Abstract
Investigations describing the ocular and lacrimal gland lesions associated with rabies are sparse. Here we characterize the pathological changes and distribution of rabies viral antigen in the eye, optic nerve, and lacrimal gland of 18 rabies cases from different mammalian species. Histology and immunohistochemistry for rabies virus, CD3, CD20, and Iba1 were performed on tissue sections of eye, optic nerve, and lacrimal gland. Polymerase chain reaction (PCR) for rabies was performed on all cases, including 7 formalin-fixed, paraffin-embedded (FFPE) and 11 frozen tissue samples of eye and lacrimal gland. Pathological changes in the eye consisted of retinal necrosis (12/18 cases) with occasional viral inclusions within ganglion cells (8/12 cases). Immunohistochemically, viral antigen was detected within the nerve fiber layer, ganglion cells, and inner plexiform layer in all 12 cases with retinal lesions and in 2 cases with no retinal lesions, as well as optic nerve (6/18 cases) and lacrimal gland epithelium (3/18 cases). CD3+ T lymphocytes were present in the retina (11/18 cases), optic nerve (2/18 cases), and lacrimal gland (11/18 cases). No CD20+ B lymphocytes or Iba1+ macrophages were detected. PCR for rabies virus was positive in 9 of 11 frozen samples but in only 2 of 7 FFPE samples. Five samples that were negative for rabies by PCR were positive by immunohistochemistry, and 2 samples were negative by both tests. These results provide evidence that rabies virus infection extends to the eye, likely via the ocular nerve, and that the lacrimal gland might be a source of viral infection.
Rabies is an infectious neurological disease caused by a neurotropic Lyssavirus of the family Rhabdoviridae. 13 Clinical disease is endemic in many parts of the world, and infection is nearly always fatal. 13 Human rabies is particularly common in Africa and Asia, where infection is overwhelmingly acquired from dogs. 12,13 The United States is considered a low-risk area for rabies, with wild animals accounting for over 90% of the confirmed cases. 12,25 Transmission typically occurs following contact with infected saliva through a bite wound or scratch. 13 After entry and intramuscular replication at the inoculated site, viral particles gain access to the neuromuscular junctions, with retrograde transport along nerves to the spinal cord and subsequently to the brain, where secondary viral replication takes place. 13,21 Once established in the central nervous system (CNS), viral particles centrifugally spread to the major and minor salivary glands and are eventually excreted in the saliva, which is the main method of transmission to the next susceptible host. 13
Although viral spread to the salivary gland is often emphasized because of the important role saliva plays in the transmission of rabies, there can also be centrifugal spread along the autonomic and sensory nerves with subsequent transfer of viral particles to other extraneural sites. 19,24,31 In human rabies, viral antigen has been demonstrated within cardiomyocytes, lingual epithelium, larynx, adrenal medulla, pancreatic islets, and hair follicles. 19 In experimental settings, rabies virus has been detected in the eye, nerve endings associated with hair follicles, lingual epithelium, intestine, adrenal medulla, olfactory neuroepithelium, pancreas, brown adipose tissue, and heart of hamsters; 26 pancreas of dogs and monkeys; 31 and heart, lung, liver, and kidney of mice and rats. 24
Although the retina is considered part of the CNS, there have been only a few studies investigating the ocular lesions associated with rabies in humans and other animals. In humans, the main pathological changes in the eye include lymphoplasmacytic anterior uveitis and choroiditis, lymphoplasmacytic retinitis, subretinal and outer plexiform layer exudation, and retinal ganglion cell necrosis. 16 A few reports investigating the extraneural changes associated with experimental rabies virus infection in rabbits, hamsters, dogs, monkeys, foxes, and skunks have revealed similar inflammatory and degenerative lesions in the eye of infected animals. 3,9,10,26,27 However, no studies have focused on the ocular and lacrimal gland lesions caused by naturally occurring rabies in domestic and wild animals. Here we describe the pathological changes in the eye, optic nerve, and lacrimal gland in domestic and wild mammals submitted for diagnosis of rabies and compare different diagnostic methods using frozen and formalin-fixed tissues from affected animals.
Materials and Methods
Animals and Tissues
Tissues were collected from animal carcasses submitted to the University of Georgia Athens Veterinary Diagnostic Laboratory or Southeastern Cooperative Wildlife Disease Study as rabies suspect cases. For diagnostic confirmation, a cross section of fresh cerebellum and brainstem was tested for the presence of rabies virus antigen following standard protocol for the fluorescence antibody test (FAT).
