Abstract
S100A4 (metastasin), a member of the S100 protein family, was initially identified in metastatic cells and is well established as a marker of aggressive human cancer. However, expression and roles of S100A4 in canine mammary tumors have not been clarified. In this study, expression of S100A4 was examined immunohistochemically in normal, hyperplastic, and neoplastic mammary glands of dogs. In all normal and benign lesions, S100A4 was restricted to a few stromal fibroblasts and inflammatory cells. However, in 7 of 57 (12%) of the malignant tumors examined, cytoplasmic and nuclear expression of S100A4 was observed in epithelial tumor cells and stromal cells. Particularly, the frequency of S100A4-positive anaplastic carcinomas was high (4/8 cases, 50%). Next, we established a novel cell line, named NV-CML, from a S100A4-positive canine mammary carcinoma. The cultured NV-CML cells and the tumors that developed in the immunodeficient mice after subcutaneous injection of the cells maintained the immunophenotype of the original tumor, including S100A4 expression. Using this cell line, we examined the cellular functions of S100A4 using RNA interference. S100A4 expression level in NV-CML cells transfected with small interfering RNA (siRNA) targeting canine S100A4 (siS100A4) was reduced to about one-fifth of those with negative-control siRNA (siNeg). Cell proliferation in WST-8 assay and cell migration in Boyden chamber assay were significantly decreased in siS100A4-transfected cells compared with siNeg-transfected cells. These findings suggest that S100A4 may be related to progression of canine mammary carcinomas via its influence on cell growth and motility.
S100 proteins were first purified from bovine brain in 1965 and named based on their solubility in saturated ammonium sulfate. 29 Currently, more than 20 proteins belong to the S100 calcium-binding protein family that contains 2 EF-hand calcium-binding motifs. These proteins can have calcium-dependent interactions with numerous target proteins that generate a variety of biological effects. 7,28 S100A4, a member of the S100 family, was initially named metastasin (mts1) due to the observation that it was specifically expressed in different metastatic cells. 7,10 Later, it was also cloned by several groups under various names, such as fibroblast-specific protein (FSP1), 18A2, pEL98, p9Ka, 42A, CAPL, and calvasculin. 13 In human breast tumors, S100A4 messenger RNA (mRNA) is present at a higher level in carcinomas relative to benign tumors. 31 The role of S100A4 in mammary epithelial cells has been investigated by several methods. Mammary tumors developing in transgenic mice expressing a high level of S100A4 in the mammary epithelial cells were more invasive and had greater metastasizing potentials compared to control tumors, and the degree of lung metastasis correlated with the levels of S100A4 expression. 1,9,21 Rat or human S100A4 gene transfection into a benign rat mammary tumor cell line conferred the property of metastasis in syngeneic transplant experiments. 8,26 In human breast cancer MCF-7 cells with S100A4 overexpression by transfection of the S100A4 gene, tumors derived from the cells injected into nude mice revealed local invasiveness and metastases, which were rarely observed in parental MCF-7 cells. 16 Thus, S100A4 has been considered to play important roles in cell migration, invasion, and metastasis formation in mammary carcinomas. Furthermore, S100A4 expression has been reported to be correlated with poor prognosis in several types of human cancers, including breast carcinoma as well as colorectal carcinoma, pancreatic carcinoma, ovarian carcinoma, bladder carcinoma, lung carcinoma, malignant melanoma, and so on. 2,19,38
Besides carcinoma cells, S100A4 protein is known to be expressed by various cell types present in the tumor microenvironment of human breast cancer (fibroblasts, macrophages, and activated lymphocytes). 3 It was also reported in canine mammary tumors that stromal cells expressed S100A4 protein. 23 However, expression and roles of S100A4 in carcinoma cells of canine mammary tumors have not been investigated. In this study, we identified the expression of S100A4 in some canine mammary carcinomas, particularly anaplastic subtypes. We established a novel cell line from a S100A4-positive highly aggressive carcinoma of the canine mammary gland and investigated the roles of S100A4 through knockdown experiments of S100A4 using an RNA interference (RNAi) technique with a WST-8 cell proliferation assay and Boyden chamber cell migration assay.
Materials and Methods
Cases
A total of 110 mammary specimens of 82 dogs were used from the archives of formalin-fixed, paraffin-embedded samples in the Department of Veterinary Pathology, Nippon Veterinary and Life Science University (Table 1). Specimens that were too small to analyze, were in a poor state of formalin fixation, or lacked the case information were excluded. Specimens of 13 lobular hyperplasias and 33 benign neoplasms from the archives of 2007–2010 and 57 malignant neoplasms from the archives of 2000–2010 were selected. Seven normal mammary tissues were obtained from mastectomy specimens with benign mammary tumors in 2009. Mammary tumors were diagnosed on the basis of the modified World Health Organization classification system proposed by Goldschmidt et al. 14 Immunohistochemistry was used in the diagnosis of some tumor types such as anaplastic carcinoma (positive for cytokeratin [CK] AE1/AE3 or CK8 and negative for desmin and Iba-1) and solid carcinoma of myoepithelial cell type (malignant myoepithelioma; positive for p63 or α-smooth muscle actin [SMA]). 46
S100A4 Expression in Normal, Hyperplastic, and Neoplastic Mammary Tissues of Dogs.
