Abstract
Secreted WNT proteins control cell differentiation and proliferation in many tissues, and NOTUM is a secreted enzyme that modulates WNT morphogens by removing a palmitoleoylate moiety that is essential for their activity. To better understand the role this enzyme in development, the authors produced NOTUM-deficient mice by targeted insertional disruption of the Notum gene. The authors discovered a critical role for NOTUM in dentin morphogenesis suggesting that increased WNT activity can disrupt odontoblast differentiation and orientation in both incisor and molar teeth. Although molars in Notum-/- mice had normal-shaped crowns and normal mantle dentin, the defective crown dentin resulted in enamel prone to fracture during mastication and made teeth more susceptible to endodontal inflammation and necrosis. The dentin dysplasia and short roots contributed to tooth hypermobility and to the spread of periodontal inflammation, which often progressed to periapical abscess formation. The additional incidental finding of renal agenesis in some Notum -/- mice indicated that NOTUM also has a role in kidney development, with undiagnosed bilateral renal agenesis most likely responsible for the observed decreased perinatal viability of Notum-/- mice. The findings support a significant role for NOTUM in modulating WNT signaling pathways that have pleiotropic effects on tooth and kidney development.
Keywords
Members of the Wingless/WNT family are secreted proteins that activate signaling cascades having central roles in developmental processes during embryogenesis. These paracrine messengers are important in maintaining homeostasis in adult tissues by regulating cell proliferation, differentiation, polarized migration, genetic stability, and apoptosis. As a result, dysregulation of WNT signaling has been linked to many diseases. 22,77 Excessive expression or aberrant regulation of WNT activity has been linked to the development of cancer, 52 whereas insufficient WNT signaling appears to be involved in the pathogenesis of neurodegenerative diseases. 26,27,47,49 Interestingly, WNT signaling also has important effects in regulating bone homeostasis, with activation of the canonical WNT pathway resulting in increased bone mass and strength while inhibition of WNT signaling has the opposite effect. 3
Signaling by WNT proteins is tightly controlled by numerous intracellular and membrane components as well as extracellular molecules, all of which guide normal development and tissue homeostasis while preventing uncontrolled division of cells that would promote the development of cancer. Secreted inhibitors include Dickkopf family members, WNT inhibitory factor 1, secreted Frizzled related proteins, sclerostin, and NOTUM. 29 NOTUM is a highly conserved secreted feedback repressor of WNT activity that has been shown to suppress WNT signaling. 15,18,19,51,76 Secreted WNTs require a posttranslational lipid/palmitoleoylate modification for receptor binding and activity. 97 NOTUM inactivates WNT proteins by hydrolyzing these palmitoleoylate adducts in the extracellular space 29 and thereby influences developmental processes. 97
Targeted gene disruption in mice is a powerful tool for gaining important insights into the molecular mechanisms underlying development, as well as disease pathogenesis, diagnosis, prevention, and treatment. In analyzing over 4600 knockout mouse lines in a high-throughput phenotyping program, 92,94 we have discovered numerous pathology phenotypes that have helped elucidate fundamental processes in biology. 1,6,12,17,24,41,50,53,55 –57,59,62,63,66,78 –88,96 The pleotropic phenotypes we observed in NOTUM-deficient mice indicate an essential (nonredundant) role for NOTUM in the development of teeth and kidneys in adult mice. This report will focus on the lesions that develop in the incisor and molar teeth of Notum-/- mice. We also briefly address the effects of NOTUM deficiency on kidney development. Our findings demonstrate the value of including routine histological evaluation of teeth when phenotyping genetically engineered mice. 65
Materials and Methods
Mouse Production
The methods used for gene trapping in embryonic stem (ES) cells, identification of trapped genes using Omnibank® Sequence Tags (OST), characterization of retroviral gene-trap vector insertion site, and reverse transcription polymerase chain reaction (RT-PCR) analysis of knockout and wild type transcripts are published. 92 Gene trapping was performed using strain 129S5SvEvBrd-derived embryonic stem (ES) cells obtained from the Omnibank library. 93 OST172035 was selected from the Omnibank library based on a BLAST search of all available sequence tags using the Notum genomic interval as a query. The mutation in this clone was determined to be intragenic using inverse genomic PCR as previously described. 23 Briefly, oligonucleotide primers complementary to the gene trap vector were used to amplify the vector insertion site from clone OST172035, which was then compared to mouse genome sequence assemblies to localize the insertion within coding exon 1 of the Notum gene. The gene trapped ES cell clone was microinjected into C57BL/6-Tyr c-Brd (albino) blastocysts to generate chimeric animals which were bred to C57BL/6-Tyr c-Brd (albino) females, and the resulting heterozygous offspring were interbred to produce homozygous gene deficient mice, being designated as Notum Gt(OST172035)Lex which will be abbreviated as Notum-/- in this report. The knockout second generation filial (F2) mice used in phenotyping studies were produced by intercrossing the first generation filial (F1) heterozygous knockout (-/+) offspring of chimeric founder parents and were therefore of mixed C57BL and 129 genetic background. Using the albino variant of C57BL/6 mice (C57BL/6-Tyr c-Brd ) permits simple visual recognition of chimeric offspring, because they have dark eyes and patches of dark hair that derive from stem cells from the agouti 129S5/SvEvBrd. Genotypes of offspring were determined by quantitative PCR as previously described. 23 Briefly, DNA isolated from tail biopsy samples was assayed by quantitative PCR for the neo gene which is present in the VICTR48 gene trapping vector used to generate the mutation described in this study. In all studies reported here, mutant mice were compared directly with their wild-type littermates used as negative controls.
