Abstract
X-linked hereditary nephropathy (XLHN) in Navasota dogs is a spontaneously occurring disease caused by a mutation resulting in defective production of type IV collagen and juvenile-onset renal failure. The study was aimed at examining the evolution of renal damage and the expression of selected molecules potentially involved in the pathogenesis of XLHN. Clinical data and renal samples were obtained in 10 XLHN male dogs and 5 controls at 4 (T0), 6 (T1), and 9 (T2) months of age. Glomerular and tubulointerstitial lesions were scored by light microscopy, and the expression of 21 molecules was investigated by quantitative real-time polymerase chain reaction with selected proteins evaluated by immunohistochemistry. No significant histologic lesions or clinicopathologic abnormalities were identified in controls at any time-point. XLHN dogs had progressive proteinuria starting at T0. At T1, XLHN dogs had a mesangioproliferative glomerulopathy with glomerular loss, tubular necrosis, and interstitial fibrosis. At T2, glomerular and tubulointerstitial lesions were more severe, particularly glomerular loss, interstitial fibrosis, and inflammation. At T0, transforming growth factor β, connective tissue growth factor, and platelet-derived growth factor α mRNA were overexpressed in XLHN dogs compared with controls. Clusterin and TIMP1 transcripts were upregulated in later stages of the disease. Transforming growth factor β, connective tissue growth factor, and platelet-derived growth factor α should be considered as key players in the initial events of XHLN. Clusterin and TIMP1 appear to be more associated with the progression rather than initiation of tubulointerstitial damage in chronic renal disease.
Keywords
Naturally occurring juvenile nephropathies that cause progressive renal injury leading to end-stage renal disease have been described in humans and several canine families, 20,26,27,41 and the primary renal defect is unknown in the majority of cases. The nephropathy of Navasota (NAV) dogs, which is on a mixed background, is an X-linked hereditary nephropathy (XLHN) resulting in the inability to synthesize the type IV collagen that is present as a principal component of the normal glomerular basement membrane. 9,30 In affected males, the structure and composition of the glomerular basement membrane is altered, resulting in juvenile-onset proteinuria (typically between 2 and 6 months of age) and rapid progression to end-stage renal disease by 6 to 15 months of age. 26,28,30 Histopathologic changes occurring in advanced stages of XLHN are typical of chronic glomerular disease with involvement of the tubulointerstitium (TI) and include nonspecific changes, such as global glomerulosclerosis, tubular dilation, interstitial fibrosis, and inflammation. 28,30 Early microscopic changes in NAV dogs and their evolution during the course of disease have thus far only been partially described. 34 The first aim of this study focused on the description of changes in renal function and morphology in early stages of canine XLHN and to characterize the evolution of lesions over time.
Investigating the molecular mechanisms responsible for canine XLHN during its initial phases and progression can be highly beneficial in designing new strategies for the diagnosis and treatment of juvenile nephropathies. Therefore, the aim of the second part of the study was to explore the gene expression of a set of molecules involved in the pathogenesis of renal disease. The genes of interest were chosen on the basis of previous literature on canine XLHN 15,39 or reported in the progression of chronic tubulointerstitial damage (TID) in primary glomerular diseases. 18,32,33,38,43,45 Five groups of genes were examined: profibrotic and antifibrotic factors, matrix metalloproteinases (MMPs), adhesion molecules of tubular epithelial cells (TECs), and a marker of renal injury.
The first group of molecules included are known to be increased in advanced stages of TID and have been associated with extracellular matrix (ECM) deposition and/or implicated in the development and progression of renal fibrosis in human and experimental animal model renal diseases—for example, transforming growth factor β (TGFβ), connective tissue growth factor (CTGF), epidermal growth factor receptor (EGFR), and members of the platelet-derived growth factor (PDGF) family. 5,38,43,45
Hepatocyte growth factor and bone morphogenetic protein 7 represented the second group of molecules. They are characterized by an antifibrotic role and were described to be nephroprotective by inhibiting the TGFβ pathway. 32,44
Different members of the MMP family and their inhibitors were part of the third group. In fact, MMP2 and MMP9 upregulation was previously associated with the remodeling of ECM in NAV dogs. 39 The fourth group of genes included adhesion molecules constitutively expressed by normal TECs (β-catenin, N-cadherin, and E-cadherin) and were investigated to explore the mechanism of epithelial-to-mesenchymal transition in XLHN dogs. Finally, clusterin (CLUST), a glycoprotein found upregulated in several human renal diseases and in NAV dogs, was selected for its involvement in chronic renal injury. 11,15,23
Selected markers with significantly increased or decreased mRNA expression in affected dogs were also investigated at the protein level via immunohistochemical staining.