Moreover, carcinomas were graded according to the Elston and Ellis criteria modified by Peña et al. 35 Briefly, each carcinoma was evaluated by quantification of anaplasia, tubule formation, nuclear pleomorphism, and mitotic activity of carcinoma cells and was assigned grade I, II, or III. The evaluation of histological grade in each specimen was performed by 2 authors (H.Y. and A.O.), and the presence of vascular/lymphatic invasion and lymph node metastasis was evaluated by 3 authors (H.Y., M.M., and K.T.).
Case History of the Original Tumor Used in Cell Culture
A 12-year-old spayed female Pomeranian dog with immune-mediated hemolytic anemia presented to the Veterinary Medical Teaching Hospital of Nippon Veterinary and Life Science University. During immunosuppressive therapy for 3 months, a mass in the right fifth mammary gland had rapidly grown to approximately 6 × 4 cm in size with clinical impression of inflammatory carcinoma. The mammary tumor and inguinal lymph nodes involved by metastases were resected surgically at the owner’s request. Partial samples of the tumor were used for cell culture, and the others were fixed for histopathological examination.
Histology and Immunohistochemistry
The tumors of the canine mammary gland and the xenograft tumors were fixed in 10% neutral buffered formalin, routinely embedded in paraffin wax, sectioned (4 μm thickness), and stained with hematoxylin and eosin. Serial sections were immunostained by the labeled streptavidin-biotin method. Details of the primary antibodies, pretreatments, and dilutions for immunohistochemical analyses are described in Supplemental Table S1. In brief, deparaffinized sections were pretreated at 121°C for 10 minutes in citrate buffer (pH 6.0). Next, the sections were placed in 0.3% H2O2/methanol for 30 minutes and incubated in Block Ace (DS Pharma Biomedical Co., Osaka, Japan) for 30 minutes. After the reaction with the specific primary antibodies overnight at 4°C, the sections were incubated with biotinylated goat anti-mouse IgG or anti-rabbit IgG (Dako Japan, Tokyo, Japan), followed by peroxidase-conjugated streptavidin (Dako Japan). Finally, the reaction with each antigen was visualized by addition of diaminobenzidine tetrahydrochloride, and the sections were counterstained with hematoxylin.
For the S100A4 immunostaining, the number of S100A4-positive carcinoma cells was expressed as the percentage of total carcinoma cells (at least 1000 cells) counted in 10 high-power (×400) fields selected randomly from the whole tumor area in each case. The results were considered positive when over 5% of the carcinoma cells were immunoreactive.
To confirm the specificity of the S100A4 antibody for canine S100A4 protein, total protein was extracted from the canine mammary carcinoma cells, and Western blot analysis was performed as described previously. 32
Establishment of the Novel Cell Line
Tissue fragments from the original mammary tumor were washed with phosphate-buffered saline (PBS) with streptomycin and amphotericin B (Invitrogen, Carlsbad, CA). Then, the tumor fragments were homogenized in collagenase solution (Wako Pure Chemical Industries, Osaka, Japan) and incubated at 37°C for 45 minutes. Thereafter, the disaggregated tissue suspension was filtered through a Falcon 35-μm pore size cell strainer (Corning, New York, NY) to remove pieces of tissue that had not disintegrated. Thereafter, the filtered cell suspension was centrifuged at 1,500 rpm at 5°C for 5 minutes, and the pelleted cells were resuspended in RPMI1640 medium (Wako Pure Chemical Industries) supplemented with 10% heat-inactivated fetal bovine serum (FBS; Invitrogen), streptomycin, and amphotericin B and incubated at 37°C in a humidified 5% CO2 atmosphere. Once the cultured cells reached near-confluence, they were washed with PBS and treated with 0.25% Trypsin-1 mmol/L EDTA 4Na solution with phenol red (Wako Pure Chemical Industries) at 37°C for 5 minutes. Trypsinization was stopped using RPMI 1640 containing 10% FBS, and a few of the dispersed cells were reseeded to a new culture flask for subculture. The same procedure was repeated continuously for over 50 passages, and the established cell line was designated as NV-CML cells.
To assess their morphology and immune phenotypes, NV-CML cells were plated in 4-well chamber slides (1 × 104 cells/well; Merck Millipore, Burlington, MA) and incubated for 14 days at 37°C in a humidified atmosphere containing 5% CO2. The cells were fixed in 4% paraformaldehyde solution for 10 minutes at room temperature. The slide was stained with May-Grünwald and Giemsa stain. Other slides were prepared for fluorescent immunocytochemistry.
Fluorescent Immunocytochemistry
The cells on chamber slides were treated with 50 mM glycine for 5 minutes, washed, treated with 0.1% Triton X-100 (Sigma-Aldrich Japan, Tokyo, Japan), washed, treated with 5% goat serum for 30 minutes, and incubated overnight at 4°C with a polyclonal rabbit anti-S100A4 antibody and monoclonal mouse anti-CK AE1/AE3, CK8, vimentin, SMA, or p63 antibodies. After PBS washing, the slides were then incubated with Alexa 488–labeled anti-rabbit IgG or Alexa 594 anti-mouse IgG antibodies (1:300 dilution) for 30 minutes and mounted in Vectashield H-1200 with DAPI (Vector Laboratories, Burlingame, CA) after washing. Fluorescence images were obtained with a BX-9000 fluorescence microscope (KEYENCE, Osaka, Japan).
