Abstract
The granuloma is the hallmark of tuberculosis and simultaneously signifies acquisition of an infection and induction of a host immune response. But who benefits more from the development of the granuloma, the host or the pathogen? Is microbe or man dictating disease course and progression? Mycobacterial diseases affect humans and animals alike, and the concepts presented in this review reflect host-pathogen interactions that influence not only mycobacterial granulomas in humans and animals but also other infectious granulomatous diseases that are encountered in veterinary medicine. Current dogma supports that an organized granuloma is a mark of an adequate and “restrictive” host immune response. However, the formation of a granuloma also provides a niche for the maturation, growth, and persistence of numerous infectious agents, and these pathogens devote some portion of their genetic machinery to ensuring these structures’ form. An understanding of pathogens’ contributions to granuloma formation can aid the development of host-directed therapies and other antimicrobial and antiparasitic therapies that can tip this balance in favor of a restrictive host response and elimination—not just containment—of the infectious organism. This review discusses animal models that have aided our understanding of pathogens’ contribution to the host response and how mycobacterial virulence genes direct host pathology in ways that may aid disease transmission and/or persistence in the form of latent infection.
Granuloma Formation Is Necessary to Control Mycobacterial Disease
In the case of mycobacteria, an adequate immune response in the form of a granuloma is absolutely necessary to control infection. 38,90 Histologically, classic human and nonhuman primate (NHP) granulomas are characterized by the presence of a necrotic “core” composed of acellular, eosinophilic homogeneous necrotic debris where extracellular mycobacteria can persist. 74 Depending on the stage of necrosis, the center of the granuloma will also contain a mixture of macrophages, dendritic cells, and multinucleate Langerhans and foreign body type giant cells that may contain intracellular bacilli. 1 Variable numbers of CD4+ and CD8+ T lymphocytes, B lymphocytes, and plasma cells will be recruited to these structures in an effort to “wall off” and contain the bacterial nidus and any tissue damage associated with bacterial replication and necrosis. During active, progressive disease in humans, lesion enlargement and progressive necrosis results in erosion of vascular and bronchial walls, allowing spillage of infectious material into the airways, thereby promoting disease transmission. 81 In the case of a restrictive granuloma, this layering of cells becomes increasingly organized and the lymphocytic rim is reinforced by the presence of organized fibrotic tissue, leading to containment of necrosis, reorganization of cellular infiltrates, and resolution of pathology. B-lymphocyte aggregates also organize into secondary lymphoid structures akin to ectopic lymphoid follicles that may be a source of mycobacterial-specific antibodies. 60 Resolved granulomas can be identified radiographically and histologically by calcification and saponification of remnant necrotic debris contained within these structures. 14,74
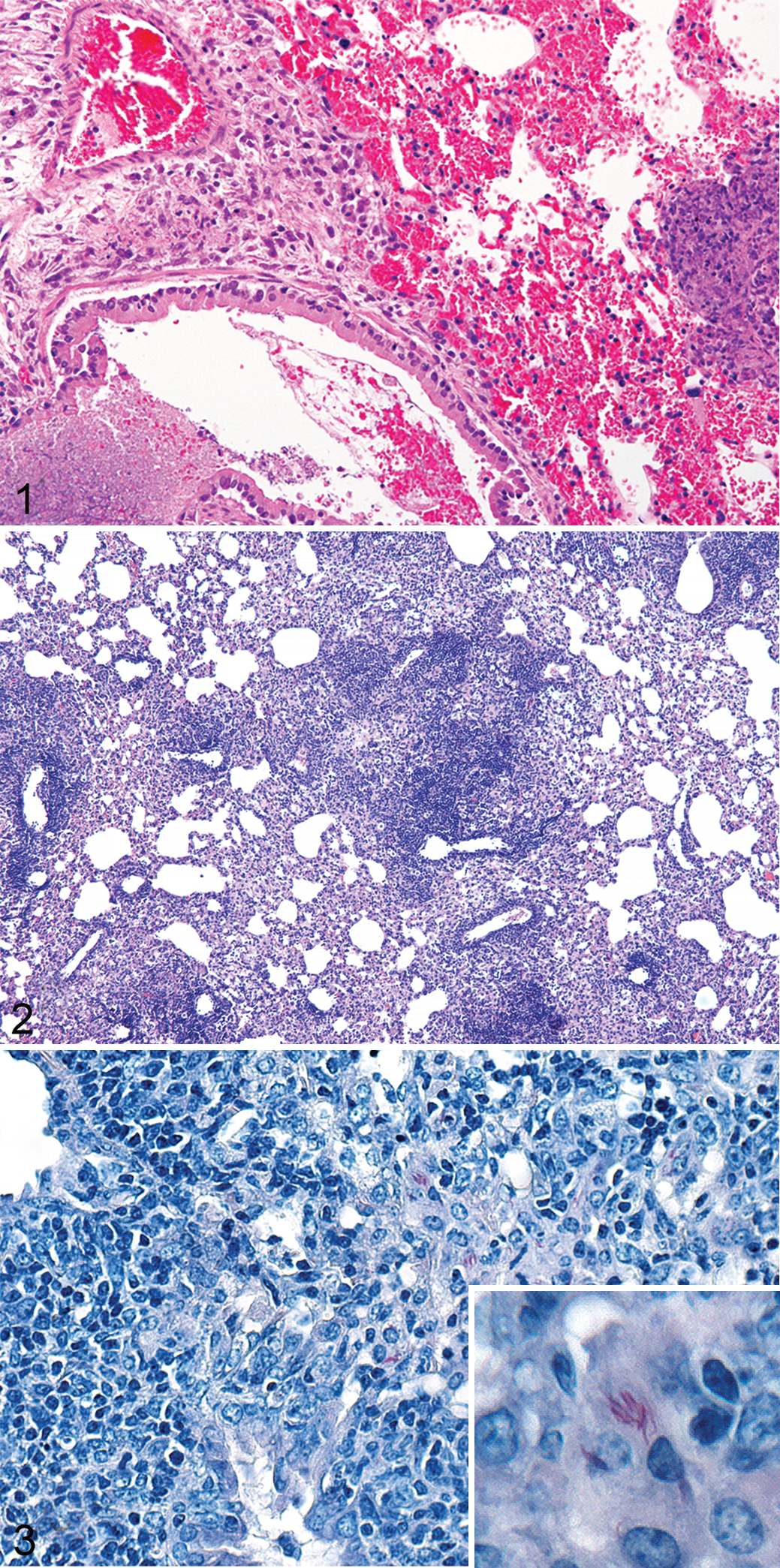
T-lymphocyte defenses are critical for the development of restrictive granulomas. Mice that lack CD4+ T lymphocytes have rapidly progressive tuberculosis (TB) characterized by massive infiltrates of neutrophils and macrophages, leading to fulminant inflammation, necrosis (noncaseous), and bacterial dissemination to numerous organ systems. 115 Likewise, CD8+-deficient animals have enhanced breakdown of immunity during chronic infection characterized by progressive interstitial pneumonia, 121 suggesting that CD4+ T cells are critical to early containment and CD8+ T cells contribute to a sustained cytotoxic response that controls bacterial replication later in disease. 39,87 Mice that are deficient in both CD4+ and CD8+ T lymphocytes, such as severe combined immunodeficiency (SCID) mice, have rapidly progressive disease characterized by infiltration and expansion of infected macrophages, leading to lung consolidation. Unchecked mycobacterial replication results in tissue necrosis, fulminant bronchopneumonia, and death (Fig. 1). What is interesting about this balance from the pathogen perspective is that Mycobacterium tuberculosis (Mtb) has evolutionarily conserved both immunodominant CD4+ and CD8+ T-cell epitopes as part of its genetic makeup. 25,27 This feature fits a scenario whereby a dominant T-cell response has been favorable to mycobacterial survival over millennia and indicates that T-cell recruitment to sites of infection may offer some advantage to Mtb. Postprimary TB disease (reactivation) in humans is thought to be the result of hypersensitivity to Mtb antigens, not bacterial replication per se, since most postprimary disease in humans is paucibacillary. 53 In many cases of postprimary disease in people, granuloma formation is secondary to subclinical obstructive bronchopneumonia that fulminates in caseous necrosis due to lipid and antigen accumulation in the absence of large numbers of bacilli, again suggesting that antigen dose, not bacillary dose, promotes tissue necrosis and Mtb spread. So “who or what” is really in charge of disease progression, and how can an understanding of mycobacterial genetics help us identify novel therapeutic targets and design novel vaccine modalities to combat TB disease in both humans and animals?
