Abstract
Bicuspid aortic valve (BAV) is the most common human congenital cardiac malformation. Although the etiology is unknown for most patients, formation of the 2 main BAV anatomic types (A and B) has been shown to rely on distinct morphogenetic mechanisms. Animal models of BAV include 2 spontaneous hamster strains and 27 genetically modified mouse strains. To assess the value of these models for extrapolation to humans, we examined the aortic valve anatomy of 4340 hamsters and 1823 mice from 8 and 7 unmodified strains, respectively. In addition, we reviewed the literature describing BAV in nonhuman mammals. The incidences of BAV types A and B were 2.3% and 0.03% in control hamsters and 0% and 0.3% in control mice, respectively. Hamsters from the spontaneous model had BAV type A only, whereas mice from 2 of 27 genetically modified strains had BAV type A, 23 of 27 had BAV type B, and 2 of 27 had both BAV types. In both species, BAV incidence was dependent on genetic background. Unlike mice, hamsters had a wide spectrum of aortic valve morphologies. We showed interspecific differences in the occurrence of BAV between humans, hamsters, and mice that should be considered when studying aortic valve disease using animal models. Our results suggest that genetic modifiers play a significant role in both the morphology and incidence of BAV. We propose that mutations causing anomalies in specific cardiac morphogenetic processes or cell lineages may lead to BAV types A, B, or both, depending on additional genetic, environmental, and epigenetic factors.
Keywords
In mammals, the aortic valve prevents the blood backflow from the aorta to the left ventricle. The normal condition, so-called tricuspid aortic valve, is characterized by the presence of 3 aortic sinuses—right, left, and dorsal (posterior in humans)—each supporting 1 leaflet (or cusp). Nevertheless, an alternative number of leaflets can exist, being considered a cardiac anomaly or malformation. This is the case of the bicuspid aortic valve (BAV), which has 2 sinuses and 2 corresponding leaflets. 2,29 BAV is the most frequent congenital cardiac malformation in humans, with an incidence of 0.5% to 2% in the general population. 3,21,46 BAV is clinically relevant because most affected patients develop functional valve defects and/or aortopathies during their lifetime. 62 In addition, the occurrence of BAV is frequently associated with other congenital cardiac malformations, such as ventricular septal defect or aortic coarctation. 13,25,36 Overall, it has been estimated that BAV disease is responsible for more human deaths than the sum of all the other congenital cardiac malformations. 66
Three different types of BAV have been distinguished 48 depending on the spatial orientation of the anatomical elements (Fig. 1). BAV type A or anteroposterior is the most frequent type in humans (∼74%). It shows 2 leaflets and 2 sinuses located at the anterior and posterior halves of the aortic root. BAV type A is also called right/left (R/L) BAV because it is the result of the anatomical fusion of the normal right and left developing leaflets to form the anterior leaflet. The remaining 2 types of BAV have 2 leaflets and 2 sinuses located at the right and left halves of the aortic root (latero-lateral BAV). They are the result of the anatomical fusion of the right and noncoronary (type B or right/noncoronary “R/N”) or the left and noncoronary (type C or left/noncoronary “L/N”) developing leaflets. BAV types B and C are less frequent in humans (B, ∼24%; C, ∼2%).
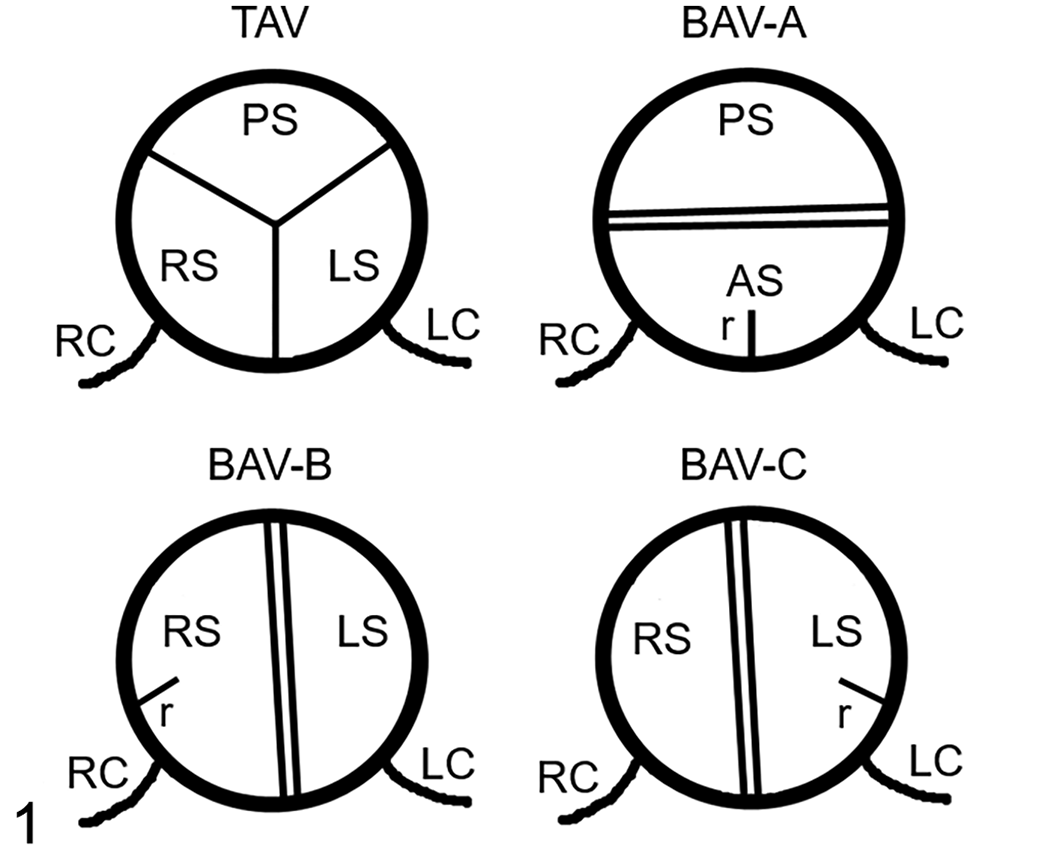
Anatomic types of aortic valves. Schematic representations of a tricuspid aortic valve (TAV) and the 3 types of bicuspid aortic valve: BAV-A or right/left [R/L], BAV-B or right/noncoronary [R/N], and BAV-C or left/noncoronary [L/N]. Cranial views. AS, anterior sinus; LC, left coronary artery; LS, left sinus; PS, posterior sinus; r, raphe; RC, right coronary artery; RS, right sinus.
