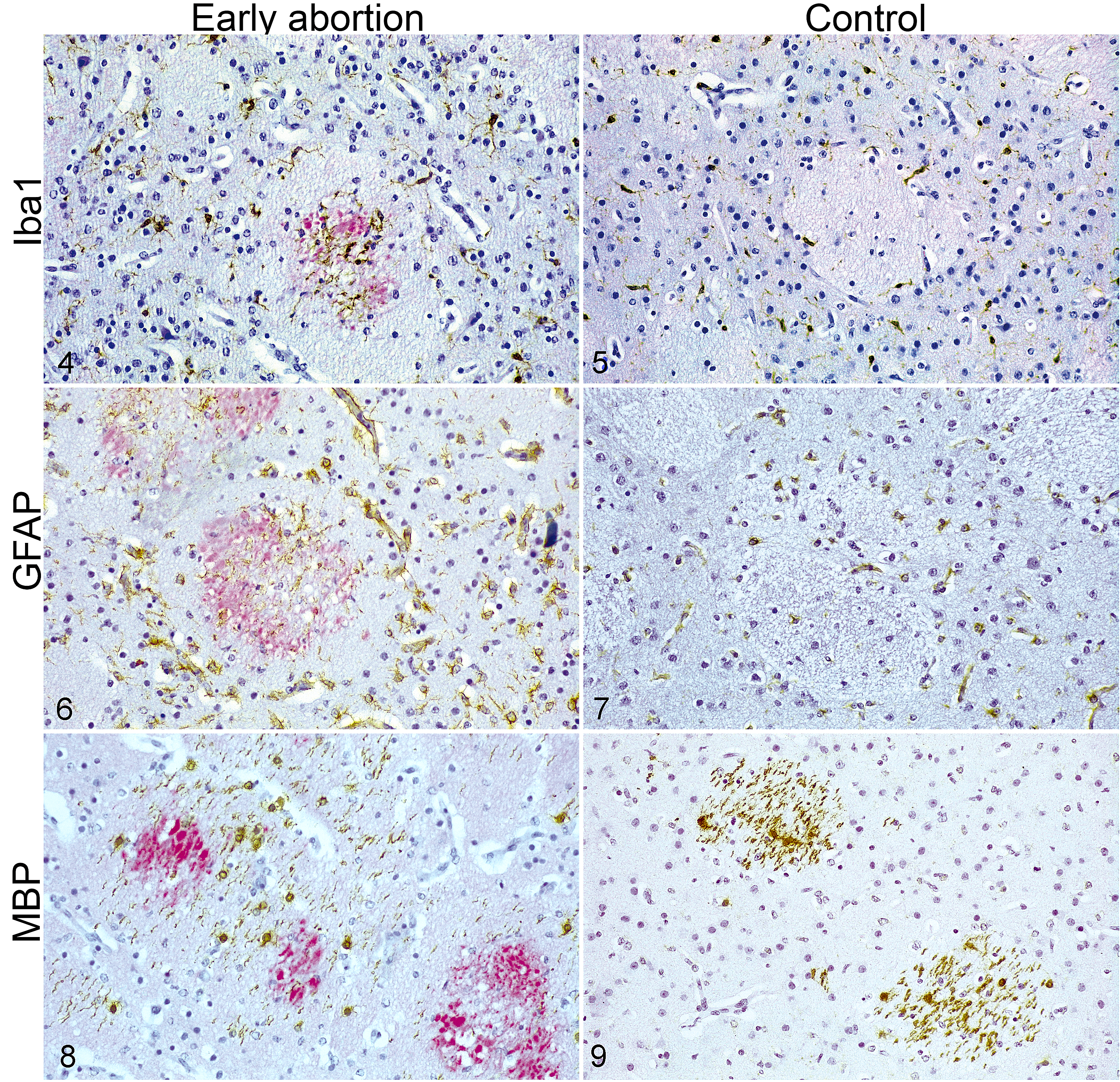
Leukomalacia, brain, fetal sheep. White matter tracts of the internal capsule, between the putamen and caudate nuclei. Double-label immunohistochemistry for βAPP (red chromogen to label leukomalacia) and cellular markers (brown chromogen) including Iba-1 (microglia/macrophages; Figs. 4, 5), (GFAP (astrocytes; Figs. 6, 7), and MBP (oligodendrocytes, Figs. 8, 9). Within areas of leukomalacia (red), there is an increased number of Iba-1-positive microglial cells and GFAP-positive astrocytes but reduced number of MBP-positive oligodendroglial cells, in the fetuses that were aborted early after Toxoplasma infection compared to similar areas of control uninfected fetuses.