Evolutionary Conservation of T-Cell Immunodominant Epitopes Suggests that T Lymphocyte–Mediated Immunity Is Important to Mycobacterial Survival
How might mycobacteria co-opt a T-cell response to favor Mtb survival? With the advent of inexpensive and high-throughput whole-genome sequencing, we can now track the evolutionary arc of mycobacterial diversification and mutation as a function of genetic and ecological pressure. By studying sequence homology of archaic Mtb genomes recovered from mummies and ancestral hominoids, we can determine which Mtb genes have been under selective pressure and likewise how coevolution of Mtb with man may have led to genetic bottlenecks reflected in both the mycobacterial and human genomes. 114 Genes that are under little genetic pressure are more susceptible to genetic drift while genes that are essential for Mtb survival in humans will be evolutionarily conserved. Frequent single-nucleotide polymorphisms (SNPs) in T-cell epitopes would be indicative of genetic pressure toward immune evasion. For this reason, it is striking that many immunodominant T-cell epitopes found in mycobacterial proteins have been hyperconserved in the genomes of Mtb and other virulent strains of the mycobacteria tuberculosis complex (MTBC), including the human pathogens Mycobacterium cannetti and Mycobacterium africanum, and virulent zoonotic mycobacterial species, such as Mycobacterium bovis, Mycobacterium microti, and Mycobacterium pinnipedi. In work by the Ernst and Gagneux groups, 25,27 sequencing of 20 bacterial strains representative of the 6 main human MTBC lineages revealed that sequence fidelity across 491 known human CD4+ and CD8+ T-lymphocyte epitopes was as high as that seen for essential Mtb genes, suggesting that T-cell recognition of mycobacterial epitopes is essential for Mtb survival.
Mtb-Secreted Proteins Can Promote Macrophage Recruitment and Maintenance of the Granuloma
Mtb virulence factors are generally divided into lipids and proteins, with secreted proteins of specific interest since the thick lipid-rich Mtb cell wall requires elaborate machinery to transport these proteins outside the cell. 48 Canonically, the focus on Mtb virulence factors has been on mechanisms of immune evasion, not immune recognition. How can we reconcile these data with the hypothesis that immune recognition of these factors favors Mtb survival based on evolutionary conservation of T-cell epitopes? What can this tell us about virulence factors orchestrating the granulomatous response? The case of early secreted antigenic target 6 (ESAT-6) is an illustrious example of this phenomenon. The ESAT-6 protein contains over 40 highly conserved T-cell epitopes (CD4+ and CD8+), and ESAT-6 is continuously expressed in vivo during infection with Mtb. 102 This immunodominant protein forms the basis for the human diagnostic test for Mtb exposure, the interferon gamma release assay (IGRA), or Quantiferon®-TB Gold (Qiagen Germantown, MD). This enzyme-linked immunosorbent assay (ELISA)–based assay detects the presence of mycobacterium-specific memory T cells in human subjects. By stimulating human PBMCs with ESAT-6, a protein found in Mtb but not bacillus Calmette-Guerin (BCG, derived from Mycobacterium bovis), clinicians can distinguish exposure to Mtb (active and latent forms of disease) from BCG vaccination. 40
ESAT-6 and its secretion machinery were initially identified as virulence factors since they are encoded on a region of the mycobacterial genome that was lost during passage of virulent M. bovis to form the vaccine strain, BCG. The RD1 region, or “region of difference 1,” is a 9.5-kb genome segment that contains a number of genes but, most prominently, the esx-1 locus. 24 This genetic element encodes for a type VII secretion system necessary for translocation of ESAT-6 across the cell wall and has been linked to a number of host responses characterized as favorable to bacterial survival. 11 Esx loci are highly conserved across virulent mycobacterial species, including Mtb and Mycobacterium marinum. Using the zebrafish model, Volkman et al 124 determined that mycobacterial strains lacking RD1 fail to induce granuloma formation. Others have shown that deletion of the RD1 locus and esx-1 in isolation leads to attenuation of virulence in mice. 72 Secretion of this immunodominant antigen has been implicated in lysis of the phagosomal membrane, 52 induction of apoptosis, 2 recruitment of macrophages, and expansion of the granuloma. 32 In addition, ESAT-6 has been show to directly activate the inflammasome. 83,84 Collectively, these responses contribute to bacterial replication, cell death, and necrosis—critical components of the mycobacterial granuloma.
One potential mechanism by which T-lymphocyte immune recognition of Mtb promotes a favorable pathological response is through tumor necrosis factor (TNF)–α secretion. As it pertains to granuloma formation and maintenance, an important function attributed to ESAT-6 secretion is induction of TNF-α production by infected macrophages. TNF-α has long been known to control TB disease in mice, 43 and numerous studies have highlighted the important role of TNF-α in maintaining granuloma integrity. 23,38 The role of TNF-α in maintaining restrictive granulomas in people has been clinically corroborated with the introduction of TNF-α antagonists, such as monoclonal anti–TNF-α immunoglobulin G (IgG), for the treatment of immune-mediated rheumatoid arthritis and noninfectious inflammatory diseases such as Crohn disease. 61 Patients with undiagnosed latent Mtb infection treated with TNF-α antagonists have a higher risk of TB reactivation, presumably via breakdown of granuloma structure. A major source of TNF-α is CD4+ T lymphocytes. 36 It has been shown that individuals with active TB disease have higher numbers or CD4+ TNF-α–secreting T lymphocytes than individuals with latent Mtb infection. 50 Engagement of T-cell receptors (TCRs) by immunodominant antigens such as ESAT-6 may initiate an immune signaling cascade that induces TNF-α secretion by T lymphocytes, leading to macrophage death and cellular necrosis. This is exemplified by studies of programmed cell death protein 1 (PD-1) deficiency in mice. PD-1 is a regulatory marker expressed on activated and exhausted T lymphocytes. PD-1 expression is critical for regulating immunopathology in the context of exuberant inflammatory responses. Mice that lack PD-1 have progressive TB disease 5,70 that correlates with elevated levels of inflammatory cytokines, including TNF-α. Thus, balanced levels of TNF-α are critical to controlling TB disease, yet expression of Mtb virulence factors may affect the timing, amount, and localization of TNF-α secretion in ways that may promote bacterial persistence and/or transmission in the context of granuloma structure.
Likewise, TB10.4 protein contains one of the most potent CD8+ T-cell epitopes known to date. 126 TB10.4, a secreted protein encoded by esxH within the esx-3 locus, has structural and functional homology to ESAT-6. The esx-3 locus, like esx-1, is essential for in vivo growth of Mtb partly due to its important role in iron acquisition during host infection. CD8+ T-cell recognition of the TB10.4 epitope after vaccination results in clonal expansion of a limited number of CD8+ T lymphocytes with high-affinity TCRs. Carpenter et al 16 showed that these memory cells were at a proliferative disadvantage compared with naive cells during Mtb challenge. One hypothesis is that Mtb benefits from an immunodominant CD8+ T-cell “decoy” response whereby naive cells outcompete memory CD8+ T lymphocytes that are more effective at bacterial killing. Immunodominance in Mtb infection may benefit the bacterium by orchestrating a granulomatous response that provides a pathologic niche for long-term persistence of the mycobacterium. In summary, secreted proteins of Mtb contribute to granuloma formation, maintenance of granuloma structure, and recruitment of both CD4+ and CD8+ T lymphocytes to sites of infection. While these responses are key to granuloma organization and local resolution of immunopathology, they may also promote establishment of latent infection.