Until recently, it was thought that the different BAV morphotypes represented mere anatomical variants of the same morphogenetic defect with no physiological significance. In 2009, experimental research with 2 animal models demonstrated that BAV types A and B form during embryonic development by 2 different morphogenetic mechanisms, indicating that each BAV type is a distinct etiological entity. 15 Further studies subsequently showed that each BAV type is differently associated with valvulopathies, aortopathies, and congenital cardiac malformations in human patients. 9,10,14,48,49,56,63
Currently, a relatively high incidence of BAV has been found in 29 strains of 2 rodent species: 2 Syrian hamster (Mesocricetus auratus) strains with BAV type A, 15,16,54,55 23 mutant laboratory mouse (Mus musculus) strains with BAV type B,* 2 mutant mouse strains with BAV type A, 20,65 and 2 mutant mouse strains with BAV types A and B. 34 These models have significantly improved our knowledge of BAV etiology. However, it is unknown how far the conclusions obtained from these rodent models can be extrapolated to humans. In this line, several questions arise: Why does each model present a specific BAV morphotype? What is the incidence and type of BAV in the 2 current model species for the disease? Although the aforementioned studies show a high incidence of BAV in mutant and spontaneous models, the morphological variation and incidence of BAV in each model species (Syrian hamster and laboratory mouse) have not been reported. These data would inform about the reliability of the extrapolation from these 2 species to humans.
The aim of the present study is to characterize the types of aortic valve anomalies and their respective incidences in different strains of Syrian hamsters and laboratory mice. To this end, we evaluated the aortic valve morphology of animals belonging to 4 inbred, 2 outbred, and 2 hybrid lines of hamsters, as well as 3 inbred lines, 4 outbred lines, and 1 hybrid line of mice. In addition, we reviewed and calculated the type and incidence of BAV in the published studies dealing with this anomaly in mice. Finally, we discuss the relevance of our results for understanding human BAV etiology.
Materials and Methods
The data included in the present study were obtained retrospectively from our databases. A total of 4340 hamsters and 1823 mice were used.
The hamsters belonged to 8 different populations (Table 1): 4 inbred strains (ID, IT, IH, and IB) generated in our laboratory, 2 outbred stocks (OC and OJ) purchased from different companies (Charles River, Wilmington, Massachusetts, and Janvier, Le Genest-Saint-Isle, France; respectively), and 2 hybrid lines (TC and TJ) generated by breeding inbred (IT) and outbred (OC and OJ) animals. The mice belonged to 7 different strains (Table 1), namely, 3 inbred (B6, Bc, and DBA) and 3 outbred (CD1, OF1, and NMRI) strains purchased from a company (Charles River) and a hybrid line (B6c) generated by breeding animals from 2 inbred strains (B6 and Bc).
Strain and Origin of 15 Lines of Syrian Hamster (Mesocricetus auratus) and Laboratory Mice (Mus musculus) Used in This Study.
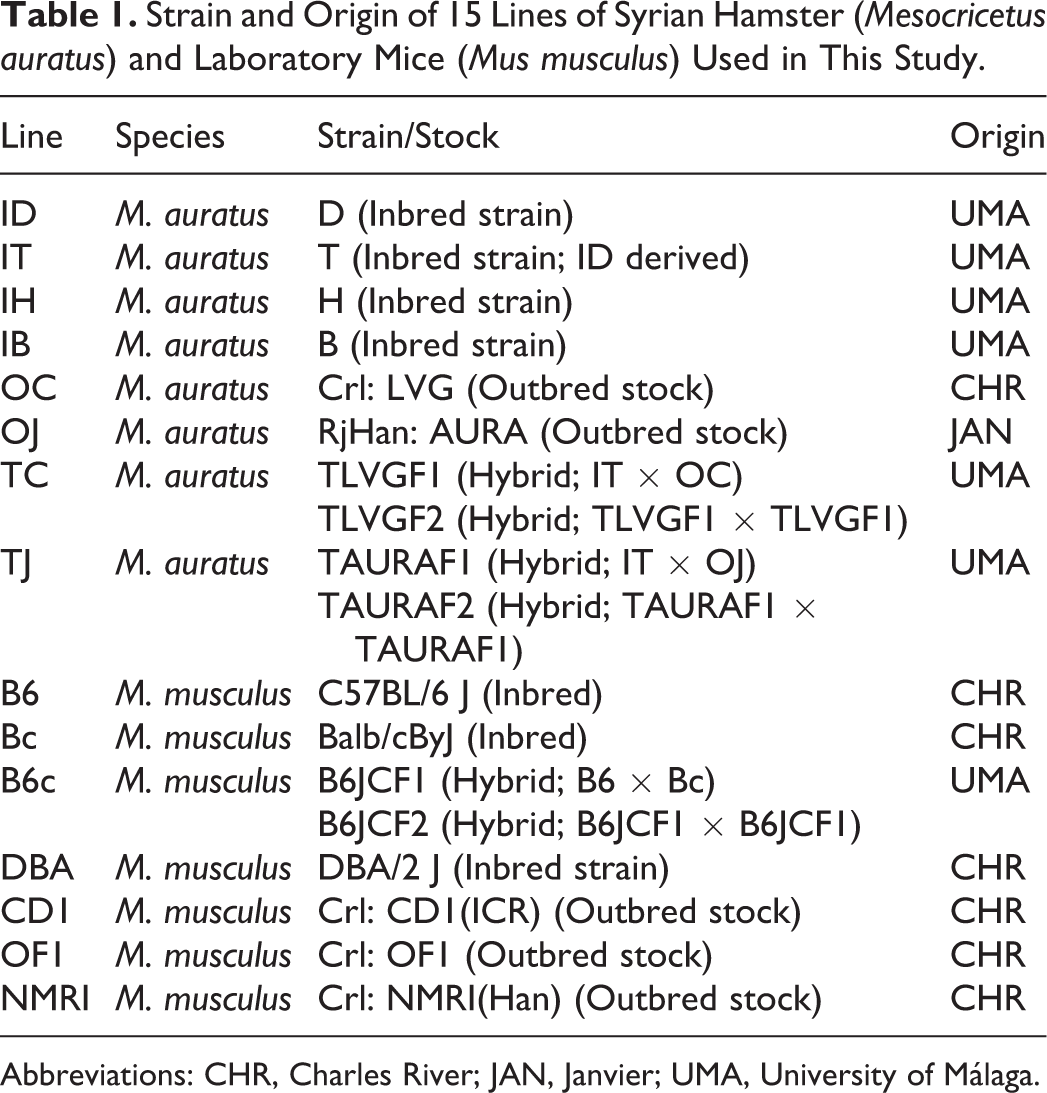
Abbreviations: CHR, Charles River; JAN, Janvier; UMA, University of Málaga.