Growth Analysis
To examine the growth of NV-CML cells, 2 × l04 cells were seeded in 60-mm culture dishes and incubated in RPMI 1640 medium with 10% FBS at 37°C. Every 24 hours, replicative dishes were trypsinized and the dissociated cells treated with 0.4% trypan blue solution (Wako Pure Chemical Industries). Unstained (viable) cells were counted using a hemocytometer. The average number of cells was calculated and a growth curve for 5 days was established. The doubling time of the NV-CML cells was estimated from its exponential growth phase.
Tumorigenicity
For the xenograft transplantation, a suspension of 1 × 105 viable NV-CML cells was injected subcutaneously into the ventral flank of 14-week-old, male nonobese diabetic/Shi-scid IL2Rγnull (NOG) mice (n = 3), obtained from the Central Institute for Experimental Animals (Kawasaki, Japan). Every 7 days, injection sites were monitored, the size of the developing tumor masses was measured by a digital caliper, and their volume was calculated using the formula V = (W2 × L)/2. After 4 weeks, the mice were sacrificed and the subcutaneous tumors that developed were harvested for histological examination. All experiments were approved by the Animal Experiments Committee of the Nippon Veterinary and Life Science University.
Transfection of Small Interfering RNA Targeting Canine S100A4
We used small interfering RNA (siRNA) specifically targeting canine S100A4 mRNA (siS100A4; s530534) designed by Applied Biosystems (Foster City, CA). The sense siRNA sequence was 5′-GCU CAA CAG AUC AGA GUU Att-3′. A siRNA that does not bind to any canine RNA (siNeg) was also used as a negative control (Applied Biosystems). According to the manufacturer’s protocol, 6 pmol of either siS100A4 or siNeg diluted in 100 μL Opti-MEM medium (Thermo Fisher Scientific, Waltham, MA) was plated in each well of a 24-well plate, followed by the addition of 1 μL Lipofectamine RNAiMAX transfection reagent (Thermo Fisher Scientific). After incubation for 20 minutes, the NV-CML cells were seeded at a density of 5 × 104 cells/500 μL in each well. To confirm the effective transfection of siRNA into cells, total RNA was prepared at 48 hours after the transfection, and S100A4 mRNA levels were determined by real-time reverse transcription–polymerase chain reaction (RT-PCR).
TaqMan Real-Time RT-PCR
Total RNA was extracted from NV-CML cells using the NucleoSpin kit (Takara Bio, Kusatsu, Japan) following the manufacturer’s protocol. The RNA concentration was measured with a DS-11 NanoPad Spectrophotometer (DeNovix, Wilmington, DE). Next, complementary DNA (cDNA) synthesis was performed using a High Capacity cDNA Reverse Transcription Kit (Applied Biosystems). Real-time PCR for S100A4 and 18S ribosomal RNA (rRNA) was performed using the 7300 real-time PCR system (Applied Biosystems) with TaqMan Gene Expression Assays (Canine S100A4, cf02622009_m1; 18S, Hs99999901_s1) and TaqMan Fast Advanced Master Mix (Applied Biosystems). Cycling conditions were as follows: 2 minutes at 50°C, 20 seconds at 95°C, and 40 cycles for 3 seconds at 95°C followed by 30 seconds at 60°C. Canine lymph node tissue was used as positive control and ultrapure water was used as negative control. Real-time PCR results were expressed as target mRNA/18S rRNA, as an internal standard concentration ratio. Relative gene expression was calculated by using the ▵▵Ct method. Gene expression levels were measured in triplicate.
WST-8 Cell Proliferation Assay
Cells were cultured in RPMI 1640 medium with 10% FBS at a density of 5 × 103 cells/100 mL/well in 96-well plates followed by incubation for 0 or 96 hours. The cells were then incubated with WST-8 cell counting reagent (Wako Pure Chemical Industries) for 2 hours. Optical density was measured using a plate reader (Bio-Rad Laboratories, Hercules, CA) at 450 to 620 nm.
Boyden Chamber Assay (Migration Assay)
Cell culture inserts (8-μm pore size and 6 mm in diameter; BD Biosciences, Franklin Lakes, NJ) were used following the manufacturer’s instructions. Briefly, 1 × 105 NV-CML cells were suspended in 500 μL serum-free medium and placed onto the upper component of the inserts. The lower compartment was filled with 750 μL of a medium containing 10% FBS, and the cells were incubated at 37°C in a humidified 5% CO2 atmosphere. After 24 hours, the cells on the upper surface of the filter were carefully removed with a cotton swab. The cells that had migrated through the membrane to the lower surface of the filter were fixed and stained with a Diff-Quick staining kit (Sysmex Corp, Kobe, Japan). The number of cells on each membrane was counted in 10 high-power (×200) fields spaced evenly over the whole area of the round membrane, and the mean value was calculated per field.
Statistical Analysis
Statistical differences in proliferation and migration assays were determined using the Student t test. Fisher’s exact probability test was used to determine statistical differences in vascular/lymphatic invasion and metastasis between S100A4-positive and S100A4-negative carcinomas. The correlation between S100A4 expression and histological grade of carcinomas was tested using Spearman’s rank correlation coefficient test. Probabilities of less than 5% (P < .05) were considered significant.