14
Eye, optic nerve, and lacrimal gland were subsequently collected from all FAT-positive cases from 2018 to 2019 for histology, immunohistochemistry, and polymerase chain reaction (PCR). Tissues were immersed in 10% neutral-buffered formalin for histology and immunohistochemistry (IHC). Two additional sets of tissue samples (a section of the other eye, including sclera, choroid, and retina, and the lacrimal gland) were separately collected for PCR testing for rabies virus. These tissues were either fixed in 10% neutral-buffered formalin (7 cases) or frozen at –20
Histology and Immunohistochemistry
Formalin-fixed tissues were routinely processed for histopathology and stained with hematoxylin and eosin. Histologic lesions in the eye were assessed in each anatomic ocular structure (conjunctiva, cornea, sclera, lens, iris, ciliary body, choroid, and retina), as well as in the optic nerve and lacrimal gland. IHC for rabies virus was performed using goat polyclonal antiserum (Light Diagnostics, Burlington, MA), 1 in 6000 dilution for 60 minutes, for confirmation of the FAT results and determination of the antigen distribution within tissues. Rabies FAT-positive bovine brain tissue was used as control for the IHC. To assess the presence and distribution of inflammatory cells, tissue sections were immunolabeled for CD3 (T lymphocytes; rabbit polyclonal, 1:1000 dilution for 60 minutes; A05452; Dako, Carpinteria, CA), CD20 (B lymphocytes; rabbit polyclonal, 1:2000 dilution for 90 minutes; 121R-18; Biocare, Pacheco, CA), and Iba1 (macrophages; rabbit polyclonal, 1:8000 dilution for 60 minutes; 019-19741; Wako, Richmond, VA). Normal canine lymph node tissue was used as control for the inflammatory cell markers. A modified scoring system 35 for the ocular lesions was used to assess the number of inflammatory cells per 2.4 mm2 (equivalent to 10 FN22/40× fields) within each ocular structure, optic nerve, and lacrimal gland. Inflammation scores were assessed