The inbred hamsters were obtained after brother-sister breeding from the 1st to the 10th (IH), 29th (IT), 39th (ID), or 56th (IB) generation of consanguinity. Their coefficient of consanguinity ranged from 0.886 (IH) to 0.999 (IT, ID, IB). All the inbred mice were obtained after breeding animals of the 20th or higher generation of consanguinity, which means that they were isogenic, with coefficients of consanguinity higher than 0.999. Hybrid hamsters (TC and TJ) and hybrid mice (B6c) belonged to the first (F1) and second (F2: F1 × F1) generations of hybrid crosses.
All the animals used in this study met the following inclusion criteria: juveniles and adults, either found dead or killed in accordance with European and Spanish guidelines for animal welfare (R.D.1337/2013; B.O.E.08.02.2013), in which the anatomy of the heart and thoracic arteries was well recognized according to the protocol detailed below. The animals were housed in standard cages, fed water and chow ad libitum, and euthanized by anesthetic overdose (ketamine/xylazine), CO2 inhalation, or cervical dislocation.
After euthanasia, the chest was opened, and the heart, together with the ascending aorta and pulmonary trunk, was dissected out. The aortic arch and the free walls of the right and left ventricles were further dissected to detect possible cases of aortic coarctation or septal defects. The aortic valve was exposed by dissection under a Leica MZ12 stereomicroscope (Leica, Heerbrugg, Switzerland) and its morphology assessed. In some cases, the specimens were analyzed by means of scanning electron microscopy. These were fixed, dehydrated, gold-coated, and observed under a Jeol JMS scanning electron microscope (Jeol, Tokyo, Japan) operated at 10 or 15 kV. 54 In other cases, hearts were fixed by immersion in 4% paraformaldehyde, dehydrated, and embedded in Histosec (Merck KGaA, Darmstadt, Germany). Serial sections, transversely cut at 8 to 10 μm, were dewaxed, hydrated, and stained with Mallory’s trichrome stain and Weigert–Van Gieson stain or resorcin-fuchsin.