Granuloma Formation May Promote Transition to Latency
Facultative intracellular organisms such as Mtb rely on a cellular niche for replication and persistence. Mycobacteria co-opt a major innate host defense, phagocytic uptake of infectious agents, to gain access to its preferred site of replication. We have discussed how virulence mechanisms may promote the influx of macrophages to sites of infection providing more target cells for mycobacterial replication 32 and how immune recognition of immunodominant T-cell epitopes during the adaptive immune response may recruit effector cells that provide structure to the granuloma while simultaneously promoting cellular necrosis. 123 Studies looking at virulence genes in mice in combination with in vitro granuloma models have begun to highlight that bacterial virulence genes can regulate both Mtb and host cellular metabolism. Macrophage effector responses, including interferon-γ secretion, phagolysosome acidification, phagolysosome fusion, and deployment of reactive oxygen species, all exert pressure on mycobacteria to engage in stringent responses that arrest growth of Mtb. Induction of the DosR regulon in vitro is associated with initiation of Mtb dormancy, defined as a nongrowing yet persistent state. As mycobacteria shift toward this metabolic state, Mtb can divert host cellular metabolism in ways that promote lipid body (LB) formation. Foamy macrophages are poorly bactericidal and have impaired autophagy and lysosomal acidification. 77,112 Necrosis of foamy macrophages is thought to directly contribute to caseum formation by spillage of intracellular lipids and LB-associated mycobacteria into extracellular spaces. 104 This is important for the long-term bacterial survival since Mtb prefers to catabolize host-cell cholesterol in vivo as a source of carbon. As the necrotic center of the granuloma forms, Mtb begins to metabolically adapt to this new harsh environment, triggering compensatory changes in host-cell metabolism that will provide a niche and long-term source of cellular cholesterol to promote Mtb persistence. This fact highlights that mycobacterial virulence genes affect not only the type and number of infiltrating immune cells to initiate granuloma formation but also the metabolic state of infected cells, again demonstrating how mycobacteria have co-opted the granulomatous response for its benefit. 71
Transposon Mutagenesis in the C57BL/6 Mouse Model for Understanding Mtb Virulence Mechanisms
What has the mouse model of Mtb infection taught us about how Mtb controls granuloma formation? In the most widely used mouse model, using C57BL/6 mice, Mtb infection leads to neither caseous granuloma formation nor the development of latent disease. Therefore, the C57BL/6 mouse model does not recapitulate 2 very important aspects of TB disease in people. Nevertheless, this mouse model has been extremely instructive for understanding Mtb virulence genes—many of which are implicated in orchestrating the host response to infection. 91,107 Generally speaking, the mouse granuloma is an “inverted granuloma” loosely composed of T lymphocytes surrounded by macrophages and occasionally fibrous connective tissue (Figs. 2, 3). These structures can be classified as “pseudogranulomas” since they lack traditional spatial organization, are not hypoxic, and, in most mouse models, are not characterized by central necrosis. Histologically, Mtb infection in the mouse results in multifocal to coalescing, lymphohistiocytic, interstitial pneumonia, and the spectrum of granulomatous disease differs across mouse strains. 90 C57BL/6 mice are considered to have a Th1 bias. 26 C57BL/6 mice are restricted to one major histocompatibility complex (MHC) class II allele, I-A b , and T-cell epitopes that are recognized by the I-A b MHC II allele in C57BL/6 mice result in strong interferon-γ secretion by memory CD4+ T lymphocytes, a response that in humans and mice alike is associated with control of mycobacterial disease. 76 In C57BL/6 mice, mycobacterial growth is characterized by an initial lag phase during the first 7 days of infection, followed by a logarithmic growth phase over the remaining 3 weeks of infection (Fig. 4). By 4 weeks postinfection, bacterial burden will plateau and the pathology associated with bacterial disease at this stage can be classified as chronic active. Mice can survive with pulmonary pathology for 3 to 6 additional months without serious complications. Eventually, disease will progress and cause death in a subset of mice. For this reason, the C57BL/6 mouse model is considered a restrictive model for studying disease pathogenesis. Any change in the ability of Mtb to survive in this model would be highly indicative of a virulence gene.
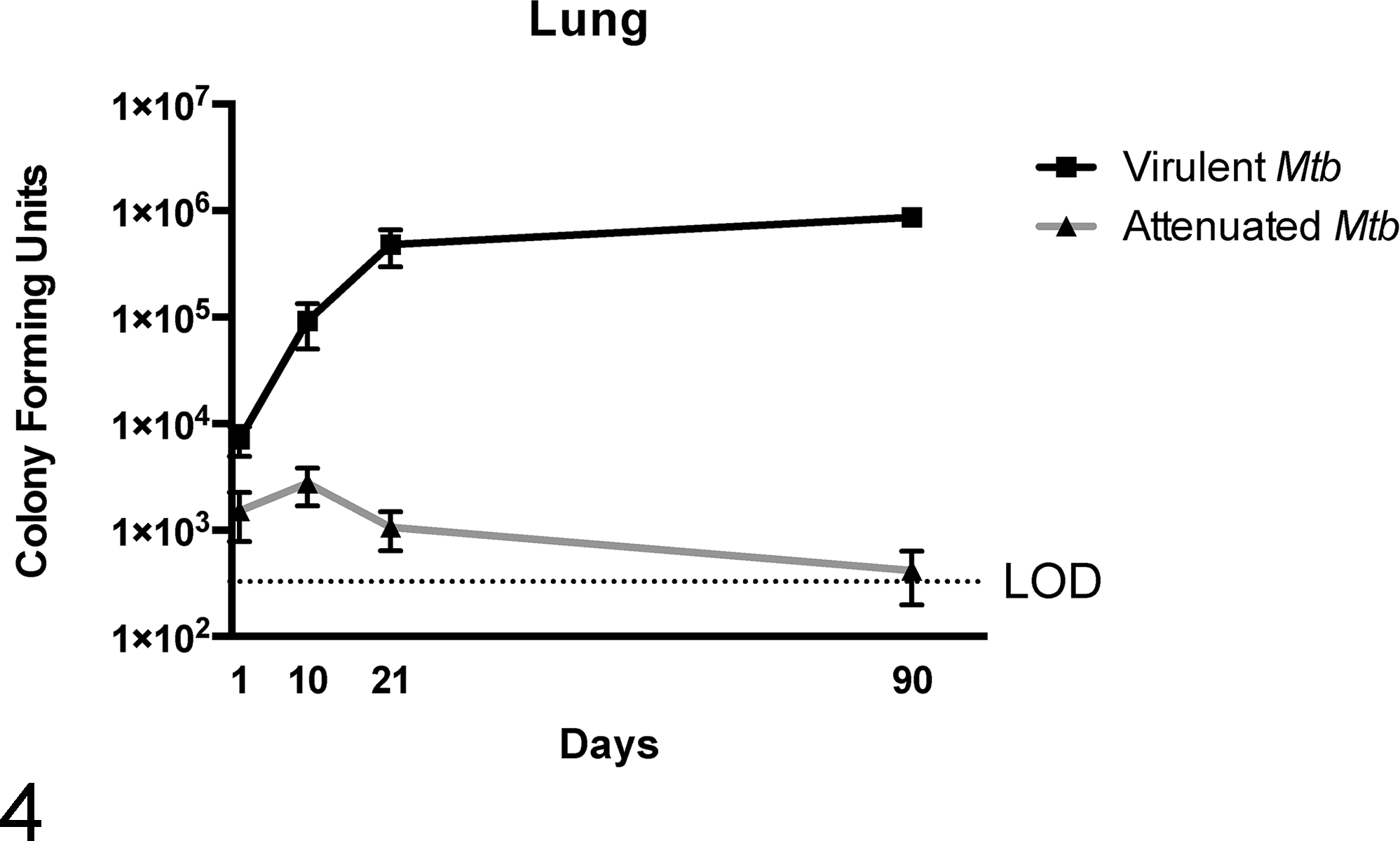
Growth curves of virulent and nonvirulent Mycobacterium tuberculosis (Mtb) in C57BL/6 mice. Mice were intravenously injected with 1 × 106 colony-forming units of Mtb H37Rv (virulent Mtb) or H37Rv Tn:: rv1410c (attenuated Mtb). After infection with virulent Mtb, exponential growth is observed during the first 7 to 21 days postinfection. By 3 weeks postinfection, the replication rate plateaus and similar numbers of virulent Mtb are recovered from lung. Attenuated Mtb strains have a different growth curve as seen by lower amounts of bacteria recovered from the lung at various time points postinfection. LOD, limit of detection.
