Abstract
Nuclear glycogen inclusions occur infrequently in pathologic conditions but also in normal human and animal tissues. Their function or significance is unclear. To the best of the authors’ knowledge, no reports of nuclear glycogen inclusions in canine parietal cells exist. After initial observations of nuclear inclusions/pseudoinclusions during routine histopathology, the authors retrospectively examined samples of gastric mucosa from dogs presenting with gastrointestinal signs for the presence of intranuclear inclusions/pseudoinclusions and determined their composition using histologic and electron-microscopic methods. In 24 of 108 cases (22%), the authors observed various numbers of intranuclear inclusions/pseudoinclusions within scattered parietal cells. Nuclei were characterized by marked karyomegaly and chromatin margination around a central optically empty or slightly eosinophilic area. The intranuclear inclusions/pseudoinclusions stained positive with periodic acid–Schiff (PAS) and were diastase sensitive, consistent with glycogen. Several PAS-positive/diastase-sensitive sections were further examined by transmission electron microscopy, also using periodic acid-thiocarbohydrazide-silver proteinate (PA-TCH-SP) staining to identify polysaccharides. Ultrastructurally, the nuclear inclusions were composed of electron-dense particles that were not membrane bound, without evidence of nuclear membrane invaginations or cytoplasmic organelles in the nuclei, and positive staining with PA-TCH-SP, confirming a glycogen composition. No cytoplasmic glycogen deposits were observed, suggesting that the intranuclear glycogen inclusions were probably synthesized in loco. Nuclear glycogen inclusions were not associated with gastritis or colonization by Helicobacter-like organisms (P > .05). Our findings suggest that nuclear glycogen inclusions in canine parietal cells could be an incidental finding. Nevertheless, since nuclear glycogen is present in several pathologic conditions, further investigations could be warranted to determine their true significance.
Intranuclear glycogen inclusions/pseudoinclusions are an uncommon histologic finding, first observed by Ehrlich in 1883. 10 In most reports, investigators have observed the inclusions/pseudoinclusions in hepatocytes, secondary to several hepatic disorders or metabolic diseases, 2,21,23,25,32 although some investigators have observed them in diseased cardiac cells, 11 in gastric tubular adenocarcinoma, 25 in human infantile type carcinoma of the pancreas, in human rhabdomyosarcoma, 13 as an incidental finding in apparently healthy humans, 7 and finally, as a normal feature in periportal hepatocytes during childhood and adolescence. 20,21
Similar to observations in humans, intranuclear glycogen inclusions/pseudoinclusions in animals occur mostly in hepatocytes and are associated with both aging and hepatic disorders. 2,4,17,26,31 However, as in humans, intranuclear glycogen inclusions/pseudoinclusions in animals have been observed also in extrahepatic tissues, such as in kidneys of diabetic Chinese hamsters (Cricetulus griseus), 6 in mast cell tumors of dogs, 32 and in chemically induced transplantable chicken sarcoma cells. 12 Parallel with observations in humans, intranuclear glycogen inclusions/pseudoinclusions occur as incidental findings in metamorphosing tadpoles and aged mice. 12,17
The significance of these findings is still unknown. Some authors have stated that intranuclear glycogen inclusions/pseudoinclusions do not appear to disrupt cell integrity 11,17 ; besides, the biochemical mechanisms leading to glycogen accumulation within nuclei as well as its biological function are unclear. Furthermore, despite the fact that they have been known for over a century, to the best of the authors’ knowledge, they have never been reported in canine gastric mucosa.
We have recently observed nuclear changes, morphologically consistent with intranuclear inclusions/pseudoinclusions, in canine parietal cells during routine histopathologic evaluation of gastric biopsies: parietal cell nuclei were enlarged with margination of chromatin and a central area optically empty or slightly eosinophilic with sharp contours.
Consequently, we sought to further characterize these inclusions/pseudoinclusions by comprehensive histologic and ultrastructural investigation. In addition, we also have investigated the association between the Helicobacter-like organisms morphologically observed in the samples and the presence of histologic signs of gastritis.