using IHC for CD3, CD20, and Iba1 and recorded as 0 = no inflammation (0–3 inflammatory cells per 2.4 mm2), 1 = mild inflammation (4–50 inflammatory cells per 2.4 mm2), 2 = moderate inflammation (51–100 inflammatory cells per 2.4 mm2), 3 = severe inflammation (101–200 inflammatory cells per 2.4 mm2), and 4 = massive inflammation (over 200 inflammatory cells per 2.4 mm2). 35
PCR
Each sample set (formalin fixed and frozen) was tested separately. For formalin-fixed, paraffin-embedded (FFPE) tissues (case Nos. 1–4, 10–11, and 13), RNA was extracted using the RNeasy FFPE Kit (Qiagen, Hilden, Germany) in accordance with the manufacturer’s recommendations. Frozen tissue samples (case Nos. 5–9, 12, and 14–18) were thawed and thoroughly homogenized with 500 μl brain heart infusion broth (Remel Products; Thermo Fisher Scientific, Waltham, MA) using a bead beater (Qiagen). Then, 300 μl Trizol (Thermo Fisher Scientific) was added in the complete homogenized sample. Further steps were performed using the Direct-zol RNA Miniprep Kit (Zymo Research, Irvine, CA) in accordance with a previously described protocol for RNA extraction of rabies virus. 15 A modified real-time reverse transcription (RT)–PCR was performed as described previously. 15 Each DNA sample was tested for Lyssavirus RNA genome 15 and for host glyceraldehyde 3-phosphate dehydrogenase (GAPDH) messenger RNA (mRNA) (internal control). 4 Each sample was run in duplicate, and each assay included a no-template control and a positive control consisting of an artificial rabies RNA virus inserted into plasmids pUC57 kanamycin and transformed into competent Escherichia coli cells (DH5α strain). Synthetic RNA plasmids were purchased from GENEWIZ (South Plainfield, NJ). For the GAPDH PCR assay, DNA from a whole-blood sample from a dog was used as a positive control.
Results
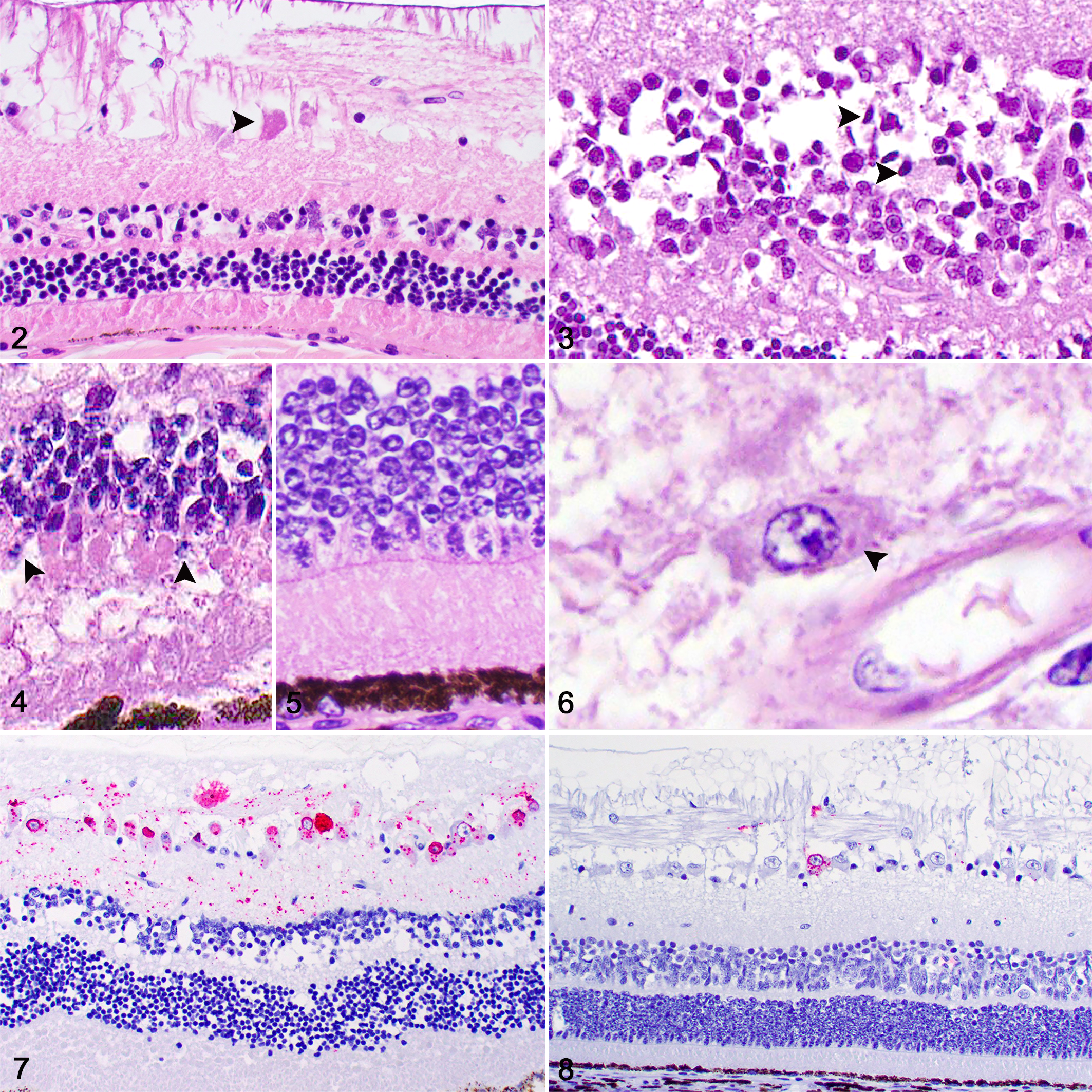
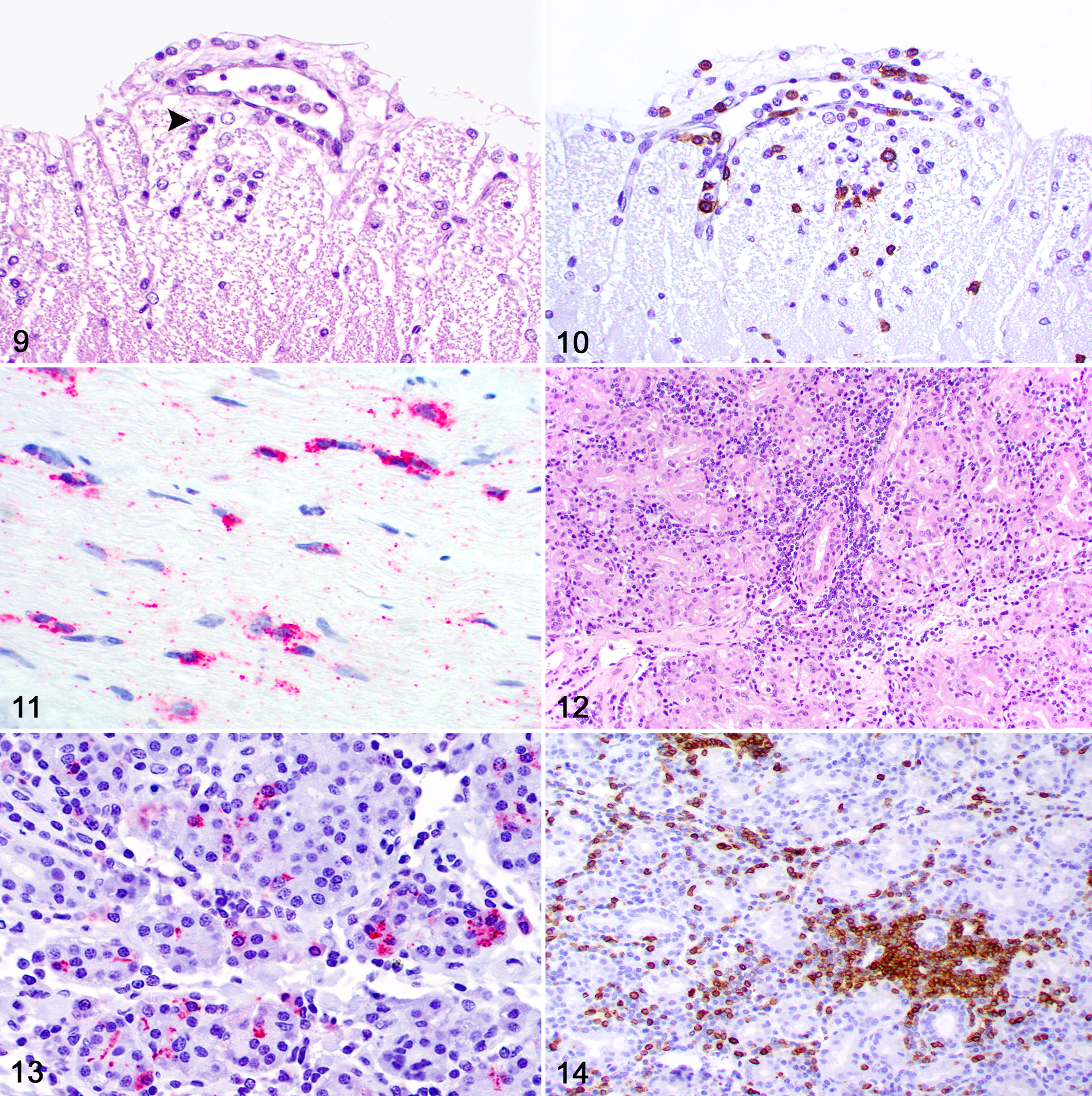
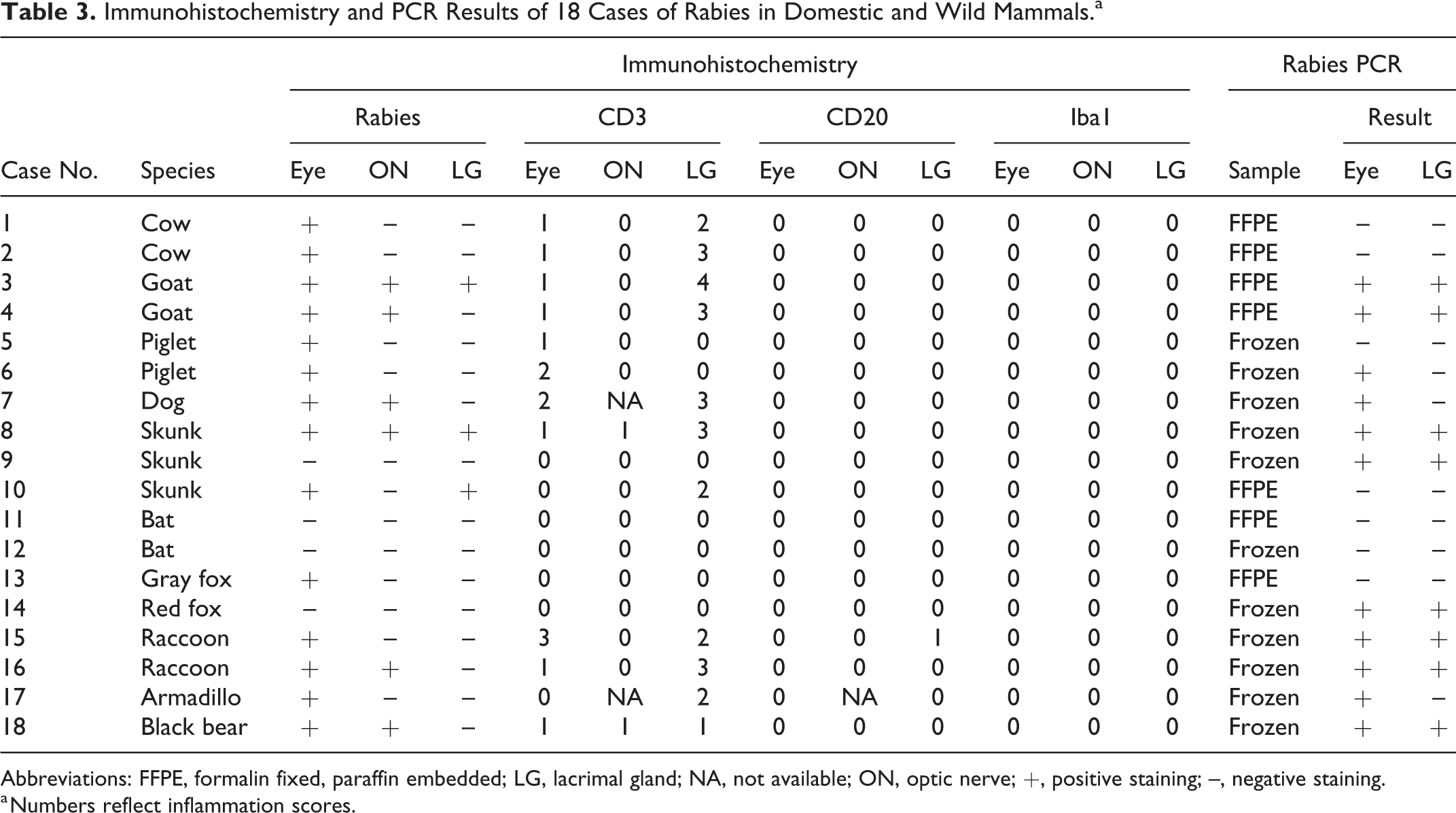