Using a forward genetics approach, the C57BL/6 model has thus elucidated numerous virulence genes by employing a genetic technique referred to as transposon (Tn) mutagenesis. Briefly, cultures of virulent Mtb are mutagenized by transduction with a mycobacterial phage, MycoMarT7, engineered to carry an antibiotic resistance gene. Upon phage infection of the mycobacterium, an antibiotic cassette will site-specifically insert at T-A nucleotide junctions randomly across the Mtb genome. Proper use of this mutagenesis system generates a library of Mtb genetic mutants that span the entire ∼4000 genes of the Mtb genome. The bacterial Tn (mutant) library is expanded and sequenced to ensure genome coverage, and the “input” Tn library is used to infect mice (Fig. 5). After 4 weeks of growth and expansion, the spleens from the mice are harvested and the surviving mycobacteria are recovered. Genetic mutations involving virulence genes will be lost during the in vivo replication of the bacteria. Using high-throughput whole-genome sequencing, the mutants that are recovered, comprising the “output” Tn library, can be annotated and output library genes are categorized as “nonessential” for growth in the mouse. 111 By comparing the genes contained in the input library with those of the output library, essential genes for in vivo growth can be identified. Using this technology, work by the Sassetti and Rubin labs have identified a set of genes that are conditionally essential for survival in the mouse (Table 1). 101,130 These seminal works helped identify a number of virulence genes in Mtb, many of which have been implicated in mycobacterial immunopathology, such as the genes involved in the esx-1 secretion system. While other inbred mouse strains such as C3H/FeJ develop caseous necrosis 66 and use of outbred mouse strains such as the collaborative cross are useful for studying mycobacterial pathology, 113 MHC class I and II restrictions on these backgrounds have limited the ability to study correlations of immune responsiveness to known Mtb epitopes and granuloma formation using these strains. Nevertheless, future screens of transposon libraries on murine background strains that develop more classical TB lesions will likely uncover novel roles of mycobacterial virulence genes in the context of granuloma formation, maintenance, and mycobacterial survival.
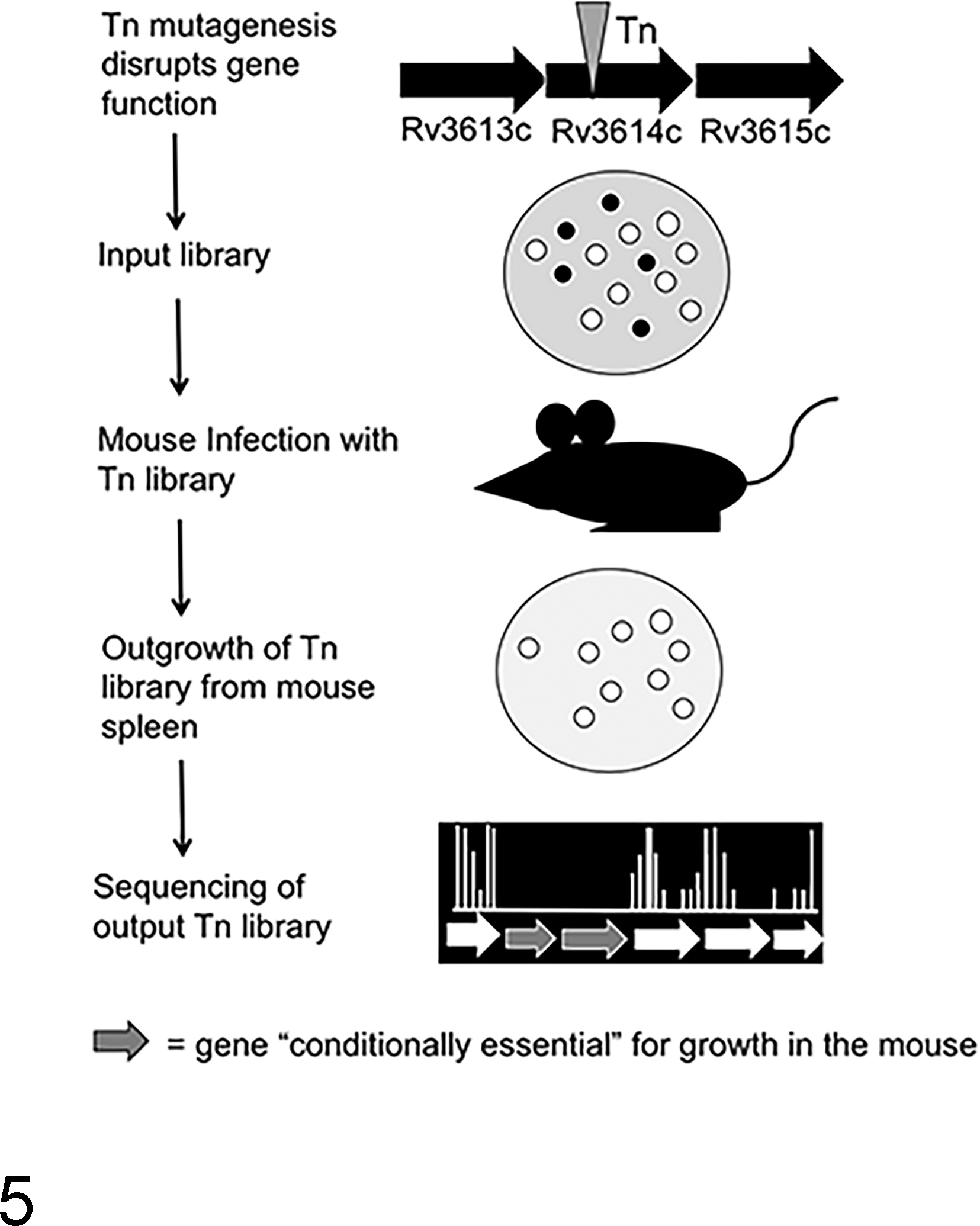
Schematic for transposon mutagenesis to generate libraries of Mycobacterium tuberculosis (Mtb) genetic mutants. Wild-type (virulent) Mtb, H37Rv, is transduced with the mycobacterial phage, MycoMar T7, carrying a transposon (Tn). Insertion of the transposon inactivates the gene or renders the gene product nonfunctional. The pool of mycobacterial mutants is plated, and each colony represents a mycobacterial strain with a Tn insertion in 1 gene. C57BL/6 mice are infected intravenously with the Tn “input” library. After 4 to 8 weeks of in vivo growth, mice are sacrificed and Mtb is isolated from splenic homogenates and plated to recover the Tn “output” library. DNA extraction and whole-genome sequencing of input and output libraries allow identification of Tn mutants present in either the input or output libraries. Mycobacteria with mutations in conditionally essential genes (black circles) will be present in the input library but will not be present in the output library after passage in the mouse.