Materials and Methods
Selection of Samples
We examined archived gastric biopsies from the Department of Veterinary Medicine of the University of Perugia (Italy), obtained from dogs presenting to the clinicians for investigation of gastrointestinal disease between 2011 and 2014. We graded these samples as adequate, marginal, or inadequate, according to the criteria suggested by Willard et al. 35 We excluded samples that we considered inadequate and dogs with a diagnosis of gastric neoplasia, since we had only a few neoplastic cases and we wished to focus attention on the inflammatory process. Age, breed, and sex were not criteria for inclusion. The samples considered marginal were used only for estimates of dichotomous outcomes (presence/absence of gastric Helicobacter-like organisms, presence/absence of nuclear inclusions/pseudoinclusions in parietal cells, presence/absence of gastritis) but were not used for morphologic descriptions.
Histology
All samples were formalin-fixed, paraffin-embedded, routinely processed, stained with hematoxylin-eosin (HE), and observed by light microscopy. Because of the lack of accepted histopathologic guidelines for the assessment of gastric biopsies, the slides were re-evaluated by the same boarded veterinary pathologist to eliminate subjective variability. The whole samples were evaluated for 4 features: inflammatory infiltration (lymphocytes, plasma cells, eosinophils, neutrophils), structural changes (atrophy, fibrosis, mucosal hypertrophy, follicular hyperplasia) according to the World Small Animal Veterinary Association [WSAVA] scheme, 8 presence of gastric Helicobacter-like organisms (GHLO) according to Happonen et al, 15 and nuclear inclusions/pseudoinclusions in parietal cells. The severity of this last finding was evaluated according to the principles of the Sydney System (the system for grading human gastrointestinal diseases) as follows: “It was recommended these parameters were to be semi-quantitatively graded as absent, mild, moderate or severe, each successive grade to represent an increase in severity of approximately one third.” 30
The samples with nuclear inclusions/pseudoinclusions in parietal cells, as well as an equal number of samples without histologic lesions, used as control cases, were stained with periodic acid-Schiff (PAS) reaction; all of the PAS-positive samples were subsequently stained with PAS after diastase pretreatment to test the diastase sensitivity. The samples with nuclear inclusions/pseudoinclusions as well as the samples used as control cases belonged to the study population; therefore, they all were gastric biopsies.
Electron Microscopy
Three formalin-fixed paraffin-embedded samples were submitted for transmission electron microscopy (TEM): 2 samples with PAS-positive/diastase-sensitive parietal cell nuclear material and 1 sample devoid of histologic lesions, which served as the control. Furthermore, 2 formalin-fixed paraffin-embedded samples with PAS-positive/diastase-resistant nuclear inclusions were examined to verify this unexpected result.
For TEM examination, the same paraffin blocks previously used for light microscopy were cut into smaller fragments and reprocessed, as previously described. 22 Semi-thin sections were stained with methylene blue and Azur II for light microscopy. Ultra-thin sections, stained with uranyl acetate followed by lead citrate, were examined under a transmission electron microscope (Philips EM 208, equipped with a digital camera).
Furthermore, ultra-thin sections of the positive and control samples were used for the periodic acid-thiocarbohydrazide-silver proteinate (PA-TCH-SP) technique for complex carbohydrate staining. 33 Sections were placed on nickel grids for electron microscopy and incubated in the following solutions: periodic acid 1% (45 minutes); washing in distilled water (5 × 10 minutes); thiocarbohydrazide 2% in acetic acid 20% (40 minutes); acetic acid 10% (10 minutes); washing in distilled water (10 minutes); silver proteinate 1% (30 minutes); and washing in distilled water before imaging.
Statistical Analysis
We first calculated absolute and relative frequencies of histologic lesions, GHLO, and parietal cell nuclear inclusions/pseudoinclusions. For these analyses, we evaluated only “adequate” samples (n = 96) to maintain a high level of accuracy. 35 We then estimated the association between nuclear inclusions/pseudoinclusions and gastritis with a chi-square test of independence, using all 108 samples. Finally, we estimated the association between GHLO colonization and the presence of nuclear inclusions/pseudoinclusions with a Fisher exact test, also using all 108 samples.
Data were analyzed using the free software R (R version 3.2.1, R commander package); P values ≤ .05 were considered statistically significant.