Results
S100A4 Expression in Canine Mammary Tumors
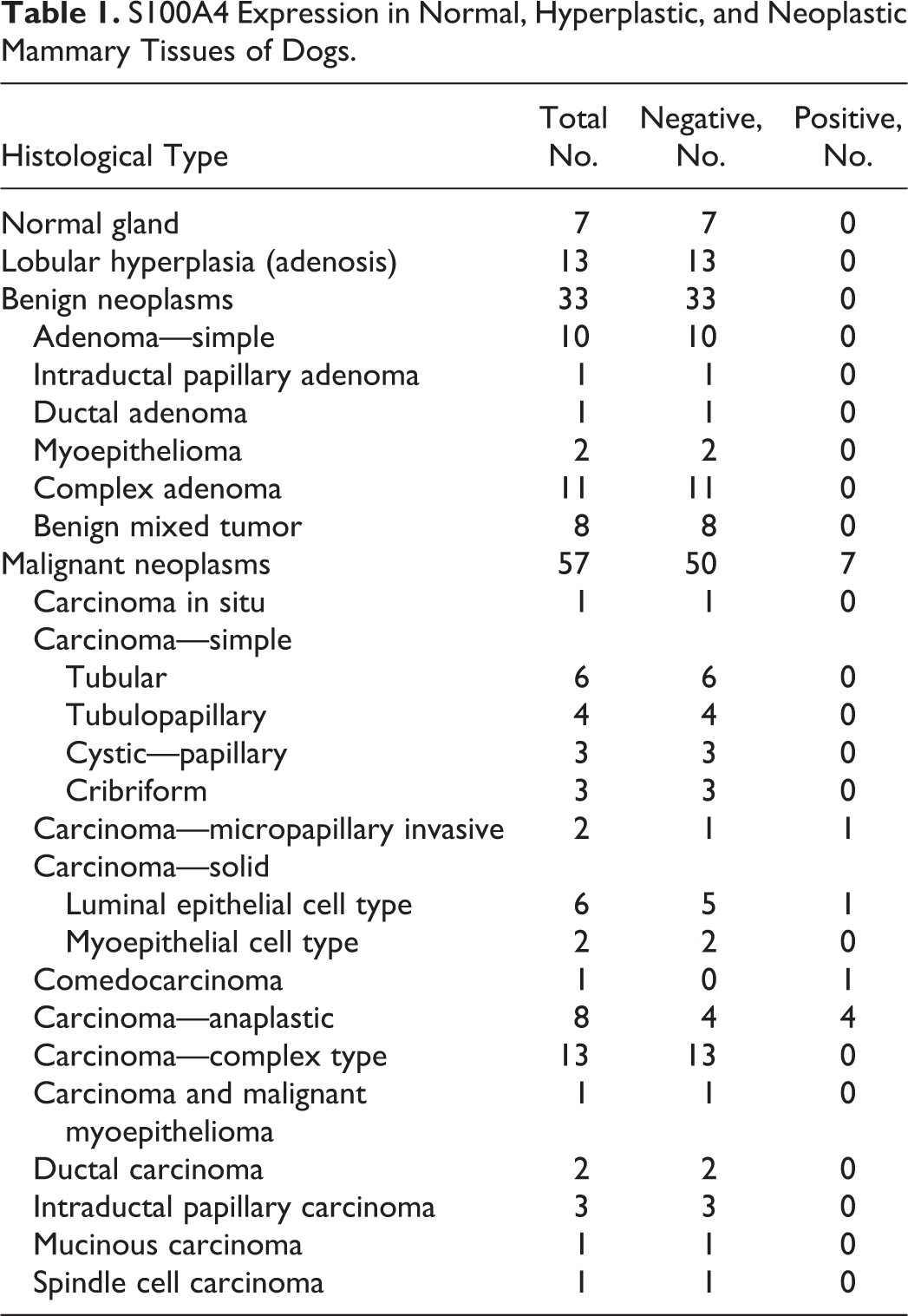
Initially, we examined immunohistochemically the expression of S100A4 in normal mammary glands and proliferative lesions in dogs. The specificity of the S100A4 antibody for canine S100A4 protein was confirmed by Western blot analysis (Suppl. Fig. S1). The immunohistochemical results are summarized in Table 1. Epithelial cells in the 7 normal and 13 hyperplastic mammary tissues lacked S100A4 expression (Fig. 1), although some stromal fibroblasts and inflammatory cells expressed S100A4. None of the 33 benign tumors had expression of S100A4 by epithelial tumor cells (Fig. 2); however, chondroid elements of benign mixed tumors were weakly stained for S100A4. In contrast, nucleic and cytoplasmic expression of S100A4 protein was observed in epithelial tumor cells in 7 of the 57 malignant tumors, including 1 of 6 solid carcinomas (percentage of positive cells, 18%), 1 of 2 invasive micropapillary carcinomas (percentage of positive cells, 71%; Fig. 3), 1 of 1 comedocarcinoma (percentage of positive cells, 8%), and 4 of 8 anaplastic carcinomas (percentage of positive cells, 46%, 60%, 81%, and 94%; Fig. 4). Stromal fibroblasts and inflammatory cells in carcinomas were often positive for S100A4.