Descriptions of the aortic valve morphology followed the nomenclature by Sans-Coma et al. 55 Briefly, the mobile components of the arterial valves are the leaflets, of semilunar shape, which are attached to the walls of the aortic sinuses. The adjacent leaflets join distally along their attachments to the sinus wall. Proximally, subvalvar interleaflet triangles are interposed between adjacent sinuses. The distal vertex of this triangular space is the point at which the leaflets join to form the commissure. Although Sans-Coma et al 55 use the terms trifoliate and bifoliate aortic valves, we prefer here the terms tricuspid and bicuspid aortic valves, respectively, which are more common in the literature.
Results
Anatomy of the Aortic Valve in Hamsters and Mice
The anatomical components of the aortic valves were similar in hamsters and mice. Generally, the valve was tricuspid, with 3 leaflets, 3 sinuses, and 3 commissures (Figs. 2 , 5, 11, 13, 14). In some tricuspid aortic valves, the right and left leaflets were partially (Fig. 6) or almost totally fused (Fig. 7) in the cephalocaudal direction. Concomitant with the degree of the fusion, the ventral commissure (the point of contact of 2 adjacent leaflets) was displaced in the caudal direction, and the interleaflet triangle between the right and left sinuses was decreased in size.
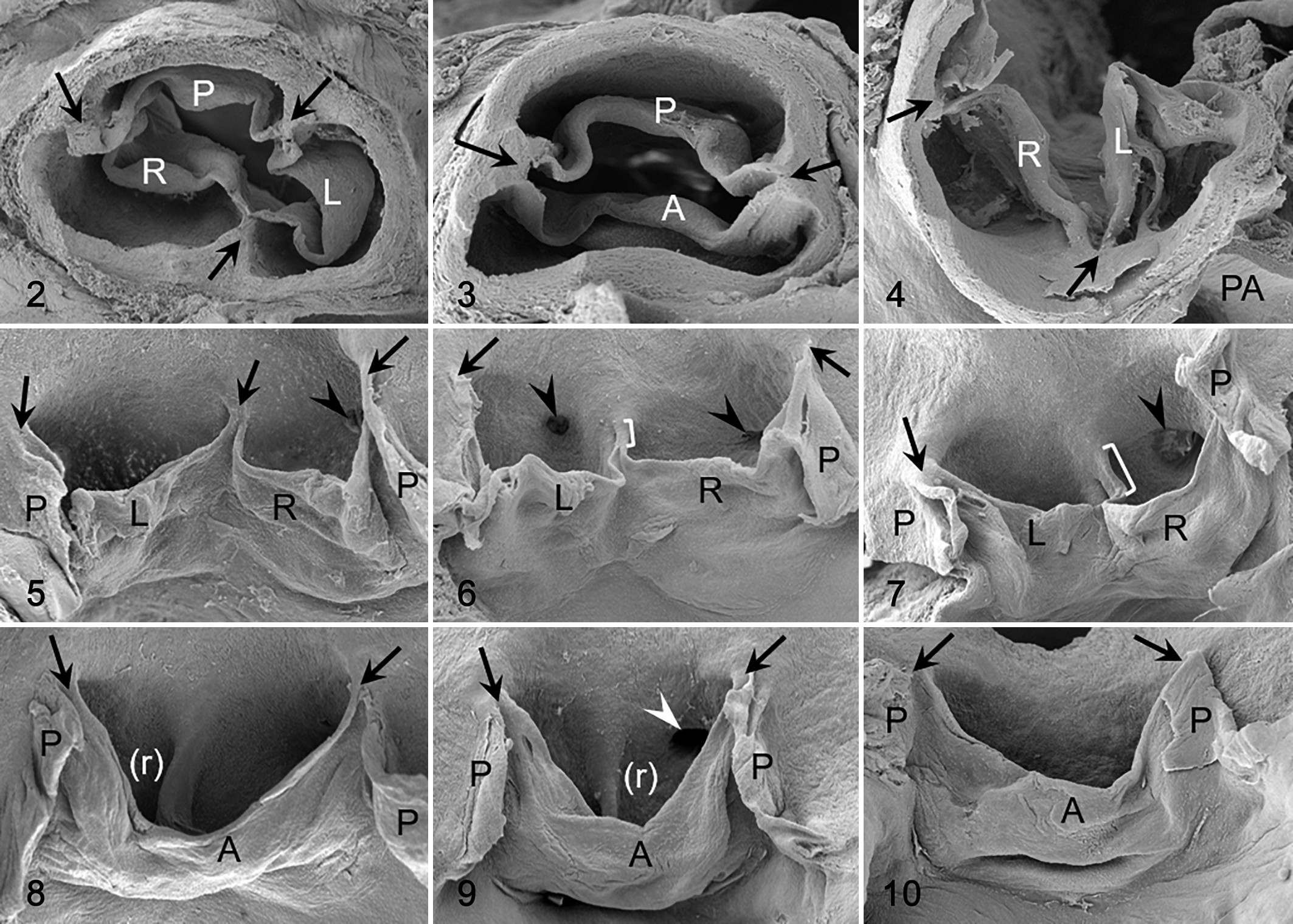
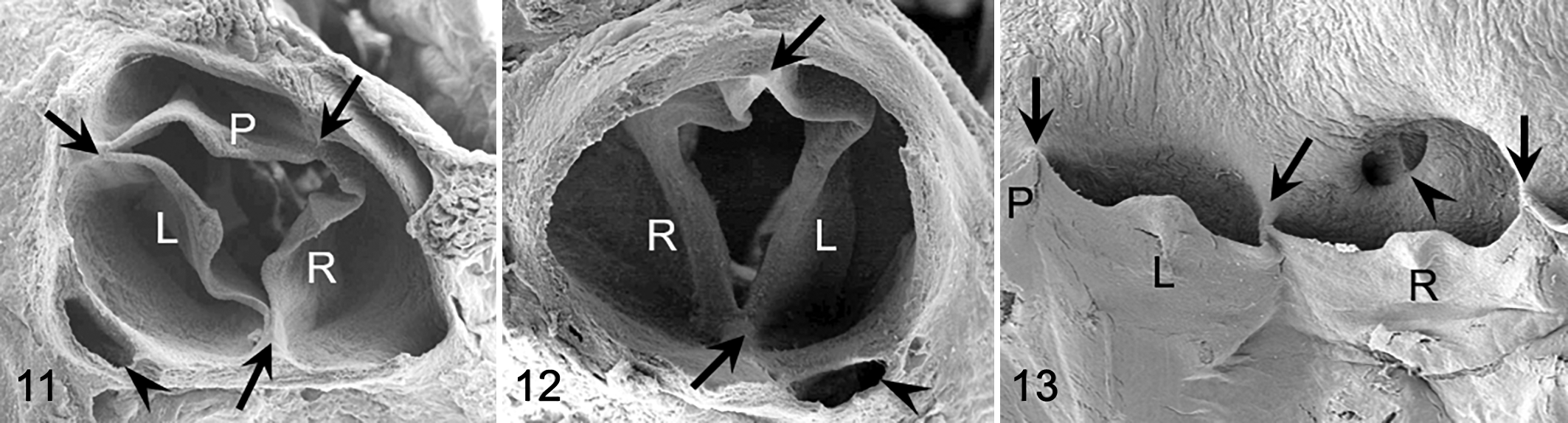
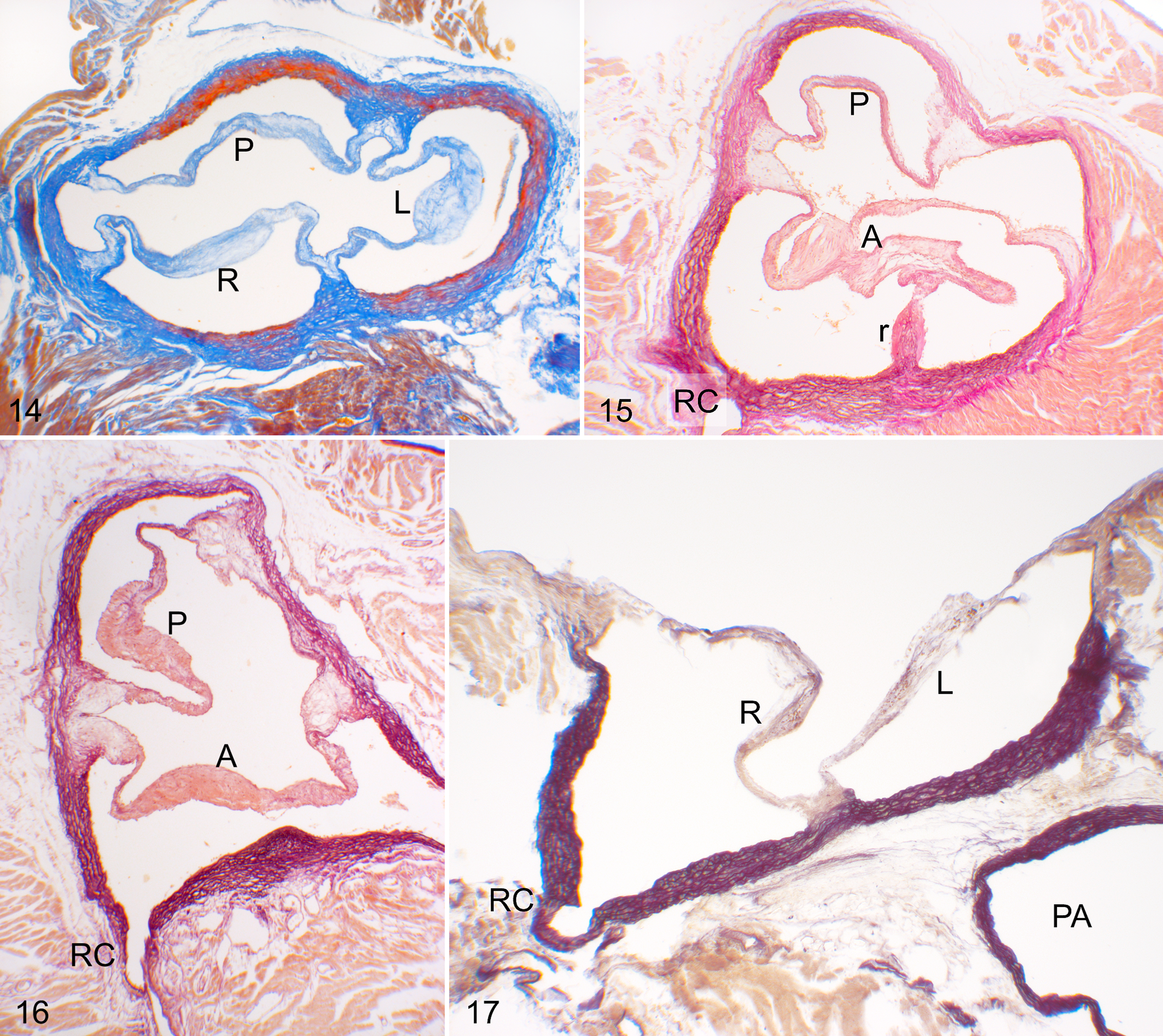
Two main types of aortic valve malformations were found in the cohorts studied: reduction (BAV) and increase (quadricuspid aortic valve) in the number of the valve components. BAVs were composed of 2 leaflets, 2 sinuses, and 2 commissures (Figs. 3, 4, 8 –10, 12, 15 –17). Two types of BAV were detected: type A (Figs. 3, 8 –10, 15, 16), with the 2 leaflets and sinuses located in ventral and dorsal position (corresponding to anteroposterior in humans), and type B (Figs. 4, 12, 17), with latero-lateral (right-left) orientation of leaflets and sinuses. Frequently, BAV type A showed a raphe located in the center of the ventral sinus (Figs. 8, 9, 15), sometimes widely encroaching toward the leaflet (Figs. 8, 15). Quadricuspid aortic valves were composed of 4 leaflets, 4 sinuses, and 4 commissures (Fig. 18). These valves occurred sporadically only in the hamster population.