Results
Study Population and Histologic Evaluation
This study included 108 samples obtained from 107 dogs (in 1 dog, a second, post-therapeutic biopsy was available), but signalment data were incomplete: age was available for only 97 dogs, whereas sex and breed were available for 100 dogs. There was a wide age distribution, with a median age of 4 years (range = 6 months to 14 years); more dogs were male (61/100) and the most frequent breeds were mixed-breed dogs (26/100), German Shepherds (10/100), and Rottweilers (8/100) (Supplemental Table 1).
Of the 108 initially identified samples, only 96 were adequate (89%) and 71% (68/96) showed histologic signs of gastritis, whereas 29% (28/96) had no lesions. Lymphoplasmacytic infiltrates were the most commonly observed histologic finding (63%, 61/96); other findings are summarized in Table 1. Gastric Helicobacter-like organisms were present on the luminal surface of 23% of samples (25/108): in 70% of the adequate samples with GHLO (14/20), they also colonized gastric pits, extending to gastric glandular lumen in 15% of the samples (3/20) (Supplemental Table 2).
Frequencies of Histologic Findings.
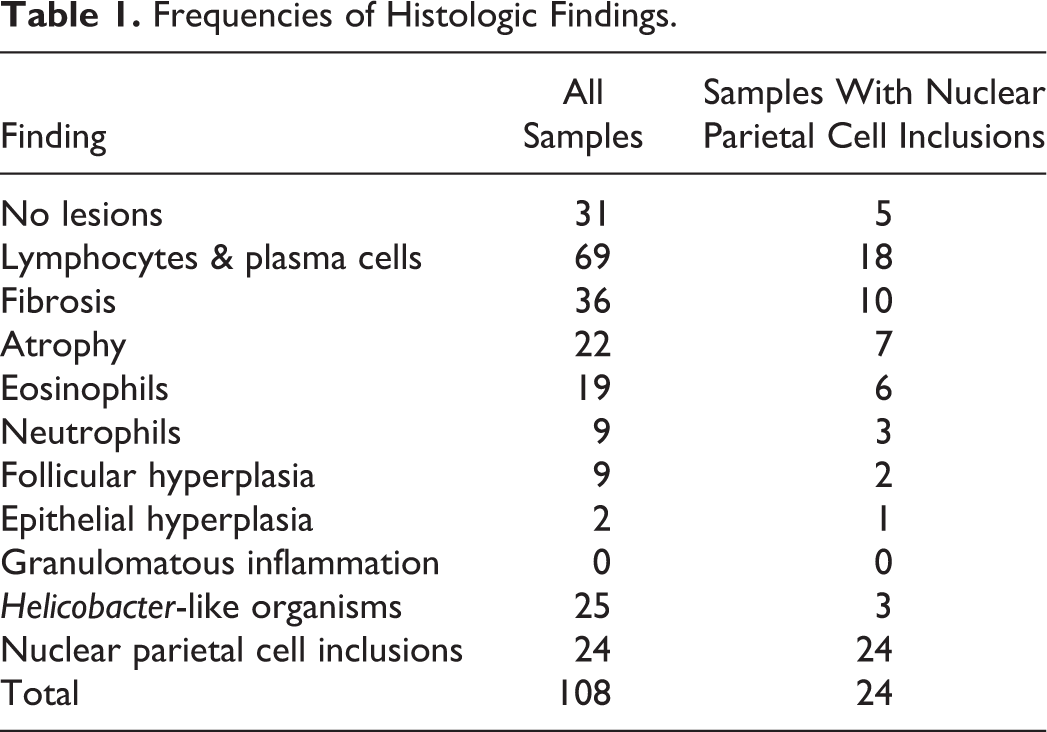
Nuclear Inclusions/Pseudoinclusions: Histology
We detected nuclear inclusions/pseudoinclusions in 22% of the samples (24/108) within scattered parietal cells, ranging from occasional (0–1/hpf) to numerous (4–5/hpf). Nuclei were characterized by marked karyomegaly and chromatin margination around a central optically empty or slightly eosinophilic area (Fig. 1). Sex and age were available for 21 dogs and the breed for 19 dogs, males and females were equally represented, the median age was 4 years (range = 11 months to 12 years), and the main represented breeds were mixed-breed dogs (26%, 5/19), followed by Cavalier King Charles Spaniels (11%, 2/19) (Supplemental Tables 1, 2). Only 19 dogs provided adequate samples (20%, 19/96). In adequate samples, we have diagnosed gastritis in 74% (14/19) of dogs with nuclear inclusions/pseudoinclusions, whereas 26% (5/19) appeared normal, similar to the proportion of gastritis in the entire sample population. Gastritis was mild in 64% (9/14) of the samples, moderate in 29% (4/14), and severe in 1 sample. All gastritis samples showed lymphoplasmacytic infiltrates; other histologic findings are summarized in Table 1. Only 13% (3/24) of samples with nuclear inclusions/pseudoinclusions had GHLO. We found no statistically significant association between the presence of nuclear inclusions/pseudoinclusions and gastritis (P = .33) or the presence of GHLO (P = .27).