Mycobacterium tuberculosis Genes That Are Conditionally Essential for Growth in C57BL/6 Mice.a
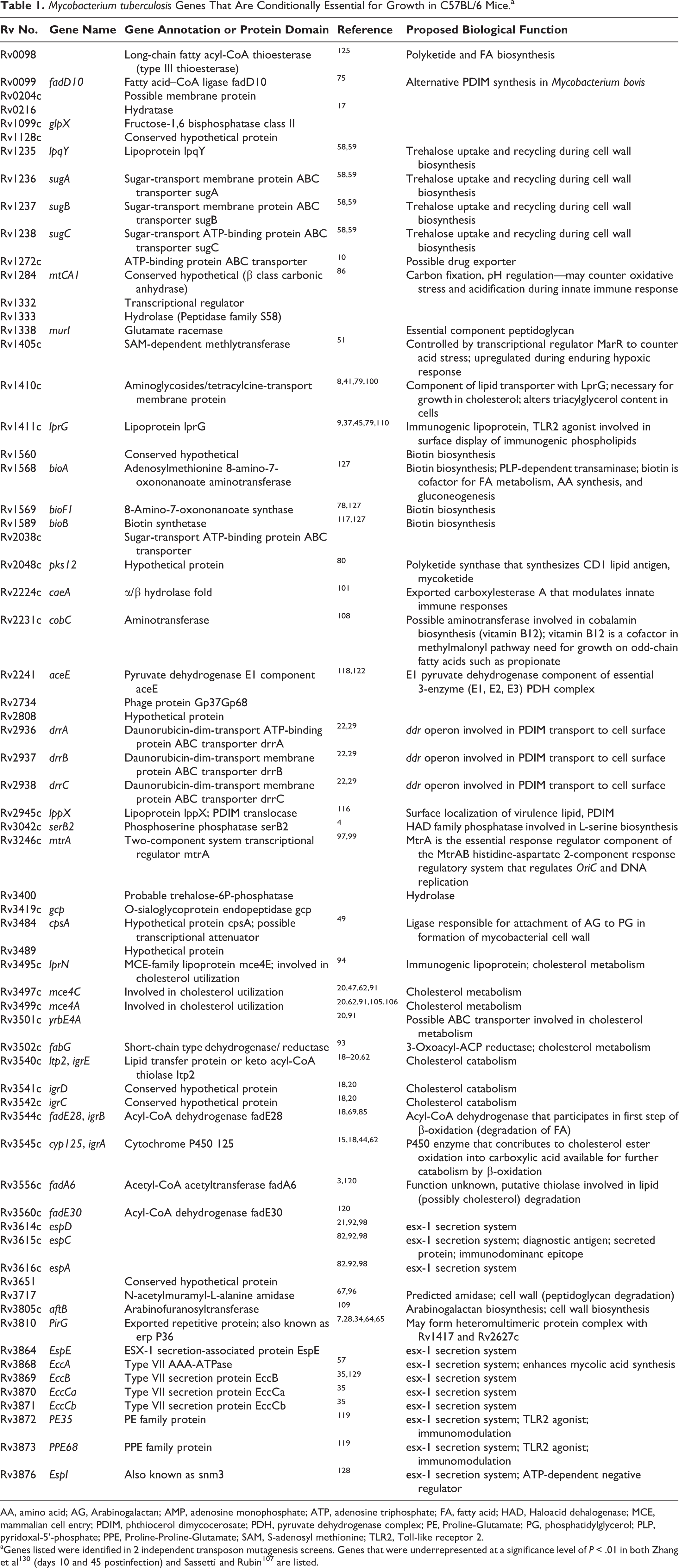
AA, amino acid; AG, Arabinogalactan; AMP, adenosine monophosphate; ATP, adenosine triphosphate; FA, fatty acid; HAD, Haloacid dehalogenase; MCE, mammalian cell entry; PDIM, phthiocerol dimycocerosate; PDH, pyruvate dehydrogenase complex; PE, Proline-Glutamate; PG, phosphatidylglycerol; PLP, pyridoxal-5’-phosphate; PPE, Proline-Proline-Glutamate; SAM, S-adenosyl methionine; TLR2, Toll-like receptor 2.
Mtb Virulence Genes Promote Metabolic Transformation and Survival on Host-Derived Lipids
This highly curated list of Mtb virulence genes needed for survival in the C57BL/6 mouse highlights that many genes necessary for in vivo growth of Mtb encode enzymes necessary for mycobacterial utilization of lipids only available in host tissues, such as cholesterol. 91 It is now widely accepted that the preferred in vivo carbon source of Mtb is host-derived cholesterol. Cholesterol is widely abundant in eukaryotic cell membranes and granuloma caseum. The mce genetic loci (mce1–mce4) encode for multigene cholesterol importers. Other conditionally essential genes, such as igrA–E, encode enzymes that degrade various components of this complex lipid into “digestible” lipid fragments. Lipid fragments in the form of acyl chains can then be further broken down into 2- and 3-carbon (C2 and C3) units available for incorporation into the complex lipids that make up the mycobacterial cell wall or shuttled into the TCA cycle for generation of adenosine triphosphate (ATP). The catabolism of host-cell cholesterol in the hypoxic, necrotic granuloma core necessitates a metabolic shift from aerobic to anaerobic functioning, and this transition likely contributes to induction of a dormant-like state. 46 Under this stress, Mtb remodels its cell wall and generates large amounts of intracellular triacylglycerol (TAG), a storage lipid. LB-laden Mtb are seen within foamy macrophages that are also characterized by high TAG content. Conditionally essential genes reflect the necessity for lipid catabolism in vivo and tight genetic control of cellular lipid content. The genes that encode for the lipid transporter LprG-Rv1410 have been identified as two of the most essential genes for in vivo Mtb growth. 79 Loss of this transporter leads to increased levels of Mtb cellular TAG levels and results in decreased growth rate of Mtb when cholesterol is the sole carbon source. Therefore, genetic elements that modulate lipid synthesis, degradation, and transport function as virulence factors that regulate Mtb growth and adaption in response to host immune pressure.