Eighteen contiguous cases of rabies from 18 mammalian species were included in this study after a diagnosis was established by FAT. The signalment, origin, clinical signs, and clinical outcome of each case are summarized in Table 1. No gross lesions were reported during the collection of brain samples for FAT. The histologic lesions and distribution of viral antigen are summarized in Table 2. Histologic lesions in the eye occurred in 12 cases and were restricted to the retina (Fig. 1). All these 12 cases had segmental degeneration and necrosis of ganglion cells, which exhibited shrunken and hypereosinophilic cytoplasm and pyknotic or karyorrhectic nuclei (Fig. 2). Similar lesions were present in the inner nuclear layer in 6 of 12 cases (Fig. 3) and in the photoreceptor cell bodies (rods and cones) of the outer nuclear layer in 4 of 12 cases (Figs. 4, 5). Small and often solitary intracytoplasmic eosinophilic viral inclusions consistent with Negri bodies were present within ganglion cells in 8 of the 12 cases with retinal lesions (Fig. 6). Immunolabeling for rabies antigen occurred in all 12 cases in which retinal lesions were present (Table 3). In addition, immunolabeling was detected in 2 cases that had no retinal lesions. Immunolabeling was restricted to the nerve fiber layer, cytoplasm of ganglion cells, and inner plexiform layer in 10 cases (Fig. 7) and to the nerve fiber layer and cytoplasm of ganglion cells in 4 cases (Fig. 8). Immunolabeling highlighted the viral inclusions and additional clusters of antigen in the cytoplasm of ganglion cells. No immunolabeling was present in 4 of the cases that had no retinal lesions. Control tissues stained appropriately. Retinal inflammation was histologically evident in only 1 case (Fig. 9) and consisted of mild perivascular accumulations of lymphocytes. However, IHC revealed scattered perivascular CD3+ T lymphocytes in the retina in 11 cases (Fig. 10). The inflammation scores are summarized in Table 3.