Regarding PTA, the expression of GFAP (W = −2.27, P < .05) or Iba-1 (W = −4.38, P < .001) was higher in the white matter bundles showing leukomalacia compared to the white matter of similar distribution in the control animals. On the other hand, MBP expression was lower (W = 7.49, P < .001) in the affected brains than in the controls (Fig. 4). When the area adjacent to the leukomalacia was compared to the white matter of the control animals, the same results mentioned above were observed for the expression of GFAP (W = −4.01, P < .001) and MBP (W = 4.24, P < .001) but no differences in Iba-1 expression were found (W = 1.95, P = .06).
Discussion
The pathogenesis of early abortions of ovine toxoplasmosis is mostly unknown, but it is characterized by the occurrence of leukomalacia in fetal brain, specifically PVL. 9 This same lesion is the hallmark finding in human infants with cerebral palsy. 27 To study this condition, different experimental models have been developed in sheep based on hypoperfusion (umbilical cord occlusion and hypoxia) or intrauterine infections with bacterial products (lipopolysaccharide). 3,13,23,32
Both placental infarcts and fetal leukomalacia are the main lesions found in cases of early abortion during the acute phase of T. gondii infection, where the burden of parasite is low or absent. 9 In fact, the absence of T. gondii was proven by polymerase chain reaction in all brains included in this study. 9,41 However, the samples from these cases frequently showed an advanced stage of autolysis that hindered histological evaluation. This degree of autolysis may have complicated the HE analysis of the samples in the current study, as it was not sensitive enough to detect the lesions in all cases. However, the lesions were clearly identified using immunohistochemical labeling for βAPP, a transmembrane glycoprotein ubiquitously expressed in the nervous system that accumulates at sites of acute disturbance of axonal transport or in transected axons. 28 The results from this study agree with previous results in humans, 2 showing that βAPP labeling is useful for detecting areas of leukomalacia and studying their distribution in the brain, even in samples with an advance degree of autolysis, such as natural cases of ovine abortion. Similarly, Ohyu et al 34 detected through βAPP-immunostaining leukomalacia in the periventricular white matter with or without variable degrees of gray matter injury in 64% of ovine fetal brains with experimental umbilical cord occlusion. On the other hand, Duncan et al 18 observed a different distribution of the lesions, as they reported βAPP expression in sites of diffuse white matter injury in all lipopolysaccharide-exposed fetuses, but not gray matter involvement. Regarding this difference of distribution, Choi et al 13 suggested that PVL caused by systemic inflammation has a different morphological appearance than that produced by hypoperfusion. In fact, Mallard et al 30 stated that asphyxia in fetal sheep induced white and gray matter injury together with microglia activation, whereas endotoxemia resulted in selective white matter damage and inflammation. However, high or moderate doses of lipopolysaccharide also induce hypotension and decreases in cerebral blood flow. 13 Thus, based on the lack of gray matter involvement in the studied fetuses, it is tempting to hypothesize that systemic inflammation in the sheep triggered after T. gondii was the major component in the fetal leukomalacia. However, the occurrence of partial hypoperfusion seems a logical consequence of the placental thrombosis characteristic of this clinical presentation, and its involvement cannot be ruled out.