Mouse Husbandry and Testing
Mice were housed in micro-isolator cages within a barrier facility at 24°C on a fixed 12-hour light and 12-hour dark cycle and were provided acidified water and Purina rodent chow # 5001 (Purina, St. Louis, MO) ad libitum. All procedures involving use of live mice were conducted in conformance with Lexicon’s Institutional Animal Care and Use Committee guidelines that are in compliance with the state and federal laws and the standards outlined in the Guide for the Care and Use of Laboratory Animals (National Research Council, 1996). Quarterly sentinel surveillance showed no evidence of pathogenic rodent viruses, Mycoplasma, or Helicobacter species in the Lexicon Pharmaceutical source colonies.
Histopathology
For histology, a minimum of 10 knockout mice (5 males and 5 females) and 10 age-matched wild type littermate control mice (5 males and 5 females) at 3 time points (5, 15, and 40+ weeks of age) were euthanized and then fixed bycardiac perfusion with 10% neutral buffered formalin. Selected tissues (to include heart, skeletal muscle, tongue, lungs, trachea, thyroid gland, liver, kidney, adrenal gland, salivary glands, lymph nodes, white adipose, brown adipose, aorta, thymus, spleen, pancreas, stomach, duodenum, jejunum, ileum, cecum, colon, urinary bladder, skin, brain, eyes, nose, teeth, ear, bone, bone marrow, testes, epididymis, prostate gland, seminal vesicles, vas deferens, or uterus, ovaries) were collected and immersed in 10% neutral buffered formalin for an additional 48 hours except for eyes, which were removed and fixed by immersion in Davidson’s fixative (Poly Scientific, Bay Shore, NY) overnight at room temperature. After overnight fixation, bones and heads were transferred into a decalcifying/fixation solution consisting of a mixture of formic acid and formaldehyde (Cal-Rite, Thermo Fisher Scientific, Waltham, MA), which allows simultaneous decalcification and fixation. All tissues were embedded in paraffin, sectioned at 4 µm, mounted on positively charged glass slides (Superfrost Plus, Thermo Fisher Scientific, Waltham, MA), and stained with hematoxylin and eosin (HE) for standard histopathologic examination.
Results
Notum-/- mice appeared grossly normal at birth but body weight and growth measurements indicated that Notum-/- mice became slightly leaner than their wild-type littermates with increasing age (data not shown). The only pathology phenotypes observed in Notum-/- mice consisted of developmental defects involving the kidneys and teeth. All other tissues appeared to be normal on histological examination. In genotyping 3391 offspring of heterozygous parental crosses at 2 weeks of age, we discovered evidence of decreased viability of knockout mice. Instead of the expected Mendelian ratio of 848 wild type: 1696 heterozygotes: 848 homozygotes, there were 931 wild type: 1825 heterozygotes: 635 homozygotes, which represented a 32% deficit of homozygous mice (P = 3 × 10-16 by chi-square test). Notably, Notum-/- mice over 2 weeks of age showed no increase in mortality. However, in evaluating several cohorts in drug studies we discovered that 23/89 (26%) Notum-/- mice displayed unilateral kidney agenesis. Either right or left kidneys could be missing, with increased individual kidney weights being observed when only 1 kidney was present (data not shown). In affected animals, no histologic abnormalities were detected in the solitary kidneys. Ureters were not examined at autopsy or by histology. In contrast to the incomplete penetrance of renal agenesis, we observed dentin dysplasia in all Notum-/- mice (100%).