Signalment, Origin, Clinical Signs, and Clinical Outcome of 18 Cases of Rabies in Domestic and Wild Mammals.
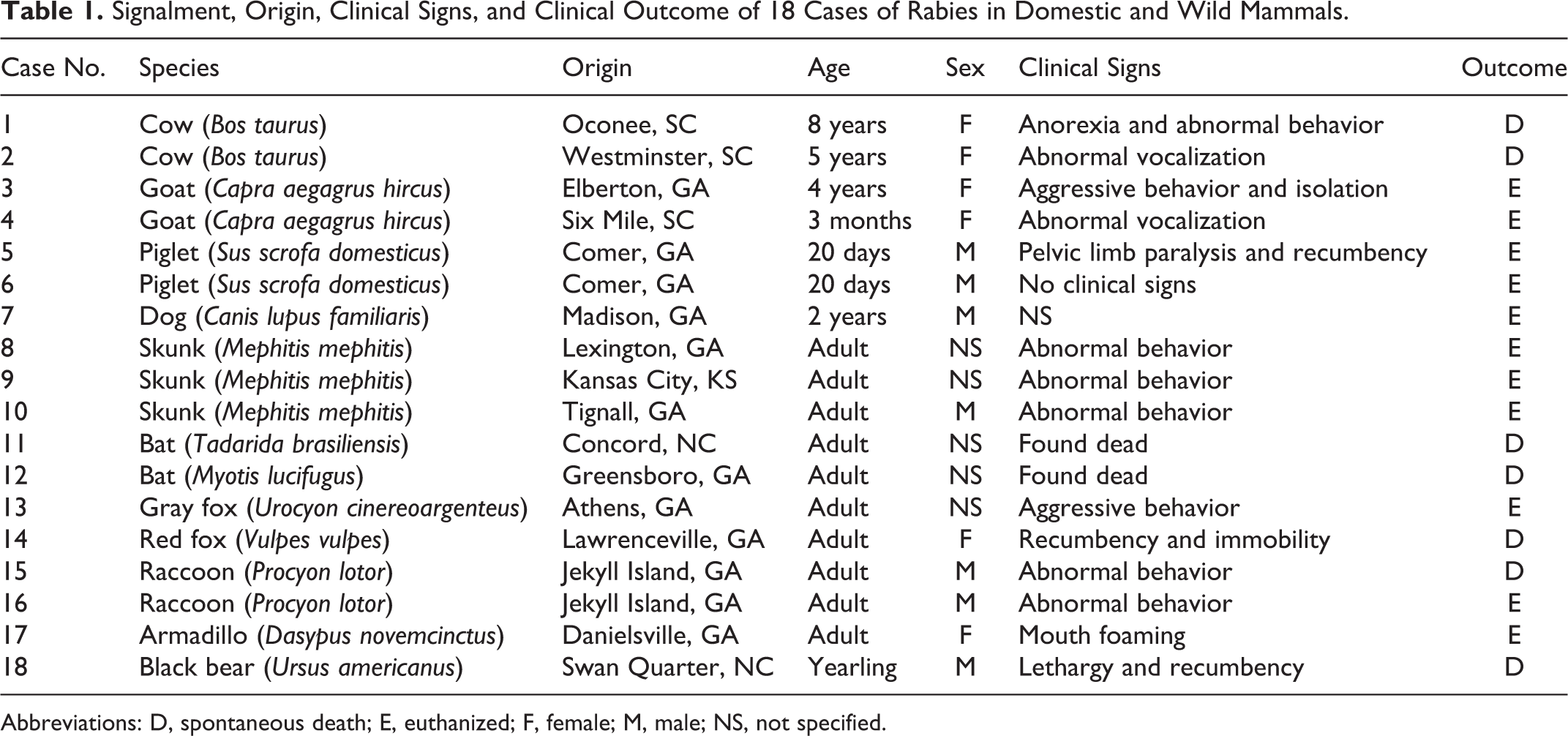
Abbreviations: D, spontaneous death; E, euthanized; F, female; M, male; NS, not specified.
Histological and Immunohistochemistry Findings of 18 Cases of Rabies in Domestic and Wild Mammals.
