Abstract
Wobbly hedgehog syndrome (WHS) is a leading cause of neurologic disease in African pygmy hedgehogs (APHs; Atelerix albiventris). This study describes the signalment, clinical signs, gross, microscopic, and ultrastructural lesions of WHS in a cohort of 12 pet APHs. Microscopically, lesions consisted of status spongiosus of the white matter, typically bilateral and symmetrical, with myelin degeneration and loss that was accompanied by neuronal/axonal degeneration plus reactive microgliosis and mild, focal astrocytosis and astrogliosis. Lesions were most severe in the cerebellum and medulla oblongata, as well as cervical and thoracic spinal cord. Less affected areas were the corona radiata, corpus callosum, corpus striatum, internal capsule, and the mesencephalon. Ultrastructurally, the lesions consisted of splitting of the myelin sheath at the intraperiod line with subsequent focal expansion, resulting in status spongiosus, disruption, dilatation, rhexis, and phagocytosis. Based on these results, WHS is best described as a “spongy myelinopathy” with widespread central nervous system involvement.
Keywords
Wobbly hedgehog syndrome (WHS) is an insidious, progressive, incurable neurologic disease that affects pet African pygmy hedgehogs (Atelerix albiventris). 6 A similar if not identical disease has been described in the European hedgehog (Erinaceus europaeus). 17 The disease is characterized by ataxia that can progress to seizures and tetraplegia. The onset of clinical signs typically occurs at less than 2 years of age. Gross lesions are not evident, and the microscopic hallmark is a spongy change (status spongiosus) involving the white matter of the central nervous system (CNS). 6,17,4,15,14,13,9,21 This study reports the lesions of WHS in a cohort of African pygmy hedgehogs with special emphasis on the microscopic and ultrastructural lesions and their neuroanatomical distribution.
Material and Methods
Twelve African pygmy hedgehogs (APHs) were submitted for necropsy to the Department of Veterinary Pathobiology at Texas A&M University between 2010 and 2016. Cases from 2010 to 2013 and 2014 to 2016 were retrospectively and prospectively evaluated, respectively. Age, sex, and clinical histories are recorded in Supplemental Table S1. Necropsies were performed and selected tissues (eye, skin, cerebrum, cerebellum, brainstem, medulla oblongata, spinal cord [10/12 cases], tongue, salivary gland, trachea, lung, heart, diaphragm, skeletal muscle, lymph node, stomach, intestine, liver, gallbladder, pancreas, kidney, adrenal gland, gonads, femoral bone marrow, and urinary bladder) were collected and fixed in 10% neutral buffered formalin and processed routinely for microscopic examination. Holmes silver nitrate impregnation and Luxol fast blue stain were conducted on selected brain sections. The inclusion criteria consisted of (a) APHs with clinical history compatible with WHS, as described above, and (b) postmortem interval <12 hours. All brains were sectioned coronally at regular intervals from rostral to caudal. For analysis of neuroanatomical lesion distribution, a template including 25 coronal brain sections was used (Supplemental Fig S1; modified from Stephan et al 20 ).
For ultrastructural examination, fresh brainstem from case No. 3 was collected and immersed in 2% glutaraldehyde, stained with 1% uranyl acetate in water, dehydrated, and infiltrated and polymerized in epoxy (Epon/Araldite with 1.5% DMP 30). For immunohistochemical analysis, a polyclonal anti-Iba (ionized calcium-binding adapter molecule) 1 antibody (Wako, Richmond, VA, USA; 1:500) was used for microglia and a polyclonal anti–glial fibrillary acidic protein (GFAP) antibody (Eurodiagnostics, Apeldoorn, Holland; 1:150) was employed for astrocytes. Internal positive controls, as well as brain sections from the same species and normal dog, served as positive and negative controls.
Results
Ages of examined animals ranged from 1 to 5 years (average: 3 years). Of the 12 cases, 7 (58%) were male and 4 (33%) were female. Most animals presented with a long and progressive history of motor incoordination and an unsteady gait (Suppl. Table S1). On physical examination, all animals had varying degrees of ataxia, paresis, paraplegia, and tetraplegia. Tremors, head tilt, and circling were also common. Proprioceptive deficits were readily evident; 1 animal (case No. 6) was obtunded and 1 animal (case No. 12) presented comatose and died shortly after. One animal (case No. 3) had seizures. Three animals had unilateral exophthalmos (case Nos. 1, 6, and 7), and 2 had keratitis (case Nos. 6 and 7). Case 6 had right-sided horizontal nystagmus.
A variety of concomitant pathologic findings were identified, with neoplasia predominating (Supplemental Table S1). Four animals had grossly evident hepatic lipidosis (case Nos. 2, 3, 5, and 6). Case 10 had bilateral polycystic kidneys. Those animals with a history of longstanding progressive ataxia had axial and appendicular skeletal muscle atrophy.