Mammary gland, dog. Immunohistochemistry for S100A4.
Correlations between S100A4 expression and pathological parameters in mammary carcinomas are summarized in Table 2. Histologically, of the 57 carcinomas, 20 had vascular/lymphatic invasion and 3 (15%) of them expressed S100A4, while 4 of the 37 (11%) carcinomas without vascular/lymphatic invasion were S100A4 positive. S100A4 was expressed in 1 of 13 (8%) of the primary carcinomas that showed lymph node metastasis and in 6 of 44 (14%) of the primary carcinomas that did not have lymph node metastasis. No significant relationships were found between S100A4 expression and vascular/lymphatic invasion or metastasis. The histological grades of carcinomas were as follows: grade I, 19 cases; grade II, 15 cases; and grade III, 23 cases. One grade II carcinoma and 6 grade III carcinomas were S100A4 positive, and there was a significant positive correlation between S100A4 expression and histological grade (P < .01).
Associations Between S100A4 Expression and Pathological Parameters in Mammary Carcinomas.
Histology of the Original Tumor Used in Cell Culture
To further study the role of S100A4 in canine mammary carcinoma, a cell line was derived from an S100A4-expressing mammary carcinoma.
The infiltrating mammary carcinoma was mainly composed of polygonal carcinoma cells arranged in solid nests (Suppl. Fig. S2). Around the nests, pleomorphic carcinoma cells resembling undifferentiated cells in anaplastic carcinoma were scattered in the stroma (Suppl. Fig. S3). Tumor emboli were observed in the lumen of the multiple lymph vessels in dermis and subcutis (Suppl. Fig. S4). Inguinal lymph nodes contained carcinoma cells. The mammary tumor was diagnosed as solid carcinoma with focal anaplastic features.
Immunohistochemically, S100A4 was intensely expressed in the cells with anaplastic features, whereas S100A4 expression in the cells arranged in the solid nests was weak and sparse (Fig. 5a). Both the solid growth cells and the anaplastic cells were diffusely positive for vimentin (Fig. 5b). Both cell types were positive for CK AE1/AE3 (Fig. 5c) and CK8; however, reduction of CK AE1/AE3 and CK8 expressions was often observed in the anaplastic cells. No expression of SMA (Fig. 5d) or p63 was observed in either cell types.
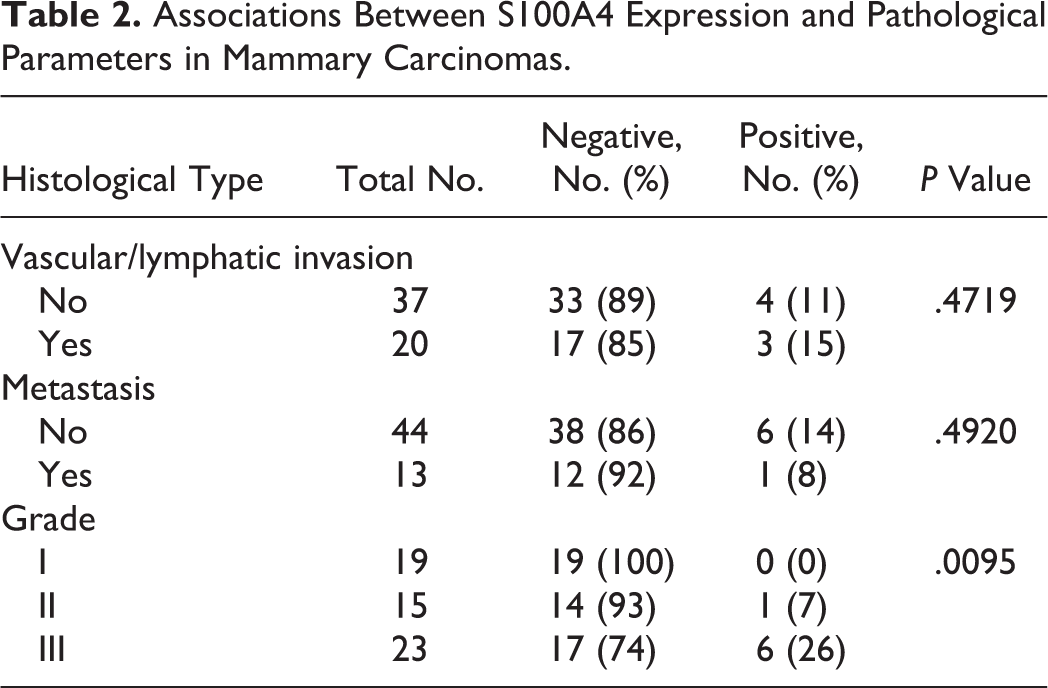
Characterization of the Novel Cell Line
The novel cell line NV-CML exhibited the following characteristics. NV-CML cells exhibited a polygonal to short spindle shape with anisocytosis (12.5–130.0 μm) and had round to ovoid nuclei with anisokaryosis (7.5–50.0 μm) (Fig. 6). Multinucleated cells were present (3.2%). The doubling time was 30 hours at passage 50 (Suppl. Fig. S5). Fluorescent immunocytochemistry (Fig. 7) showed that NV-CML cells were positive to S100A4 and vimentin, weakly positive to CK AE1/AE3 and CK8, and negative to SMA and p63.
