Abstract
Wood frogs (Rana sylvatica) are highly susceptible to infection with Frog virus 3 (FV3, Ranavirus, Iridoviridae), a cause of mass mortality in wild populations. To elucidate the pathogenesis of FV3 infection in wood frogs, 40 wild-caught adults were acclimated to captivity, inoculated orally with a fatal dose of 104.43 pfu/frog, and euthanized at 0.25, 0.5, 1, 2, 4, 9, and 14 days postinfection (dpi). Mild lesions occurred sporadically in the skin (petechiae) and bone marrow (necrosis) during the first 2 dpi. Severe lesions occurred 1 to 2 weeks postinfection and consisted of necrosis of medullary and extramedullary hematopoietic tissue, lymphoid tissue in spleen and throughout the body, and epithelium of skin, mucosae, and renal tubules. Viral DNA was first detected (polymerase chain reaction) in liver at 4 dpi; by dpi 9 and 14, all viscera tested (liver, kidney, and spleen), skin, and feces were positive. Immunohistochemistry (IHC) first detected viral antigen in small areas devoid of histologic lesions in the oral mucosa, lung, and colon at 4 dpi; by 9 and 14 dpi, IHC labeling of viral antigen associated with necrosis was found in multiple tissues. Based on IHC staining intensity and lesion severity, the skin, oral, and gastrointestinal epithelium and renal tubular epithelium were important sites of viral replication and shedding, suggesting that direct contact (skin) and fecal-oral contamination are effective routes of transmission and that skin tissue, oral, and cloacal swabs may be appropriate antemortem diagnostic samples in late stages of disease (>1 week postinfection) but poor samples to detect infection in clinically healthy frogs.
Keywords
The Iridoviridae is a family of large double-stranded DNA viruses that infect insects (Iridovirus and Chloriridovirus genera) and poikilothermic vertebrates (Lymphocystivirus and Ranavirus genera). 6 Ranavirosis is a recognized emerging disease 6 and, since 2008, one of only 2 amphibian pathogens reportable to the World Organization for Animal Health. 34 Frog virus 3 (FV3), the type species of the genus Ranavirus, and FV3-like viruses have been responsible for epizootics in fish, amphibians, and reptiles. 7,22 In North America (United States and Canada), at least 60 species of frogs, toads, and salamanders (orders Anura and Caudata) are known to be susceptible to infection with FV3. Confirmed mortalities due to FV3 infection have been reported in 36 of those species. 26 Collapse in amphibian communities associated with the presence of the common midwife toad virus, a species of Ranavirus present in continental Europe, may be the first indication that ranaviruses can result in population declines and, potentially, extirpations (local extinctions). 28 Mathematical population modeling provides further support for the ability of ranaviruses to cause local extinction: using susceptibility data and demographic parameters to predict the likelihood of local extinction of wood frogs (Rana sylvatica or Lithobates sylvaticus), extirpation has been hypothesized to occur as quickly as 5 years after introduction of Ranavirus sp to an isolated population. 11 Whether ranaviruses have only recently turned into “emerging cold-blooded killers” 6 or we simply failed to recognize their effect in the past is uncertain. A recent emergence would seem perhaps more likely, given how they were first isolated from nondiseased frogs in the 1960s 13 and remained orphan viruses for several years afterwards. A report of mortalities in wild tadpoles infected with tadpole edema virus, an icosahedral virus that would later be recognized as a member of the Ranavirus genus, 17,33 and the possibility that at least some “red leg syndrome” mortalities associated with septicemia with Gram-negative bacteria 35 could have been initially caused by ranaviral infection, argue against it. If we are to decipher the emergence of ranaviruses and determine their current and future impact on wild populations, we first need to describe their pathogenesis in an amphibian model species.
Wood frogs, members of the large Ranidae family, are commonly used in the study of Ranavirus sp infection and were proposed as a good experimental animal model by participants at the First International Symposium on Ranaviruses. 22 This species is widespread across North America, reaches the farthest north in Canada, and is sympatric with many other amphibians susceptible to Ranavirus sp infection. Adult wood frogs infected with a lethal dose of FV3 develop the gross and histologic lesions most consistently reported in other amphibians infected with various species of Ranavirus 3,4,9,20,26 : epidermal ulceration, dermal and visceral hemorrhages, and necrosis of the glomeruli, renal tubules, and hematopoietic tissue in various organs. 12
Although the clinical signs, lethal dose, and terminal lesions developed by adult wood frogs infected with FV3 have been described, 12 the pathogenesis of the infection remains unknown, making it difficult to establish sampling recommendations that result in effective surveillance protocols. Research on aquatic African clawed frogs, Xenopus laevis, has partially elucidated the pathogenesis of Ranavirus sp infection in that species. A time-series experiment exposing adult African clawed frogs to a nonlethal dose of FV3 via a water bath found that viral DNA load (estimated by quantitative polymerase chain reaction [PCR]) was most abundant in intestinal tissue as soon as 3 hours postexposure, suggesting that ingestion was the main route of infection. 30 The virus was also detected in blood, skin, liver, and kidney. Evidence of active viral transcription (by reverse transcriptase PCR) was most consistently detected in the intestine and kidney. The authors concluded that the intestine acts as a route of infection and transmission and that the kidney is a main site of replication of FV3 in African clawed frogs. 30 Because lesions due to different Ranavirus infections may vary depending on the specific pathogen and the host species, 18 further research must be conducted before extrapolating the findings in African clawed frogs to native North American species, such as the wood frog. The objective of this study was to describe the pathogenesis of fatal FV3 infection in adult wood frogs by sequential collection and analysis of samples, using a previously established infection model for FV3 in adult wood frogs. 12
Materials and Methods
Capture, transport, husbandry, and experimental procedures followed recommendations of the Canadian Council for Animal Care 5 and were approved by the Animal Care Committee of the University of Prince Edward Island (Protocol 12-014, file 6004702).
Origin and Husbandry of Experimental Frogs
Wood frog adults were collected from an urban vernal pool (n = 56) and a rural pond (n = 3) in Prince Edward Island, Canada (May 17 and 22, 2012). The frogs were housed individually or in small groups in polycarbonate tanks with shallow water dishes on bleach-free paper towel (Suppl. Figs. S1, S2) and fed a combination of crickets (Acheta domesticus), mealworms (Tenebrio molitor), and earthworms (Lumbricidae sp). The temperature and humidity of the room where the animals were housed before and during the experiment were recorded daily, both as the value recorded immediately after entering the room in the morning and as the minimum and maximum values of the previous 24 hours. The frogs were maintained at an average of 20°C to 22°C, with a minimum-maximum range of 18°C to 23°C. Humidity varied substantially and reflected the seasonal ambient conditions (average minimum-maximum of 46%–61% during acclimation and 28%–44% during the experiment).
All mortalities that occurred during the 6.5-month acclimation prior to the experiment were examined grossly and histologically to determine cause of death, with a particular emphasis on any lesions suggestive of a ranavirus infection since there is no reliable method to detect subclinical infection in live animals. As none of the mortalities had any histological evidence of a ranaviral infection, we assumed that our wild-caught adults were free of the virus. After acclimation, 40 frogs were randomly assigned to 1 of 7 infection groups (n = 33 frogs) or served as uninfected controls (n = 7 frogs) and were moved to individual tanks (1 frog per tank).
FV3 Culture
The viral strain used in this study originated from the 1965 FV3 isolated from a Northern leopard frog, Rana [Lithobates] pipiens. 13 The isolate had been cultivated in vitro by various researchers in a variety of cells: fathead minnow cells (at least 15–25 passages; G. Chinchar, personal communication), Xenopus A6 cells, and baby hamster kidney fibroblasts (1–2 and 2 passages, respectively; J. Robert personal communication, 2012). The viral stock used in this study was propagated by the authors in epithelioma papulosum cyprini cells (2 passages) at room temperature (18°C–20°C) in minimal essential medium (MEM) supplemented with 2% fetal bovine serum and 1% antibiotic antimycotic (Invitrogen, Burlington, Canada). Titration of viral stock was performed in a 6-well plate following standard methods to determine the number of plaque-forming units (pfu). 10
Infection, Termination, and Sample Collection
Three days after being moved to individual tanks, the frogs were randomly assigned to 1 of 7 infection groups and a nonexposed control group. The dose of 104.43 pfu of FV3 and period from infection to euthanasia (end point) of each group were predetermined and ranged from 0.25 to 14 days postinfection (dpi), based on a previously established infection model for kinetics of ranavirus infection in adult wood frogs. 12 This ensured that the trial covered the early and late stage of infection but could be terminated before natural death occurred and thus reduce animal suffering. On infection day, each frog was inoculated orally with 104.43 pfu of FV3 13 contained in 0.25 ml of MEM (see above) using a graded pipette (Suppl. Fig. S3). Uninfected control frogs (1 per group) received the same volume of MEM. Frogs were visually inspected 2 or 3 times daily to detect and record clinical signs; checks lasted from 1 to 2 hours and were performed through the transparent wall of their enclosure except when the frogs were not readily visible (hiding under paper towel or cardboard tube), and it was necessary to open the lid and search for them. Crickets and mealworms were fed ad libitum; every other day the consumption of prey was recorded and any dead insects were removed; water was added as required and water dishes were changed every other day or sooner if they contained feces, skin sheds, or dead insects. All handling started with the controls and continued through the infection groups; equipment (ie, plastic gloves, metal forceps) was disinfected with sodium hypochlorite (5% bleach solution, containing 5000 ppm of chlorine) after handling each frog or enclosure.