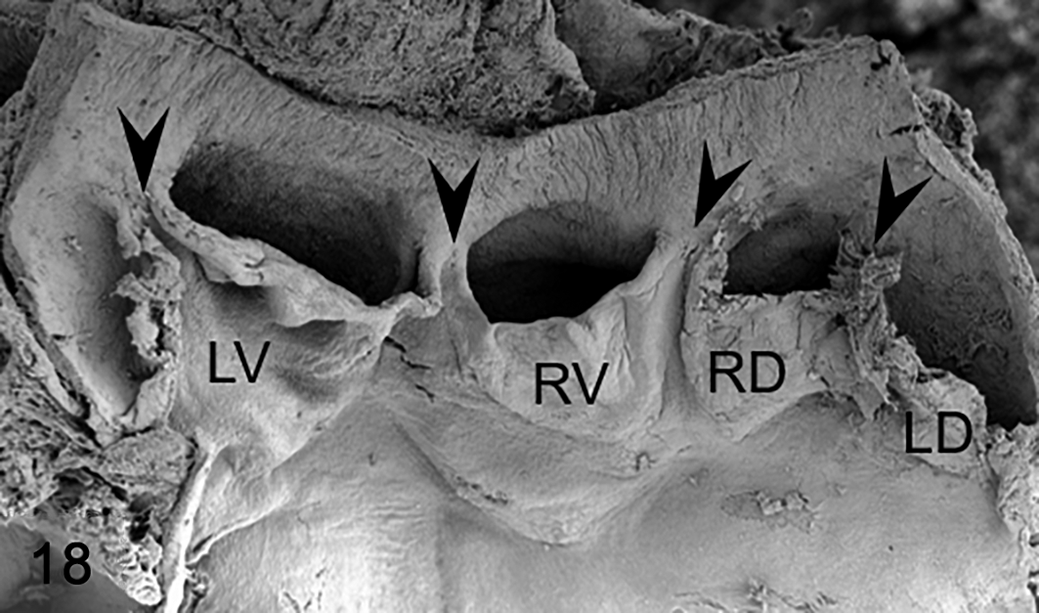
Abnormal valve anatomy, aortic valve, Syrian hamster. Scanning electron microscopy. Frontal view of a quadricuspid aortic valve. The dorsal aspect of the aortic root was dissected for a clear observation of leaflets and sinuses. Arrowheads indicate the commissures. LD, left dorsal leaflet; LV, left ventral leaflet; RD, right dorsal leaflet; RV, right ventral leaflet. Reprinted from López-García et al.31 Copyright 2015 by the Japanese Journal of Veterinary Research. Reprinted with permission.
Anatomical Types of the Aortic Valve in Hamsters
A total of 4340 hamsters were analyzed. These belonged to 4 inbred strains (ID, IT, IH, and IB), 2 outbred stocks (OC and OJ), and F1 and F2 hybrids of the IT strain and the 2 outbred stocks (TC and TJ). The incidences of the different valve phenotypes in the 8 hamster cohorts are given in Table 2.
Number and Incidence of the Different Valve Morphologies Found in the 8 Lines of Hamsters Studied.

Abbreviations: BAV-A, bicuspid aortic valve type A; BAV-B, bicuspid aortic valve type B; BAV-C, bicuspid aortic valve type C; QAV, quadricuspid aortic valve; TAV, tricuspid aortic valve.
The 2 outbred stocks (OC: n = 127 and OJ: n = 87) showed a relatively low (4.7%) or a null incidence of BAV type A, respectively. These BAVs presented a ventral raphe. Most OC (n = 121, 95.3%) and all OJ (n = 87, 100%) animals had a tricuspid aortic valve, with a relatively small proportion of them (OC: n = 11, 9.1%; OJ: n = 7, 8.1%) showing a variable degree of fusion of the right and left leaflets. Tricuspid aortic valves with leaflet fusion were distinguished from BAVs with raphe because the latter lacked the ventral interleaflet triangle, and raphes did not reach the cranial level of the commissures. 16
Two of the inbred strains of hamsters (IB: n = 1480 and IH: n = 34) also showed a relatively low (n = 68, 4.6%) or null incidence of BAV type A, respectively. Most (n = 57, 83.8%) of these BAVs presented a ventral raphe. In addition, 1 (n = 1, 0.1%) animal of the IB strain had a BAV type B without a raphe. Most (n = 1411, 95.3%) animals of the IB strain and all (n = 34, 100%) of the IH strain had a tricuspid aortic valve. The tricuspid aortic valves showed a fusion of the right and left leaflets in 267 (n = 267, 18.9%) specimens of the IB and 2 (n = 2, 5.9%) of the IH strains. Neither BAV type C nor quadricuspid aortic valve were found in these 2 strains.
The 2 remaining inbred strains of hamsters (ID: n = 1500 and IT: n = 895) showed a relatively high incidence of BAV type A (n = 795, 53% and n = 269, 30.1%, respectively), many of them with a ventral raphe (n = 466, 58.6% and n = 158, 58.7%, respectively). One (n = 1, 0.1%) animal of the IT strain had a BAV type B without a raphe. In addition, 3 (n = 3, 0.3%) animals of the IT strain and 1 (n = 1, 0.1%) of the ID strain presented a quadricuspid aortic valve. The remaining 704 (n = 704, 46.9%) hamsters from the ID strain and 622 (n = 622, 69.5%) from the IT strain had tricuspid aortic valves, many of them (ID: n = 273, 38.8%; IT: n = 272, 43.7%) with fusion of the right and left leaflets. No BAV type C was found in these 2 strains.