Novel canine mammary carcinoma cell line, NV-CML cells.
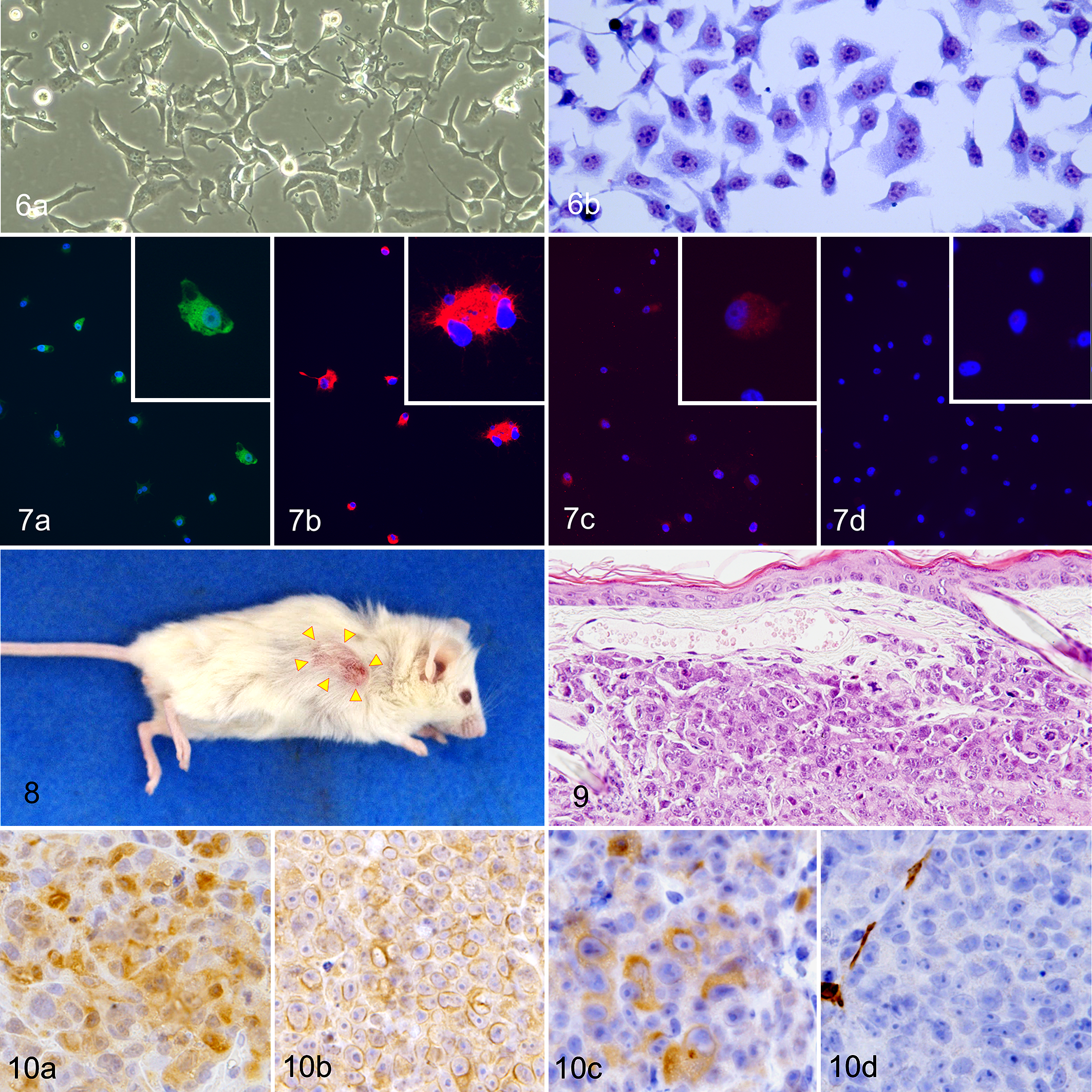
To confirm the tumorigenicity of NV-CML cells, we injected these cells into the subcutis of NOG mice. Tumor masses developed at the injection site in 3 of 3 NOG mice. The masses first became palpable at 1 week and reached a mean size of 494 mm3 4 weeks later (Suppl. Fig. S6). They showed an ellipsoidal shape, and the covering skin showed thinning hair and became red (Fig. 8). The mice showed no other clinical signs throughout the whole experimental period. Microscopically, the tumors were located in the dermis to the subcutis. Tumor cells were pleomorphic and individualized or arranged in sheets separated by fine stroma (Fig. 9). Focal necrosis in the center of tumor masses was observed. Tumor cells had a moderate amount of pale eosinophilic cytoplasm and had round to ovoid, hypochromatic nuclei containing large prominent nucleoli. Anisocytosis and anisokaryosis were severe, and mitotic figures were found at the rate of 91 per 10 fields (at ×400 magnification). Immunohistochemically, tumor cells were positive for S100A4, vimentin, CK AE1/AE3, and CK8 and were negative for p63 and SMA (Fig. 10).
