Abstract
Migration of vertically transmitted Toxocara canis larvae through the liver and lungs is poorly documented as a cause of periparturient mortality in puppies. This case series describes 4 cases of fading puppies in 2 litters from 2 different bitches owned by the same breeder. Of the 4 cases, 4 had verminous pneumonia, 2 had fibrinoid necrosis of pulmonary arterioles, 4 had hepatic necrosis and inflammation, 2 had hepatic thrombophlebitis, and 1 had tracheal occlusion. These lesions were associated with migrating nematode larvae morphologically consistent with T. canis. The identity of the larvae was confirmed by sequencing of a portion of the ITS-2 region of nuclear ribosomal DNA. The tissues involved are consistent with the known migration pathways of this parasite. The dam of the first litter was negative for Toxocara spp. and other intestinal parasites by fecal floatation. This report highlights the need to consider T. canis migration in the differential diagnosis of fading puppies.
Mortality in neonatal puppies has a wide variety of causes, including congenital defects, dystocia-associated hypoxemia, various viral and bacterial infections, trauma, and homeostatic immaturity, especially of the cardiopulmonary system. 4,17 Death in pups older than 48 hours is sometimes attributed to the so-called fading puppy syndrome, a diagnosis of exclusion for which no single cause has been established. 1 Toxocara canis infection can be a cause of periparturient disease, with “inflammation of lung tissue” accounting for 51% of mortality within the first 5 days of birth in one study. 15 T. canis is a common nematode parasite with worldwide distribution and zoonotic potential, for which canids are the definitive host. 3 Transmission of the parasite can occur by ingestion of embryonated fecal roundworm eggs (horizontal infection) or ingestion of a paratenic host. In addition, activation of tissue-encysted larvae in a pregnant or lactating bitch may lead to vertical transmission of T. canis larvae via transplacental 18 and transmammary routes. 11,18 Transplacental transmission is the dominant vertical route in most cases, although the relative importance of the transmammary route increases when a naive bitch is infected in late gestation or at or after parturition. 2
Following transmission from the dam during late gestation, the fetal liver and other parenchymal organs contain inactive third-stage T. canis larvae. 6,14,15 Within 30 minutes to 72 hours of parturition, larvae exit the organs, enter the circulation, and extravasate in the pulmonary parenchyma. 14,15 Larvae initially occupy alveoli, then move up the airways to the oropharynx, where they are swallowed and establish intestinal infection 2 to 6 days after parturition. 6,14,15 Microscopic lesions of larval migration include eosinophil-rich necrotic foci in the liver and lung. 18 Most of the early reports on T. canis focused on experimental infection and life cycle definition. 2,6,16 Natural T. canis larval migration is not commonly recognized as a cause of clinical disease or mortality in periparturient or fading puppies. 1,11
Two Scottish Terrier littermates delivered via caesarean section from a 3-year-old bitch appeared healthy at birth, but their mentation and physical activity began to decline soon after, until they became nonresponsive and died 3 days postdelivery. Three days following the birth of the first litter, 3 puppies were delivered by caesarean section from a second Scottish Terrier bitch kept on the same property. Within 7 days, 2 of the 3 puppies had “faded” similarly to the first litter and were euthanized. Necropsies were performed on all 4 of the deceased puppies from the 2 bitches. Both bitches were in good health throughout. One week following the deaths of these puppies, fecal egg floatation testing of the first dam was negative for T. canis and other parasitic ova. The single surviving puppy was lost to follow-up.
On postmortem examination, case No. 1 (male) and case No. 2 (female) from the first litter both had viscous fluid adherent to the visceral pleura, congestion of the lungs, few 1-mm-diameter white-tan gritty nodules in the lungs, and similar lesions on the edges of some liver lobes. In the second litter, case No. 3 (male) had a mildly enlarged and mottled liver, and case No. 4 (female) had mottling of the edges of the liver similar to case Nos. 1 and 2. The 2 euthanized pups had milk in their stomachs, and all 4 appeared in good nutritional condition.