Nuclear inclusions in parietal cells, corpus stomach, dog.
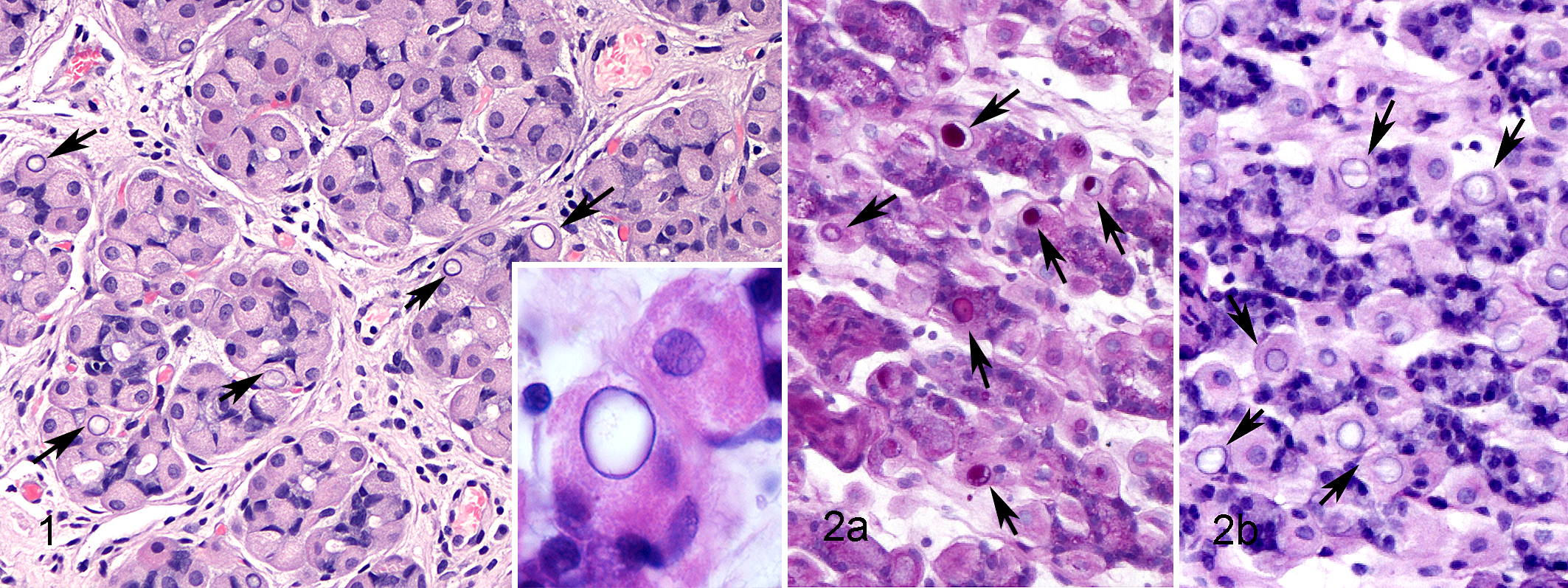
We stained only 68% (13/19) of available adequate samples containing nuclear inclusions/pseudoinclusions within parietal cells with PAS because of the lack of enough material to obtain new sections in 6 samples (Supplemental Table 3): parietal cell nuclei in 77% (10/19) of the samples were PAS positive (Fig. 2a), varying from mild to marked, but did not stain with PAS in 23% (3/19) of the samples. All of the parietal cell nuclei in the control samples were PAS negative. All PAS-positive samples were stained using the PAS/diastase method, and for 50% (5/10) of sections, PAS staining of inclusions was eliminated with diastase pretreatment, indicative of diastase sensitivity (Fig. 2b).