There were no gross lesions detected in the brain or spinal cord. Microscopic lesions in the CNS were of variable severity and chronicity (Table 1). Lesions were bilateral and symmetrical in all cases, except for some sections of case Nos. 1, 3, and 5 (Figs. 1, 2). All animals had status spongiosus of the white matter that was characterized by well-delineated, round to oval, single to coalescing, 15- to 75-μm, clear spaces (Figs. 3–5). In acute stages, consisting of minimally affected foci of status spongiosus, axonal swelling (spheroids) and fragmentation were a common finding (Figs. 3, 4). These changes coexisted with neuronal changes that included central chromatolysis (case Nos. 2 and 12) and shrinkage of cell bodies with nuclear pyknosis (Fig. 5). Rarely, neurons had hypereosinophilic cytoplasm with karyorrhectic and karyolytic nuclei. Axonal loss predominated in chronic stages, along with focal myelin degeneration and loss (Fig. 6), and there was a mild reduction of neuronal cell bodies in degenerative foci of adjacent gray matter. The same changes occurred in all of the deep cerebellar nuclei (medialis, interpositus, and lateralis) and in the ventral horn of the spinal cord. In case Nos. 1, 5, and 9, a spongy change also occurred in the cerebellar cortex, predominately the inner granular cell layer with lesser involvement of the inner molecular layer. Case No. 5 had marked, focal, unilateral neuronal degeneration and necrosis in the pyramidal cell layer of cornu ammonis CA-2 and CA-3 segments.
Central Nervous System Histopathologic Findings Associated With Wobbly Hedgehog Syndrome in 12 Pet African Pygmy Hedgehogs.
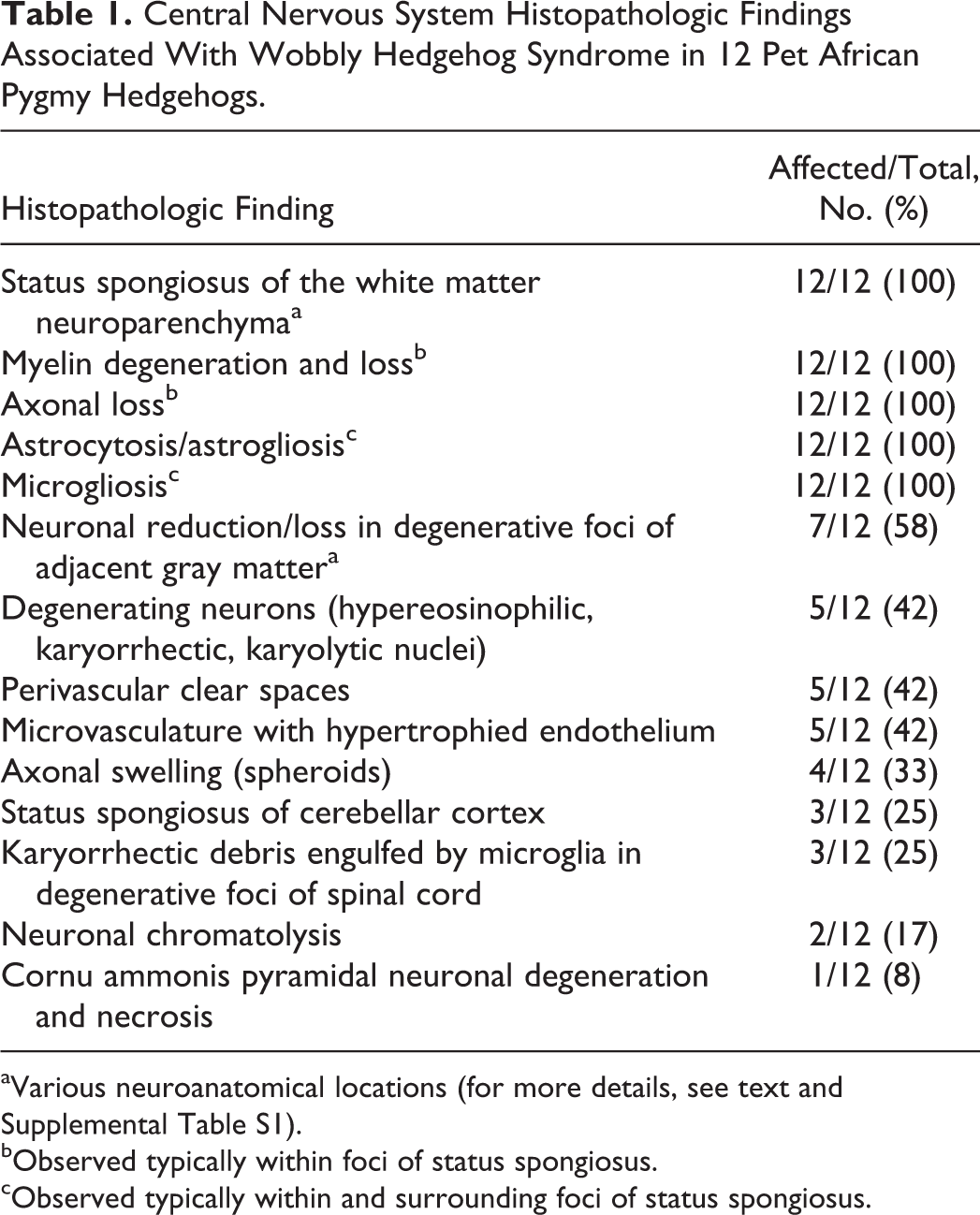
aVarious neuroanatomical locations (for more details, see text and Supplemental Table S1).
bObserved typically within foci of status spongiosus.
cObserved typically within and surrounding foci of status spongiosus.