In one of the hybrid hamster cohorts (TJ: n = 130), the incidence of BAV (n = 15, 11.5%) was intermediate between that of the 2 parental lines, whereas the presence of a raphe (n = 6, 40%) did not differ significantly from the IT strain. However, in the other hybrid hamster population (TC: n = 87), the incidence of BAV (n = 5, 5.8%) did not differ from that of the control parental line (OC), whereas the presence of a raphe (n = 3, 60%) did not differ significantly from the IT strain. The remaining 115 (88.5%) animals of the TJ hybrid line and 82 (94.3%) animals of the TC line had a tricuspid aortic valve. In both hybrid populations, tricuspid aortic valves with fusion of the right and left leaflets were equally represented (TJ: n = 32, 27.8%; TC: n = 21, 25.6%). BAV types B and C and quadricuspid aortic valves were not found in hybrid hamsters.
Anatomical Types of the Aortic Valve in Mice
A total of 1823 mice were examined. They belonged to 3 inbred strains (B6, Bc, and DBA), F1 and F2 hybrids of the B6 and Bc strains (B6c), and 3 outbred stocks (CD1, OF1, and NMRI). The incidences of the different valve phenotypes in the 7 mouse populations are detailed in Table 3.
Number and Incidence of the Different Valve Morphologies Found in the 7 Lines of Mice Studied.

Abbreviations: BAV-A, bicuspid aortic valve type A; BAV-B, bicuspid aortic valve type B; BAV-C, bicuspid aortic valve type C; QAV, quadricuspid aortic valve; TAV, tricuspid aortic valve.
The aortic valve was tricuspid in the 433 mice belonging to the 3 inbred strains (B6, Bc, and DBA) and 2 outbred stock (CD1 and NMRI).
A BAV type B (Figs. 12, 17) was detected in 1 of 85 (n = 1, 1.2%) mice of the OF1 outbred stock and in 8 of 1305 (n = 8, 0.6%) hybrid B6c mice. These BAVs showed no raphe. The remaining 1381 mice of the OF1 and B6c cohorts (n = 1381, 99.6%) had tricuspid aortic valves. Fusions of the right and left leaflets as was described for hamsters were not found in any of the mouse cohort studied (compare Figs. 5 –7 with Fig. 13).
In both hamsters and mice, BAVs and quadricuspid aortic valves were found as isolated nonsyndromic anomalies. Congenital cardiac anomalies frequently associated with BAV, such as ventricular septal defect and coarctation of the aorta, were not detected in any of the animals examined. Anomalies in the origin of the coronary arteries were detected in hamsters of the ID (n = 495), IT (n = 168), and IB (n = 146) strains, as well as in mice of the B6 (n = 55) strains, with similar incidences to those described in previous studies. 16,32,51 A case of dissection of the ascending aorta was found in a hamster with a tricuspid aortic valve of the TJ line. Dilatation of the aortic sinuses occurred in 5 mice with a tricuspid aortic valve belonging to the OF1, NMR1, and B6c lines.
Literature Review
We performed a literature review of studies on nonhuman mammals in which BAV was reported. Most of them refer to the hamster (Mesocricetus auratus) and the mouse (Mus musculus), which constitute the main model species for BAV disease. The studies on BAV in the hamster have been performed by our own group, 15,51 –55 and our data of BAV incidence and anatomy are included in the results shown above.
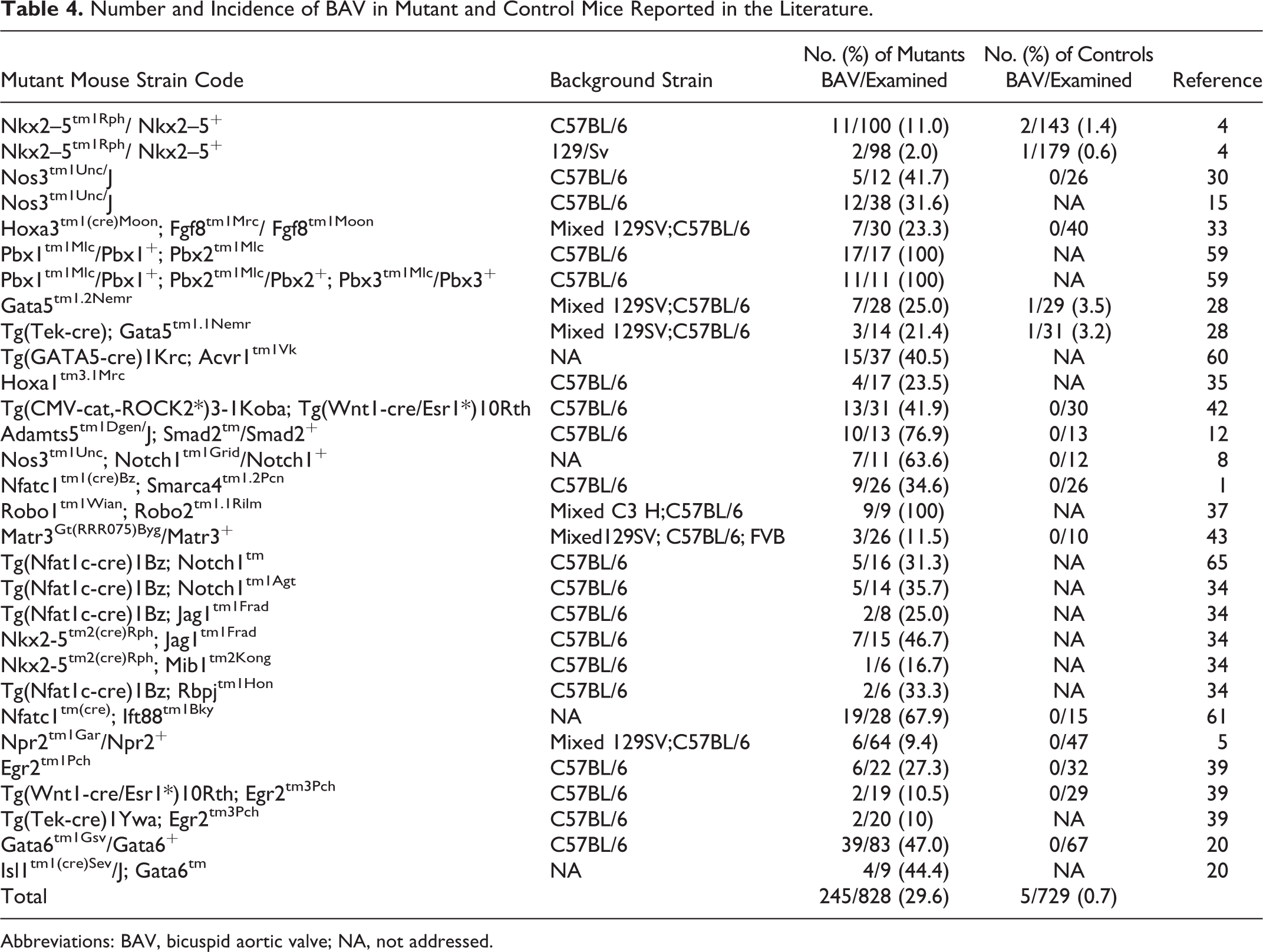
With respect to the mouse, a total of 20 studies were identified (Table 4), which included 250 mice with BAV. Most of these animals (n = 245) belonged to 27 transgenic mouse strains with a variety of genetic modifications and genetic backgrounds, whereas the remaining 5 mice with BAV were wild-type controls. In 18 of these transgenic strains, † BAV was associated with other cardiac and extracardiac malformations, whereas in the remaining nine, 5,8,12,28,33,39,61,65 BAV appeared as an isolated defect.