Materials and Methods
Dogs
Dogs were from a colony at Texas A&M University referred to as the NAV kindred, which is on a mixed background, in which a 10 base-pair deletion in the gene encoding the α5 chain of type IV collagen causes XLHN. Affected males (n = 10) were monitored to a standardized end point of serum creatinine (SCr) concentration >5.0 mg/dL (upper reference limit ≤1.2 mg/dL) or onset of uremic signs (anorexia and vomiting on 2 consecutive days), whereupon each dog was euthanized. Randomly selected unaffected male littermates (n = 5) evaluated under the same protocol served as age-matched controls. All studies of these dogs were approved by the Texas A&M University Institutional Animal Care and Use Committee.
Tissue Samples
Samples of kidney were collected at established time points: 4 (T0), 6 (T1), and 9 (T2) months of age. Samples were collected via an ultrasound-guided needle biopsy technique, 16 unless the dog reached the study end point at or prior to the time points, in which case samples were obtained immediately posteuthanasia. Tru-Cut renal biopsies were examined under a dissecting microscope with 20× to 40× magnification to verify presence of glomeruli. 29 At each time point, renal cortex was fixed in 10% buffered formalin and embedded in paraffin within 24 hours.
Renal tissue placed in RNAlater Solution (Life Technologies, Foster City, CA, USA) immediately after collection and stored at –80°C was available for most affected dogs (21 samples from 7 dogs at T0, 8 at T1, and 6 at T2) and from all control dogs.
Renal Histopathologic Evaluation
Three-micrometer-thick sections were cut from 2 levels of each biopsy 100 μm apart to represent different planes within the biopsy. Sections stained with hematoxylin and eosin, Masson trichrome, and periodic acid–Schiff were evaluated. Glomeruli and twenty 400× fields of TI were evaluated and scored according to a grading system (Suppl. Tables 1, 2). In addition, the number of obsolescent and cystic atrophic glomeruli were counted, and their sum was classified as “nonfunctional” glomeruli.
Clinical Pathology
Blood and voided midstream urine were collected in association with the renal biopsy. Serum creatinine and urine protein:creatinine ratio (UPC) were determined as previously described. 34 In addition, urine albumin concentration was determined by ELISA as previously described. 37
Gene Expression
RNA isolation from homogenized cortical renal tissue and reverse transcription were performed with the RNeasy Mini Kit (Qiagen, Valencia, CA, USA) and the High Capacity cDNA Reverse Transcription Kit (Applied Biosystems, Foster City, CA, USA), respectively. Expression of 21 genes was quantified with quantitative real-time polymerase chain reaction in a Light Cycler 480 Instrument (Roche Diagnostics, Basel, Switzerland) as previously described. 3 Primers (Suppl. Table 3) were designed with Primer Express 2.0 (Life Technologies). Calibration curves based on a 6-fold serial dilution (1 in 2) of a cDNA pool revealed PCR efficiencies near 2.0 and error values <0.2. Six candidate reference genes (BACT, GAPDH, TBP, HPRT, 1RPL32, 2RPL32) were selected and tested per the existing literature. 6,36 TBP was chosen as the reference gene based on its stable expression in healthy and pathologic samples. Moreover, its amplification efficiency was approximately equal to those of the target genes. The ΔΔCt method 31 was used to determine the relative quantification of the target mRNA.
Relative quantification values were analyzed with MultiD-Genex software. 4 Hierarchical clustering (HCL) and principal component analysis (PCA) were performed, adopting the following settings: mean center scaling, complete linkage, and euclidean for the evaluation of all samples and Ward algorithm and Manhattan distance for the evaluation of T0 samples.