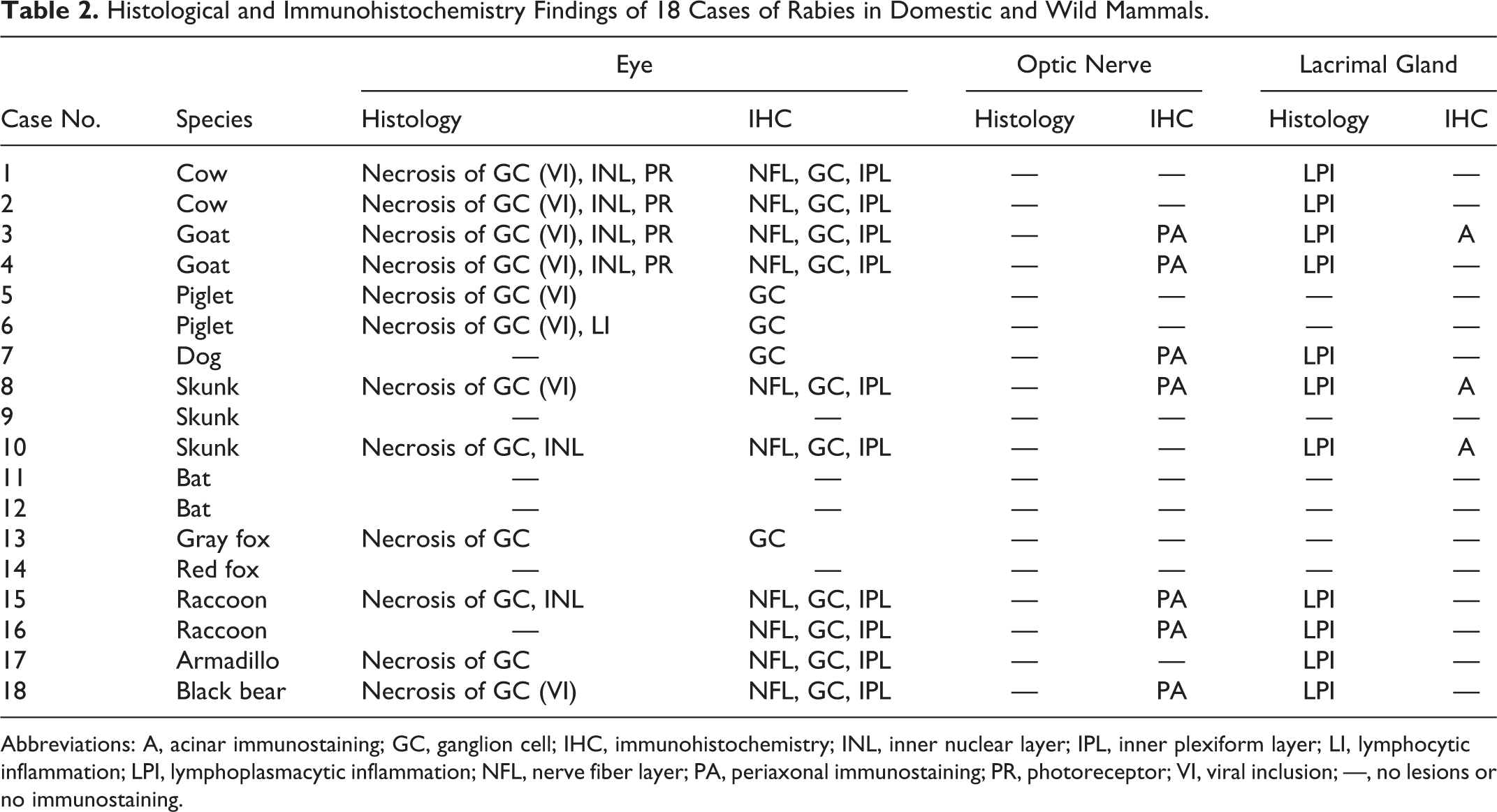
Abbreviations: A, acinar immunostaining; GC, ganglion cell; IHC, immunohistochemistry; INL, inner nuclear layer; IPL, inner plexiform layer; LI, lymphocytic inflammation; LPI, lymphoplasmacytic inflammation; NFL, nerve fiber layer; PA, periaxonal immunostaining; PR, photoreceptor; VI, viral inclusion; —, no lesions or no immunostaining.
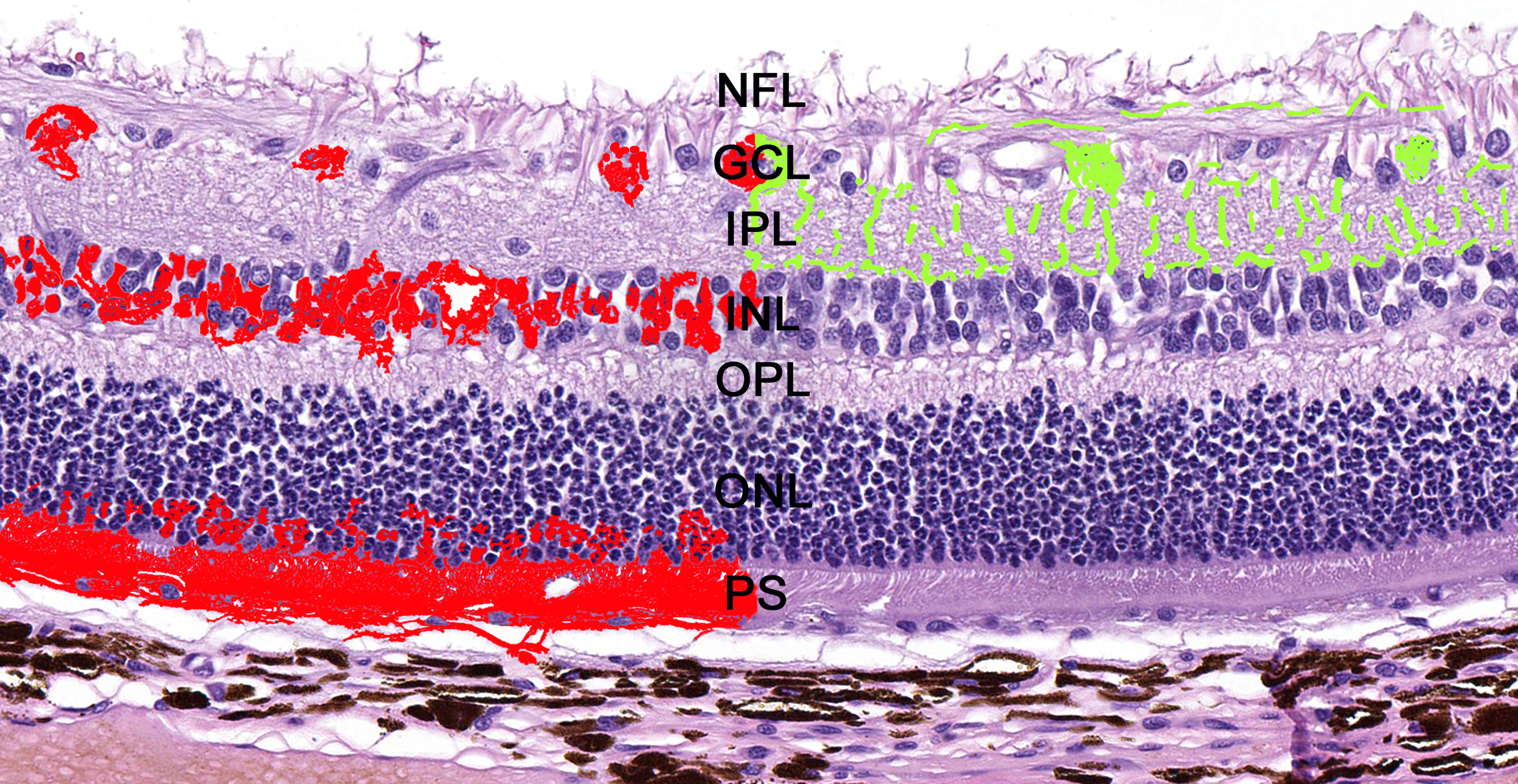
Schematic distribution of the lesions and viral antigen in the retina of rabid mammals. Degeneration and necrosis (left, in red) occurred in the ganglion cell layer (GCL), inner nuclear layer (INL), outer nuclear layer (ONL), and photoreceptor segments (PS). Viral antigen was detected by immunohistochemistry (right, in green) within the nerve fiber layer (NFL), GCL, and inner plexiform layer (IPL). OPL, outer plexiform layer. Hematoxylin and eosin.
Immunohistochemistry and PCR Results of 18 Cases of Rabies in Domestic and Wild Mammals.a
Abbreviations: FFPE, formalin fixed, paraffin embedded; LG, lacrimal gland; NA, not available; ON, optic nerve; +, positive staining; –, negative staining.
a Numbers reflect inflammation scores.
No optic nerve lesions were evident histologically, but immunolabeling for rabies virus antigen was present within periaxonal cells (likely Schwann cells) in 6 cases (Fig. 11). Perivascular CD3+ T lymphocytes were present in the optic nerve in 2 cases.
Lacrimal gland lesions were histologically evident in 11 of 18 cases and consisted of interlobular and lobular accumulations of lymphocytes and rare plasma cells (Fig. 12). Immunolabeling with rabies virus antigen was detected in acinar epithelial cells in 3 of 18 cases (Fig. 13). CD3 IHC revealed a predominantly T-cell infiltration in the 11 cases in which inflammation was evident histologically (Fig. 14). No CD20+ B lymphocytes or Iba+ macrophages were observed in the eye, optic nerve, or lacrimal glands. External and internal control tissues stained appropriately.
PCR for rabies was positive in 11 samples of eye (9/11 frozen samples and 2/7 FFPE samples) and in 8 samples of lacrimal gland (6 frozen samples and 2 FFPE samples). Of the 7 cases that tested negative by PCR (5 frozen samples and 2 FFPE samples), 5 were immunohistochemically positive for rabies. All DNA samples yielded a positive result for the internal control (ie, amplification of GAPDH by PCR).
Discussion