Regarding the distribution of the lesions, leukomalacia was detected in the 4 levels of the brain studied by βAPP immunolabeling. Within these sections, lesions were found not only circumscribed to the periventricular area but also in the corona radiata. This distribution differs from that described in PVL in human infants, which only affects the dorsal and lateral white matter of the external angles of the lateral ventricles. 45 In this sense, Ohyu et al 34 detected necrotic lesions in the periventricular white matter as well as in the cerebral cortex and thalamus in the sheep model of hypoperfusion, and Duncan et al 17 observed white matter damage in all frontal, parietal, and temporal brain sections after repeated endotoxin exposure in a sheep model. In human preterm fetuses, it has been suggested that the distribution of these lesions may be a consequence of the susceptibility of the immature penetrating cerebral vasculature (eg, trigone of the lateral ventricles and foramen of Monro) to impaired perfusion, which would explain why focal periventricular necrosis is commonly found at border zones. 19,33 However, the distribution found in sheep is different, as the leukomalacia is found in these regions but also in the white matter of the brain hemispheres, both in experimental hypoxic and inflammatory models of PVL as well as in early T. gondii abortions. It is possible that anatomical or physiological differences between human and ovine brain might explain such differences, but the reasons behind this variation are not known and warrant further investigation into the mechanisms responsible for leukomalacia in sheep.
Previous characterization of these lesions has usually been carried out subjectively, and few works have performed a quantitative assessment of axonal damage. 44 Our results provide a quantitative value that shows a high individual variability in the size of βAPP-labeled areas (0.1% to 34%) without significant differences between sections, similar to that described in experimental models of PVL, 17 where the percentage of white matter occupied by PVL ranged from 0% to 6.4%. Another relevant finding of the current study is that the dose of inoculation was not associated with the severity and/or distribution of the lesion. This fact might be explained because all brains were collected in a short period of time (4 days). That is, sheep were euthanized when fetal death was detected by ultrasonography. This may suggest that once the inflammatory response triggered by the infection exceeds a given threshold where it becomes harmful for the fetus, the pathogenesis of the abortion is similar regardless the number of oocysts inoculated to the mother.
In the classical presentation of ovine toxoplasmosis, fetal brain lesions are characterized by foci of gliosis, frequently with central areas of necrosis. 5 It is believed that these lesions are triggered by the presence of the parasite and the fetal immune response to infection. 7 The lesions of fetal leukomalacia found in the early abortions of ovine toxoplasmosis show different histological features, which suggest a different pathogenesis. 5 Due to the lack of knowledge on the pathogenesis of early abortion in ovine toxoplasmosis, characterizing the glial cell populations might identify mechanisms responsible for these lesions.
In the current study, a higher expression of Iba-1 and GFAP as well as the apparent loss of MBP expression are the main changes affecting glial cells. Iba-1 is a marker for microglial cells and macrophages, and both have been described in brain lesions. 39 The lack of labeling for calprotectin 12 suggests that all the Iba-1 positive cells were microglia and that there has been no time, or stimulus, for blood monocytes to infiltrate into the white matter and differentiate to macrophages. Two distinct populations of microglial cells have been described according to their functions. M1 microglial activation in response to altered levels of cytokines is crucial for inflammation and free radical injury to cells, that is, neurons and oligodendrocytes due to secretion of cytokines that exert toxic effects. 4,14,29 Conversely, alternatively activated microglia (M2) may also contribute to dissolution of the necrotic tissues at an early stage as well as protection against cell injury. 4,34,43 The activated microglia found in the current study might play any of these roles and further phenotypical characterization is needed to clarify the role of microglia in early abortions. Regarding GFAP expression, the reactive astrogliosis reported here is in contrast to a previous study that described a loss of astrocytes in PVL sheep models. 13 However, astrogliosis is a defensive response aimed at limiting damage. 42 In addition, there was loss of oligodendrocytes, denoted by the reduction on MBP expression when compared to nonaffected fetuses which might reflect death of oligodendrocytes through apoptosis. 34
Intraamniotic endotoxin infusion in pregnant sheep causes regional inflammatory changes that are associated with the occurrence of hypoxia and hypovolemia in fetal sheep in utero. 13,30,40 The hypoxia appears to directly damage the vulnerable oligodendrocyte system and astrocytes. 13,40 On the other hand, systemic inflammation after in utero inoculation of an infectious agent has been also associated with reduced MBP expression within regions of overt microglial activation. 35 In fact, Ophelders et al 35 found that white matter injury was the result of activated microglia, which produced cytokines, free radicals, nitric oxide, and excitotoxic amino acids that could be toxic to different cell populations. These finding suggest 2 possible mechanisms the fetal brain lesions of PVL in the current study.