Mtb Lipids Are Adjuvants and Major Modulators of Tissue Inflammation During Mtb Infection
The mycobacterial cell wall is composed of a complex layer of highly specialized lipids and is extremely immunogenic. The lipid-rich cell wall serves as a permeability barrier for Mtb and limits the amount and types of antimicrobial drugs that can be used to treat mycobacterial infections. Screening transposon libraries has highlighted the importance of some mycobacterial lipids for survival in vivo. One of the best-known Mtb lipid virulence factors is phthiocerol dimycocerosate, or PDIM. PDIM was genetically identified as important for Mtb virulence when it was discovered that Mtb strains lacking genes for PDIM biosynthesis and transport were significantly attenuated for growth in mice. 29 PDIM, an immunogenic mycobacterial lipid present on the outermost surface of the thick mycobacterial cell wall, has been implicated in a number of biological functions important to virulence. PDIM has been show to influence Toll-like receptor binding and activation status of macrophages. 13,33 PDIM mutants have altered cell wall permeability that is linked to enhanced killing by nitric oxide (NO) and other antimicrobials. 12,103 Last, PDIM biosynthesis is metabolically coupled with synthesis of other virulence-associated methyl-branched lipids, and alterations in PDIM transport have been linked to altered ex vivo growth rate of Mtb in macrophages. 56 The importance of PDIM as a virulence factor is reflected by the number of conditionally essential genes that regulate its transport across the cell wall, including the lipoprotein, LppX, and the ABC-like transporters, ddrA–C, which together are implicated in the proper positioning of PDIM on the cell surface. Numerous works have confirmed that loss or decreased levels of PDIM result in attenuation of mycobacterial strains. 107 Furthermore, alterations in PDIM levels may result in compensatory alterations in other cellular lipids that result in a loss of fitness for Mtb in vivo. 56
Another important mycobacterial lipid, trehalose dimycolate (TDM), also functions as virulence factor–modulating granuloma formation during TB disease. TDM or cord factor has been widely described as a direct stimulus for granulomatous inflammation. 55 Administration of purified TDM leads to formation of sterile lung granulomas indistinguishable from Mtb-induced granulomas in mice. 6 Interestingly, TDM-induced necrosis is dependent on the bacterial burden and lipid content of the cellular environment. It has been proposed that the shedding of TDM in a lipid-rich environment precipitates caseous necrosis within granulomas. 54 TDM is also the main component of the well-known adjuvant, complete Freund’s adjuvant (CFA). The importance of trehalose for in vivo survival of Mtb is reflected by the conditional essentiality of genes responsible for recycling of the trehalose moiety of many glycolipids. The LpqY-SugABC importer is formed by the periplasmic sugar-binding lipoprotein, LpqY, and a series of proteins that compose a transmembrane channel (Sug A, B, and C) that together mediate retrograde uptake of trehalose. Together, these proteins scavenge extracellular trehalose released during conversion of trehalose mono-mycolate (TMM) to TDM by the antigen 85 complex of enzymes that have mycolic acid transferase activity during cell wall synthesis. 59 These 2 examples of bioactive lipids and the conditional essentiality of genes related to their export and import illustrate how genetic regulation of these processes by Mtb may dictate disease course and progression. 53
Mycobacteria Have Co-opted Host Responses to Promote Bacterial Dissemination—Insights From the Zebrafish Model
The development of the zebrafish model of mycobacterial disease has become a powerful tool for dissecting the roles of virulence-associated genes and host response in mycobacterial pathogenesis. M. marinum, a causative agent for mycobacterial disease in fish, is characterized by necrotizing granulomas. Natural infection with M. marinum typically results in generalized mycobacteriosis with granulomas containing numerous acid-fast bacilli present in many organs. 88 Zebrafish larvae are transparent during the initial stages of development. Since the adaptive immune system in zebrafish is not fully developed until 4 to 6 weeks postfertilization, 68 use of this model allows investigators to study host-pathogen interactions in the context of the innate immune response, without using models of immune deficiency as are typically employed in mice. In brief, zebrafish larvae are injected intravenously with virulent or knockout strains of M. marinum via the caudal vein or into the hindbrain ventricle (Fig. 6, upper). Using fluorescent mycobacteria and host-cell markers, trafficking of macrophages and mycobacteria can be followed in real time using time-lapse fluorescent microscopy. By 3 days postinfection, macrophages can be visualized trafficking to sites of infection (Fig. 6, lower). Adult zebrafish experimentally infected with M. marinum develop classic caseous granulomas in numerous organs, including the kidney, testes, and pancreas (Fig. 7). 31 Important work by Davis and Ramakrishnan 32 demonstrated for the first time the role of the granuloma in potentially promoting dissemination of mycobacterial infection. Subsequent investigations using this model have elucidated the role of vascular supply on granuloma structure and bactericidal function 89 and the role of host-derived inflammatory lipid mediators in promoting disease severity. Using the zebrafish model, it was shown that coordinated expression of mycobacterial virulence lipids, such as PDIM, directs the phagocytic uptake of mycobacteria while simultaneously modulating the capacity of phagocytic cells to kill mycobacteria. 13 An elegant study by Cronan et al 30 elucidated the role of the macrophage cell-cell adhesion molecule E-cadherin in promoting epithelioid transformation of macrophages. Zebrafish with defective E-cadherin had disorganized granulomas and were better able to limit bacterial growth. This result correlated with increased immune cell access to the interior of the granuloma. Collectively, data generated using the zebrafish model for TB pathogenesis research have challenged the notion that granulomas are solely protective during the host immune response to mycobacterial infection and have helped to generate models for how conditionally essential genes might orchestrate the granulomatous response to promote establishment of mycobacterial infection.
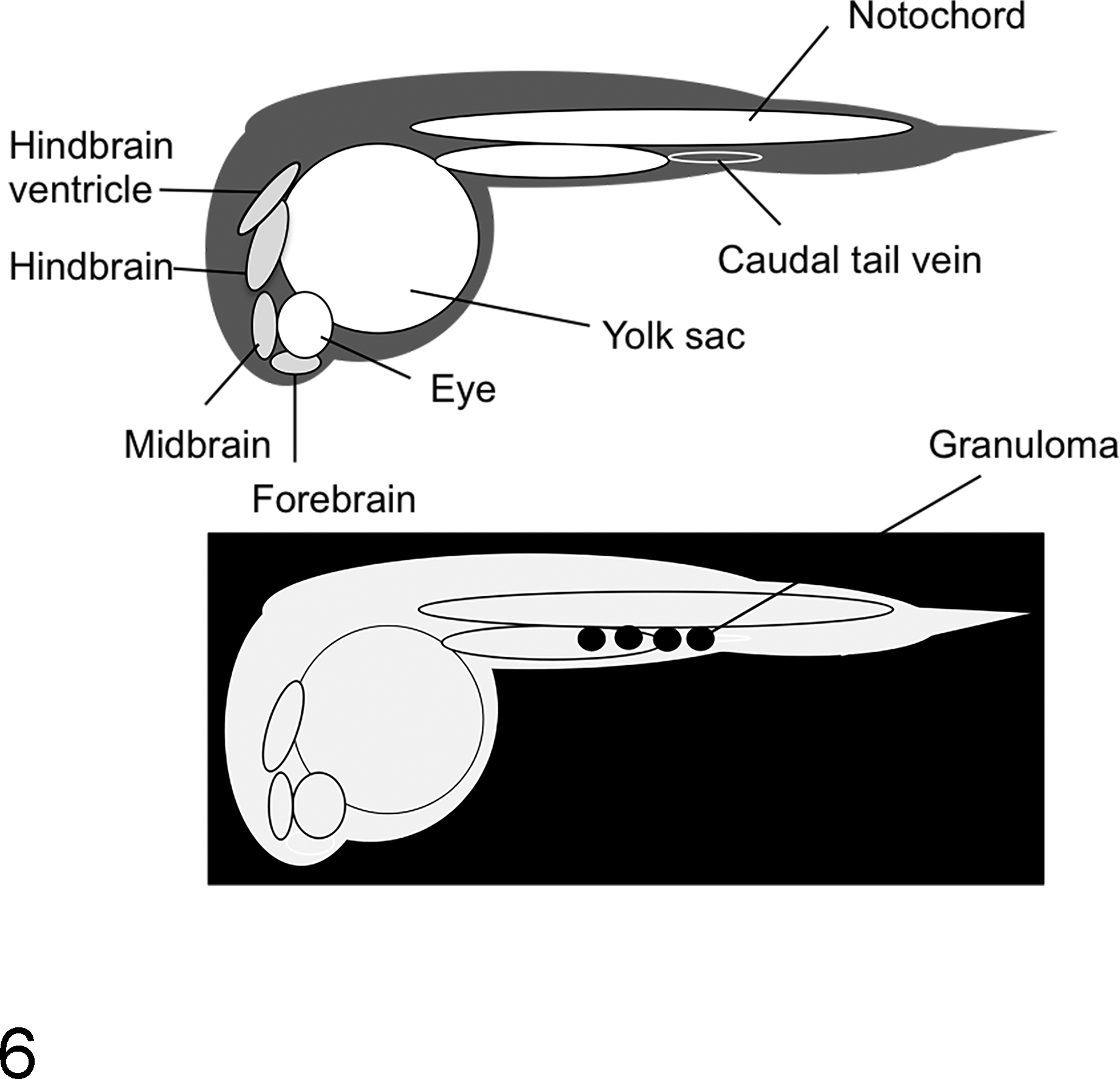
Schematic of experimental Mycobacterium marinum infection in the zebrafish model. (Upper) Anatomical features of 24-hour zebrafish larvae. M. marinum infection is via injection into either the caudal tail vein or hindbrain ventricle. (Lower) Day 3, transparent zebrafish larvae showing foci of macrophage aggregation or “granuloma” (black circles) after infection with M. marinum.