Microscopically, all 4 puppies had evidence of larval migration within the lung and liver. The lungs had patchy filling of alveoli with foamy macrophages, eosinophils, neutrophils, occasional multinucleated giant cells, and erythrocytes, as well as necrotic basophilic and eosinophilic debris that occupied alveoli and adjacent bronchioles (Fig. 1). Occasional arterioles had fibrinoid necrosis (Fig. 4). Many nematode larvae were within alveoli (Fig. 2). Larvae were also within the lumena of some bronchi and bronchioles (Fig. 3). The tracheal lumen contained multiple (case No. 2) or few (case No. 1) larvae within a cellular and proteinaceous inflammatory exudate. Case Nos. 3 and 4 had nematode larvae within the small intestinal lumen, often nestled between villi (Fig. 5).
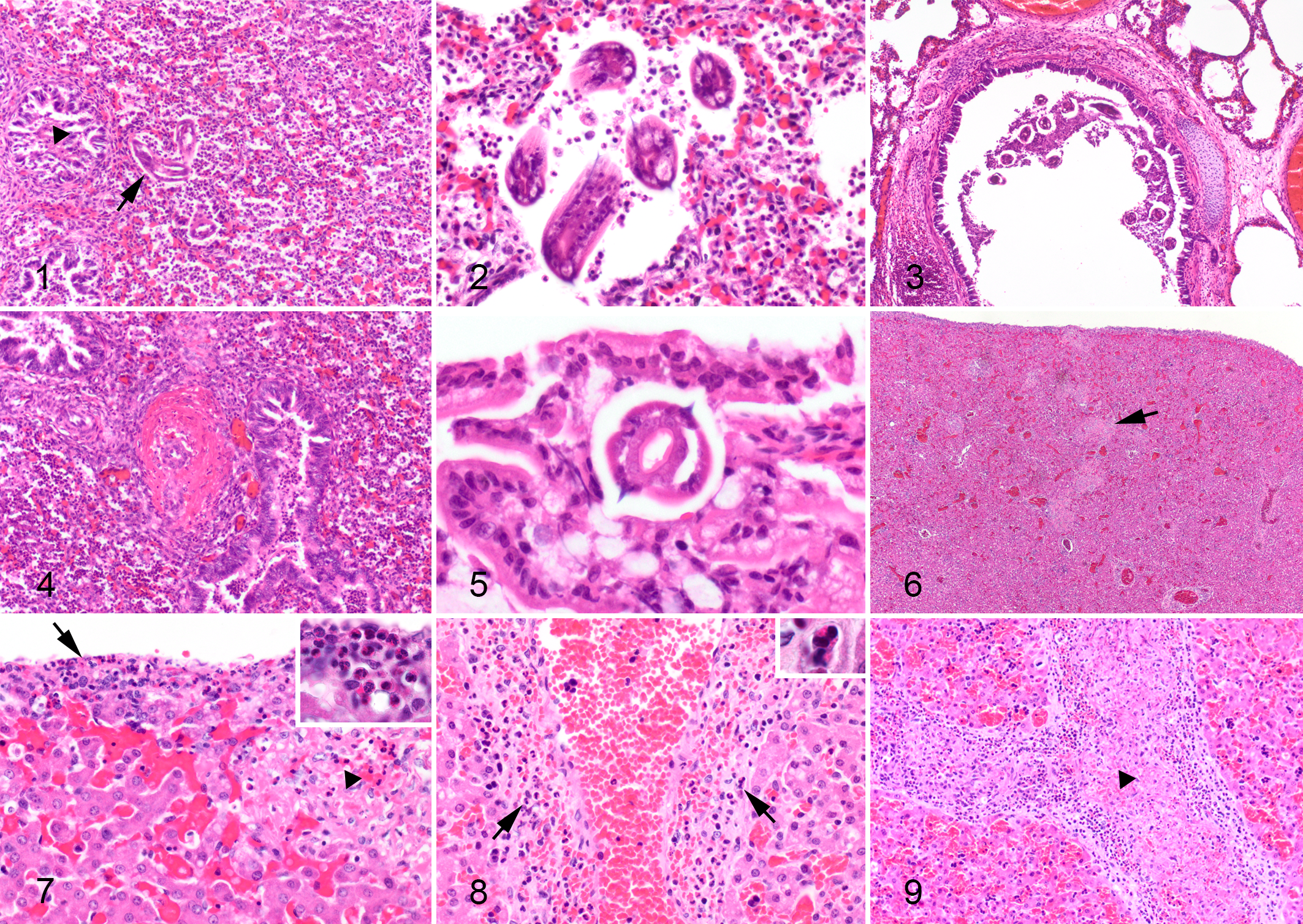
Toxocara canis larval migration, puppies. Hematoxylin and eosin (HE).