Dentin defects in both incisor and molar teeth were characterized by a markedly disrupted odontoblast layer, with replacement of dentin tubules by thick irregular trabeculae of osteodentin-like stroma containing variable numbers of entrapped cells in lacunae. In both incisors and molars, disruption of odontoblasts and dentin formation depended on both the location and the stage of dentin development. Normal dentin architecture in wild type mice was characterized by parallel dentin tubules that extended from the dentin-enamel junction on a base of linearly arranged odontoblasts in the dental pulp (Fig. 1). This contrasted with findings in Notum-/- mice, in which both molar and incisor teeth retained a relatively thin layer of mantle dentin but much of the underlying dentin was disorganized and atubular. In the incisors, dentin dysplasia was restricted to the lingual and labial quadrants of teeth (Figs. 2 and 3), whereas the odontoblasts and dentin on lateral and medial quadrants were generally normal (Figs. 2 and 3). At higher magnification, the regular arrangement of tubular predentin and dentin was produced by a relatively uniform layer of odontoblasts (Fig. 4). Normal dentin mantle was present adjacent to the enamel protein layer but disorganized dentin surrounded and entrapped odontoblasts at the lingual and labial quadrants (Figs. 5 and 6). As the incisors continued to mature (Fig. 6), tubular dentin formation often resumed on the interior dentin surface, leaving a linear band of disrupted dentin and trapped odontoblasts. In maturing normal teeth, a circumferential layer of dentin was formed and the epithelial elements on the surface of the labial quadrant were replaced by periodontal ligament attachments (Fig. 7). In contrast, variable amounts of irregular dentin with entrapped odontoblasts were formed within the lingual quadrant of incisors in Notum -/- mice (Figs. 8 and 9). In many cases, a layer of enamel epithelium or even ameloblasts was retained on the lingual surface of incisor teeth (Figs. 8 and 9). In some cases, this epithelial layer was complete and prevented the formation of periodontal attachments to bone (Fig. 8). Sometimes the epithelial layer on the lingual surface produced a thin layer of enamel (Fig. 8) while in others there was only a small cluster of epithelial cells with minor localized disruption of toot attachments (Fig. 9).