The frogs were euthanized at the predetermined end point for their corresponding infection group: 0.25, 0.5, 1, 2, 4, 9, or 14 dpi. The exception was 1 frog that had to be euthanized 13 dpi because it showed clinical signs of serious illness compatible with terminal FV3 infection, that is, petechial hemorrhages in the ventral skin with a focus of white necrosis (ulcer) in one of the legs and severe depression with loss of normal posture. 12 Euthanasia for all frogs consisted of immersion in a 10% solution of tricaine methanesulfonate (TMS; Syndel Laboratories, Nanaimo, Canada) (Suppl. Fig. S4). A rigorous aseptic technique protocol was followed to avoid cross-contamination during processing of carcasses and tissue samples. Briefly, handling of the frogs for euthanasia and sample collection was restricted to 1 person (M.J.F.), who exchanged gloves between individual frogs. Collection instruments (scalpels, forceps, scissors) and the stainless steel tray where the carcasses were laid were disinfected in 5% bleach for at least 2 minutes between individuals (multiple sets of instruments and trays were used to expedite the process). A clean person (K.M.J.) recorded measurements and handled the specimen collection containers, opening and closing vials and tubes, carefully avoiding touching a carcass, tissue, or swab; if the clean person accidentally touched a sample, she exchanged gloves immediately. Immediately after euthanasia, the snout-vent length was measured and any abnormal gross findings recorded. Weight, being extremely variable due to hydration status, food in the stomach, and bile in the gallbladder, 35 was intentionally not recorded. During postmortem examination, a cloacal swab and tissue samples of abdominal skin, left liver lobe, and cranial half of left kidney were obtained, frozen at –80°C and later tested for ranavirus DNA by PCR (Pisces Molecular, Boulder, CO). The spleen of 1 frog in each infection group and of 4 control frogs was similarly obtained and preserved for PCR testing. The rest of the carcass and internal organs were preserved in 10% buffered formalin and, within 2 weeks of collection, trimmed and processed routinely for histological examination. Tissues examined were stained with hematoxylin-eosin and included 1 forefoot, 1 hindfoot, a medial section of the head and jaw, a mid-shaft cross section of the thigh, and sections of the heart, lungs, abdominal fat body, liver (2 sections), kidneys, urinary bladder, stomach, intestine, colon, reproductive organs (ovary and oviduct or testicles), and spleen (except in those frogs where spleen was collected for PCR testing). Sections that included bone (feet, head, jaw, and thigh) were trimmed together and decalcified in a commercial solution of formic acid, methyl alcohol, formaldehyde, and water (Cal-Ex II Fixative/Decalcifier; Fisher Chemical, Ottawa, Canada) for 24 to 36 hours prior to processing. To ensure at least some of the skin would be examined without having gone through decalcification, a slice from the abdomen was trimmed along with other soft tissues.
PCR for Ranavirus (FV3) DNA
Portions of abdominal skin, liver, kidney, and spleen were individually transferred into tissue lysis buffer; total DNA was extracted from all samples using a spin-column DNA purification procedure (Qiagen DNeasy 96, mouse-tail protocol; Qiagen, Valencia, CA); and tissues were tested for the ranavirus major capsid protein (MCP) gene with single-round PCR amplification using primers 4 and 5 (forward: 5′-GACTTGGCCACTTATGAC-3′ and reverse: 5′-GTCTCTGGAGAAGAAGAA-3′; amplicon length = 528 bp), 23 which cover the same region of the MCP gene as the MCP1 assay recommended by the Aquatic Animal Health Code. 24,34 PCR reaction parameters were as follows: 1× GeneAmp Core buffer, 1 U AmpliTaq Gold (Life Technologies, Carlsbad, CA) 2 mM Mg2+, 800 nM dNTPs (New England Biolabs, Beverly, MA), 500 nm each forward and reverse primers, and 2 μL template DNA in a 20-μL reaction. Thermal parameters were as follows: 9 minutes × 94°C, then 35 cycles of [30 seconds × 94°C, 1 minute × 53°C, 1 minute × 72°C], then hold at 15°C. For the cloacal swabs (assumed to contain a combination of feces and urine), DNA preparation was slightly different: ATL lysis buffer (Qiagen) was added into the sample tubes and vortexed at 55°C 4 times within 1 hour; then, 10 μg carrier DNA (Sigma-Aldrich, St Louis, MO) was added to the lysis buffer, and the lysis buffer was then transferred to newly labeled microfuge tubes for spin-column DNA extraction as above. All PCR tests were run with positive and negative controls. Only samples with a strong and clearly visible amplified fragment of the expected size of 528 bp were scored as positive. Samples with a barely detectable band at this molecular weight on an agarose gel were conservatively interpreted as negative and ascribed to possible cross-contamination during sample collection or processing based on when the samples were collected and handled in relation to positive samples.
Immunohistochemical Staining
The tissues from 2 frogs from each of the infection groups and 1 negative control frog were stained immunohistochemically to detect the presence of viral antigen in affected tissues using a rabbit antibody against epizootic hematopoietic necrosis virus (anti-EHNV); 29 antisera against the ranaviruses are broadly cross-reactive with viruses in the genus. 1,2,17 The immunohistochemical (IHC) staining was first performed manually on 1 frog from each infection group. For the manual staining, 5-μm sections were deparaffinized by immersion in 2 separate baths of xylene (2 minutes each), 3 separate baths of 100% ethanol (1 minute each), and rinsed in running tap water (1 minute). After antigen epitope retrieval was achieved by boiling in Tris/EDTA pH 8.5 (TBS) solution for 20 minutes using an 850-W microwave, the slides were washed with tap water and carefully dried, and a well was created around the tissue sections to hold the IHC solutions. Slides were washed 3 times with TBS, blocked with enzyme-linked immunosorbent assay (ELISA) buffer containing casein (30 minutes at room temperature), and incubated (overnight at 4°C) with 100 μl rabbit anti-EHNV antibody, diluted 1:500 in TBS. Slides were then washed 3 times with TBS, incubated in a solution of 0.3% hydrogen peroxide and 0.1% sodium azide in TBS (15 minutes) to inactivate endogenous peroxidases, washed 3 more times with TBS, and incubated (1.5 hours at room temperature) with 100 μl goat anti-rabbit–horseradish peroxidase conjugate antibody diluted in TBS. Following another 3 washes with TBS, the slides were developed with the addition of 100 μl of the chromogenic solution (0.005% 3-amino-9-ethylcarbazole and 0.001% hydrogen peroxide in substrate buffer, 20 minutes at room temperature), then rinsed in running tap water, counterstained with hematoxylin (3 minutes), and rinsed again with tap water. Once dried, the slides were cover-slipped using aqueous mounting media. Nonspecific binding and endogenous peroxidase control slides were produced by following the above method and omitting the primary and secondary antibodies, respectively.
Using an automated stainer, IHC staining was repeated and expanded to include a second frog from each infection group. The automated staining followed previously published methods.
29
Briefly, 5-μm sections mounted on SuperFrost Plus G/Edge slides (Menzel-Gläser, Braunschweig, Germany; HD Scientific, Kings Park, Australia; cat. HD 041300 72P3) were deparaffinized in a xylene bath and rehydrated through graded ethanol solutions. The following steps were completed using an autostainer at room temperature (Autostainer Plus LV-1; DAKO, Carpinteria, CA). Slides were rinsed in Tris buffer, 3% hydrogen peroxide was added and incubated for 5 minutes and after rinsing, and slides were incubated in Proteinase K solution (ready-to-use solution, cat. S3020; DakoCytomation, Glostrup, Denmark) for 10 minutes. After rinsing in Tris buffer, primary antibody (affinity-purified rabbit anti-EHNV, lot M708, OIE Reference Laboratory for EHN Virus, University of Sydney) diluted 1:1500 in antibody diluent (code S0809; DAKO) was added and incubated for 30 minutes. Sections were gently air blown. Labeling of anti-EHNV antibodies was achieved using a commercial kit (DAKO LSAB
A tissue was considered positive if strong and distinct staining with anti-EHNV (Ranavirus sp) antibody was present in the cytoplasm of 1 or more cells and background staining was either absent or clearly distinct from true specific staining; if background staining was significant and specificity was questionable, the tissue was considered negative.
Results
Frogs measured an average of 52.6 mm in snout-vent length (median, 52.9 mm; range, 24.9–57.9 mm); all were male. One frog was euthanized 1 day ahead of its scheduled euthanasia (13 dpi) due to severe clinical signs consistent with terminal FV3 infection. 12 The results for this frog are included with those from the other frogs euthanized at 14 dpi. All other frogs survived until their planned euthanasia time. Gross lesions (Table 1), evident only in frogs euthanized at 13 to 14 dpi, included petechiae in ventral skin, splenomegaly, and hemorrhage in the wall of the gastrointestinal tract. Histologic lesions (Table 1) consistent with FV3 infection were more common in frogs euthanized toward the end of the trial (ie, 9 and 14 dpi) than those euthanized soon after inoculation. Mild lesions in the skin (hemorrhages and mononuclear inflammation), kidney (interstitial edema), and bone marrow (necrosis) were present in some frogs between 6 and 48 hours after oral inoculation with FV3 (0.25–2 dpi) (Figs. 1–6, 39–50). Except for mild renal interstitial edema and an increase in mononuclear infiltrates in mucosal epithelia, lesions were mostly absent at 4 dpi (Figs. 7–12). Lesions were observed in frogs euthanized at 9 dpi and similar, but more severe, in frogs euthanized at 14 dpi. These lesions consisted of necrosis of hematopoietic and lymphoid tissues (bone marrow, spleen, thymus, kidney, and liver), epidermal and glandular necrosis (skin), mucosal necrosis (oronasal cavity, tongue, lung, and gastrointestinal tract), and renal glomerular and tubular necrosis (Figs. 13–38, 45–50, 51–58). Infiltrates of mostly mononuclear cells in the oronasal mucosa and tongue were present in infected frogs at all sampling times, but their size increased with incubation time, and only in frogs euthanized after 9 dpi did the infiltrates include necrotic/apoptotic cellular debris (Figs. 15–17). Intracytoplasmic viral inclusions were first observed in the hematopoietic tissue (bone marrow and kidney) in the group of frogs euthanized at 9 dpi. At 14 dpi, viral inclusions were present in the oronasal mucosa, tongue epithelium, liver, pancreas, gastrointestinal mucosa, and, in very large numbers, epidermis. By transmission electron microscopy, characteristic hexagonal viral particles were detected in hepatocytes, keratinocytes, endothelium, and fibroblasts of a frog euthanized 14 dpi (Figs. 26, 32).
Gross and Histologic Lesions and Other Findings in Adult Wood Frogs Orally Infected With 104.43 pfu of Frog Virus 3 and Euthanized at Different Days Postinfection (dpi).