Nuclear Inclusions/Pseudoinclusions: Electron Microscopy
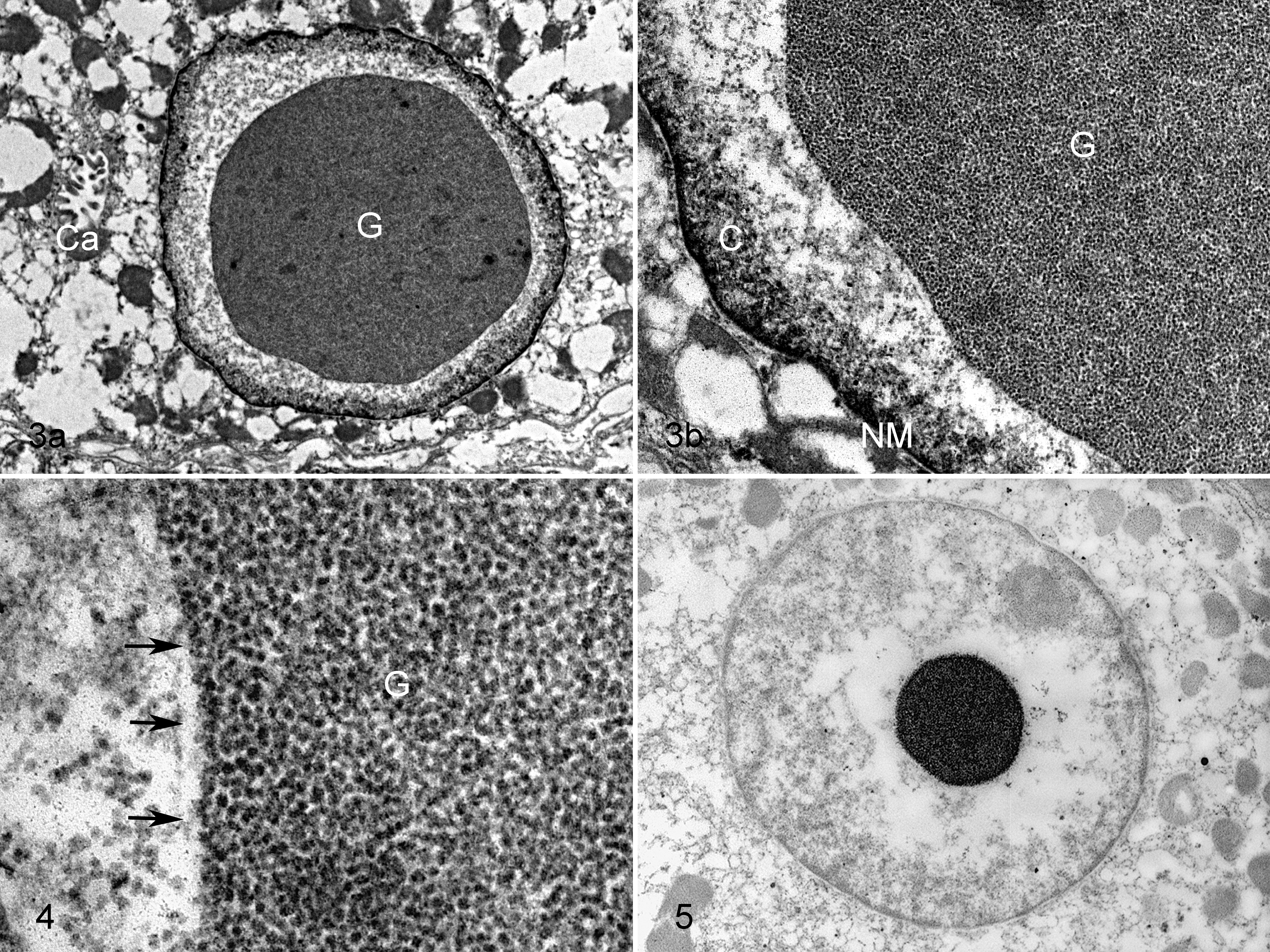
We used 3 formalin-fixed paraffin-embedded samples for TEM investigation: although these reprocessing techniques could affect the quality of the ultrastructural features (vacuolations, disruption of membranes, tissue degradation), 5,22 with an optimal fixation in 10% neutral buffered formalin, the preservation of tissues is reported to be similar to that obtained with glutaraldehyde. 22 Our samples were rather well preserved, without fixation artifacts, and we evaluated them adequate for ultrastructural evaluation. In PAS-positive/diastase-sensitive samples, TEM showed the presence of a central, round to ovoid to irregularly shaped, moderately to intensely electron-dense deposit with sharp margins within parietal cell nuclei (Fig. 3a). The deposits were composed of small particles (range = 9–20 nm), consistent with plumbophilic glycogen β-particles, that formed single masses, sometimes strictly close to the marginated chromatin but, in most instances, surrounded by an electron-lucent halo of nuclear matrix with chromatin at the periphery (Fig. 3b); occasionally, small groups or solitary monoparticulate glycogen were seen near the widest deposit. We did not find evidence of nuclear membrane invaginations or cytoplasmic organelles within the nuclear contour; furthermore, the nuclear glycogen was not membrane bound, even though particles tended to line up at the marginal area of the main deposit (Fig. 4).