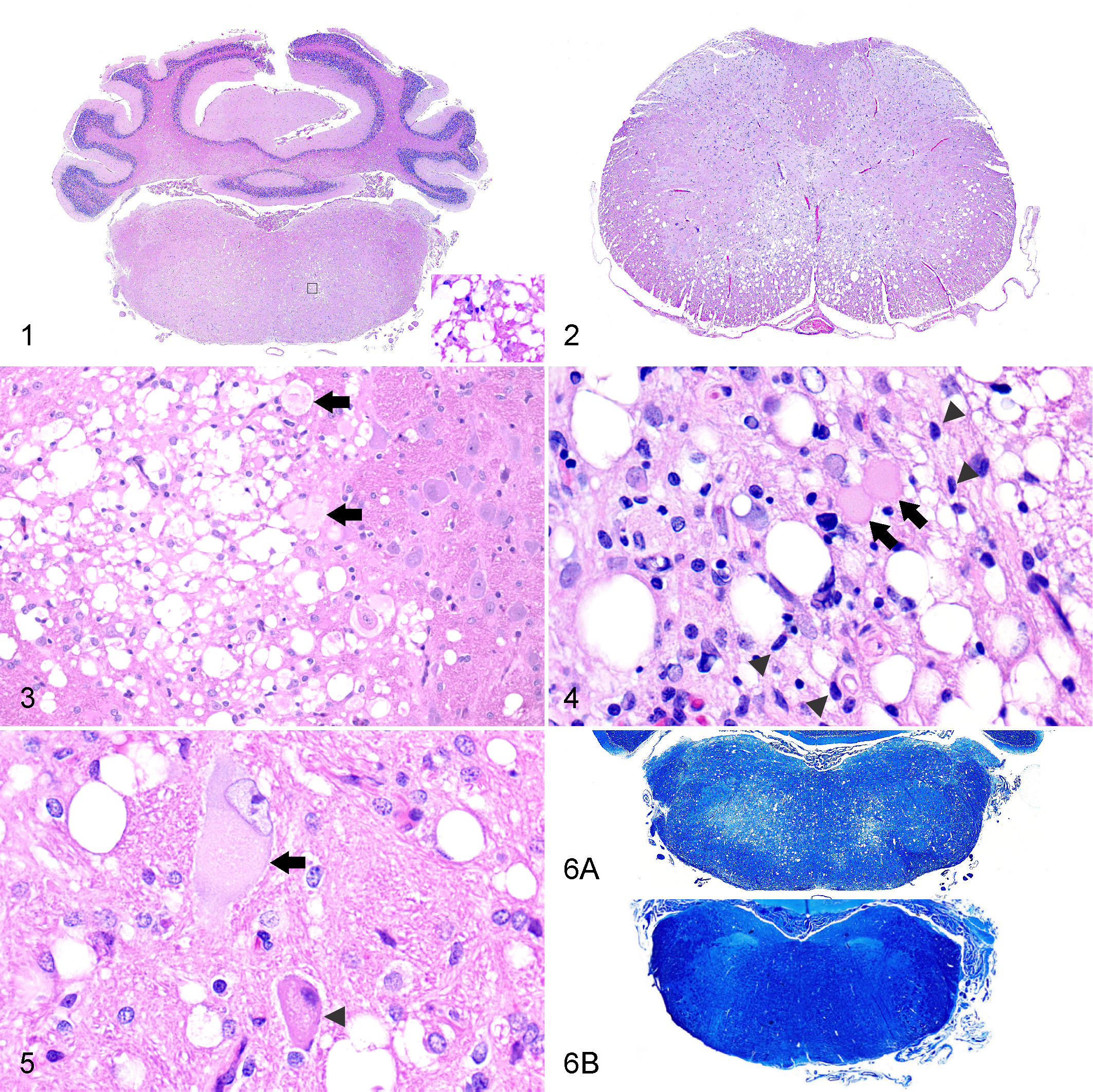
Wobbly hedgehog syndrome, African pygmy hedgehog, case No. 10.
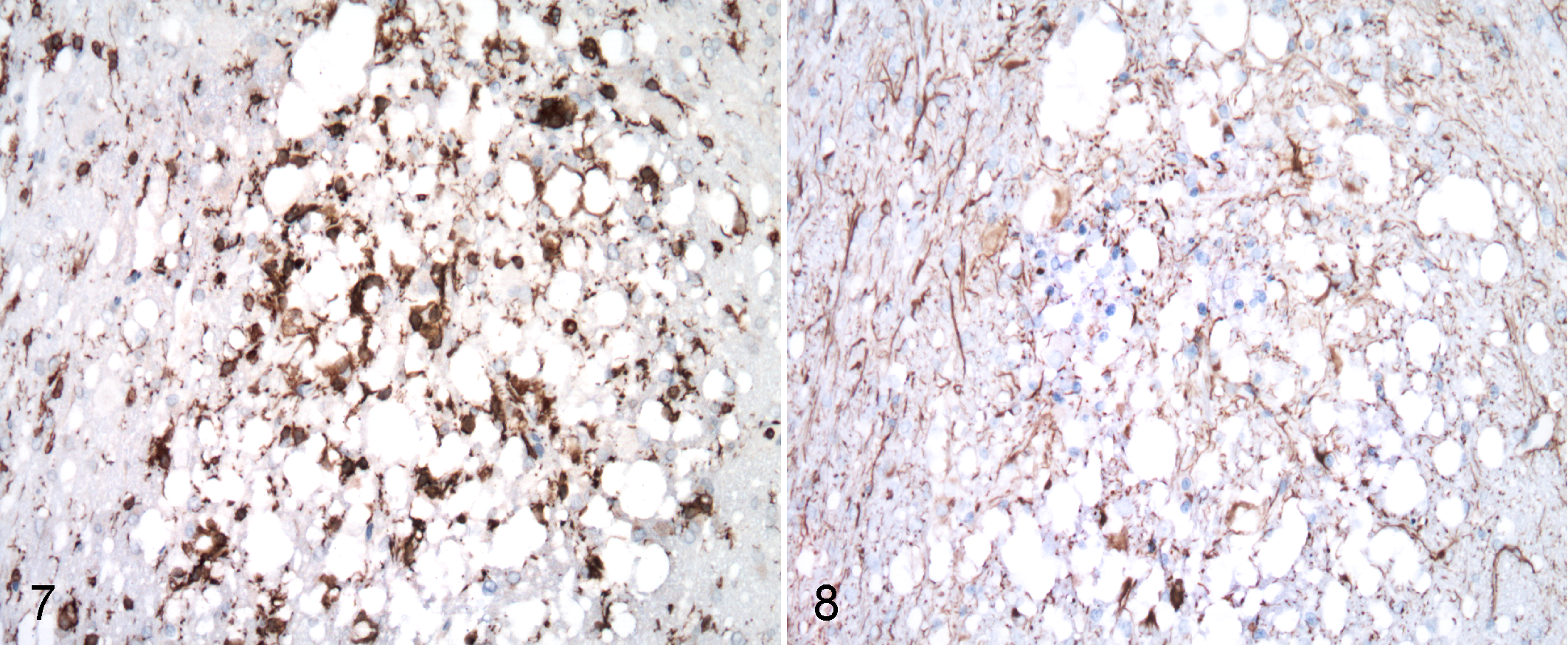
Affected white matter and occasional gray matter had small multifocal areas of microgliosis characterized by increased numbers of hypertrophied microglia, as illustrated by Iba1 immunolabeling (Fig. 7). Small clusters of astrocytes in affected areas often had enlarged nuclei with rarely visible cytoplasm with H&E staining. While relatively few hypertrophic astrocytic processes were present in central areas of severe and extensive status spongiosus, they were increased peripherally, as highlighted by GFAP immunostaining (Fig. 8). In small foci, considered to be early lesions, surrounding enlarged areas of degeneration, astrocytic cell bodies and processes had more intense GFAP immunostaining. Varying degrees of astrogliosis and microgliosis, from minimal to moderate, were associated with spongiosus in all animals. Other less prominent and uncommon microscopic features within degenerative foci included scattered pyknotic debris, microvasculature with hypertrophied endothelium, and perivascular clear spaces.
Wobbly hedgehog syndrome, medulla oblongata, African pygmy hedgehog, case No. 5. White matter adjacent to interface between vestibular and trigeminal nerve nuclei.
Lesions in the spinal cord in case Nos. 1, 5, and 8 included karyorhectic debris engulfed by microglia associated with foci of degeneration. Case No. 10 had mild spongy change involving some ventral cervical spinal rootlets. Case Nos. 11 and 12 had scattered foci of mineralization in the neuroparenchyma and capillary walls. Case No. 3 had minimal scattered lymphoplasmacytic perivascular cuffs in the leptomeninges of the ventral brainstem.