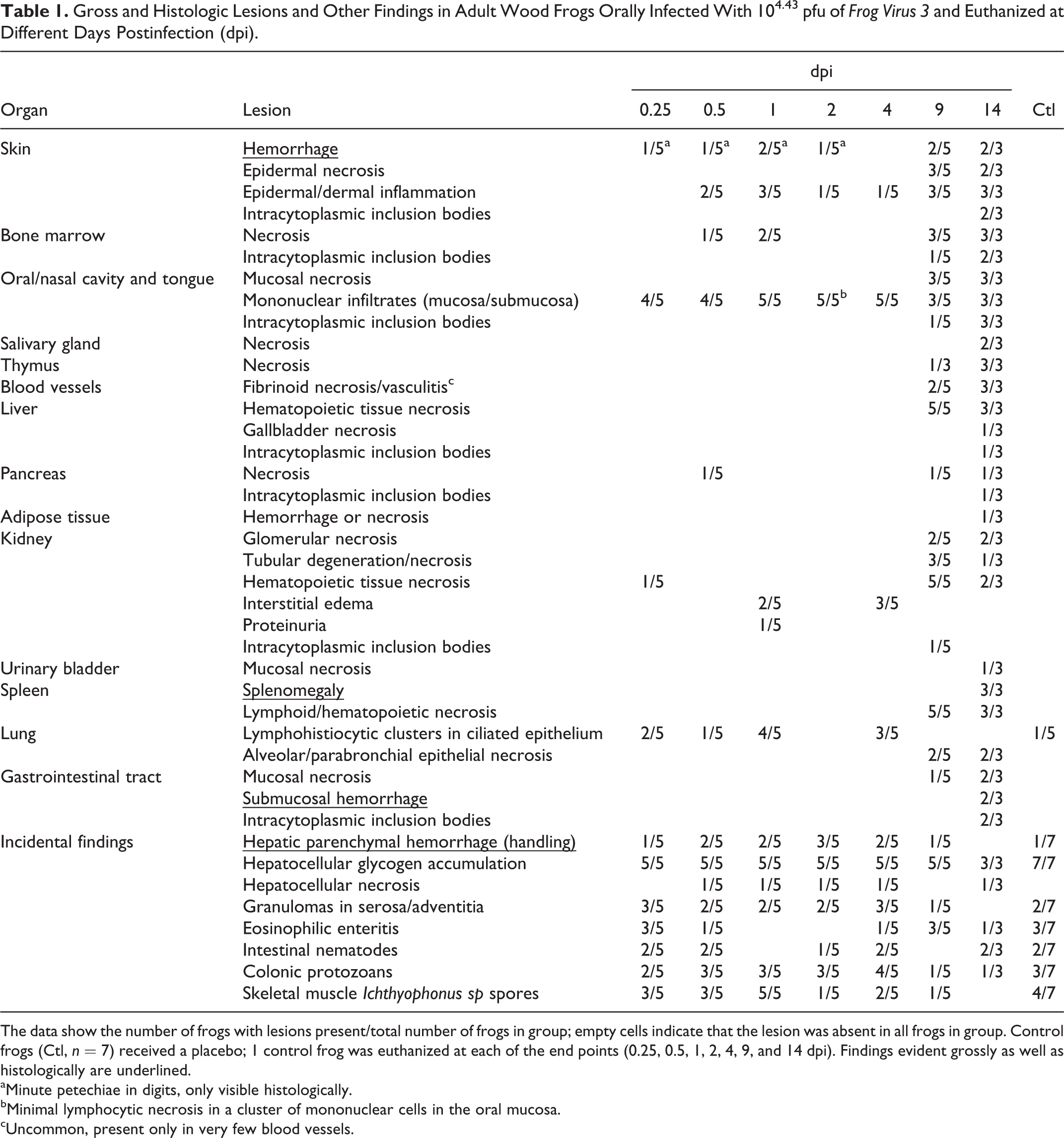
The data show the number of frogs with lesions present/total number of frogs in group; empty cells indicate that the lesion was absent in all frogs in group. Control frogs (Ctl, n = 7) received a placebo; 1 control frog was euthanized at each of the end points (0.25, 0.5, 1, 2, 4, 9, and 14 dpi). Findings evident grossly as well as histologically are underlined.
aMinute petechiae in digits, only visible histologically.
bMinimal lymphocytic necrosis in a cluster of mononuclear cells in the oral mucosa.
cUncommon, present only in very few blood vessels.
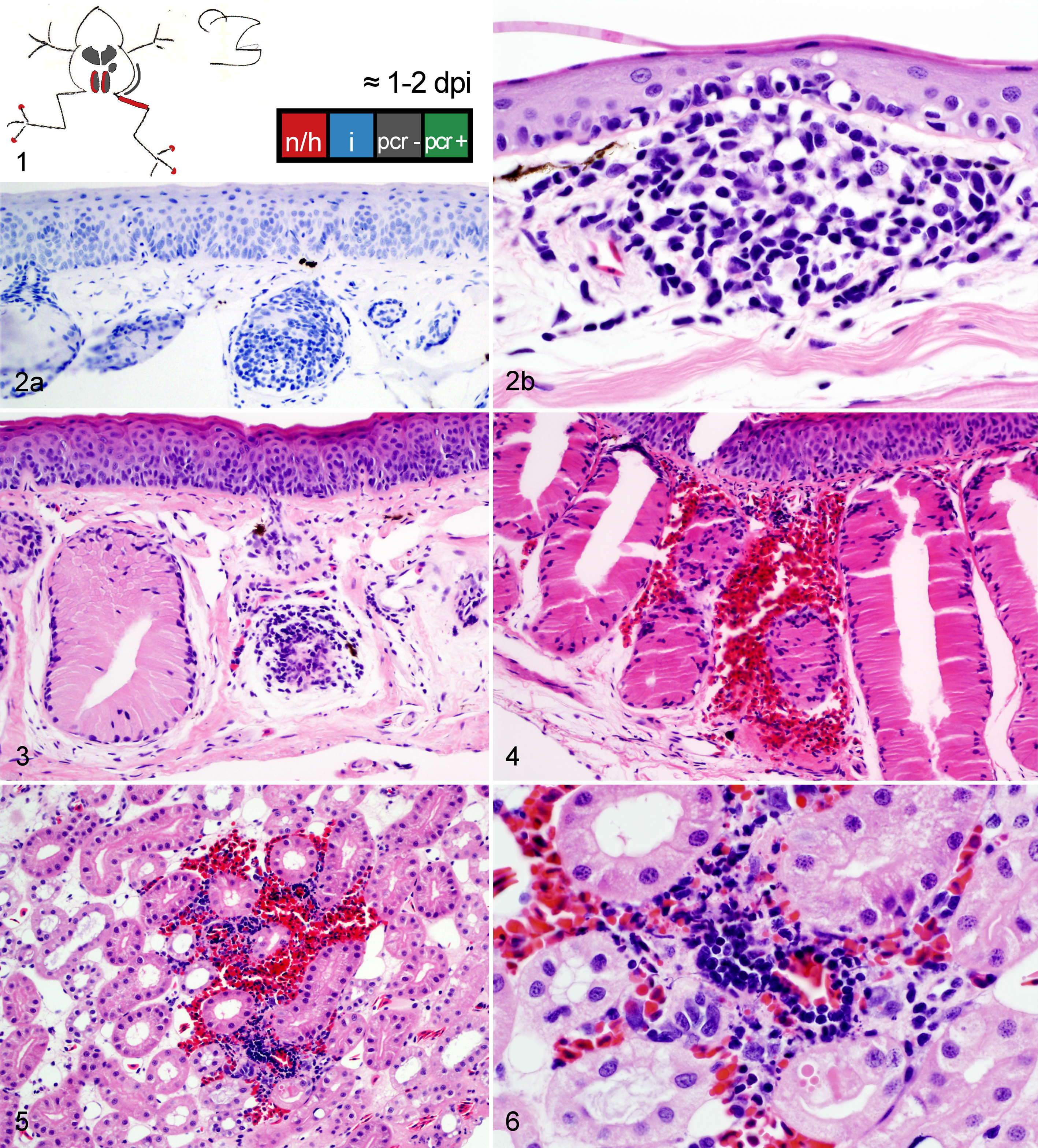
Wood frog adults euthanized 0.25 to 2 days postinfection (dpi) with 104.43 pfu of Frog virus 3.
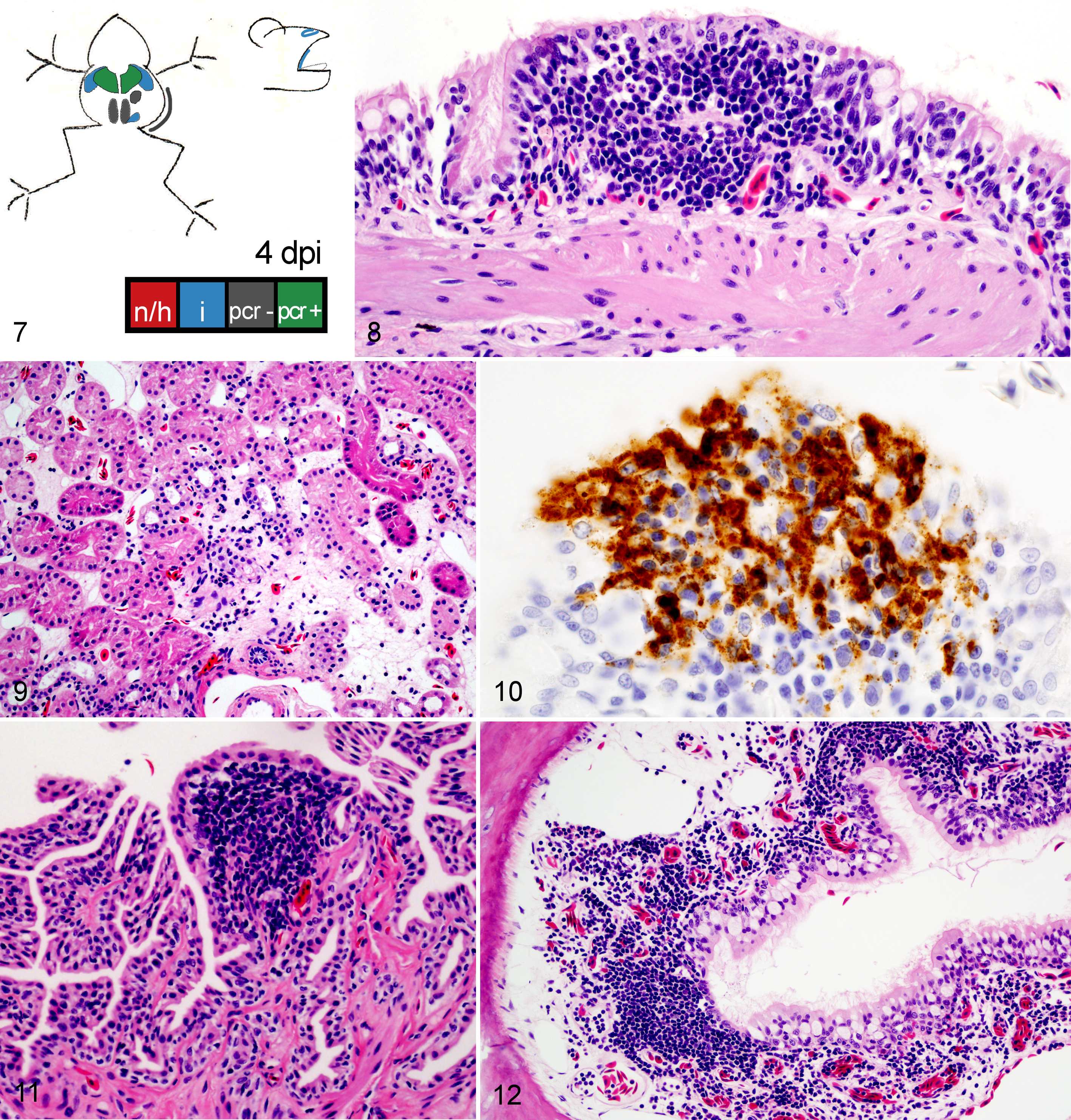
Wood frog adults euthanized 4 days postinfection (dpi) with 104.43 pfu of Frog virus 3.