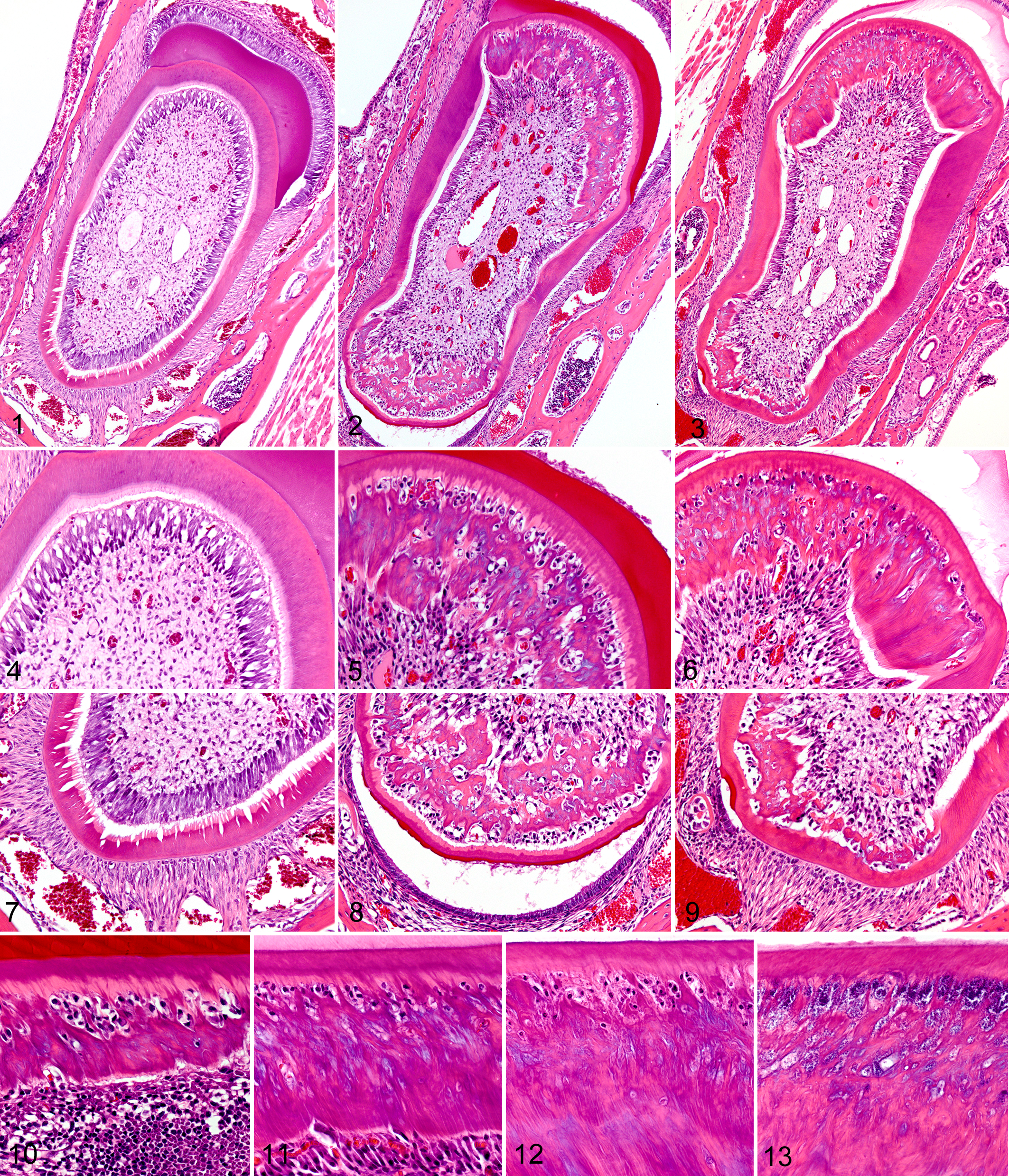
In all teeth, defective dentinogenesis appeared to occur after the thin initial layer of mantle dentin was formed, and the progression of the lesion could be tracked along longitudinal sections of the incisor teeth (Figs. 10–13). Initial disruption of the odontoblast layer first became evident at approximately the same stage of development where enamel production began. The earliest lesions in incisors consisted of increased collagen fiber deposition in the subodontoblastic dental pulp; with the rapid accumulation of irregular dense collagenous material separating, disrupting, and eventually isolating odontoblasts that remained attached to the thin layer of predentin underlying the mantle dentin (Fig. 10). This defective dentin was characterized by a superficial layer of loose disorganized mineralized dentin matrix with open spaces containing trapped odontoblasts and the debris of necrotic cells. A thick layer of irregular woven reactive dentin later formed under the spongiform layer as an irregular zone of newly differentiated odontoblasts formed on the interior surfaces of the collagen bands (Fig. 11). In both incisor and molar teeth, this second layer of odontoblasts eventually lined up and nearly normal arrangements of columnar odontoblasts and tubular dentin were restored at a fairly uniform distance from the enamel layer (Fig. 12). This process resulted in the development of a linear defect consisting of irregular osteodentin-like stroma trapped between normal mantle dentin and deeper layers of nearly normal tubular dentin. In areas near the tip of the incisor teeth, the odontoblasts/osteoblasts trapped in this linear defect underwent necrosis/apoptosis and the gaps in the dentin often contained bacteria (Fig. 13).