This study characterized the ocular and lacrimal gland lesions in naturally occurring cases of rabies in 7 domestic mammals and 11 wild mammals. The pathological changes in the eye, optic nerve, and lacrimal gland were similar in terms of distribution and nature across the cases. A few experimental studies have briefly documented the ocular lesions following rabies virus inoculation in dogs, monkeys, foxes, and skunks. 3,9,27 Similar studies of rabies in humans mainly examined the recovery of infective virus from the cornea in the context of disease transmission and diagnostics. 16,18
One difference between the current investigation and previous studies involves the distribution of the pathological changes in the eye. 3,10,27 In the current study, ocular lesions were restricted to the retina and were subtle in all cases, consisting of degeneration and necrosis of ganglion cells, inner nuclear layer cells, and photoreceptor cell bodies of the outer nuclear layer. This finding differs from previous studies in humans and other animals, in which degenerative changes occurred predominantly in the nerve fiber layer and ganglion cell layer. 3,9,16,27 Furthermore, ocular lesions in other animal species also occurred in other parts of the eye, including corneal and vitreous clouding with choroidal hemorrhage in experimentally infected rabbits 10 and lymphocytic anterior uveitis in experimentally infected monkeys. 27 In addition, intramuscular inoculation in foxes revealed viral antigen but no pathological changes in the corneal epithelium. However, no other ocular structures were examined in that investigation, and the true extent of the pathological changes in those cases remains unknown. 3,9 The reason for these differences remains elusive but could have resulted from the distinct clinical course between different individuals and species, site of inoculation, nature of inoculation (natural vs experimental), and particularities of distinct viral strains. 5,13
Although direct rabies virus infection of the eye with subsequent spread to the CNS has been reported in experimental and natural infections, 13,23 the distribution of the pathologic changes and viral antigen in our cases, which was restricted to the retina, supports ocular infection secondary to centrifugal viral spread through the optic nerve after CNS infection. 6 Viral infection of retinal ganglion cells has been demonstrated in experimentally infected mice at 6 days after inoculation, 6 and this mechanism was supported by the presence of viral antigen in the optic nerve using IHC in 6 of our cases. The lack of immunostaining in the other optic nerves may be a result of a low antigenic load or to differences in the progression and clinical course of the disease. 6 Given that these were natural cases of rabies, time of inoculation and interval between inoculation and euthanasia or death are unknown.