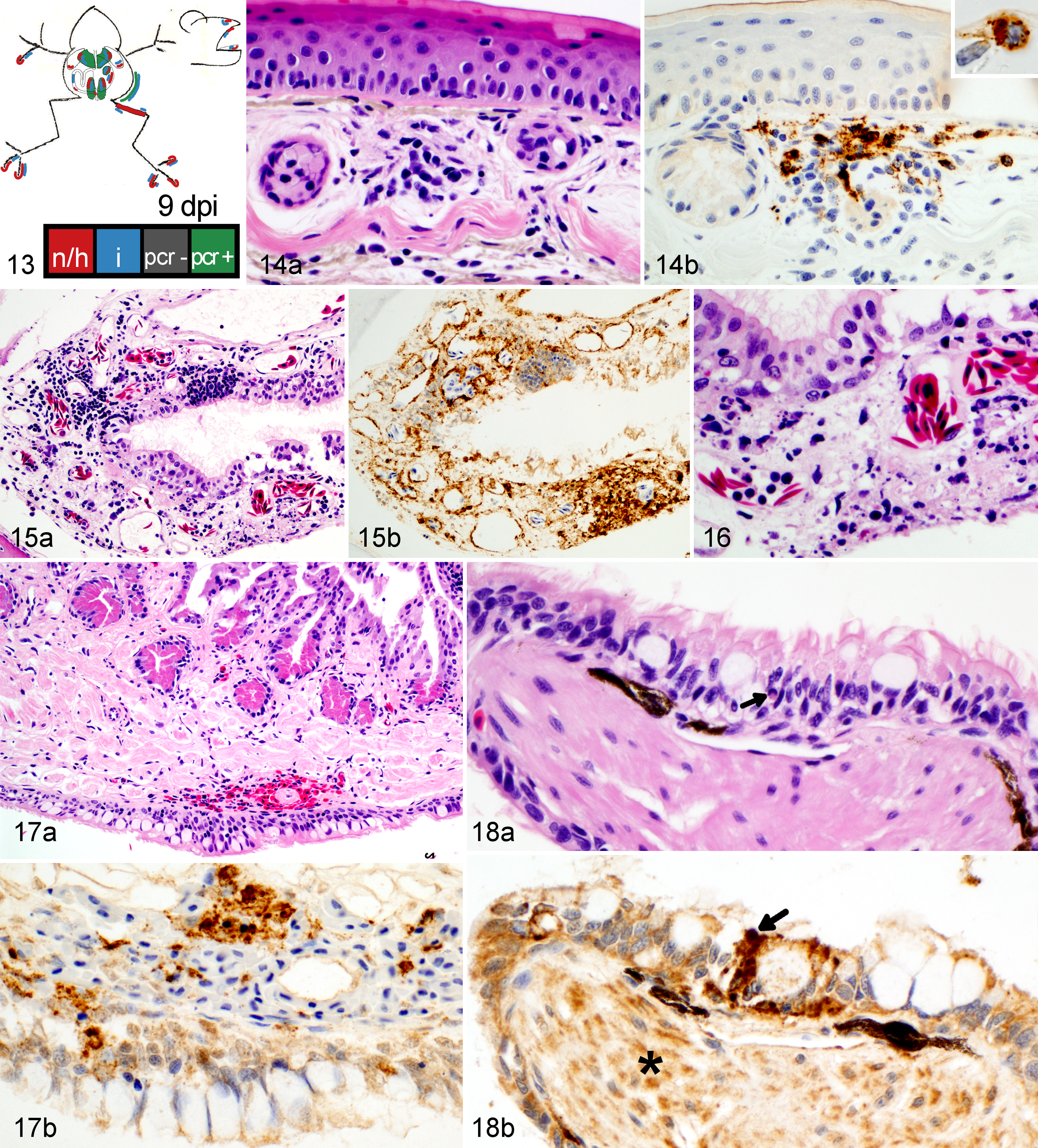
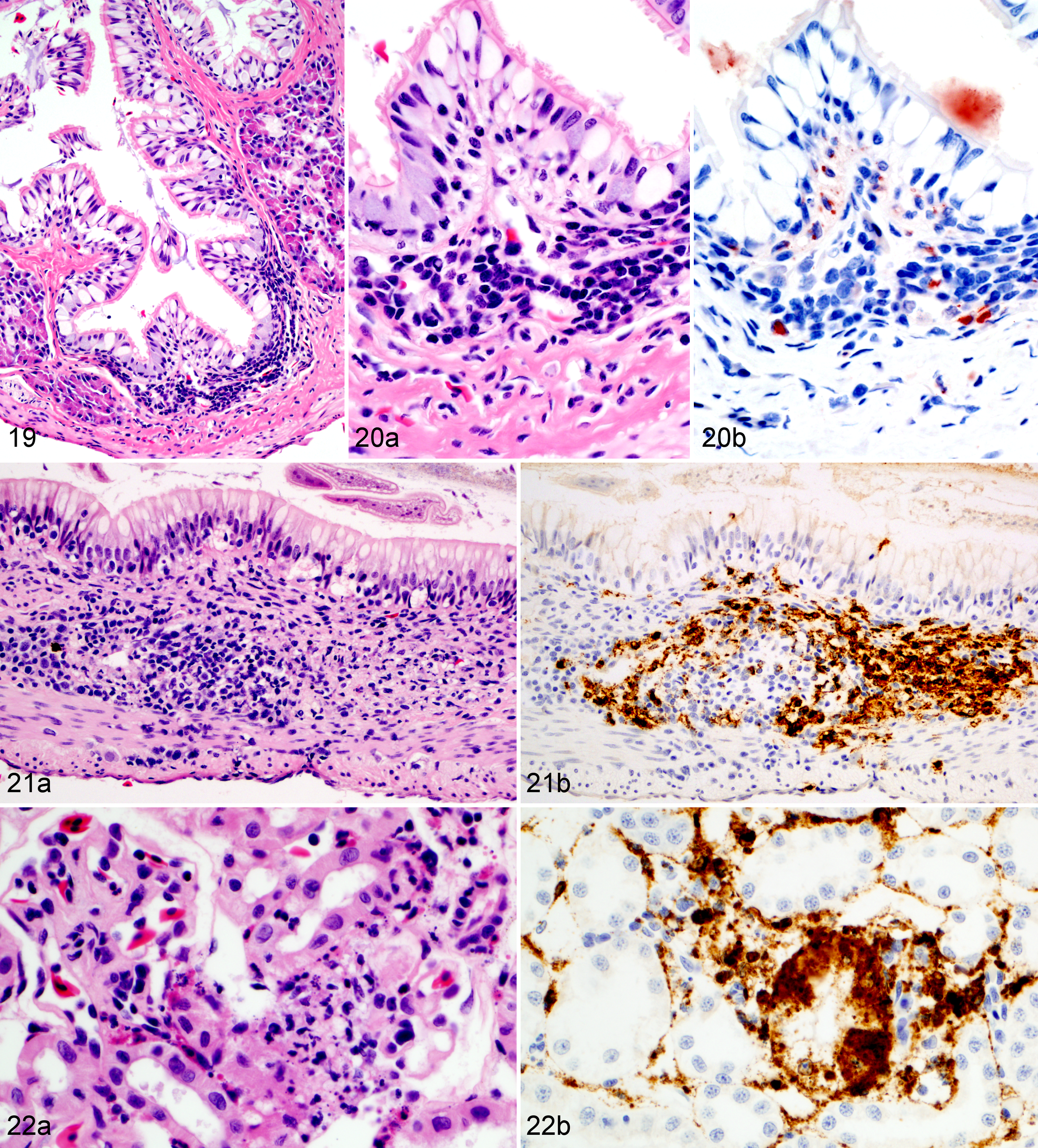
Wood frog adults euthanized 9 days postinfection (dpi) with 104.43 pfu of Frog virus 3.
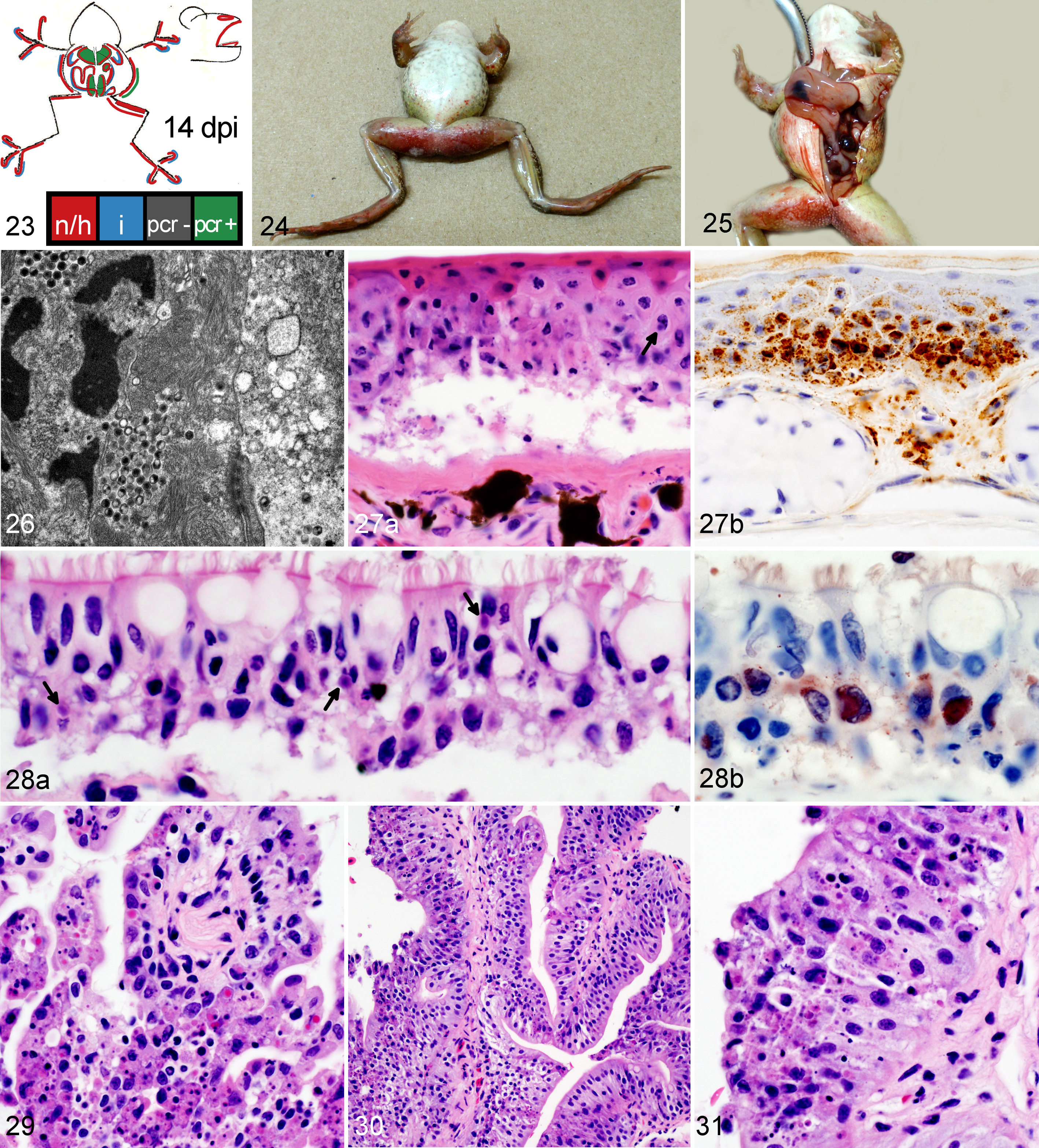
Wood frog adults euthanized 14 days postinfection (dpi) with 104.43 pfu of Frog virus 3 (FV3).