Immunohistochemistry
Expression of selected proteins was investigated by immunohistochemistry in affected and control dogs to subjectively investigate whether protein expression corresponded with RNA expression. Evaluation consisted of qualitative assessment of cell types and location (cytoplasmic/membranous or nuclear). Additionally, immunohistochemistry for smooth muscle actin (αSMA) was performed to evaluate the role of myofibroblasts in the progression of fibrosis in XLHN. Briefly, paraffin sections (3 μm) were placed on surface-coated slides (Superfrost Plus). Slides were incubated at 37°C for 30 minutes before the immunostaining procedures. Staining was performed with an automatic immunostainer (Ventana Benchmark XT, Roche Diagnostics), which uses a kit with a secondary antibody with a horseradish peroxidase–conjugated polymer that binds mouse and rabbit primary antibodies (ultraViews Universal DAB, Ventana Medical System). All reagents were dispensed automatically except for the primary antibody, which was dispensed by hand. Tissues known to express the specific protein were used as positive controls. Negative controls were performed by replacing the primary antibody with antibody diluent. Antibody details are listed in Supplemental Table 4. For αSMA, 20 high-power fields (400×) were evaluated and scored: Grade 0: monolayer of αSMA-positive cells surrounding <50% of tubules Grade 1: monolayer of αSMA-positive cells surrounding >50% of tubules Grade 2: multilayer of αSMA-positive cells surrounding tubules Grade 3: areas of distortion of tubular architecture with multiple aggregates of αSMA-positive cells in the interstitium
Statistical Analysis
Data were analyzed with a commercially available statistical package (SAS 9.1; SAS Institute, Cary, NC, USA). Normal distribution was confirmed by the Shapiro-Wilk test and the equality of variance by the Levene test. A linear repeated mixed model was used to analyze morphologic and clinicopathologic parameters and gene expression over time, with the group (renal disease vs control dogs) as the fixed effect; time and animal effects were included as repeated and random effects, respectively. Interaction between group and time was inserted in the models, and the Bonferroni post hoc pairwise comparison test was used if statistically significant differences were detected. Spearman’s rho coefficient was used to assess the degree of association between all the variables included in the study. P < .05 was considered statistically significant.
Results
Morphologic and Clinicopathologic Findings
Among affected dogs, 2 subjects reached the study end point at 6 months of age, 4 dogs at 7 to 9 months, and the remaining 4 dogs at >10 months.