Effects of S100A4 Knockdown in the Novel Cell Line
Finally, we performed knockdown of S100A4 in NV-CML cells using the siRNA technique and evaluated whether S100A4 knockdown influenced cell proliferation and migration. S100A4 expression level in NV-CML cells transfected with siS100A4 was decreased to approximately 20% of that with siNeg (Fig. 11). First, cell proliferation after a 96-hour culture was evaluated by a WST-8 assay. The absorbance measured after treatment with WST-8 reagent was significantly lower in siS100A4-transfected cells than in siNeg-transfected cells (Fig. 12). Next, a cell migration assay was conducted using the Boyden chamber technique. The siS100A4-transfected cells migrated more slowly than the siNeg-transfected cells, and the number of migrating cells in the siS100A4-transfected cells was significantly fewer by comparison with that in the siNeg-transfected cells (Fig. 13). Therefore, S100A4 at least partially seems to mediate the proliferation and migration of canine mammary carcinoma cells.
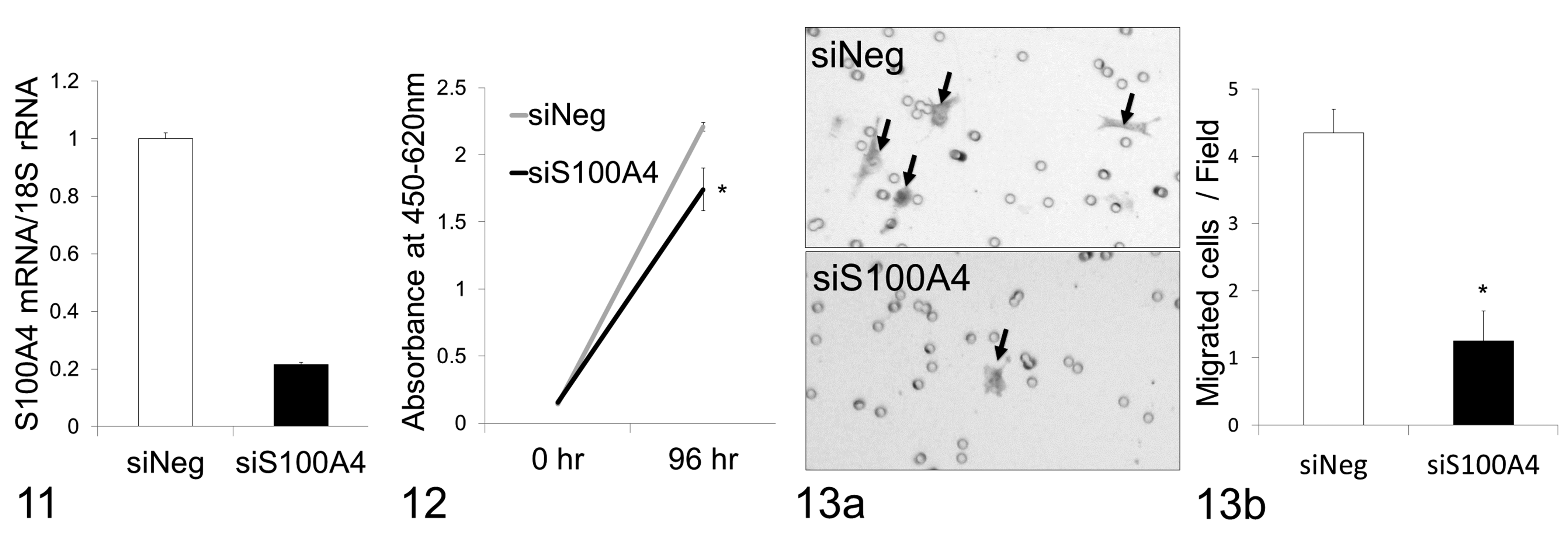
S100A4 messenger RNA (mRNA) levels measured by reverse transcription–quantitative polymerase chain reaction. S100A4 mRNA level in novel canine mammary carcinoma cell line (NV-CML) cells transfected with small interfering RNA (siRNA) targeting S100A4 (siS100A4) is reduced to one-fifth of that of the negative control siRNA (siNeg).
Discussion
We examined S100A4 expression in normal, hyperplastic, and neoplastic canine mammary tissues, as an association between S100A4 expression and human breast cancer progression has been indicated in several studies. 34,36,38 In our study, all of the examined normal glands and benign lesions were S100A4 negative in epithelial cells, but 7 of 57 malignant tumors were positive for S100A4, suggesting that S100A4 protein may be involved in malignant progression of some canine mammary tumors. This is consistent with the findings of other studies, which showed that immunolabeling for S100A4 is associated with poorer survival in human breast cancer. 36,38 The S100A4-positive canine mammary carcinomas were categorized into subtypes known to have a worse prognosis (anaplastic, simple solid, comedo, and invasive micropapillary) compared to other subtypes. 12,37 It is particularly notable that half of the 8 anaplastic carcinoma cases expressed S100A4 protein. Anaplastic carcinoma is a subtype having the most unfavorable outcome and showing a distinct morphology. 27,37 Furthermore, in a case of simple solid carcinoma with focal anaplastic features, immunostaining for S100A4 was positive in the anaplastic cells with higher intensity as compared to the solid growth cells. These findings suggest that S100A4 could be related to the development of anaplastic tumor cells. In addition, we examined the relationship between S100A4 expression and pathological parameters in carcinomas. Although there were no significant differences in S100A4 expression between carcinomas with and without vascular/lymphatic invasion or metastasis, S100A4 expression correlated significantly with high histological grade. Histological grade is known as a good independent prognostic parameter. 35
Furthermore, we attempted to study the S100A4 function in canine mammary carcinoma cells in vitro. First, we established a novel cell line NV-CML from the tissue of the case of simple solid carcinoma with focal anaplastic features. The tumor exhibited the clinical impression of inflammatory mammary cancer and histologically showed the dermal tumor embolization of superficial lymphatics that was the hallmark of inflammatory mammary cancer. Recently, canine inflammatory mammary cancer has drawn attention as a natural model for human inflammatory breast cancer. 4 The novel cell line is useful for in vitro research of aggressive mammary carcinoma. As we confirmed, the established cell line maintained the immunophenotypes of the anaplastic cells of the original mammary tumor, such as S100A4 positivity. The cell line was tumorigenic when the cells were injected into the immunodeficient NOG mice. 20 The tumors that developed shared similar histological and immunohistochemical features, including S100A4 expression, with the anaplastic cells of the original tumor. Thus, we considered that the novel cell line would be useful for investigating S100A4 function in canine mammary cells.