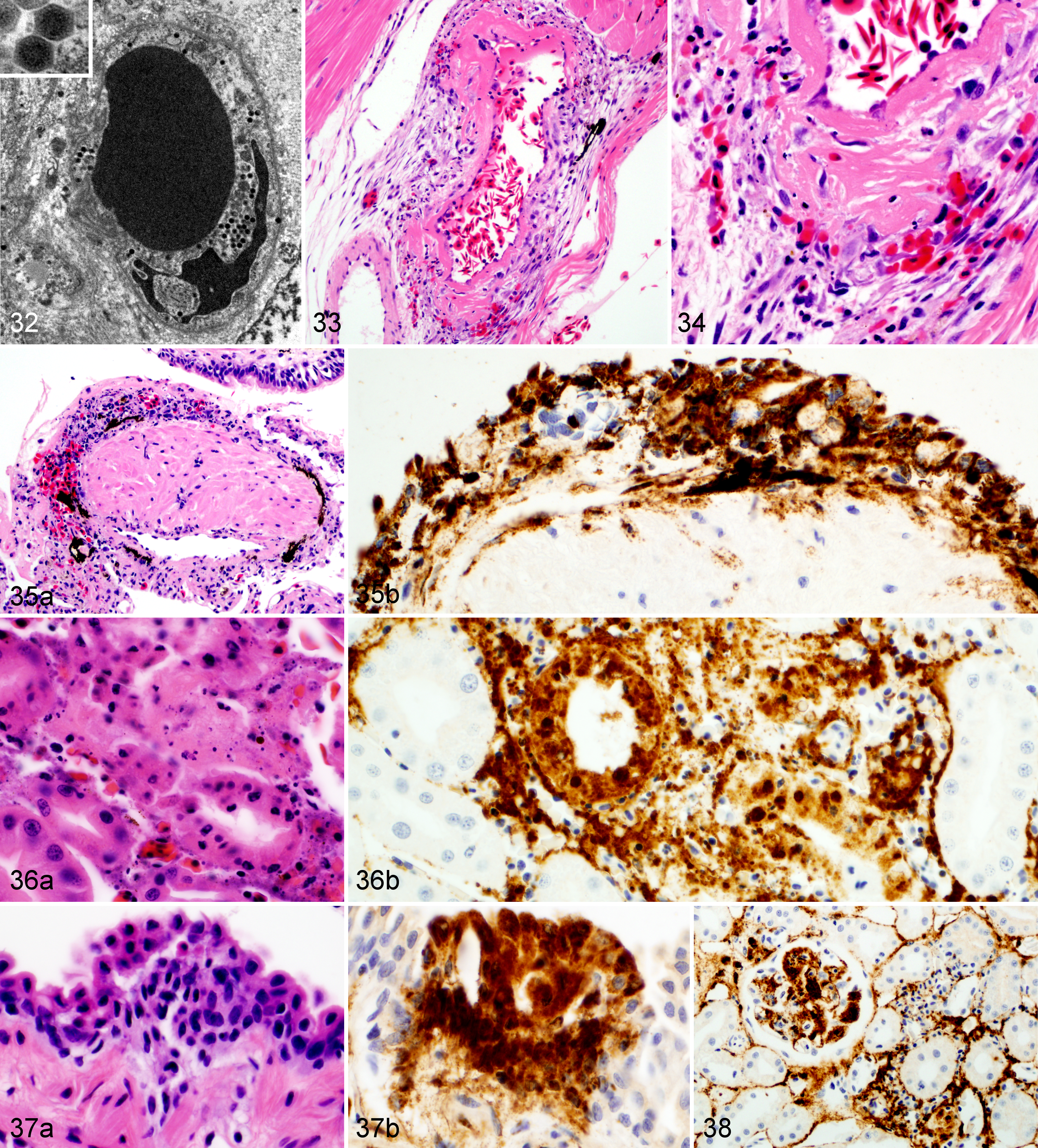
Wood frog adults euthanized 14 days postinfection (dpi) with 104.43 pfu of Frog virus 3 (FV3).
Findings were classified as “incidental” if present in frogs from all infection groups, including both challenged (infected) and negative (uninfected) controls (Figs. 59–63). These incidental findings included hepatocellular glycogen accumulation, peracute hepatic hemorrhage, chronic granulomas in coelomic cavity, intestinal parasitism, and fungal spores in the skeletal muscle. Periodic acid–Schiff (PAS) reaction confirmed the accumulation of glycogen in hepatocellular cytoplasm. Of note was the paucity of extramedullary hematopoietic tissue in the renal and hepatic parenchyma of those frogs that had no lesions and of the uninfected controls.
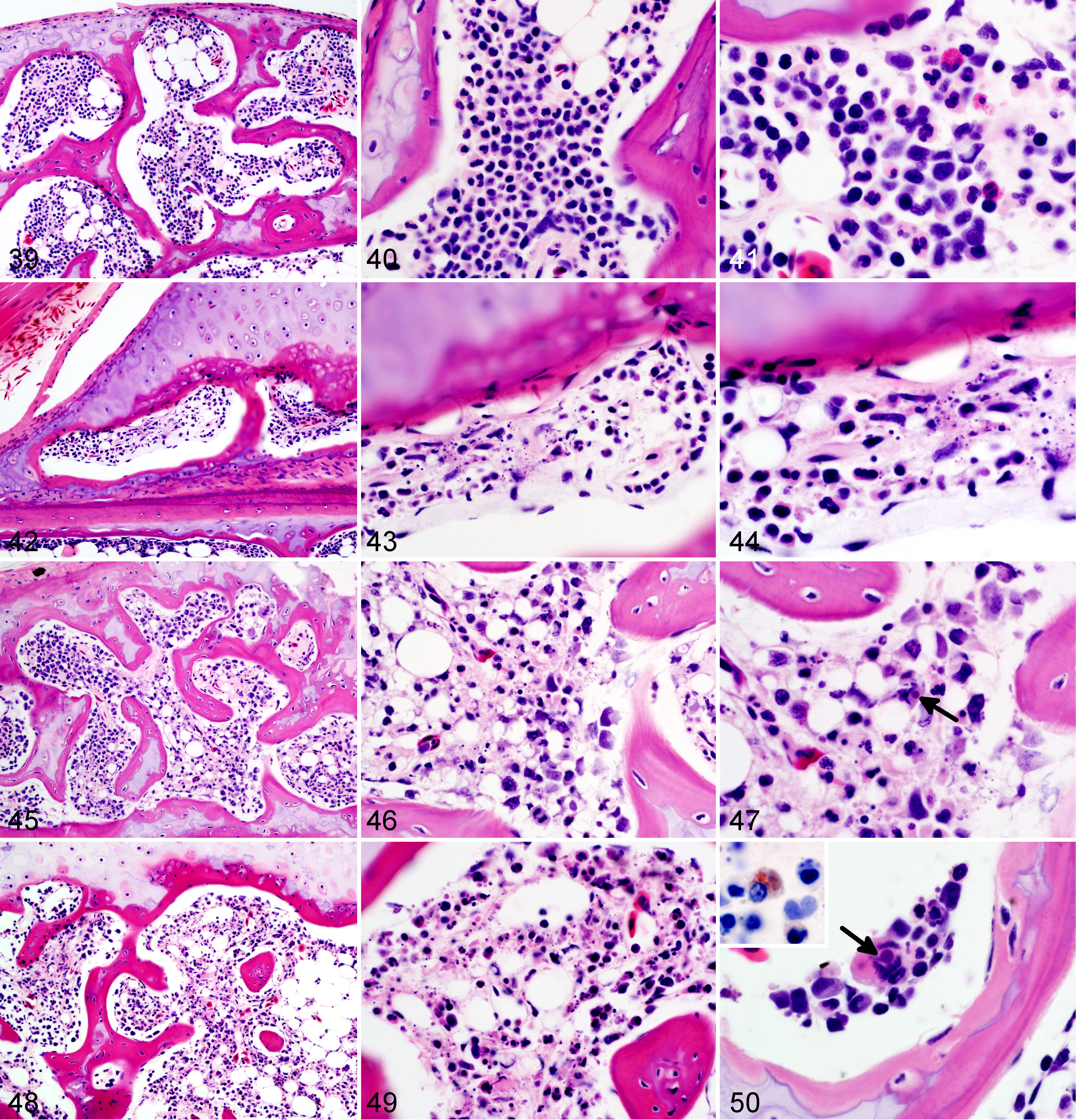
Wood frog adults, uninfected control (Figs. 39–41) and frogs euthanized 1 (Figs. 42–44), 9 (Figs. 45–47), or 14 (Figs. 48–50) days postinfection with 104.43 pfu of Frog virus 3, bone marrow. Necrosis of the hematopoietic tissue increases in severity with time. Note the intracytoplasmic inclusion bodies, probably of viral origin, present in an unidentified cell (arrow, 47) and in a multinucleated cell (either an osteoclast or a megakaryocyte; arrow, 50). Hematoxylin and eosin (HE). Intracytoplasmic inclusion in a mononuclear cell is labeled as ranavirus positive (50, inset, immunohistochemistry for epizootic hematopoietic necrosis virus).