Number and Incidence of BAV in Mutant and Control Mice Reported in the Literature.
Abbreviations: BAV, bicuspid aortic valve; NA, not addressed.
Four of the 27 mutant mouse strains included animals with BAV type A (n = 52). These animals were defective in Gata6, Notch1, or Jag1. 20,34,39 BAV type B was found in transgenic mice (n = 193) belonging to 25 strains with altered expression of a variety of genes, namely, Adamts5, Acvr1, Egr2, Fgf8, Gata5, Gata6, Hoxa1, Ift88, Jag1, Matr3, Mib1, Nkx2.5, NOS3 Notch1, Npr2, Pbx1, Pbx2, Pbx3, RBPJ, Robo1, Robo2, Rock2, Smad2, and Smarca4, ‡ and in wild-type mice with pure or hybrid C57BL/6 and 129/Sv genetic backgrounds (n = 5). 4,28
The incidence of animals with BAV in the mutant strains ranged between 2% and 100%, depending on the genetic modification and the genetic background of the mutant strain. In most of these studies, no morphological variability of the aortic valve was described. Only 1 study reported a spectrum of valve morphologies depending on the size of the noncoronary leaflet and sinus. 60
Apart from human, hamster, and mouse, BAV has been found in only 3 species: the pig (Sus scrofa), 57 the California sea lion (Zalophus californianus), 27 and the dog (Canis lupus familiaris). 40,50,64 Oliveira et al 40 performed a large systematic study of the incidence of congenital cardiac malformations in 4480 dogs, and only 1 (0.02%) presented a BAV, although the anatomical type or the presence of a raphe was not detailed. The remaining cases were incidental findings of 3 BAV type A and 1 latero-lateral BAV in pig embryos, 57 1 BAV type B in a German Shepherd dog, 50 and 2 BAV type C in an English Bulldog 64 and in a Californian sea lion. 27
Discussion
In the present study, the incidence of BAV recorded in the Syrian hamster control strains ranged between 0% and 4.7%, with a mean value of 2.4% (Table 2). These percentages barely differ from those of the general human population (0.5%–2%). 3,21,46 In humans, BAV type A is the most frequent morphological variant (∼74%), followed by type B (∼24%) and type C (∼2%). 48 However, the proportion of each BAV morphotype in hamsters (type A: 99%; type B: 1%) diverged notably from that in humans. Between the tricuspid and the bicuspid condition, the aortic valve of hamsters shows a wide spectrum of intermediate morphologies, also found in human valves, 58 characterized by different degrees of fusion of the valve leaflets (Figs. 6, 7) and presence of raphes of different size (Figs. 8, 9, 15). 16,36,52,54,55 In addition, hamsters showed a low incidence of quadricuspid aortic valves (Fig. 18), with a similar morphology to that of humans (see also López-García et al 31 ). In the lines of mice examined, the incidence of BAV (from 0% to 1.2%, with a mean value of 0.3%) was lower than that of hamsters and humans (Table 2), and BAV type B was the only aortic valve anomaly detected. The morphology of the murine aortic valve was less variable than in humans and hamsters, with no commissural fusions and raphes (Figs. 11 –13, 17). These results are based on the analyses of 4340 Syrian hamsters and 1823 laboratory mice belonging to 8 and 7 distinct genetic backgrounds, respectively. We assume that this sample size is sufficiently representative of each species.
The studies reported in the literature (Table 4) described the presence of BAV in 250 animals belonging to 27 genetically modified mouse strains. In most of these strains, mice deficient in 24 possible genes showed exclusively BAV type B, whereas BAV type A was identified in only 4 strains, with defective expression of 3 possible genes (Gata6, Notch1, or Jag1). In addition, in most of these studies, the presence of raphes or leaflet fusions was not described. Therefore, our data, together with those from the literature, indicate that, in mice, the aortic valve shows little morphological variation, and BAV type B without a raphe is the predominant valve malformation. Compared to humans, both the hamster and the mouse show interspecific differences in BAV morphology and incidence, although murine differences are much more pronounced. These differences must be taken into consideration when studying aortic valve disease in animal models. In addition, although data on BAV morphology and incidence in other mammalian species are scarce, studies mainly performed in dogs also point to interspecific differences between this species and humans. 40
Animal models are unquestionably useful tools for research on human disease. However, possible interspecific differences must always be taken into consideration when aiming to extrapolate results from one species to another. Even though minimal or negligible interspecific differences may exist for a specific trait or biological process, other possible intrinsic constraints of the selected animal model should be considered. One of these constraints may be named intraspecific difference when the trait or biological process to investigate varies among different populations of the animal model species. Intraspecific differences are particularly relevant in biomedical research, where the majority of studies involving animal models rely on a few inbred strains of laboratory mouse (see Garner et al 19 ). By definition, an inbred (isogenic) strain is an animal population with minimal genetic and phenotypic variation, given that each strain is genetically uniform, with all animals sharing the same genotype, being comparable to clones. 17,18 Thus, when we extrapolate the experimental results obtained from a specific inbred animal strain to humans, we are comparing a genetically and phenotypically uniform animal population with the wide spectrum of genotypes and morphological heterogeneity of human populations.