Subsequently, we examined the canine mammary carcinoma cells when their S100A4 expression was knocked down by siRNA. We found that the knockdown of S100A4 expression led to decreased cell proliferation in the WST-8 assay. Similar observations have been also reported in several human tumor cells, including bladder, gastric, laryngeal, and thyroid carcinomas and osteosarcoma. 5,18,22,24,42 S100A4 has been considered to promote cell proliferation by its interaction with various ligands, including the epidermal growth factor and the tumor suppressor protein, p53. 6,28
In the Boyden chamber assay using mouse mammary carcinoma cell lines, their migration abilities correlated with the levels of S100A4 expression. 21 In an experiment of siRNA-mediated knockdown of S100A4 expression in a human breast carcinoma cell line, cell migration in the Boyden chamber assay was also decreased in a S100A4 expression level-dependent manner. 41 We also demonstrated in the Boyden chamber assay with the siRNA technique that S100A4 was involved in migration of canine mammary carcinoma cells. In human breast carcinoma cells, S100A4 is considered the effector of actin cytoskeletal changes and affects cell morphology and migration. 15 S100A4 also enhances matrix metalloproteinase 13 (MMP13) expression, leading to increased migration of human breast carcinoma cells. 41 S100A4 has been also known to play a key role in epithelial-mesenchymal transition (EMT), which enhances cell motility. 40 Several studies have reported that S100A4 expression is inversely associated with the expression of the epithelial marker, E-cadherin, in several carcinoma cell lines and carcinoma tissues. 2,25,30,33,43 Canine mammary anaplastic carcinoma is characterized by high invasiveness, pleomorphic morphology, and expression of mesenchymal markers. 27 Since S100A4 has been thought to play crucial functions in cell migration, morphology, and EMT as described above, these functions may contribute to the formation of anaplastic carcinoma cells.
Stromal fibroblasts in canine mammary carcinomas expressed S100A4 more frequently compared to those in benign lesions. Murine models have indicated that S100A4-positive fibroblasts promote the metastatic capability of tumor cells. 17,39 It has been suggested that S100A4-positive fibroblasts within canine mammary tumor stroma are the primary source of extracellular matrix protein tenascin-C, which has been implicated in the progression of canine mammary tumors. 23,44,45
In human cancer patients, S100A4 has been shown to serve as a promising candidate biomarker for the prediction of metastasis, as well as a therapeutic target for cancer. 11,28 Various therapeutic strategies targeting S100A4 in human cancer have been evaluated in preclinical experiments, including siRNA-mediated silencing of S100A4, S100A4-specific antibodies, and small molecule–based interference of S100A4-protein interactions. 11 siRNA silencing has been suggested to have tremendous potential as a therapeutic strategy for cancer by downregulating oncogenic genes. 47 Treatment of siRNA targeting canine S100A4 mRNA resulted in a decrease in proliferation and migration potential of canine mammary carcinoma cells in vitro, indicating that S100A4 might become a potential molecular target for gene therapy of canine mammary carcinomas. However, its mechanism and effectiveness remain to be fully verified. Therefore, future research should focus on clarifying the underlying gene pathways responsible for S100A4-mediated progression of canine mammary carcinomas and verifying the effects of the inoculation of S100A4-targeting siRNA into immunodeficient mice bearing canine mammary carcinoma xenografts.
Supplemental Material
Supplemental Material, DS1_VET_10.1177_0300985818823772l - Expression and Roles of S100A4 in Anaplastic Cells of Canine Mammary Carcinomas
Supplemental Material, DS1_VET_10.1177_0300985818823772l for Expression and Roles of S100A4 in Anaplastic Cells of Canine Mammary Carcinomas by Hisashi Yoshimura, Aya Otsuka, Masaki Michishita, Masami Yamamoto, Minori Ashizawa, Manami Zushi, Maiko Moriya, Daigo Azakami, Kazuhiko Ochiai, Yoko Matsuda, Toshiyuki Ishiwata, Shinji Kamiya and Kimimasa Takahashi in Veterinary Pathology
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