The nematode larvae were approximately 20–50 μm in diameter, with a thick, smooth cuticle; coelomyarian musculature; lateral alae; lateral chords; an intestine composed of uninucleate cells with a brush border; and absent mature reproductive organs (Figs. 2, 5), consistent with the larval stages of T. canis.
In the liver of all 4 puppies, there were random small foci of hepatocellular necrosis (Figs. 6, 7) with neutrophils and eosinophils as well as mild eosinophilic and mononuclear periportal and subcapsular hepatitis (Fig. 7). In case Nos. 1 and 2, some small and large hepatic veins had eosinophilic and neutrophilic infiltrates (Fig. 8) with associated thrombosis (Fig. 9). There was subjectively increased granulopoiesis within the periportal extramedullary hematopoietic tissue in case Nos. 1 and 2. Case Nos. 3 and 4 both had small foci of necrotizing and hemorrhagic pancreatitis, associated with large numbers of eosinophils, macrophages, and neutrophils. No larvae were seen within the hepatic or pancreatic parenchyma of any of the pups. In case No. 3, there were a few small foci of coagulative necrosis in the myocardium.
Paraffin-embedded blocks of formalin-fixed lung that contained nematode larvae were used as source material for the extraction of DNA. Using a block from each puppy, 10-μm sections were cut, and DNA was extracted using a commercial kit (Roche High Pure FFPET DNA isolation kit; Roche, Basel, Switzerland), per the manufacturer’s instructions. An aliquot of the extracted DNA was amplified using 2 different primer sets sourced from the literature, 5,8 both targeting the ITS-2 region of nuclear ribosomal DNA for T. canis. A positive control, consisting of DNA extracted from a morphologically confirmed adult T. canis, and a negative control containing no DNA were run simultaneously. Polymerase chain reaction (PCR) was performed using 5× HOT FIREPol Blend Master Mix 10 mM MgCl2 (Solis Biodyne, Tartu, Estonia) following the manufacturer’s recommendations, with 1 μl of template in a total volume of 20 μl. Touchdown PCR was performed on a Labcycler (SensoQuest, Göttingen, Germany) with a starting annealing temperature of 60°C and a final annealing temperature of 54°C. The PCR product was separated by electrophoresis on a 1% w/v agarose gel (Bioline, Nottingham, UK) using RedSafe (iNtRON Biotechnology, Seoul, South Korea) and visualized under UV light. Size of PCR products was estimated in comparison to a HyperLadder 100-bp molecular ladder (Bioline, Nottingham, UK).
The positive control and samples from all 4 puppies produced a clear band of the appropriate size for each primer set (∼390 bp for Tcan1-NC2 and ∼330 bp for YY1-NC2). To confirm the identity, positive bands were cut from the gel and submitted for bidirectional Sanger sequencing. The resultant sequence was subjected to BLAST analysis through the National Center for Biotechnology Information database (https://www.ncbi.nlm.nih.gov) and aligned to the ITS-2 region of T. canis (MF495480) with 100% coverage and pairwise identity, as well as an e-value of 5e–81, supporting the morphological identification of the larvae.
Following the deaths of these pups, the first bitch was rebred, with aggressive deworming with praziquantel (dosage unknown) every 2 weeks as well as daily fenbendazole (dosage unknown) treatment from day 42 of gestation. The resulting litter contained 3 pups delivered by caesarean section that began fading at 4 days postdelivery. All pups were dosed with fenbendazole (30 mg/kg once daily × 3 days) and again at 16 days. At 22 days, 1 pup became dyspneic and dehydrated with decreased rectal temperature and a distended abdomen. The pup was euthanized, but no autopsy was performed. The other 2 puppies recovered and were healthy at 6 weeks.