Nuclear inclusions in parietal cells, corpus stomach, dog.
In the sample without histologic lesions, used as control and examined by TEM, we did not see glycogen particles either in the nuclei or in the cytoplasm of parietal cells.
Subsequently, we processed these 3 samples using the PA-TCH-SP technique (Thiéry method), a method analogous to PAS staining for light microscopy. The inclusions within parietal cell nuclei, previously morphologically identified as glycogen particles, showed positive reactivity, confirming the polysaccharide composition of inclusions (Fig. 5). We did not observe any PA-TCH-SP reactivity within the cytoplasm of these parietal cells.
In the negative control sample, we did not see PA-TCH-SP reactivity either in the nuclei or in the cytoplasm of parietal cells.
Since 50% of our samples were diastase resistant, we have performed TEM evaluation using 2 of them, in order to verify whether the ultrastructure of the deposits was consistent with the diastase-sensitive samples. Ultrastructurally, the PAS-positive/diastase-resistant samples have shown similar features compared with PAS-positive/diastase-sensitive samples.
Discussion
Our retrospective study of canine gastric mucosa demonstrates, for the first time, the presence of intranuclear glycogen inclusions within parietal cells of dogs investigated for gastrointestinal disease with and without gastric histologic lesions. The inclusions do not appear to be associated with gastritis or gastric Helicobacter-like organisms.
Nuclear glycogen storage is an infrequent finding, described in several pathologic conditions and occasionally in normal subjects. The biochemical mechanism leading to glycogen accumulation within nuclei, as well as its biological function, is unclear. Some researchers stated that glycogen particles could be translocated inside the nucleus from the cytoplasm throughout nuclear pores, 29,32 but no experimental evidence supports this theory. 2,13 Another possibility is the presence of a pseudoinclusion: in this case, the nuclear membrane forms invaginations, entrapping a small amount of cytoplasm and its components (glycogen, mitochondria, ribosomes), 2 such as in chronic pyrrolizidine alkaloid poisoning. 1,31 Recently, Bennett et al 2 reported nuclear inclusions of glycogen in liver of western barred bandicoot with papillomatosis and carcinomatosis syndrome in which there was no evidence of nuclear membrane invaginations or cytoplasmic organelles. Finally, glycogen can be synthesized in interchromatin regions of the nucleus 2,4,14,25 by glycogen synthase, a key enzyme of glycogen synthesis. 3,24,28 Some authors have demonstrated nuclear enzyme activity in neoplastic cells, suggesting that other mammalian cells with nuclear glycogen storage also could have nuclear glycogen synthase. 14,18
We detected nuclear inclusions in parietal cells of 22% of dogs: most of these inclusions were PAS positive and some were diastase sensitive, typical of glycogen. However, 23% of the samples with inclusions were PAS negative. The negativity to PAS staining might be due to the loss of glycogen during formalin fixation and processing, 2 as we used archival samples without knowledge of the time of fixation. Bennett et al 2 reported 8% of negativity to PAS staining in their archival samples: this difference might be due both to the variability of the time of fixation and to the different method of evaluation of PAS-positive percentage, since we have calculated the number of cases, and they have calculated the number of nuclear inclusions. Furthermore, only 50% of our samples exhibited diastase sensitivity: despite these unexpected histochemical observations, we confirmed the nature of nuclear glycogen inclusions with TEM evaluation, including a processing technique used specifically to detect polysaccharides. Moreover, since TEM has shown that the material in the diastase-resistant nuclei has similar ultrastructural features with respect to the diastase-sensitive samples, we argue that this result was likely a consequence of an unexpected event that occurred during technical procedures. Similar to observations by Bennett et al, 2 the glycogen in canine parietal cells was not membrane bound and the nuclei lacked membrane invaginations and cytoplasmic organelles. Furthermore, TEM showed a lack of cytoplasmic glycogen in cells with nuclear glycogen inclusions, suggesting that the translocation of cytoplasmic glycogen into the nucleus through nuclear pores, as suggested by some authors, is unlikely. 29,32 Based on these findings, we can suspect that the PAS-positive/diastase-sensitive material in parietal cell nuclei is a real nuclear inclusion of glycogen, most likely synthesized in loco by nuclear glycogen synthase. 2,4,14,18,25