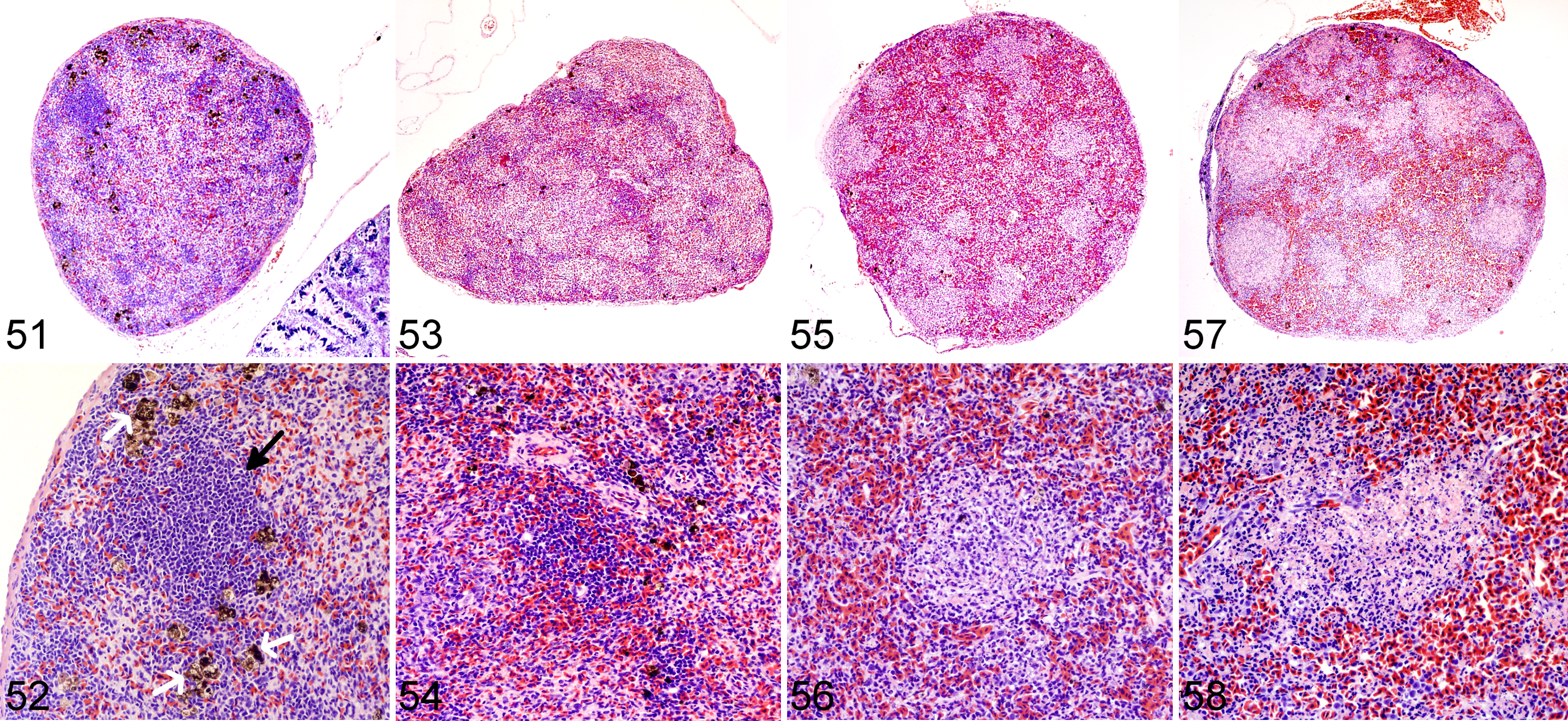
Wood frog adults, uninfected control (Figs. 51–52) and frogs euthanized 1 (Figs. 53–54), 9 (Figs. 55–56), or 14 (Figs. 57–58) days postinfection with 104.43 pfu of Frog virus 3, spleen.
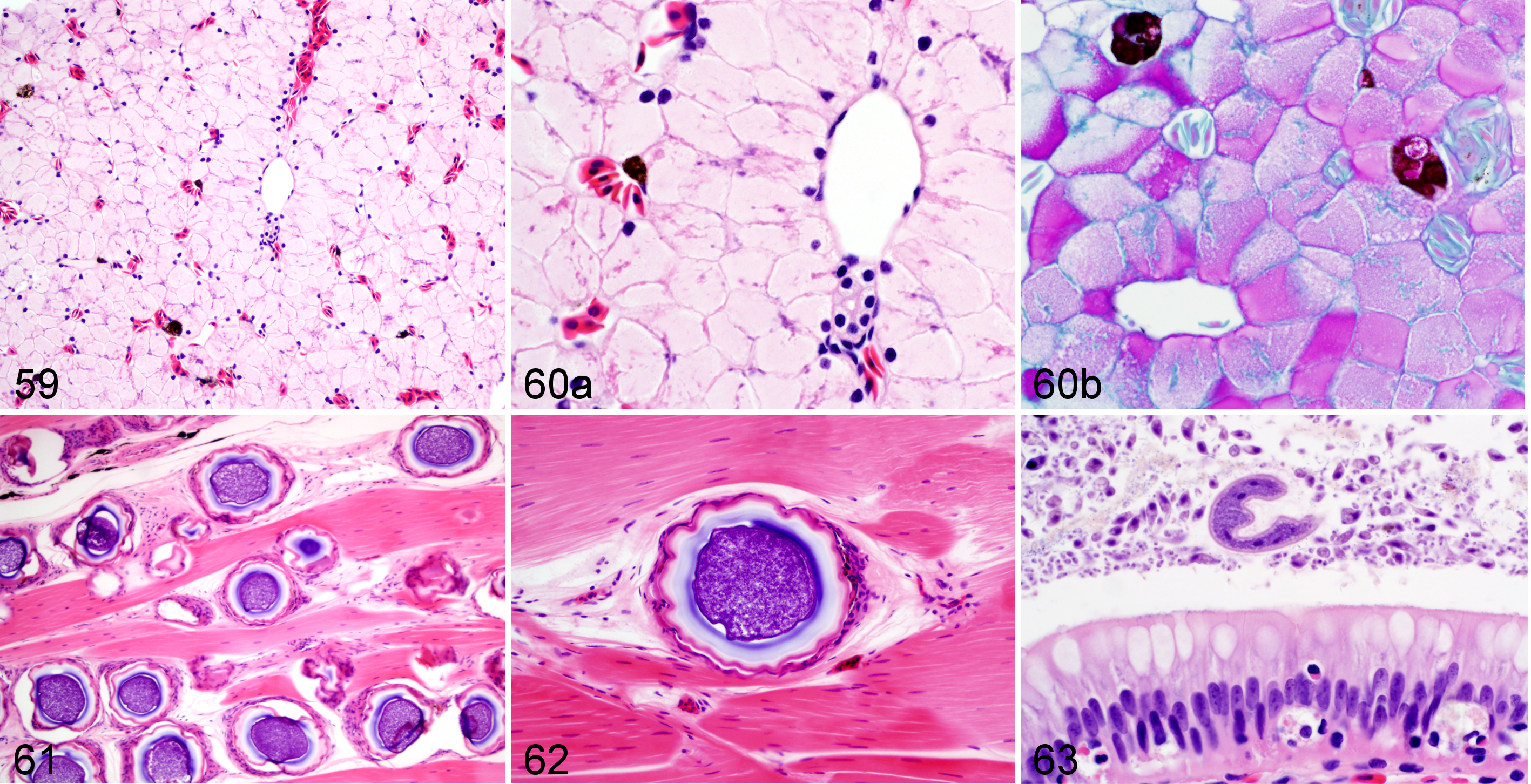
Wood frog adults, control individuals with incidental findings at necropsy.
Detection of Ranavirus sp DNA in tissues via PCR was, in general, consistent with the appearance of lesions. PCR for ranavirus DNA was negative in all samples (abdominal skin, liver, kidney, spleen, and cloacal swab) from frogs euthanized at 0.25, 0.5, 1, and 2 dpi. The earliest positive PCR signal was recorded in the livers of 2 frogs at 4 dpi. At 9 and 14 dpi, all organs tested (ie, skin, liver, kidney, and spleen), as well as the cloacal swab obtained immediately postmortem, were strongly positive for Ranavirus sp DNA (Table 2). Except for the livers of frogs euthanized on dpi 4, all positive PCR signals corresponded to positive IHC staining (Table 3). All tissues from all control frogs (n = 7) were PCR negative for Ranavirus sp DNA.
PCR Testing for Ranavirus sp DNA in the Tissues of Adult Wood Frogs Euthanized at Different Number of Days (dpi) After Oral Inoculation with 104.43 pfu of Frog Virus 3 (Ranavirus sp).
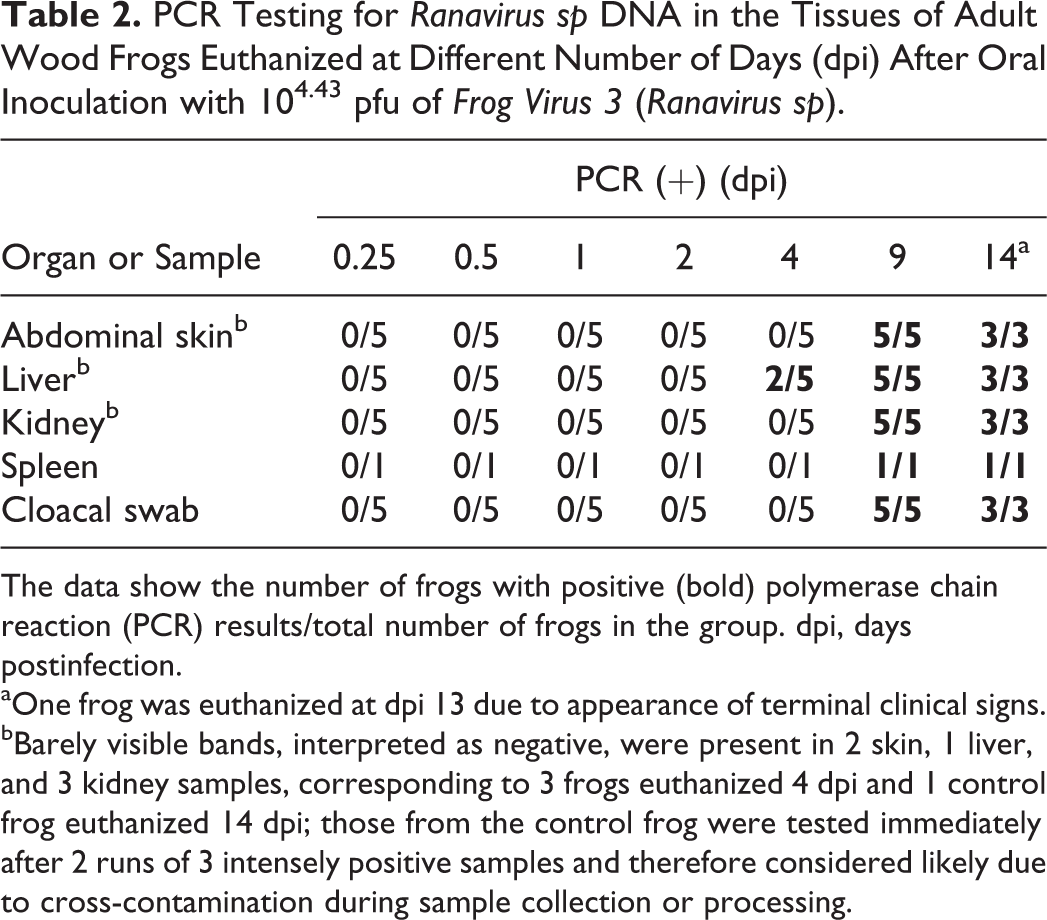
The data show the number of frogs with positive (bold) polymerase chain reaction (PCR) results/total number of frogs in the group. dpi, days postinfection.