Parasitic pneumonia combined with necrotizing hepatitis and pancreatitis were considered to have resulted in the death or euthanasia of the pups in this report. Given the ages of these pups, transplacental and possibly transmammary transmission were the most likely routes of infection.
References to T. canis migration-associated morbidity and mortality are common, but primary reports are scarce. Stillbirths may be due to primary hepatic or even pulmonary damage by larval migration in utero. 12,15 According to Levine, 7 verminous pneumonia associated with large numbers of prenatally acquired larvae can be a major cause of mortality in puppies within the first 2 to 3 days of birth. Reported lesions were inflammatory foci of leukocytes, fibrin and haemorrhage, as well as larvae within alveoli and bronchioles. 7 When small numbers of larvae are present, petechiae on the pulmonary pleura may be the only gross lesions. 7 Puppies that survive a heavy prenatal larval burden may develop progressive emaciation, abdominal distension, diarrhea (sometimes containing mature worms), and constipation and frequently die 2 to 3 weeks after birth. Vomition is also frequent, and vomitus may also contain adult Toxocara. 7 There is a recent report of a fading puppy syndrome in 12-day-old German Shepherd puppies that suffered anorexia, emaciation, hypothermia, and death ascribed to obstruction by intestinal burdens of T. canis (up to 75 per puppy); tissues were not examined histologically. 13 Infected puppies may also die of obstruction of the gallbladder, bile duct, or pancreatic ducts; jaundice; intestinal rupture; peritonitis; or pneumonia. 1,13,14 Hepatic changes similar to our cases, but without necrotizing vasculitis, have been described experimentally in dogs (ages not stated) that were part of a euthanized time series. 18 Many references addressing this syndrome are at least half a century old. More recent articles 4,17 on canine neonatal mortality (deaths during the first week of life) do not mention Toxocara larval migration.
No larval nematodes were seen in sections of liver or pancreas. T. canis larvae are postulated to cause the hepatic and pancreatic lesions based on the eosinophils within lesions and the known migration cycle of T. canis. It is likely that the nematode larvae completed their hepatic and pancreatic transit prior to the death of the pups. In the older pups from the second litter, larval nematodes had reached the small intestine and had invaded the intervillous spaces. There was no systemic vasculitis in any pup, and no other cause was found to explain the vascular or hepatic parenchymal lesions. Hepatic necrosis and hemorrhages are common in neonatal canine herpesvirus and adenovirus (infectious canine hepatitis) infections, 19 but other lesions consistent with these infections were not present.
The prevalence of T. canis infection in various countries ranges from 3% to 83%. 3 The highest prevalence is in dogs under 1 year of age. The dam of case Nos. 1 and 2 tested negative on fecal egg floatation soon after parturition, suggesting a lack of patent intestinal infection despite evidence of vertical transmission. This is consistent with reports that female dogs are less likely than their male age-matched counterparts to have patent intestinal infections, yet they have a high predilection for reactivation of tissue (mostly muscle)–encysted larvae during pregnancy. 14,15,18 Pregnancy and lactation are thought to induce immunological suppression in bitches. 9 Furthermore, immature larval stages passed in the feces of puppies are ingested by the dam and may result in heavy intestinal adult worm burdens, 9,10 meaning that even bitches treated with anthelmintics prior to pregnancy may eventually shed fecal eggs and contribute to environmental contamination.
Persistent infection in treated dogs may be due to increased anthelminthic resistance of tissue-encysted larvae. 11 Although 3-day consecutive fenbendazole and albendazole treatments have been shown to reduce the numbers of tissue-encysted larvae in nonpregnant bitches, the treatment of infected bitches during pregnancy is not recommended. 11
In conclusion, practitioners and pathologists investigating fading puppies should consider verminous pneumonia secondary to T. canis larval migration as a possible differential diagnosis.
Footnotes
Acknowledgements
We thank Mike Hogan and Evelyn Lupton for technical assistance; final-year veterinary students Nicola Bell, Megan Hero, Nan Joo (Grace) Kim, and Tom Sarjant; veterinarians Genevieve Rogerson and Margaret Young; and the breeder for assisting with follow-up and providing deworming protocols and a thorough history. Grateful thanks to Nutnapong (Sam) Udomteerasuwat and Keren Dittmer for assistance with the images.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