The main represented breeds with nuclear inclusions in our sample population were mixed-breed dogs and Cavalier King Charles Spaniels; it is surprising that no German Shepherds showed nuclear inclusions, despite being one of the most frequent breeds in our study. However, we had a small number of positive cases and a large variety of breeds, making any assessments of breed association impossible.
The proportion of samples with nuclear glycogen inclusions of parietal cells and gastritis (73%) did not differ from that without inclusions (71%); therefore, we can hypothesize that these glycogen accumulations are related to functional modifications not associated with the inflammation.
Similarly, we found GHLO in a small proportion of samples with nuclear glycogen inclusions, which did not differ from the prevalence of GHLO in the entire study population. This is not surprising, since degenerative lesions are reported to be rare even in parietal cells with intracytoplasmic GHLO. 19 In humans, Helicobacter pylori is considered an invasive and facultative intracellular micro-organism, and this characteristic can affect its pathogenicity. 9
Our retrospective study has some limitations. First, because of the invasive nature of endoscopy, only dogs with evidence of gastrointestinal disease undergoing a routine diagnostic evaluation were available, without healthy animals as controls. Nevertheless, in a wide study on incidental findings in healthy laboratory Beagles, Sato et al 27 did not report a similar finding among the 155 typical findings in any of the 1188 dogs examined; it seems unlikely that the finding we observed in 22% of unhealthy dogs had never been observed in such a large number of healthy dogs.
Second, complete clinical details that would have been useful to investigate a possible activation of cell metabolism induced by, among others, gastrointestinal active peptides (e.g., gastrin) were not available. This is a common issue with retrospective studies that use archived materials. Furthermore, we excluded samples with gastric neoplasia. Nuclear glycogen inclusions have been described in the cells of several tumors (human Ehrlich ascites tumor, human glycogen-rich hepatocellular carcinoma, human gastric tubular adenocarcinoma, human infantile type carcinoma of the pancreas, human rhabdomyosarcoma, chemically induced transplantable chicken sarcoma, canine mast cell tumors). 7,12,13,25,34 Although their role is unclear, the presence of glycogen particles could suggest the activation of nuclear glycogen synthase and synthetic pathways in tumors, because glycogen seems to be involved in the formation of a higher order chromatin intermediate. 16 Finally, due to insufficient samples, techniques with higher sensitivity for the detection of GHLO were not attempted, potentially underestimating the incidence of GHLO in this study.
We have demonstrated that optically empty areas observed in the nuclei of parietal cells of dogs are glycogen nuclear inclusions. The intranuclear inclusions occur in the gastric parietal cells in approximately 20% of the biopsy samples from dogs evaluated for possible gastrointestinal signs and show no association with inflammation or GHLO colonization. However, because of the lack of a control group, we can draw no definitive conclusions about the biological or pathophysiological significance of this finding that are worthy of further investigation.
Footnotes
Acknowledgements
We are grateful to Prof. Mark Rishniw for his invaluable assistance in reviewing the manuscript. Finally, we thank Luca Stefanelli and Paola Coliolo for technical support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