There was a clear neuroanatomical distribution of the lesions described above (Fig. 9). The lesions, which were largely bilateral and symmetrical, were most severe and extensive in the following decreasing order of severity: cerebellum, medulla oblongata, cervical and thoracic spinal cord, cerebrum, mesencephalon, and spinal nerves and sensory ganglia. In the cerebellum, the hemispheric white matter was most severely affected, with variable involvement of the cerebellar nuclei. The molecular and granular layers of the cerebellar cortex were affected only in the most severe cases. In the medulla oblongata, most commonly affected areas were the reticular formation, inferior cerebellar peduncle, and nucleus of the spinal tract of the trigeminal nerve. In the spinal cord, the ventral and lateral funiculi were most commonly affected, with minimal involvement of the dorsal corticospinal tract. No lesions were observed in the lumbar spinal cord. Dorsal and ventral spinal nerves and sensory ganglia were spared in all but case No. 10, where rare ventral spinal rootlets had identical spongy change. In the cerebrum, the structures most frequently affected were the periventricular and neocortical white matter, mostly involving the corona radiata, corpus callosum, corpus striatum, and internal capsule. In the diencephalon, the ventrolateral and dorsal thalamus, zona incerta, and cerebral peduncles were commonly affected. In the mesencephalon, the reticular formation was consistently involved. Additional significant concurrent diagnoses are recorded in Supplemental Table S1.