First, the fetal brain lesions might result from systemic immune activation in the mother after oral inoculation with oocysts, which might have resulted in the cerebral damage in the fetus. This mechanism would not require infection of the brain by a viable pathogen, as some experimental sheep models for PVL are based on intrauterine exposure to lipopolysaccharide. 13 In this scenario, multiplication of T. gondii in maternal tissues might trigger a systemic immune response that damages cells in the fetal brain. Comparatively, higher levels of interferon (IFN)-γ, tumor necrosis factor (TNF)-α, and transforming growth factor (TGF)-β have been reported in the serum of pregnant women in the acute phase of T. gondii infection. 31 In the same way, TNF-α released by active macrophages or microglia during ischemia and/or infection are thought to induce damage to pre-myelinating oligodendrocytes by oxidative injury. 21 In fact, in the hypoxic sheep model microglial cells related to necrotic foci in white matter express TNF-α and may contribute to the subsequent tissue injury. 34 In this context, we speculate that after inoculation of ewes, circulating tachyzoites of T. gondii during the early phase of infection might have triggered a systemic and dysregulated proinflammatory response in the mother, leading to the production of different mediators of inflammation such as proinflammatory cytokines, that may have reached fetal circulation, causing an early abortion without the need for an actual invasion of the fetus by the parasite.
Second, the fetal brain lesions might result from hypoxia caused by the thrombosis or hemodynamic changes in the placenta, which could disturb neuronal or glial metabolism resulting in axonal damage. Hypoperfusion of the fetal brain has been shown to be an important contributor to white matter injury. 13 In fact, an existing sheep model for PVL is based on umbilical cord occlusion in near-term fetal sheep, and this results in fetal hypoxia, acidosis, brain hypoperfusion, free radical generation, and development of brain lesions. 34 The cardiovascular response to hypoxic-ischemia involves a redistribution of cardiac output to the brain, 22 which leads to congestion, hemorrhages, cytotoxic cerebral edema, and ischemic neuronal injury. 15,37 Placental thrombosis has been described in the early abortions of ovine toxoplasmosis, 9 which might cause hemodynamic disturbances that contribute to fetal hypoxia. The mechanisms by which T. gondii infection causes thrombosis is uncertain.
Although the experimental sheep models for PVL are based on different principles (cord occlusion vs inflammation), both hypoxia-ischemia and exposure to infection or lipopolysaccharide lead to a prominent neuroinflammatory response in the fetal brain. 11,24,25 Histological and immunohistochemical changes observed in the fetal brains from early abortions after inoculation with T. gondii are similar to those described in sheep models of PVL. Thus, the pathogenic mechanisms described in both experimental models might also be involved in the pathogenesis of early abortions in ovine toxoplasmosis. Based on these results, it is tempting to speculate that hypoxia and mediators of inflammation induced by T. gondii infection may lead to a loss of oligodendrocytes without neuronal damage, with subsequent activation of microglia. Nevertheless, the role played by the systemic immune response, as well as the hemodynamic changes in the mother, need to be further investigated.
Footnotes
Acknowledgements
We thank all the employees at the “Instituto de Ganadería de Montaña.” We acknowledge Carmen Agudín, María Teresa Carro, Luis Miguel Ferrer, and Jose María González, specifically, for their excellent technical assistance. We also acknowledge Dr Jose Espinosa for his help in the statistical analysis. We also acknowledge The Animal Experimentation Service (AES) at the University of Zaragoza for providing their facilities to carry out the reproductive program in sheep.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by research projects from the “Junta de Castilla y León” (LE080U16) and Spanish Ministry of Sciences (AGL2016-75935-C2-1-R and AGL2016-75935-C2-2-R). Daniel Gutiérrez-Expósito is the recipient of a postdoctoral contract from the Ministry of Science and Innovation (Grant No. FJCI-2017-32020).