In this context, previous studies in mutant mice have shown that the penetrance of the BAV type B phenotype is genetic background dependent, being found preferentially in mutant mice in a B6 genetic background. 4 Interestingly, most of the mice with BAV in the present study were hybrids (B6c) with 50% of the B6 genetic background. These data support the notion of Biben et al 4 that allelic differences among genetic backgrounds can modify BAV incidence in the laboratory mouse. Thus, not only interspecific but also intraspecific differences must be considered in studies on BAV disease involving murine models. With respect to hamsters, the incidence of BAV type A in animals resulting from hybrid crosses also varied widely depending on the genetic background of the parents, suggesting that modifier alleles for BAV type A incidence also operate in the Syrian hamster. We propose that current strategies to identify alleles responsible for phenotypic differences between mouse strains (linkage genetic studies) can be implemented to identify new genes involved in BAV etiology.
In the hamster model, BAV type A shows a complex mode of inheritance 52,55 and is associated with one particular genetic background 16,54,55 existing in the 2 related inbred strains ID and IT, whereas BAV type B sporadically appeared in IT and IB strains. These data suggest that the genetic mechanism involved in BAV formation in the ID and IT strains specifically alters the morphogenetic mechanisms involved in the formation of BAV type A. It remains to be elucidated whether the ID and IT genetic backgrounds contain either BAV type A specific mutations or BAV-causing mutations together with type A–specific modifier alleles.
Evidence of the influence of genetic modifiers on BAV morphology in humans already exists. Previous comparative studies on BAV embryology between hamsters with BAV type A (IT strain) and mice with BAV type B (NOS3-deficient mice) concluded that each type of BAV relies on a distinct etiology. 15 These findings led to the hypothesis that each BAV type results from a different genotype. 9,11,22,26 However, subsequent studies in human cohorts revealed discordant BAV morphologies in first-degree relatives. 9,47 In addition, BAV types A and B have been described in fetuses with Turner syndrome caused by the same genetic abnormality. 63 These data suggest that some genotypes may lead to BAV type A or B depending on genetic modifiers, 7,63 although the influence of environmental and epigenetic factors on BAV morphology cannot be discarded.
The finding in the present study that mice mostly develop BAV type B encourage the revisitation of our knowledge of BAV etiology driven by murine studies. Would a mutation leading to BAV type B in mice give rise to different valve phenotypes in other species such as humans or hamster? It has been shown that rare sequence variants of Gata5 in humans are associated with the development of both BAV types A and B, 6,41 whereas Gata5-defective mice develop only BAV type B. 28 Thus, we must conclude that some of the gene mutations identified in the mouse model can give rise to BAV type A or B in humans, depending on additional factors, probably genetic modifiers. Still, some mutations leading to BAV are probably type specific. This seems to be the case in the hamster model and in some mouse models (present results), and it is predictable for humans according to the results of human familiar and genetic studies. 6,9,38,47,63
Further research is required to determine the morphogenetic basis of a single mutation leading to 2 morphological types of BAV (ie, how a single genetic mutation can alter the morphogenesis of the cardiac outflow tract, resulting in 2 possible BAV phenotypes). According to the knowledge obtained from animal models, BAV type A results from a defective septation of the embryonic cardiac outflow tract, in particular from an excessive fusion of the 2 opposing conotruncal ridges. 15,34,65 BAV type B results from an abnormal formation and/or remodeling of endocardial cushions, leading to the absence of the noncoronary accessory cushion or its fusion with the right conotruncal ridge. 1,12,15,28,34,65 Both developmental processes, conotruncal septation and endocardial cushion formation, rely on the behavior of 2 different cell lineages (ie. neural crest and second heart field) 15,20,22,54,65 and on the regulatory functions of distinct signaling pathways (eg, Jagged/Notch and BMP). 23,24,44,45 On one hand, a mutant gene, expressed early during endocardial cushion formation and later during outflow tract septation, could lead to BAV type A or B, depending on additional genetic factors (modifier genes expressed during each of these embryonic processes), environmental factors (embryonic blood flow), and/or epigenetic factors. This hypothesis seems to be consistent, since Jagged and Notch conditional mutant mice develop BAV types A and B. 34 On the other hand, the abnormal behavior of neural crest or second heart field cells (caused by a variety of possible genetic defects) could affect endocardial cushion formation or outflow tract septation, leading to BAV type A or B depending again on genetic, environmental, and/or epigenetic factors. This hypothesis seems also consistent, since defective cardiac neural crest cell migration into the cardiac outflow tract has been shown to cause BAV type A in inbred hamsters 15,54 and BAV type B in Egr2, 39 Fgf8, 33 Hoxa1, 35 and Rock 42 deficient mice.
Conclusions
The present study uncovers previously undetected interspecific and intraspecific differences in the phenotypic expression of aortic valve malformations between humans and animal models. The Syrian hamster can develop a wide spectrum of aortic valve anomalies, including fusion of the valve leaflets and bicuspid and quadricuspid aortic valves, similar to those found in humans, although BAV type A is overrepresented. Unlike humans and hamsters, the laboratory mouse shows a narrow spectrum of aortic valve defects, predominantly BAV type B without raphe. Thus, interspecific differences must be considered when studying aortic valve disease using animal models. In addition to interspecific differences, intraspecific differences in both mouse and hamster strains point to the existence of genetic modifiers of BAV incidence.
Our data allow the reinterpretation of studies on BAV etiology to conclude that among BAV-associated mutations, some may influence the development of BAV type A or B depending on genetic modifiers, environmental factors, or epigenetic factors, whereas other may be type specific.
We raise the hypothesis that mutations in a single gene expressed in 2 key developmental processes (ie, endocardial cushion formation and outflow tract septation) or in 2 key embryonic cell lineages (ie, neural crest and second heart field) may lead to BAV type A or B, depending on additional genetic, environmental, and/or epigenetic factors.
Footnotes
Acknowledgements
We thank Mr. Luis Vida, Málaga, for his technical assistance and Mr. Gregorio Martín, Málaga, for assistance in operating the scanning electron microscope.
Notes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Junta de Andalucía (grant P10-CTS-6068); Ministerio de Ciencia, Innovación y Universidades (grant CGL2017-85090-P and fellowships FPU15/03209 to M.A.L.-U. and PRE2018-083176 to M.T.S.-N.); Sistema Nacional de Garantía Juvenil (contract UMAJI75 to M.T.S.-N.); and FEDER funds.