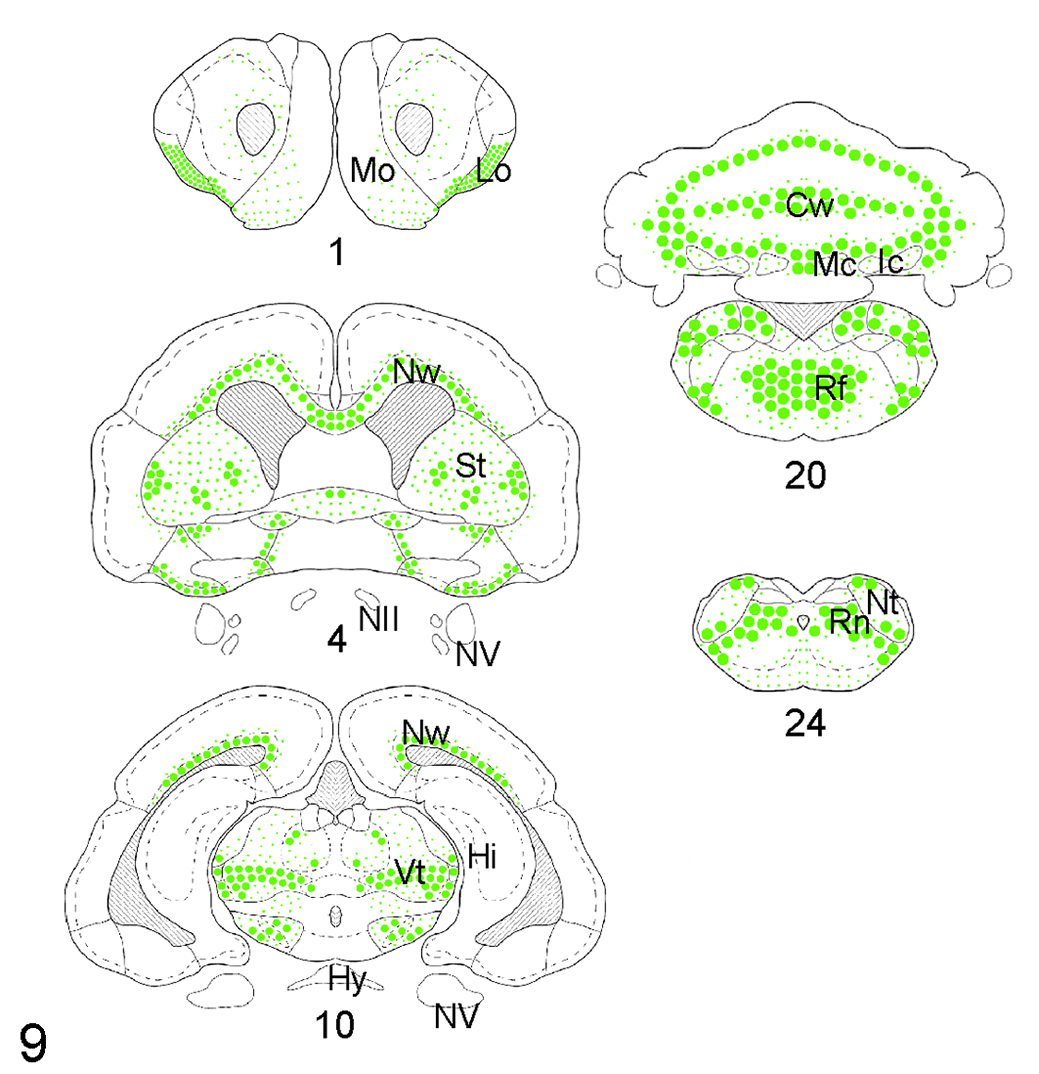
Neuroanatomical distribution and severity of status spongiosus in African pygmy hedgehogs with wobbly hedgehog syndrome. The size and density of dots indicates the severity and extent of the lesions. Abbreviations: Cw, cerebellar white matter; Hi, hippocampus; Hy, hypophysis; Ic, interpositus cerebellar nucleus; Lo; lateral olfactory tract; Mc, medial cerebellar nucleus; Mo, main olfactory bulb; NII, optic nerve; Nt, nucleus of the spinal tract of the trigeminal nerve; Nw, neocortical white matter; NV, trigeminal nerve; Rf, reticular formation; Rn, reticular nuclei of medulla oblongata; St, striatum; Vt, ventroposterior thalamus. Numbers below the diagrams indicate craniocaudal sectioning level as recorded in Supplemental Figure S1.
Ultrastructurally, the myelin sheaths were variably split, forming areas of status spongiosus, and some were separated from axons that were segmentally degenerated or totally lost (Fig. 10). Degenerated myelin sheaths often had irregular, disfigured contours and thickness with multifocal splitting initiated at the intraperiod line (Fig. 11). Affected myelin sheaths also contained fine to coarse granular electron-dense aggregates and had focal out-pouchings forming clear spaces of status spongiosus that displaced axons that were variably enlarged and had degeneration or loss of axonal components. Rarely, there was a separation of the inner myelin sheath from the axolemma, forming a space that contained occasional degenerating cellular debris of unknown origin. Focally, macrophages had engulfed fragments of myelin sheaths (Fig. 12). Occasional astrocytes had a mild to moderate increase of ribosomes plus clumping of nuclear chromatin. No other ultrastructural changes were noted in astrocytes or oligodendrocytes. Overall, the ultrastructural and microscopic lesions were compatible with this disease being classified as a spongy myelinopathy.