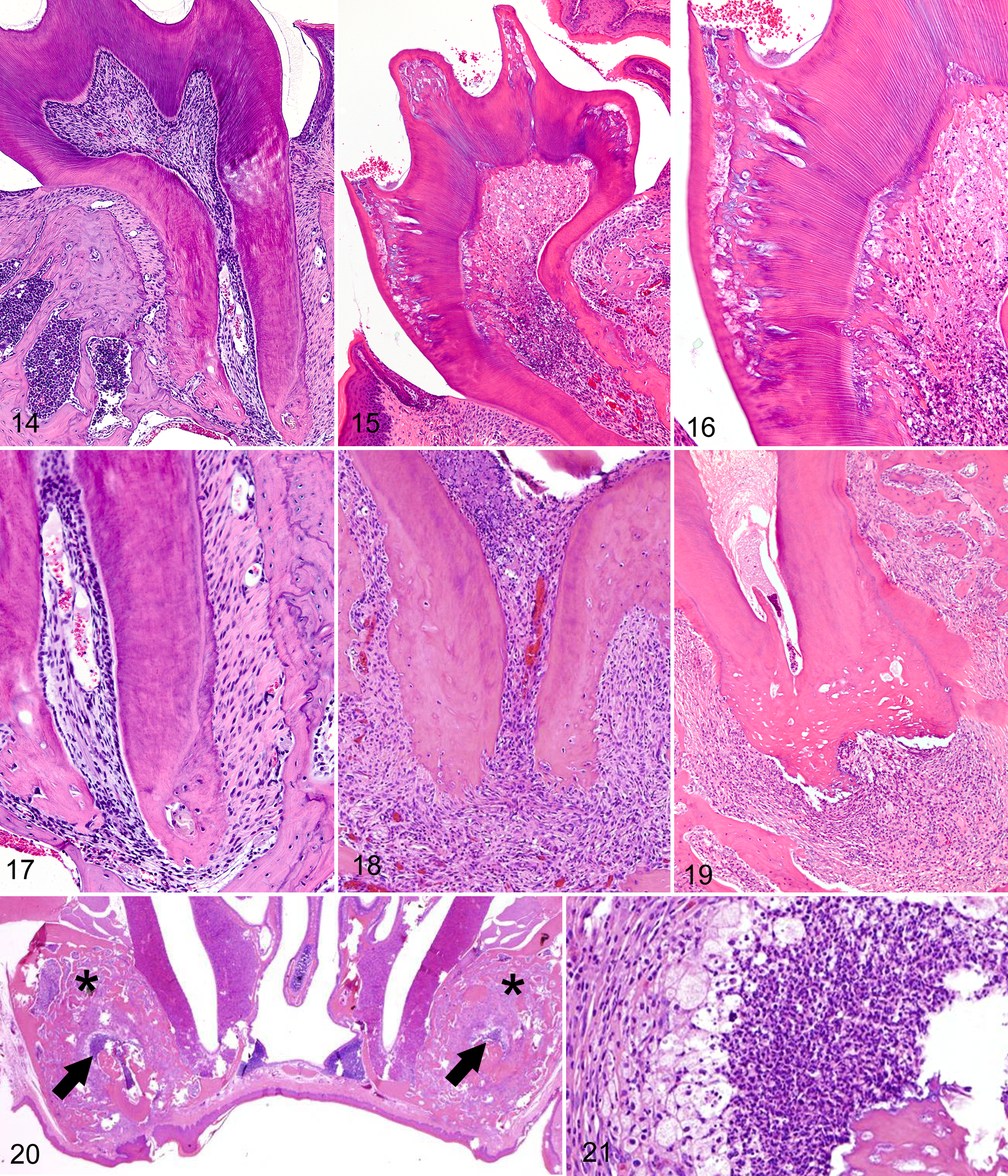
Despite the presence of severe dentin defects and some hypoplastic roots, all molar teeth in Notum-/- mice appeared to have normal crowns and enamel. Similarly, dentin dysplasia in molar teeth was also stage- and site-specific; in contrast to the diffuse regular arrangement of dentin tubules in normal molar teeth (Fig. 14), zones of severe dental dysplasia typically developed adjacent to mantle dentin in Notum-/- mice. Interestingly, dentin lesions in molar teeth were located under cusps, whereas defects were generally mild to absent under fossae and fissures (Fig. 15). Bacteria were often present within dentin defects at the cusps, and inflammation, fibrosis, and necrosis usually extended from the crown areas into the deeper pulp (Fig. 16).
The roots of molar teeth in wild type mice were attached to periodontal bone by relatively short and parallel arrangements of periodontal fibers (Fig. 17). In contrast, molar roots in Notum-/- mice were often shorter, thinner and more irregular in shape than those of wild type littermates (Fig. 18) and were often connected to more distant periosteum by thick irregular layers of periodontal fibers (Fig. 18). In young mice, the development of mild to moderate periodontal inflammation and osteolysis/proliferation was restricted to those teeth and roots containing necrotic pulp. In older mice, inflammation and ischemic necrosis were present in essentially all molar teeth, with associated breakdown of root dentin and periodontal attachments (Fig. 19). Extensive areas of bone remodeling and alveolar bone formation were associated with the decay of molar teeth (Fig. 20). Pulp necrosis was a prominent feature in all molar teeth of aged mice and suppurative inflammation with periapical abscess formation were typically present in these lesions (Fig. 21). In the aged mice (over 40 weeks), there was widespread disintegration of molar teeth, with the outer surfaces of roots being covered with irregular aggregations of dysplastic cementum which were in turn attached to abnormal periodontal ligaments composed of irregular thickened bands of collagen.