aOne frog was euthanized at dpi 13 due to appearance of terminal clinical signs.
bBarely visible bands, interpreted as negative, were present in 2 skin, 1 liver, and 3 kidney samples, corresponding to 3 frogs euthanized 4 dpi and 1 control frog euthanized 14 dpi; those from the control frog were tested immediately after 2 runs of 3 intensely positive samples and therefore considered likely due to cross-contamination during sample collection or processing.
Immunohistochemical Staining of Tissues From Selected Adult Wood Frogs Orally Infected With 104.43 pfu of Frog Virus 3 (Ranavirus sp) and Euthanized at Different Days Postinfection (dpi).
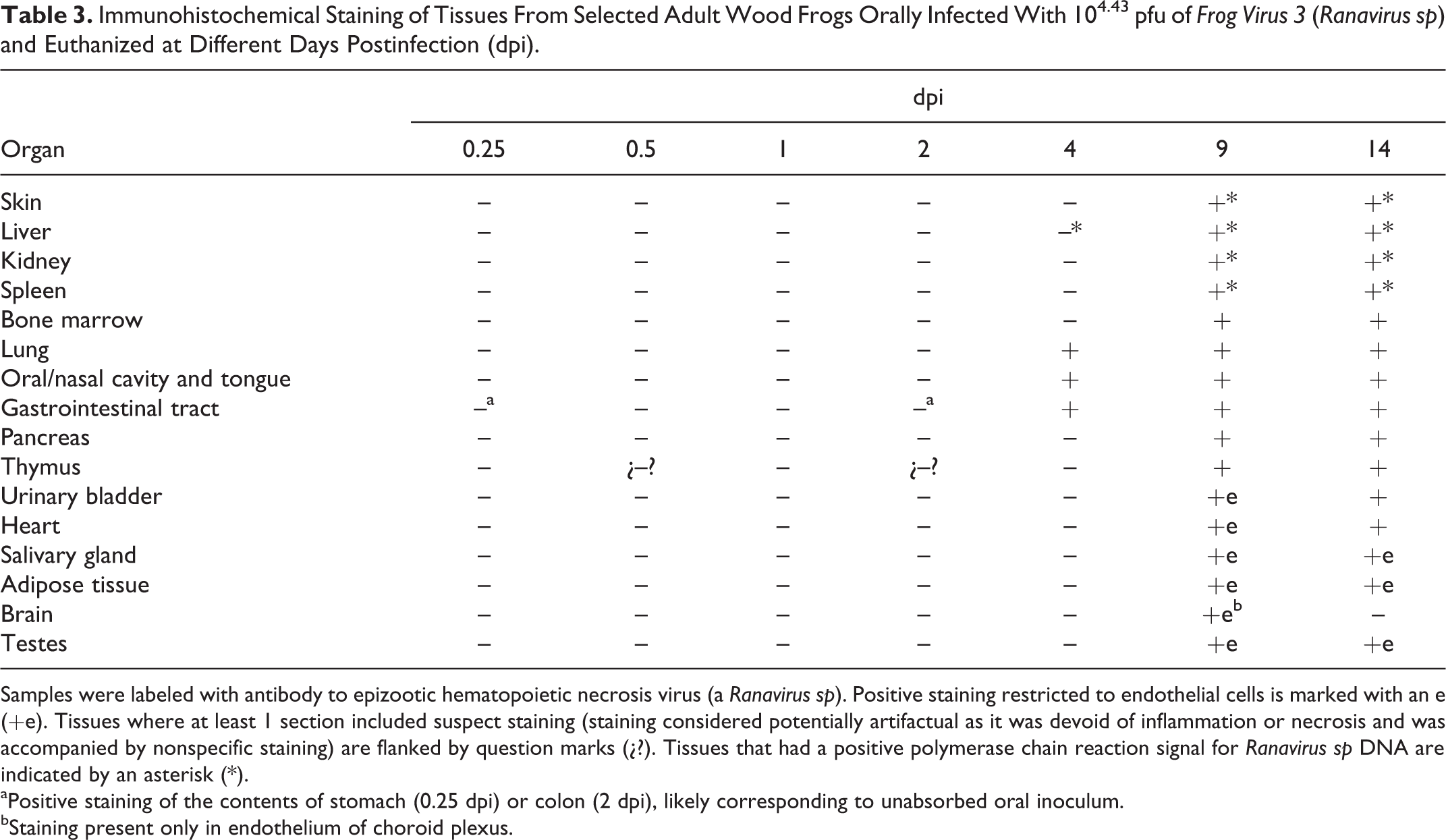
Samples were labeled with antibody to epizootic hematopoietic necrosis virus (a Ranavirus sp). Positive staining restricted to endothelial cells is marked with an e (+e). Tissues where at least 1 section included suspect staining (staining considered potentially artifactual as it was devoid of inflammation or necrosis and was accompanied by nonspecific staining) are flanked by question marks (¿?). Tissues that had a positive polymerase chain reaction signal for Ranavirus sp DNA are indicated by an asterisk (*).
aPositive staining of the contents of stomach (0.25 dpi) or colon (2 dpi), likely corresponding to unabsorbed oral inoculum.
bStaining present only in endothelium of choroid plexus.
Manual and automated immunohistochemical staining yielded similar and repeatable results. The only exception was the staining of tissue sections located along the edge of the glass slide, which failed to stain with the manual technique but were clearly positive when the automated stainer was used. We also noted a small and subjective difference in the sharpness and hue of the staining: the manual technique sometimes achieved crisper staining than the automated one (eg, Figs. 20b, 21b). Skin sections that had to be decalcified since they were attached to bone (hand, feet, head, thigh) appeared to stain similarly to ventral skin, which had been processed along with other soft tissues, suggesting that decalcification had little to no effect on IHC staining.
In frogs euthanized 4 dpi, there were distinct but rare foci of positive IHC staining in the oral cavity (mucosal epithelium of the lip junction) and lung (respiratory epithelium), as well as in a few individual cells within the mucosal epithelium and submucosa of the colon; no necrosis or inflammation was evident in the positively stained foci. Positive staining could be found in all tissues examined from frogs euthanized at 9 and 14 dpi, often clearly corresponding to intracytoplasmic inclusion bodies (Figs. 27, 28). Staining at those times was restricted to scattered endothelial cells (urinary bladder, heart, salivary gland, adipose tissue, brain, and testes) or present also in parenchymal and hematopoietic cells (skin, liver, kidney, spleen, bone marrow, lung, oral and nasal cavities, tongue, gastrointestinal tract, pancreas, and thymus). Positive staining in the contents of stomach (0.25 dpi) and colon (2 dpi) likely corresponded to unabsorbed oral inoculum, and thus staining of the organ was considered negative. Suspect IHC staining was present in a few cells in the thymus at 0.5 and 2 dpi but was considered artifactual as it was devoid of inflammation or necrosis and was accompanied by nonspecific staining. No IHC staining for Ranavirus sp was present in any tissue from the uninfected controls (Table 3).
Discussion
Adult wood frogs were experimentally challenged with FV3 and euthanized at scheduled intervals to obtain samples reflecting the temporal pathogenesis of a natural infection.
This longitudinal study of a ranavirus infection in adult wood frogs identified important sites of viral replication and shedding throughout the infection and can guide which diagnostic samples are effective in the early and late stages of infection.
Lesions caused by FV3 infection in these adult wood frogs were consistent with what we have recently described in fatally infected wood frogs. 12 Although present in multiple organs, lesions reflected a predilection for certain tissues: hematopoietic and lymphoid tissues, epidermal, renal tubular and mucosal epithelia, and vascular endothelium. Mild and sporadic lesions consistent with damage to epithelium, endothelium, and hematopoietic tissue occurred in the first 48 hours after inoculation (0.25–2 dpi) and resulted in minimal petechiae in skin of digits, mild renal interstitial edema, and minute foci of bone marrow necrosis. These early lesions were not evident in samples collected after 2 dpi, suggesting they were sites of primary replication from where the virus spread systemically, during a subclinical stage, for a second and more intense wave of replication a few days later, eventually leading to clinical disease. During this subclinical stage, mononuclear infiltrates began to increase in the epithelia of oral, respiratory, and intestinal mucosae, and IHC staining identified rare small foci of viral replication in the epithelium of the oral cavity, lung, and gastrointestinal tract (4 dpi). When the lesions were again visible in samples collected on 9 dpi, they were more widespread and severe compared to those observed in samples collected on day 4 dpi: hemorrhages in the skin were larger and more frequent, found not only in digits, and accompanied by foci of epidermal necrosis. Hematopoietic necrosis was more extensive in the bone marrow and present also in extramedullary hematopoietic tissue in the kidney, liver, and, most severely, the spleen. Along with the exacerbation of lesions in the skin and hematopoietic tissue, necrosis of lymphoid tissue (spleen, thymus, oronasal, and gastrointestinal mucosae), mucosal epithelium (oronasal cavity, tongue, stomach, and intestine), and renal glomeruli and tubules had developed.
Our findings suggest that FV3 infection in adult wood frogs first targets hematopoietic tissue in the bone marrow and endothelial cells in the skin, causing very mild microscopic lesions. Then, after a few days during which lesions are no longer histologically visible, FV3 causes severe lesions in medullary and extramedullary hematopoietic tissue, lymphoid tissue, and epithelial cells of skin and mucosae throughout the body. Abundant IHC staining in the skin and gastrointestinal mucosal epithelium points to these tissues as important secondary sites of viral replication and shedding. Replication in epidermal and mucosal epithelia and shedding from skin and oral and gastrointestinal mucosae may facilitate transmission through direct contact and a fecal-oral route. Shedding of infected renal tubular cells in urine, which would have been also collected with a cloacal swab, may also contribute to transmission.