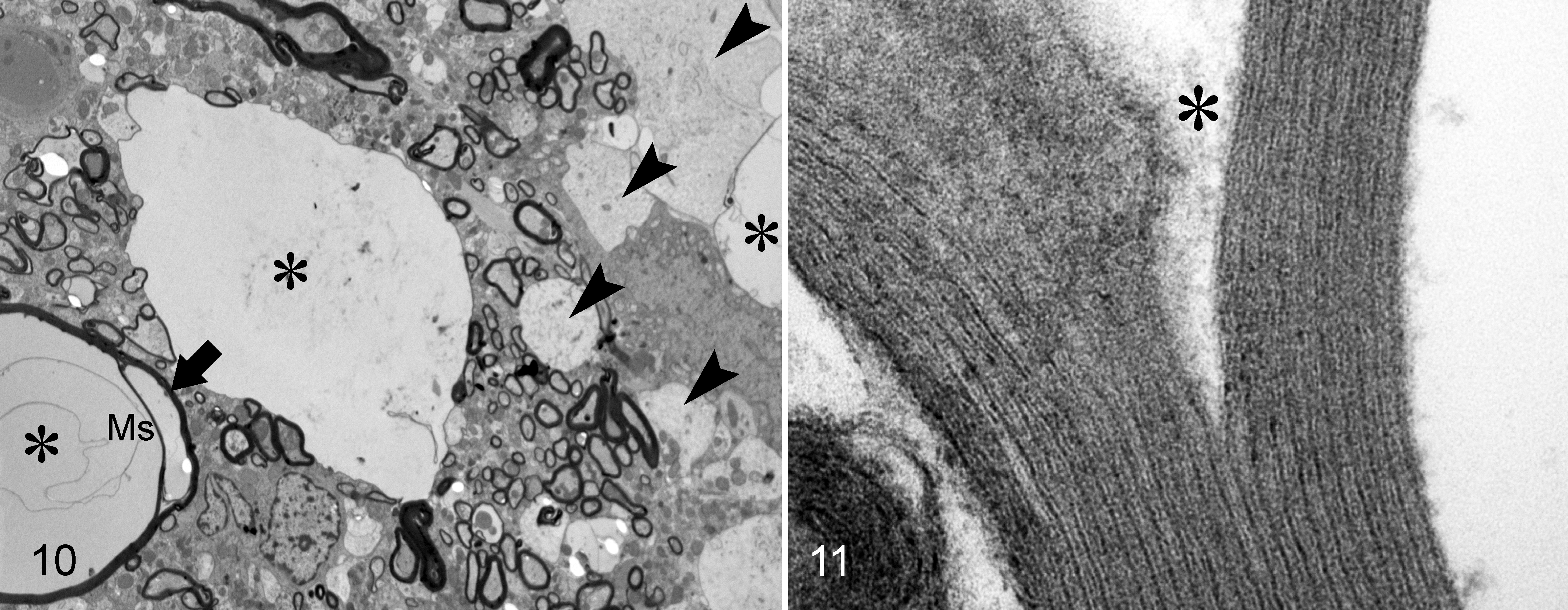
Spongy myelinopathy; brainstem; African pygmy hedgehog, case No. 3; transmission electron microscopy.
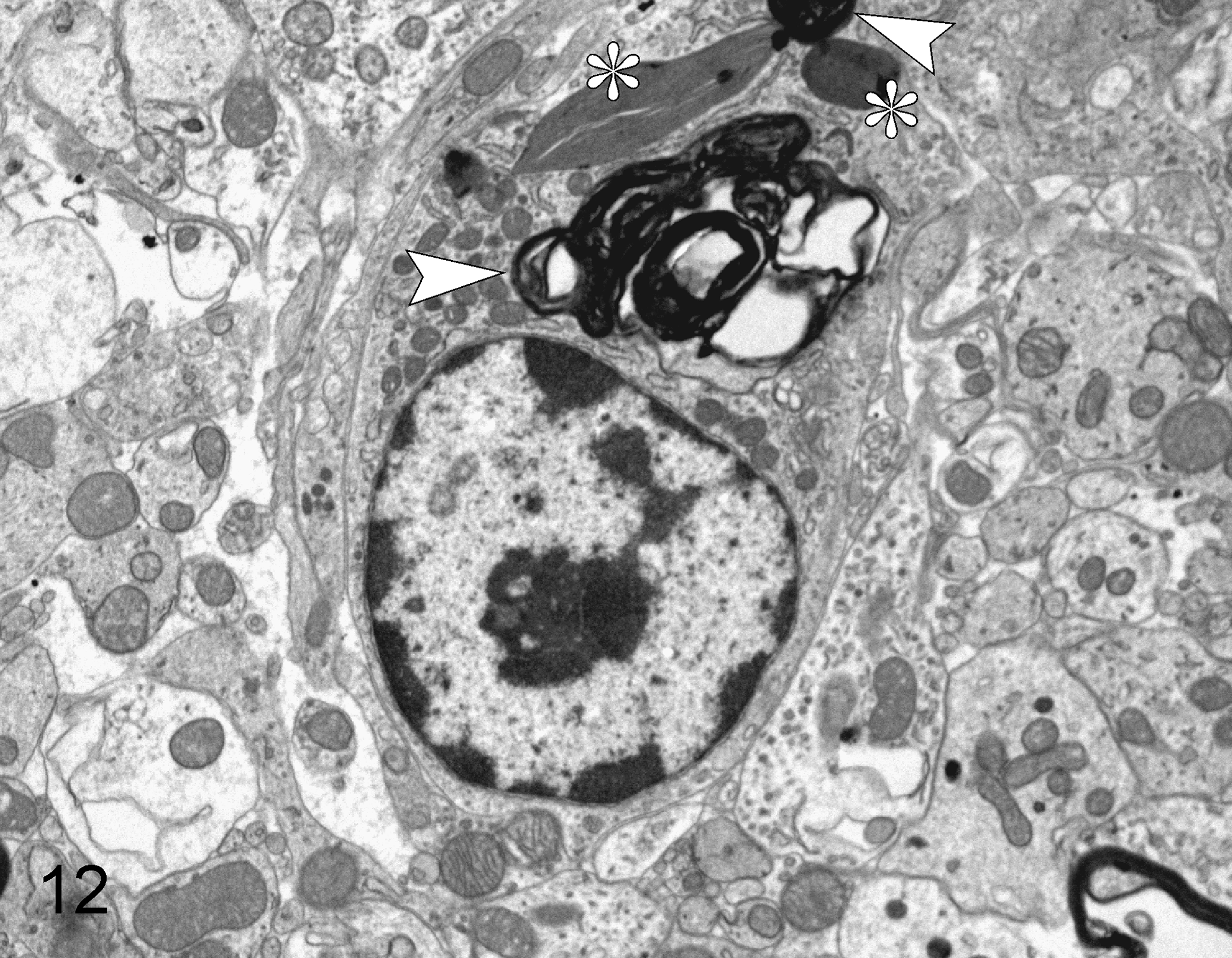
Spongy myelinopathy; brainstem; African pygmy hedgehog, case No. 3; transmission electron microscopy. In the center of the image is a macrophage that contains 2 accumulations of myelin debris (arrowheads) and 2 structures of unknown origin with some similarities to collagen (asterisks).