Discussion
Mutations involving components of the WNT/ß-catenin pathway have pleiotropic effects on a wide range of developmental processes. The phenotypes that we observed in teeth and kidney of Notum-/- mice can all be attributed to the effects of increased WNT signaling in the absence of NOTUM, and indicate that this secreted enzyme plays a critical role in the development of teeth and kidneys.
The continuous growth of rodent incisors permits detailed study of the highly conserved and complex interactions involved in tooth morphogenesis and enamel/dentin formation. Mouse models have proven useful in elucidating many processes involved in tooth development and the pathogenesis of dental diseases. 8,14,45 Tooth development involves complex reciprocal interactions between oral epithelium and dental pulp mesenchyme and provides a useful system to study organogenesis and mineralization defect diseases. 11,42,98 The terms used to describe distinct stages of tooth development are the dental lamina, bud, cap, bell, and crown formation stages. During the transition from cap stage to bell stage, some dental pulp mesenchyme cells undergo dentinogenesis, a process in which they differentiate into preodontoblasts, polarizing odontoblasts, secretory odontoblasts, and finally, terminally differentiated odontoblasts. 72 The secretory odontoblasts synthesize and secrete dentin extracellular matrix which includes many types of collagen and noncollagen proteins. Dentin sialophosphoprotein (DSPP) and dentin matrix protein 1 (DMP1) are noncollagen proteins that are markers of odontoblast differentiation and defects in these proteins in humans and mice result in dentin-related diseases, such as dentinogenesis imperfecta and dentin dysplasia. 42
Inherited dentin defects in humans have been divided into 5 types, including 3 types of dentinogenesis imperfecta and 2 types of dentin dysplasia. 68 Dentinogenesis imperfecta sometimes occurs in association with osteogenesis imperfecta, which is generally caused by mutations in the genes encoding type I collagen (COL1A1 and COL1A2). 4 However there are many other genes involved in the posttranslational modification and assembly of collagen, and mutations in these genes can also result in bone disease occasionally accompanied by defective dentin. Further classification of dentin dysplasia is based on the most severely affected areas of the tooth, with type I primarily involving the tooth roots (DDI; radicular dentin dysplasia) and type II affecting the tooth crowns (DDII; coronal dentin dysplasia). 32,89 The underlying genetic defects in DDII involve mutations in DSPP, which encodes the major noncollagenous dentin proteins (DSPP). Significantly, the genetic etiology of DDI (OMIM#125400) has not been identified. 4,32 In humans, DDI is characterized by normal appearing crowns but with defective dentin development, short roots (resulting in hypermobility and premature loss) and frequent dental abscesses. 4 Histologically, the enamel and the mantle dentin appear to be normal but deeper layers of dentin are atubular and irregularly organized. 75
Even though DDI generally shows an autosomal-dominant mode of inheritance in humans, 32 our findings in Notum-/- mice raise the possibility that defects in the WNT/ß-catenin pathway could be involved in the pathogenesis of DDI in humans. Support for this hypothesis is provided by the numerous similarities between the dental lesions in Notum-/- mice and those reported in DDI. These include normal-shaped crowns, normal mantle dentin with underlying zone of dentin dysplasia, short blunt roots, hypermobile teeth, and frequent abscesses. The extent of reactive periodontal alveolar bone formation in young mice was not as severe as in older mice, suggesting that the bone changes were secondary to pulp necrosis and tooth decay (occurring at a relatively older age) rather than vice versa. The periodontitis combined with the short narrow roots of molar teeth in Notum-/- mice would likely contribute to tooth mobility, which would also result in disorganization of the periodontal ligaments and increased reactive alveolar bone formation. Because the blood vessels that supply the tooth pulp in molars must pass through a narrow apical foramen, and because vessels in the tooth pulp consist of thin-walled capillaries that are easily compressed by edema or exudate, ischemic necrosis of tooth pulp could be exacerbated by periodontal edema and inflammation. The severe periodontal inflammation arising as a consequence of the dentin dysplasia was believed to be the most likely indirect cause of the lean phenotype observed in mature Notum-/- mice (data not shown).
Taken together, our findings suggest that the severe lesions that develop in teeth are the result of a primary developmental defect in dentinogenesis rather than compromised integrity of the periodontal membrane, cementum, or alveolar bone. The loss of dentin integrity compromises the strength of the enamel, making the tooth prone to fracture and infection. In addition, it is possible that the formation of excessive amounts of abnormal dentin and fibrosis in affected teeth compromises the vascular supply to the dental pulp cavity of molar teeth. The apparently reduced diameter and length of molar roots is another factor that could contribute to the prominent changes in periodontal bone and connective tissues because increased movement of teeth during mastication with consequent physiological loading would produce mechanical strains that stimulate osteoblasts/osteocytes and bone formation.