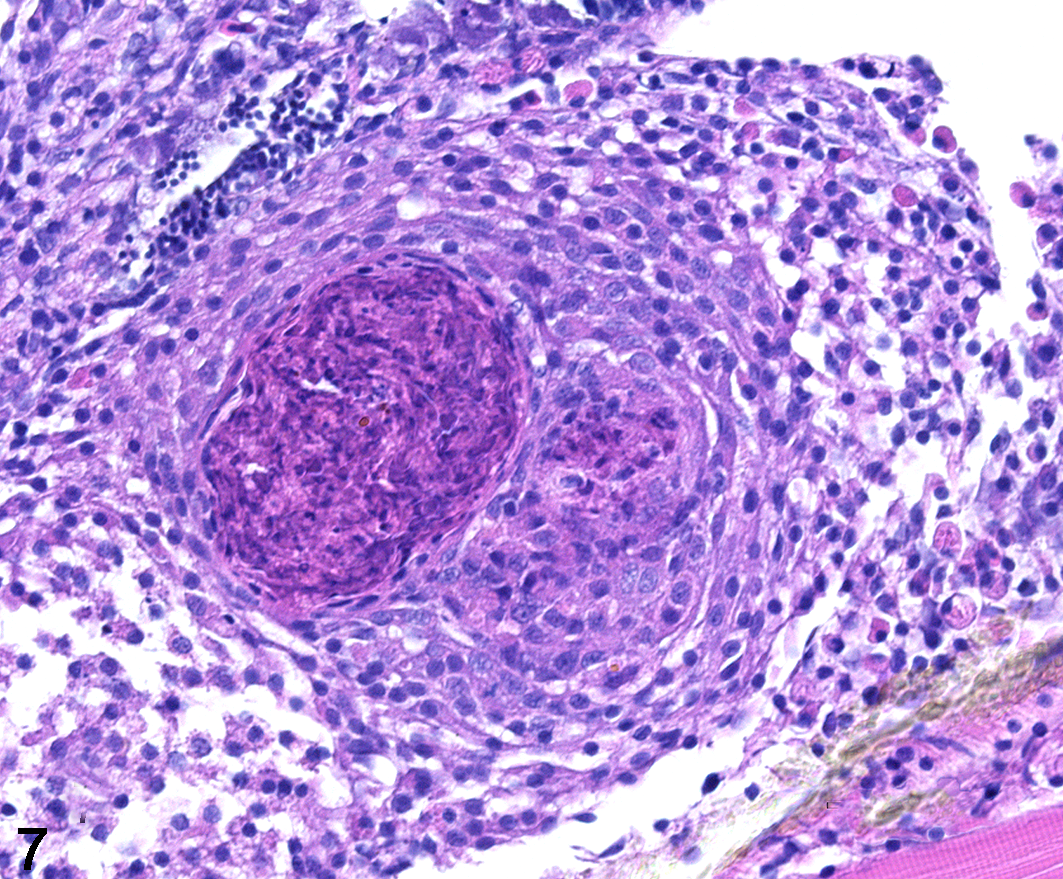
Mycobacterium marinum experimental infection, testis, adult zebrafish. A solitary granuloma within the testis is characterized by a core of acellular, necrotic caseum surrounded by a rim of epithelioid macrophages. (Image courtesy of Dr Mark Cronan, Duke University)
Gaining Control of the Ball in the TB Granuloma Game—What the NHP Model Has Taught Us
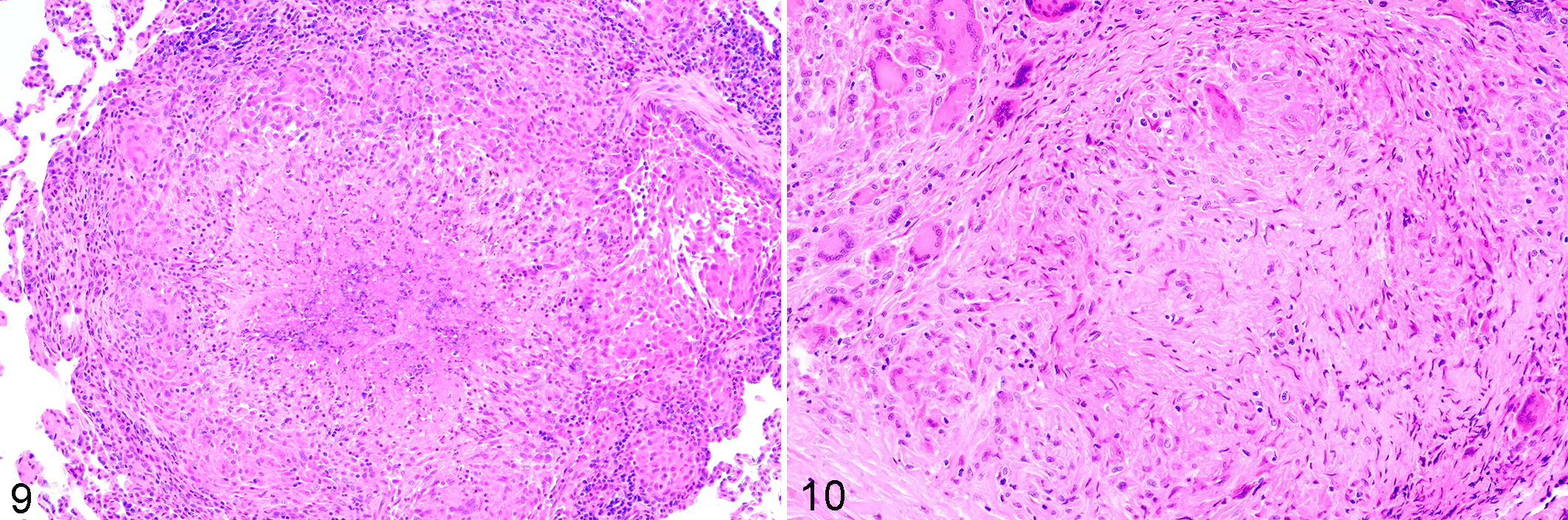
Everything discussed thus far has highlighted how, indeed, it does seem that mycobacteria may have the upper hand in the granuloma game. But one of the biggest breakthroughs in our understanding of mycobacterial immunopathogenesis is the result of the NHP model of Mtb infection. 42,95 It is becoming increasingly evident that the clinical manifestation of TB disease reflects a summation of heterogeneous pathological responses within a single infected animal or person. The development of the cynomolgus macaque model has shed light on this very important aspect of TB immunopathology. Canonically, individuals are clinically classified as having active vs latent disease. “Latently” infected individuals are thought to make up the majority of the population or 95%, compared with the roughly 5% of actively infected individuals. In the cynomolgus macaque model, low-dose pulmonary infection with Mtb leads to clinically active disease in approximately 40% of animals and latent disease in 60% of animals. Latently infected animals have culture-negative bronchoalveolar lavages and gastric aspirates and no radiographic evidence of pulmonary disease (Fig. 8). Use of this model has led to the understanding that individual lesions within the same host can differ in their pathologic trajectory. In a clinically defined “active” Mtb-infected macaque, some lesions will have replicating mycobacteria, high bacterial burden, and necrosis within granulomas (Fig. 9). Other lesions in the same animal will have few mycobacteria, fibrosis, and resolution of pathology (Fig. 10). This is also evident from metabolic labeling of granulomas. Within a single actively or latently infected NHPs, both metabolically active (hot) and metabolically inactive (cold) granulomas can be detected using 18F-fluorodeoxyglucose positron emission tomography coupled with computed tomography (FDG/PET CT). 73 This shows that clinical TB disease is a spectrum rather than a dichotomy of “active” vs “latent” infection where the clinical manifestation of disease is a function of the predominant lesion type. “Latently” infected NHPs have more restrictive or sterilized granulomas compared with animals with active disease. Although some heterogeneity in local immune responses within a single host may exist, overall the ability to control Mtb infection within a single person or animal should be consistent. This suggests that the lesion heterogeneity seen in the NHP model is more likely a result of stochastic differences in metabolic state and expression of virulence factors by the mycobacteria within individual lesions.