Discussion
The clinical histories and signs in this cohort of hedgehogs were similar to those previously described with WHS in both African pygmy hedgehogs 6 and European hedgehogs. 17 The clinical signs were primarily progressive ataxia and paresis resulting in paraplegia, tetraplegia, and death. Some previously reported signs that were not seen in the present study include dysphagia, self-mutilation, aggression, intestinal stasis, urine retention, and curvature of the spinal column (scoliosis). 6 A possible explanation for such variation is that most of the animals in this study were euthanized within 3 months of clinical onset; thus, very chronic stages were not represented.
Microscopically, areas of prominent status spongiosus had microgliosis with microglial hypertrophy and numerous branching processes. An increase of astrocytic processes surrounded spongy areas, representing a mild degree of astrogliosis. In addition, there was degeneration and loss of myelin, followed by degeneration and loss of neuronal cell bodies and axons. 6 Two cases had neuronal chromatolysis. The apparent low incidence of chromatolysis in this set of APHs precluded drawing any further conclusions about the potential implication of chromatolytic events in the pathogenesis of WHS. Chromatolysis in these cases is most likely secondary to myelin/axon damage. Acute and chronic foci of status spongiosus, largely defined by their extent and severity, were identified in all APHs included in this study. The timing of the various clinical signs identified in these APHs with WHS ranged between <1 week and more than 3 months. A precise timeline distinction between acute and chronic lesions in this cohort of APHs is troublesome. Nonetheless, it is reasonable to think that this spongy myelinopathy process progresses with aging and although new degenerative foci of status spongiosus may be identified at any time, more severe and extensive microscopic changes are expected in longstanding cases. Peripheral nerves or appendicular muscles were not examined microscopically in this study, so the degree to which a peripheral neuropathy caused neurogenic muscle atrophy could not be evaluated. 6,12
To the authors’ knowledge, this study provides the first detailed description of the distribution of WHS lesions. The most severe and extensive lesions occurred in the cerebellum, medulla oblongata, and cervical and thoracic spinal cord, followed by milder involvement of the corona radiata, corpus callosum, corpus striatum, internal capsule, and the mesencephalon. A previous study reported severe spongiosus of the optic chiasma, olfactory tract, and lateral lemniscus, as well as a predilection for the subpial parenchyma of the spinal cord. 17 In the current study, there was minimal involvement of the optic chiasm, lateral lemniscus, and the subpial spinal cord parenchyma.
Except for the cerebellum, there was no association between neurologic signs and microscopic findings. The location, severity, and extent of the cerebellar lesions could potentially explain the clinical signs compatible with cerebellar and vestibular disturbances; however, more in-depth data and a rigorous neurological study design are required to better define clinicopathological correlations in WHS. Although the central vestibular nuclei were examined, the inner ear and vestibulocochlear nerves were not examined, so any role for peripheral vestibular involvement could not be determined.
The authors compared findings in these cases to 2 adult APHs with no clinical history of WHS. In these animals, rare single clear vacuoles were observed in the white matter of the telencephalon and myelencephalon. The relevance of these vacuoles is unknown. We think they are most likely age related and incidental, as seen in humans, 22 rodents, 5 and other animal species. 4 These isolated vacuoles were not observed at the white-gray matter interface, in brain nuclei, or in the spinal cord.
The ultrastructural results of the present study support the microscopic findings that WHS is best defined as a spongy myelinopathy. Spongy myelinopathies are generally characterized by status spongiosus involving the white matter and often, in contrast to the present study, do not have degeneration of myelin with phagocytosis or any astrocytic response. 2 Status spongiosus or separation in the white matter of the CNS may result from several different disease processes, including splitting of the myelin sheath, accumulation of extracellular fluid, swelling of cellular processes, and axonal injury when spheroids are no longer detectable within distended spaces. Furthermore, status spongiosus should be distinguished from primary demyelination (loss of the myelin sheath). 21 In light of the present results, splitting of the myelin sheath, followed by axonal swelling and loss, seems the most plausible pathogenic mechanism in WHS. In addition, astrocytes have been reported to experimentally have an increase of ribosomes with cerebral degeneration. 2
In humans, the classic spongy myelinopathy is Canavan disease, 1 and in animals, recognized spongy myelinopathies include hereditary neuraxial edema in Hereford calves, 3 branched-chain ketoacid dehydrogenase deficiency (BCKD) in polled Hereford and Hereford calves, 8 congenital brain edema of Hereford calves, 11 canine spongiform leukoencephalomyelopathies, 14,16,25 spongy degeneration of kittens, 13 spongy myelinopathy of silver foxes, 7 and epizootic vacuolar myelinopathy of bald eagles and American coots. 23 These conditions are generally idiopathic, and many have a presumed genetic basis.