Because the experimental infection was conducted in autumn, outside of the seasons when mortalities from ranaviruses occur in the wild (ie, spring and summer), 15 the relevance of our findings could be questioned. Fortunately, the progression of disease we document here is consistent with the findings of a dose-dependent infection trial conducted in adult wood frogs in mid-spring. 12 Also, room temperature was controlled to resemble spring and summer temperatures in the wild. Whether there are differences in susceptibility between males and females could not be explored since all the frogs we collected were male. As long as experimental subjects are wild-caught adult wood frogs, this bias will be difficult to avoid: females stay in vernal pools only long enough to mate and lay a clutch of eggs, whereas males remain there for days, even after mating, hoping to increase their reproductive successes.
Our conclusions are based on a combination of visible histopathologic changes and detection of viral presence through indirect and direct methods (ie, IHC staining and PCR detection of DNA). In the late stages of infection, each technique supported the findings of the other two: in most instances, histologic evidence of necrosis in a tissue was consistent with positive IHC staining and a positive PCR signal. In the early stages, however, the skin petechiae and hematopoietic necrosis observed histologically were not accompanied by positive PCR or IHC staining. The apparent discrepancy in the PCR results could be simply because the lesions were observed in tissues not tested: the petechiae were noted exclusively in the skin of digits and absent from abdominal skin, and the hematopoietic necrosis was present only in the bone marrow but not in any extramedullary hematopoietic site. Perhaps if we had tested bone marrow or the skin from digits instead of abdomen, positive PCR signals would have been detected earlier in the infection. Lack of IHC staining could have been due to insufficient amount of antigen (viral proteins) in those mild early lesions to produce visible staining. A more sensitive technique, such as in situ hybridization, 21 could perhaps have been more successful. Alternatively, it could be that the virus was indeed absent from the bone marrow and the damage to the hematopoietic tissue was indirectly due to FV3 infection. An induction of apoptosis through an increase of proapoptotic cytokines such as tumor necrosis factor α, which is upregulated in African clawed frogs infected with Ranavirus sp, 27 could have produced hematic cell necrosis without actual viral presence in the tissue.
A consistent finding in all frogs, infected or uninfected controls, was the glassy appearance of the cytoplasm in hepatocytes. Due to the abundance of glycogen (confirmed with PAS stain), the finding probably reflected the time of year when the experiment was conducted: mid to late autumn. The frogs, even with no access to outdoor light and maintained at a fairly constant temperature, seemed to have been able to sense the change in seasons and duly prepared for hibernation. Because wood frogs must tolerate freezing temperatures during the winter, cryoprotectants, of which glucose is perhaps the most important, are crucial for survival. 8 Reaching the concentration of glucose necessary to prevent freezing damage to all tissues depends on previous accumulation of sufficient glycogen in the liver. 31 Wild wood frogs in spring have much lower concentrations of liver glycogen (100–200 μmol/g fresh weight) than their counterparts in the autumn (700–1000 μmol/g fresh weight). 19 Glycogen accumulation, which renders the liver friable, also explains the presence of peracute (handling-associated) hemorrhages in several frogs, including 1 uninfected control. Neither the glycogen accumulation nor the liver hemorrhages were, therefore, considered to result from FV3 infection but were incidental findings associated with the natural history of wood frogs.
Other incidental findings, inevitable when working with wild-caught adults, included intestinal nematodiasis and skeletal muscle ichthyophoniasis. Nematode infection was mild, perhaps partly because of the deworming of all frogs 5 months before to treat an intense pulmonary rhabdiasis (0.02 mg ivermectin per frog, transcutaneous). The small granulomas found on visceral serosal and adventitial surfaces of some frogs were likely associated with larval migration (in some, nematode larvae were still present). Ichthyophoniasis is not uncommon in wild-caught frogs from Prince Edward Island (Canadian Wildlife Health Cooperative necropsy records, unpublished) and has been reported also in wood frogs from Québec. 25 Although massive infections of Ichthyophonus fungi have been documented as the cause of death of green frogs (Rana [Lithobates] clamitans) and bullfrogs (R.[L.] catesbeiana), mild endemic infections are more commonly observed. 15 Nematodiasis, nematode larva-associated granulomas, and ichthyophonosis had no apparent deleterious effect on the health of the wood frogs involved in our experimental trial. Avoiding incidental infections with parasites or fungi in experimental subjects would require captive breeding or at least captive rearing from eggs. No captive-reared, let alone commercially available specific pathogen-free, wood frogs are available at present, but captive rearing from eggs or tadpoles is possible. 12 To ensure that wild-caught frogs are not already infected with the pathogen in question, necropsy and histopathological examination of all mortalities in captivity, along with PCR or other specific ancillary tests, are indispensable. Based on such an examination of mortalities during the long acclimatization period prior to the experimental trial, the lack of recorded ranavirus outbreaks in the ponds from which the animals were collected, and from the PCR, histopathology, and IHC staining of all controls, we are confident that our experimental subjects did not harbor a natural infection with Ranavirus sp.
The pathogenesis FV3 infection in adult wood frogs resembles what occurs in other adult frogs infected with this or other species of ranavirus. Adult Australian tree frogs (Litoria splendida and Litoria caerulea) that succumb to natural infections with Mahaffey Road (Bohle-like) virus develop widespread necrosis of epidermal and mucosal epithelia, as well as lymphoid and hematopoietic tissues. 20 Unlike wood frogs in which necrotizing vasculitis is uncommon and which lack any evidence of involvement of the central or peripheral nervous tissues, widespread vasculitis with endothelial necrosis, meningoencephalitis, myelitis, and ganglioneuritis are prominent lesions reported in Australian tree frogs. 20 Based on IHC staining, Bohle-like virus infection in Australian tree frogs is strongly endotheliotropic, as FV3 seems to be in wood frogs, but lesions preferentially develop in hematopoietic, lymphoid, and epithelial tissues rather than blood vessels. In African clawed frogs infected with FV3 and euthanized at various times postinfection in a manner resembling the methods of our study, a strong PCR signal for viral DNA was detected in the skin at 1 dpi and in the kidney at 3 and 7 dpi, but it was absent or barely detectable in the liver at similar times. 30 Comparable findings were present in our wood frogs if we include histopathology and IHC staining results: mild lesions in the skin at 1 to 2 dpi and strong DNA signals in the kidney at 9 dpi. Unlike African clawed frogs, no DNA was detected in wood frogs at 1 dpi, even though mild petechiae were observed histopathologically. A possible explanation is preferential viral targeting of distal capillaries at early stages of infection: the lesions (petechiae) at 1 to 2 dpi were observed only in the skin of digits, while our sample for PCR testing was obtained from the ventral abdomen, where no lesions were present. Also, unlike African clawed frogs, wood frog livers were positive for viral DNA as early as 4 dpi and, along with skin and kidney, strongly positive at 9 dpi. Different tissue tropism could be due to characteristics of each host species. However, given the histopathological appearance and IHC staining of these tissues, the positive PCR signal in the liver and kidney in wood frogs was at least partly due to virus in local hematopoietic and lymphoid tissues and, in the case of the liver, resident melanomacrophages. Histopathology was not performed in the African clawed frog study, so the precise location of the virus in the tissue was not determined. Perhaps, as lymphoid tissue is often present in the kidney of African clawed frogs, 32 viral DNA detected in renal tissue corresponded to lymphocytes infected with FV3. Whether differences in tissue tropism suggested by comparing our study with those in Australian tree frogs or African clawed frogs are due to characteristics specific to the host, the pathogen or both would require experimental exposure of adults from each species of frog to the various Ranavirus sp.
One of the challenges in the study of disease ecology of Ranavirus spp is the lack of a sensitive diagnostic test that can detect infection in live animals. Cloacal and oral swabs, as well as toe and tail clips, have been used in an attempt to diagnose infection through nonlethal sampling. 14,16 Studies on the validity of PCR tests on samples that can be obtained from live animals have found a failure to detect pre- or subclinical infections: tail clips in salamanders are negative except during active viremia; 16 tail clips and oral and cloacal swabs of bullfrog tadpoles fail to identify approximately one-fifth of infected individuals. 14 Our findings indicate that PCR testing on cloacal swabs of wood frogs fails to detect viral DNA until just a few days before death (when clinical signs become evident) and that survivors do not seem to shed detectable virus as early as 2 weeks after clinical signs disappear. 12 Sampling of liver is appropriate in wood frogs, as it allows viral DNA detection earlier than other commonly sampled organs (skin, kidney, and spleen). Further studies on nonlethal alternatives to detect Ranavirus sp infection are urgently needed. Until then, any nonlethal sampling of infected but clinically healthy individuals is likely to yield a false-negative result.
We report a progression of lesions in adult wood frogs infected with a lethal dose of FV3. Mild lesions were first detected at around 1 to 2 dpi in the skin (probably associated with damage to capillary endothelia) and bone marrow. Slow viral replication without obvious tissue lesions in oral, gastrointestinal, and pulmonary epithelia followed. Severe, ultimately fatal, lesions arose approximately a week postinfection and consisted of necrosis of medullary and extramedullary hematopoietic tissue, lymphoid tissue in spleen and throughout the body, and epithelium of skin and mucosae. Oral, gastrointestinal, and pulmonary mucosae were important sites of secondary viral replication, before fatal lesions developed. Once ranavirosis became clinically evident and lesions were widespread, the skin was also an important secondary site of viral replication and shedding. Direct contact (skin) and fecal-oral contamination are likely effective routes of transmission; thus, skin and cloacal swabs may be reliable sources of antemortem diagnostic samples but only in late stages of disease, soon before or at the time of appearance of clinical signs.
Footnotes
Acknowledgements
We thank Dr Alexandra Reid, who kindly provided the isolate of FV3 used in the infection; Dr Thijs Kuiken, who advised on the experimental design; Dr Dorota Wadowska, who performed the electron microscopy; Wytamma Wirth, who advised on the manual immunohistochemistry technique; and Drs Marion Desmarchelier, Shannon Martinson, and Jonathan Spears, Mr Chris MacQuaid and Mr Maciez Zawadzki, and in particular Dr Jessica Thompson and Ms Sara Vazquez Quiroga, whose efforts were indispensable to the successful rearing of the wood frogs.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was partly funded by an Alexander Graham Bell Graduate Scholarship-Doctoral and the Canadian Wildlife Health Cooperative (formerly the Canadian Cooperative Wildlife Health Centre).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