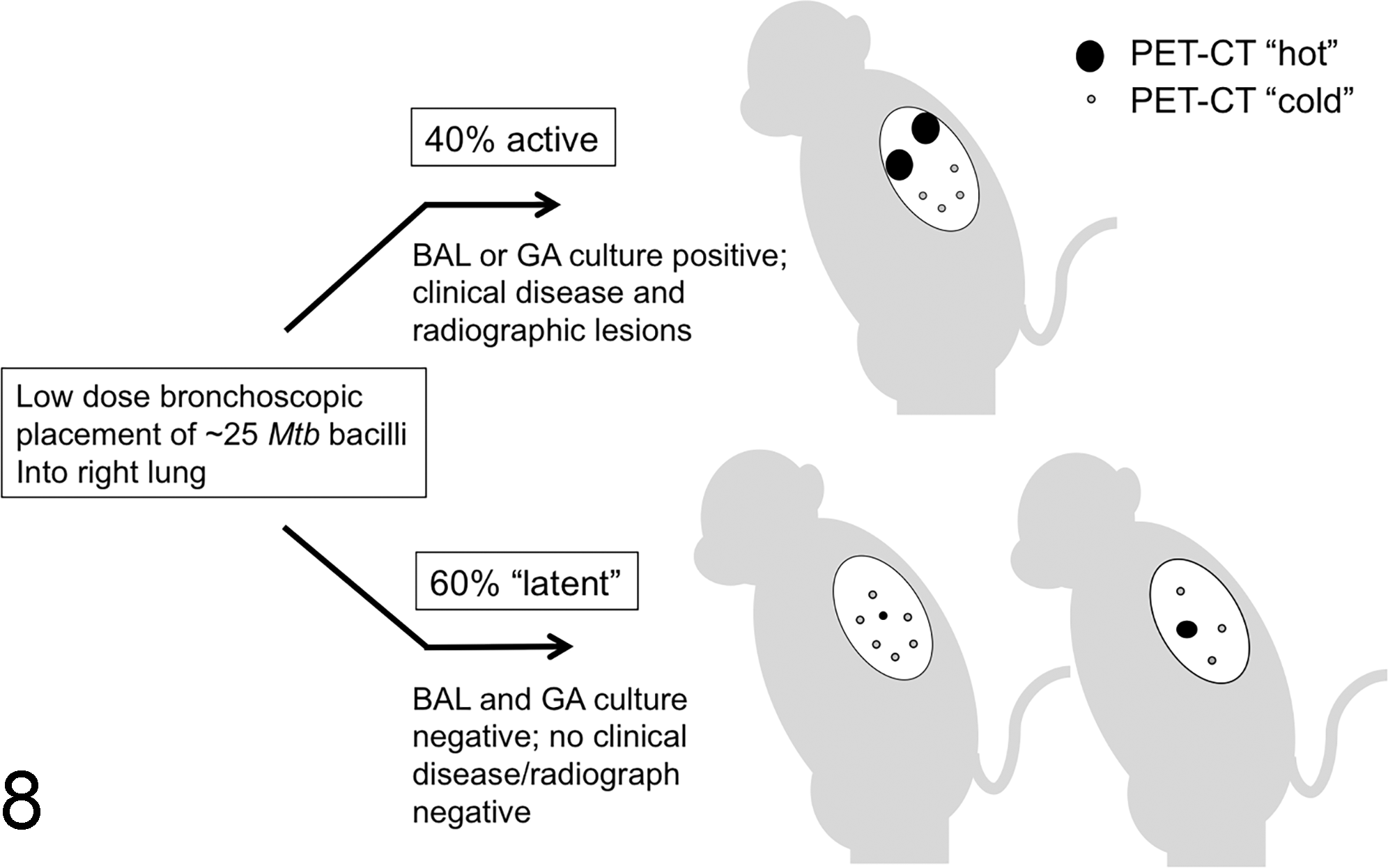
Schematic of cynomolgus macaque model of Mycobacterium tuberculosis (Mtb) infection. Monkeys receive a low-dose inoculum (∼25 colony-forming units) of virulent Mtb Erdman via bronchoscopic placement into the right caudal or right middle lung lobes. Approximately 40% of animals will have bronchoalveolar lavages (BALs) or gastric aspirates (GAs) that are culture positive for Mtb and are classified as having “active” disease. Animals with culture-negative BALs and GAs and no radiographic or clinical disease are considered “latently” infected (approximately 60%). 18F-fluorodeoxyglucose (FDG) positron emission tomography coupled with computed tomography (FDG/PET CT) allows metabolic labeling of granulomas. Animals with “active” TB also have “cold” lesions (white) and animals with “latent” disease also have “hot lesions” (black) indicating that granulomas within a single host can behave differently.
Given how effective Mtb is in orchestrating an immunopathologic response that favors its persistence, how can we shift the odds of the game to favor host response in clearing bacteria? One of the most compelling new concepts in the field of tuberculosis research is that of host-directed therapies (HDTs). This topic has been thoroughly covered in detail elsewhere 63 but in essence involves use of broad classes of therapeutics that target inflammation, metabolism, vascularization, and tissue remodeling to assist the host in depriving Mtb of its preferred niche. Despite the large armamentarium of mycobacterial virulence genes, 90% to 95% of individuals are able to control or sterilize TB granulomas with an adequate host response. Yet, only one active lesion is necessary to initiate the full spectrum of clinical disease in both humans and NHPs. Interventions that shift host-offensive mechanisms to become 99% to 100% effective may completely thwart the effects of bacterial virulence factors, preventing clinical disease and Mtb transmission.
Conclusion
Animal models of tuberculosis are at the forefront of basic science research and product development for countering TB disease worldwide. Scientists and pathologists should continue to challenge interpretations of host response to Mtb infection and investigate whether exuberant innate or adaptive host immune responses are truly beneficial. These considerations are extremely important in the context of vaccine development for human TB. Genetically, mycobacteria have conserved the immunodominant antigens that are incorporated in most of the vaccines that make up the current TB vaccine pipeline. It is likely that host responses to these antigens provide some benefit to the establishment of Mtb infection, to dissemination of TB disease, or the establishment of Mtb latency. Alternatively, shifting the host immune response to subdominant antigens may represent one way to stack the odds in favor of host response against this pathogen. Most important, we need to define correlates of host response and control or elimination of mycobacterial infection to truly understand who is calling the shots in the TB granuloma game. Development of new pharmacological interventions that target both bacterial growth and host response may help improve disease outcomes in the absence of complete sterilization. Regardless of whether this is accomplished through host-directed therapies, vaccines, or other antimicrobials, the result in terms of global health betterment could be profound given that one-third of the world’s population, or 2 billion individuals, are latently infected with Mtb. Expanding our understanding of both host and microbial determinants of granuloma formation and function has tremendous potential to advance the health of both humans and animals alike.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