WHS shares many features with most spongy myelinopathies in other species in having a predilection for the white matter and splitting of the myelin sheath at the intraperiod line, but there are significant differences. Lesions in the present study that differ from other spongy myelinopathies include the following: (1) spinal cord involvement is not a feature of BCKD 8 and spongy myelinopathy of silky terriers, 19 (2) degeneration and loss of myelin and axons is not a feature of spongy myelinopathy in border terrier puppies, 16 and (3) involvement of ventral and lateral spinal cord funiculi is not a feature of hereditary neuraxial edema in Hereford calves. 3 Two additional lesions that occur in other species but not in WHS include congenital brain edema in Hereford calves that is characterized by hydropic degeneration (severe cellular swelling) of astrocytes 11 and spongy myelinopathy of silver foxes characterized by large cytoplasmic vacuoles in oligodendrocyte cytoplasm. 7
The etiology of WHS remains unknown. In the 1 report of WHS in European hedgehogs, 17 mild inflammation was seen in some cases, suggesting a possible infectious etiology, but attempted virus isolation and testing for at least 1 agent (canine distemper virus) were negative. A paramyxovirus (antigenically related to canine distemper virus) was isolated from a hedgehog showing neurologic signs in another report, but the lesions more closely resembled canine distemper than WHS. 24 Recently, a murine pneumovirus (Paramyxoviridae) was identified as a cause of nonsuppurative encephalitis and vacuolation of the white matter in an APH with suspected WHS, 15 but the significance of this finding is unclear. In the present study, inflammation was limited to only case No. 3, and it involved leptomeninges of the brainstem. No infectious agents were identified histologically or ultrastructurally in the present study.
Given the lack of evidence of an infectious, nutritional, or toxic etiology and the similarity to spongy myelinopathies in other species, a genetic basis seems most plausible. Since the US importation ban on APHs in 1991, subsequent inbreeding has led to a lack of genetic variability, 18 which supports a possible heredofamilial component. 6 Also, WHS has not been reported in wild APHs.
Although a genetic etiology is most likely, other causes should still be considered. The lesions of WHS resemble the lesions of hepatic and renal encephalopathy. 3 Previous WHS studies have reported concurrent hepatic and renal lesions in some animals, including hepatic lipidosis in up to 20% of cases and frequent renal cortical infarcts. 6,17 In the present study, 5 of 12 animals had gross or histologic evidence of hepatic lipidosis, but it was considered severe in only 1 case. Other hepatic or renal lesions in animals of this study included single cases of severe centrilobular hepatic necrosis, renal infarction, membranous glomerulonephritis, lymphoplasmacytic interstitial nephritis with fibrosis, and severe polycystic kidneys. Given the nonuniform incidence of hepatic and renal disease in this study and in previous reports, hepatic and renal diseases are considered unlikely causes of WHS.
Neoplasia is reported to be a common cause of death or euthanasia in APHs. 9 Neoplasms identified in this study that have previously been reported in this species are listed in Supplemental Table S1. Other neoplasms that occurred in the present study and are not previously reported in APHs included renal adenoma, urinary bladder leiomyoma, urinary bladder papilloma, and disseminated pulmonary adenocarcinoma. Some of the concurrent neoplastic and nonneoplastic conditions in this set of APHs may have contributed to the neurologic signs. These conditions include myeloid leukemia (case No. 2), eosinophilic leukemia (case No. 4), multicentric histiocytic sarcoma (case No. 9), disseminated pulmonary adenocarcinoma (case No. 12), low-grade cerebral astrocytoma (case No. 3), subependymal giant cell astrocytoma (case No. 4), spinal astrocytoma (case No. 11), calvarial spindle cell sarcoma (case No. 8), and botryomycosis (case No. 6). The exophthalmos (case Nos. 1, 6, and 7) and keratitis (case Nos. 6 and 7) appeared unrelated to WHS in these cases. In case No. 10, there was thoracic and lumbar intervertebral disc disease (IVDD). Although IVDD is reported in adult hedgehogs, 10 there are no reports on IVDD-induced spinal cord lesions. A potential role for IVDD in spongy change in cervical spinal rootlets in this case cannot be ruled out; however, there was no evidence of extradural hemorrhage, fibrocartilaginous embolism, hemorrhagic myelomalacia, or any inflammatory reaction at this level. IVDD was not clinically reported or grossly evident upon longitudinal sectioning of the vertebral column in the remaining animals.
In summary, this study reports the lesions and their neuroanatomical distribution in 12 APHs with WHS. The primary lesion was a widespread symmetrical status spongiosus resulting from splitting of the myelin sheath at the intraperiod line. Based on these results, WHS is best described as a spongy myelinopathy with widespread CNS involvement. Future studies should focus on the potential genetic basis of the disease. The high prevalence of WHS makes the disease a potential model for studying similar myelinopathies.
Supplemental Material
Supplemental Material, DS1_VET_10.1177_0300985818768033 - The Pathology of Wobbly Hedgehog Syndrome
Supplemental Material, DS1_VET_10.1177_0300985818768033 for The Pathology of Wobbly Hedgehog Syndrome by Josué Díaz-Delgado, Derick B. Whitley, Ralph W. Storts, Jill J. Heatley, Sharman Hoppes, and Brian F. Porter in Veterinary Pathology
Footnotes
Acknowledgements
We thank Dr. Andy Ambrus for the immunohistochemical staining and Ross Payne for the electron microscopy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