Intracytoplasmic viral inclusions (Negri bodies) were detected within ganglion cells in 8 of the 18 cases. Viral inclusions have also been reported in the ganglion cells of experimentally infected rabbits 10 and naturally infected humans, 16 as well as in the corneal epithelium of experimentally infected foxes. 3 The detection of eosinophilic cytoplasmic inclusions within neurons is highly indicative of rabies. 13 However, inclusions are not present in all cases, and the diagnosis should not rely solely on histopathology. 11,27 Furthermore, viral inclusions in the current study were small, often solitary, and difficult to detect. IHC was useful to identify rabies viral antigen in the retina and confirm the diagnosis, similar to what has been described in the CNS. 20,30
Ocular inflammation restricted to the retina is rare in animals and occurs mainly as part of neurotropic viral infections, including rabies. 34 In our study, scattered CD3+ T lymphocytes were found in 11 cases, with only 1 case that had sufficient lymphocytic infiltration to be detected on routine histopathology. Lymphocytic retinal inflammation has been demonstrated following experimental rabies virus infection in dogs and mice. 6,27 While the retinal inflammation associated with rabies and other neurotropic viral infections typically reflects the lymphocytic and plasmacytic inflammation present in the CNS, 34 our results revealed a minimal and predominantly T-cell response throughout the examined cases, with no B-cell component within inflammatory infiltrates. A lack of B-cell response has been occasionally found in cases of rabies and other viral infections of the eye. Experimental rabies in athymic nude mice resulted in a higher incidence and severity of ocular disease when compared to control BALB/c mice, indicating that T lymphocytes play an important role in the immune response to rabies virus infection. 6 T lymphocytes have also been shown to have a major antiviral effect during retinal necrosis following human herpesvirus 1 infection in mice. 2 The mechanism involved in the protective T-cell response to these viral infections remains poorly understood but has been attributed to T lymphocyte stimulation by proinflammatory cytokines such as p55TNFR. 6
Centrifugal spread of rabies virus from the CNS through various nerves can infect a wide variety of extraneural tissues. 19,24,31 Viral infection of salivary glands most likely occurs through the parasympathetic nerves originating in the medulla oblongata or the sympathetic nerves arising in the thoracic spinal cord. 5,7 Similar to the findings reported in the salivary gland of experimentally infected dogs and naturally infected humans, 13,27 interlobular and lobular inflammation was present in 11 of 18 lacrimal glands in the current study. In our cases, IHC revealed that inflammatory cells consisted primarily of CD3+ T lymphocytes, mirroring the observations in the retina and optic nerve. Furthermore, acinar epithelial cells in 3 of 18 lacrimal glands displayed positive immunolabeling for rabies virus. Similarly, viral antigen has been reported from the lacrimal gland in natural cases of human rabies, 22 as well as in the salivary gland of naturally infected dogs 29 and experimentally infected raccoons. 17 These findings suggest that, comparable to the salivary gland, the lacrimal gland could potentially act as a source of viral infection. 8,13,17,22 This hypothesis would need to be evaluated by the demonstration of infective virus in the lacrimal secretion of live animals with rabies.
The FAT is the standard diagnostic test recommended by the World Health Organization and the World Organisation for Animal Health for diagnostic confirmation of rabies. 13 While FAT results are typically reliable and fast, with a turnaround time of 2 to 3 hours, its sensitivity is highly dependent on the overall quality of the sample and the degree of postmortem autolysis. Therefore, other diagnostic modalities, such as IHC and PCR, have been optimized and may eventually replace FAT after proper validation has been completed. Furthermore, molecular testing offers additional advantages over FAT, especially sensitivity and the capacity to identify specific viral species and strains in more detail. 13 In the current study, we performed PCR in 2 sets of samples. Only 2 of 7 cases with FFPE tissues and 9 of 11 cases with frozen tissues were positive. DNA extraction from FFPE tissues is routinely performed given that formalin is the most commonly used fixative in pathology laboratories. However, RNA extraction from FFPE is more difficult and inefficient due to RNases, chemical modification of the RNA, and cross-linking of nucleic acids and proteins. 28 Despite extracting RNA within few months after embedding and using an RNA extraction kit for FFPE tissues that is suitable for real-time PCR, 32 we amplified Lyssavirus RNA from only 2 of 7 FFPE samples. Our results indicate that fresh eye and lacrimal gland tissues are more suitable than FFPE tissues for rabies virus testing using real-time PCR. Five samples that were negative by PCR had positive immunostaining for rabies virus. In addition, 2 samples of bat tissues (case Nos. 11 and 12) were negative for rabies using IHC and PCR. This could be explained by a potential false-positive FAT result or by a different genotype of the viral strain infecting these bats. However, the PCR assay used in this study amplifies and detects a region of the genome that is conserved across divergent Lyssavirus species. This assay was validated using a panel of 88 diverse Lyssavirus isolates belonging to different phylogroups, including several samples originated from bats. 33 Despite the broad reactivity of this assay, further research is warranted to clarify whether the bats were infected by a different genotype. Furthermore, negative IHC and PCR results could have resulted from the small eye and lacrimal gland tissue size in some species, low antigenic load, or advanced postmortem autolysis (which was present in these 2 cases). 1 These results confirm that more than 1 diagnostic modality should be used for diagnostic confirmation of rabies when adequate samples are not available for 1 specific laboratory test to be performed. 11
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