The pleiotropic effects associated with NOTUM deficiency in these mice reflect the essential, nonredundant roles of NOTUM on WNT protein-mediated cell-to-cell signaling during development of teeth and kidneys. The complex process of tooth development is highly conserved in vertebrates and involves the integrated actions of several different pathways, including WNT/β-catenin 71,91 and transforming growth factor β (TGFβ) 44,61,69,100 signaling. Many studies support an essential role for the canonical WNT signaling in tooth development (reviewed by Liu and Millar 40 ).
During tooth morphogenesis, canonical WNT ligands and signaling regulators mediate the epithelial-mesenchymal interactions. Their importance in the tooth development process is supported by studies demonstrating that tooth development is blocked with complete disruption of canonical WNT signaling by β-catenin deletion. 39 WNT/β-catenin mediated activation of Fgf4 is necessary for developing teeth to transition from the bud to the cap stage. 101 Conversely, increased β-catenin signaling in the oral epithelium results in ectopic tooth formation. 28,39 Multiple WNT ligands, including WNT-3, -4, -5a, -6, -7b, -10a, and -10b, are expressed in developing teeth, with loss-of-function experiments suggesting that WNTs regulate the maturation of dental mesenchymal stem cells into dentin-secreting odontoblasts. 95 WNT signaling promotes early tooth development, whereas excessive WNT attenuates late tooth development. 64,91 WNT signaling also controls dentin thickness, 37 and WNT10b specifically regulates odontoblast differentiation and expression of DSPP. 90
The time- and site-specific distribution of dentin dysplasia lesions in Notum-/- mice suggest that NOTUM’s inhibitory effects on WNTs help mediate delicately balanced processes of odontoblast differentiation and orientation. Mesenchymal stem cells first differentiate into odontoblasts under the influence of the inner enamel epithelium, with differentiation starting at the cusp tips and proceeding downward to cervical and intercuspal directions. Interestingly, dentin dysplasia in Notum-/- mice also begins at cusps. The ameloblast basement membrane is important for normal polarization and differentiation of the odontoblasts and was proposed to serve as a reservoir of signal molecules. 61 Signals from TGFβ/BMP (Transforming growth factor β/Bone morphogenetic protein) family members have been implicated in odontoblast induction. 60 Odontoblasts are prevented from terminal differentiation and are unable to deposit dentin when the TGFβ/BMP signaling pathway is conditionally knocked-down (gene Smad4). 35
Until recently, it was believed that neural crest cells were the primary source of dental mesenchymal stem cells that differentiate into pulp cells and odontoblasts. 8,99 However, recent findings indicate that a significant proportion of the mesenchymal stem cells in the dental pulp that can differentiate into odontoblasts are derived from peripheral nerve-associated glia (Schwann cell precursors and Schwann cells). 31 Regardless of their source, WNT reporter activity in odontoblasts supports a role for WNTs in the process of differentiation. 71
The dentin dysplasia in Notum-/- mice was characterized by irregular dense bands of collagen surrounding cells trapped in lacunae. Because of its histological similarities to bone, this dysplastic dentin might best be described as osteodentin. These findings suggest a role for NOTUM in determining whether stem cells differentiate into odontoblasts or osteoblasts. Enhanced canonical WNT signaling directly stimulates Runx2 gene expression, 16 and RUNX2 has important roles in tooth development and in the transdifferentiation of odontoblasts into osteoblasts. 9,46 The osteodentin in Notum-/- mice was morphologically very similar to well-mineralized areas of dysplastic dentin that develop in Osr2-IresCre;Smad4fl/fl mice, 35 in which the loss of Smad4 in dental mesenchyme upregulates the canonical WNT signaling pathway (similar to loss of NOTUM in our mice). In SMAD4-deficient mice, odontoblasts are converted into osteoblasts, resulting in the replacement of dentin by ectopic bone-like material. 35 Three other independent conditional Smad4 knockout mice also exhibited impaired odontoblast maturation and dentin defects in both the crowns and roots of their molar teeth. 33 TGFβ1 is another factor involved in the differentiation of pulpal cells into odontoblasts. 13,44 Overexpression of active TGF-β1 in transgenic mice resulted in similar disruptions of the odontoblast and formation of irregular dentin containing a significant number of cellular inclusions. The incisors of these transgenic mice also retained a thin layer of relatively normal mantle dentin supported by areas of markedly abnormal atubular dentin. The coronal areas of their molar teeth were filled with disorganized dentin. 74 Taken together, these studies show that TGF-β/BMP signaling is required for odontoblast maturation and dentin formation in a stage- and site-dependent manner, 33 and suggest a role for NOTUM in modulating this process through the WNT pathway. Root dentin is also produced by odontoblasts, and the short irregular roots in Notum-/- mice resemble those comprising a critical diagnostic feature of DDI (radicular dysplasia) in humans. Root formation follows crown development at the late bell stage. First, the inner and outer enamel epithelium in the cervical loop extend apically to form Hertwig’s epithelial root sheath, and then pulpal mesenchymal cells differentiate into odontoblasts to produce root dentin. 73 Again, WNT/β-catenin signaling plays an essential role in root morphogenesis. Conditional knockout of the β-catenin gene (Ctnnb1) in odontoblasts and cementoblasts of developing teeth resulted in rootless molars as well as incomplete incisors. 95 Since NOTUM deficiency increases WNT/ β-catenin signaling activity, reports that excessive WNT/ β-catenin signaling disturbs tooth-root formation 2 and that stabilization of β-catenin in dental mesenchyme increases formation of dentin and cementum 34 are particularly relevant.
In addition to dentin dysplasia, Notum-/- mice displayed a developmental phenotype involving the kidneys and we believe that the decreased viability of neonatal Notum-/- mice is most likely due to unobserved bilateral renal agenesis in some offspring. Similarly to tooth development, formation of the mammalian kidney is dependent on reciprocal inductive tissue interactions between different cell types, which in the kidney consist of the metanephric mesenchyme and the ureteric bud. 67 These tissue interactions result in outgrowth and branching of ureteric-derived tubules that differentiate into collecting ducts and in the formation of nephrons from mesenchyme-derived metanephric blastema. 58 However, the metanephric mesenchyme can only respond to signals from the ureteric bud within a narrow time window. 38 Several components of the WNT/ β-catenin signaling pathway are required for normal kidney development 54 and the critical role of WNT signaling in regulating kidney organogenesis has been demonstrated in several knockout mouse models. 25 For example, WNT9b is a primary ureteric bud-derived factor and WNT9b-deficient KO mice display renal agenesis and die soon after birth. 7 WNT4 is located downstream from WNT9b, and WNT4 knockout mice develop only vestigial kidneys. 70 WNT11 promotes development of collecting ducts, and both the numbers of nephrons and kidney size are markedly reduced in WNT11 deficient mice. 43
However, it is not only the loss of specific WNTs that can result in renal agenesis. For example, stabilization of β-catenin in the ureteric bud before the onset of kidney development is also associated with renal aplasia caused by disruption of ureteric branching. 5 While WNT signaling promotes normal branching of the ureteric bud, the BMP pathway appears to inhibit branching (reviewed in Karner et al 30 ). If the ureteric bud fails to reach the mesenchyme in this time window the kidney fails to develop. 20,21
The incomplete penetrance of renal agenesis that we observed in Notum-/- mice is most similar to that reported previously in low density lipoprotein receptor-related protein 4 knockout Lrp4-/- mice. 30 This observation is interesting because LRP4 is a multifunctional receptor involved in the integration of WNT and BMP signaling. 48 LRP4 inhibits LRP5/6-mediated activation of canonical WNT signaling. 36 LRP4 is a critical regulator of ureteric budding in both mice and humans. 30 In the developing kidney, Lrp4-/- mice show delayed ureteric bud formation, which results in unilateral or bilateral kidney agenesis. 30 Interestingly, LRP4 serves as a receptor for the WNT inhibitor sclerostin, which regulates bone metabolism in mice 10 and thus LRP4 is also involved in the regulation of bone mineral density. Because WNT signaling is upregulated in both LRP4- and NOTUM-deficient mice, these findings suggest that NOTUM and LRP4 may interact within the same WNT signaling pathways in the kidney and bone. In conclusion, our findings in Notum-/- mice support a significant role for NOTUM in modulating WNT signaling pathways that have pleiotropic effects on tooth and kidney development.
Footnotes
Acknowledgements
We wish to thank Mary Thiel, Kathy Henze, Ryan Vance, and June Wingert for their invaluable necropsy and histology support, and Jeff Liu, Andrea Y. Thompson, and Melanie K. Shadoan for essential contributions to skeletal phenotyping and pharmacology (all employees of Lexicon Pharmaceuticals).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