Control dogs were characterized by absence of noteworthy renal lesions and clinicopathologic alterations throughout the course of the study.
All XLHN dogs were proteinuric at T0 (UPC, 1.84–18.66; urine albumin, 9.8–290 mg/dL), despite the presence of negligible light microscopic morphologic changes (Fig. 1). Occasionally, minimal segmental mesangial hypercellularity and mild mesangial matrix expansion were present in focal glomeruli and classified as mild focal and segmental glomerulosclerosis. Both UPC ratio and albuminuria were positively correlated with mesangial matrix expansion (r = 0.73 and r = 0.76, respectively; P < .01).
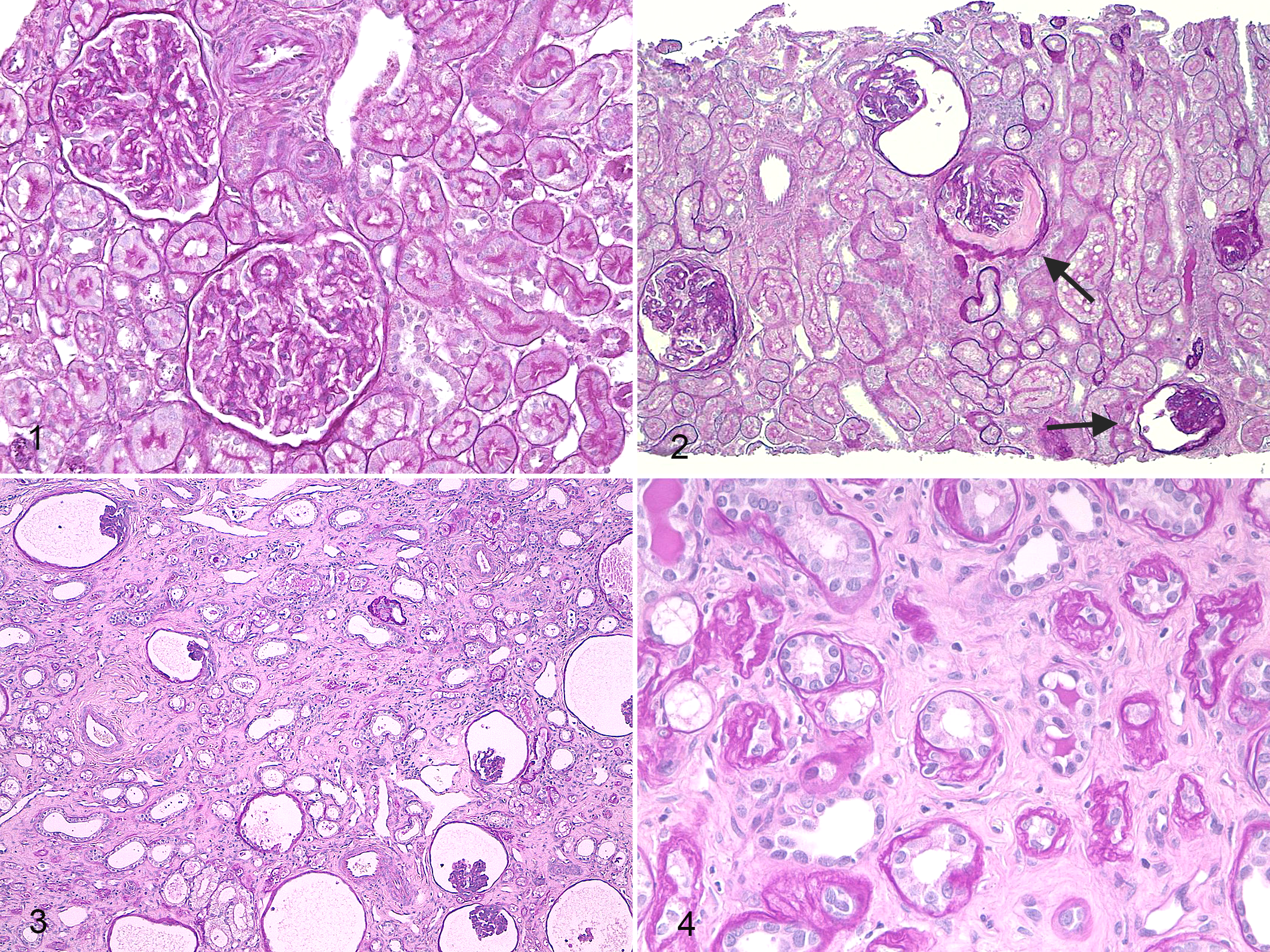
X-linked hereditary nephropathy, kidney, dog No. 3. Periodic acid–Schiff.
At T1, segmental to global matrix expansion and mesangial hypercellularity were present in most glomeruli, and a significant number of glomeruli were classified as “nonfunctional” (0%–80%; mean, 40.1%; Fig. 2). The TI showed multifocal lesions mainly consisting of degeneration and necrosis of TECs and mild to moderate interstitial fibrosis, frequently associated with atrophy of adjacent tubules. Impairment of renal function manifested by increased albuminuria and UPC and decreased serum albumin concentration was more prominent at T1 in affected dogs compared with controls and with XLHN dogs at T0 (P < .05). A mild increase in SCr concentration was also noted in affected dogs. Albuminuria and SCr were positively correlated with the percentage of “nonfunctional,” obsolescent, and cystic glomeruli, as well as with tubular atrophy and dilation, necrosis, and interstitial fibrosis (0.64 < r < 0.92; P < .05).
Overall, glomerular and TI lesions were more severe at T2 compared with T1. The percentage of “nonfunctional” glomeruli ranged from 67.1% to 100% (mean, 85.2%) with a greater prevalence of cystic atrophic glomeruli (mean, 48%) and lesser proportion of obsolescent glomeruli (mean, 36.5%; Fig. 3). Compared with controls, fibrosis, tubular atrophy, and inflammation (mostly lymphocytes, plasma cells, and macrophages) were more severe at T2 in affected dogs (Fig. 4). UPC and albuminuria were significantly greater at T2 (mean, 15.63 and 272.38 mg/dL, respectively; P < .01) in affected dogs compared with T1 and were positively correlated with the percentage of “nonfunctional,” obsolescent, and cystic glomeruli as well as with tubular atrophy and dilation, necrosis, fibrosis and the number of inflammatory cells (0.62 < r < 0.96; P < .05).
Significant morphologic and clinicopathologic findings in XLHN dogs compared with age-matched controls are shown in Figures 5 and 6, respectively.
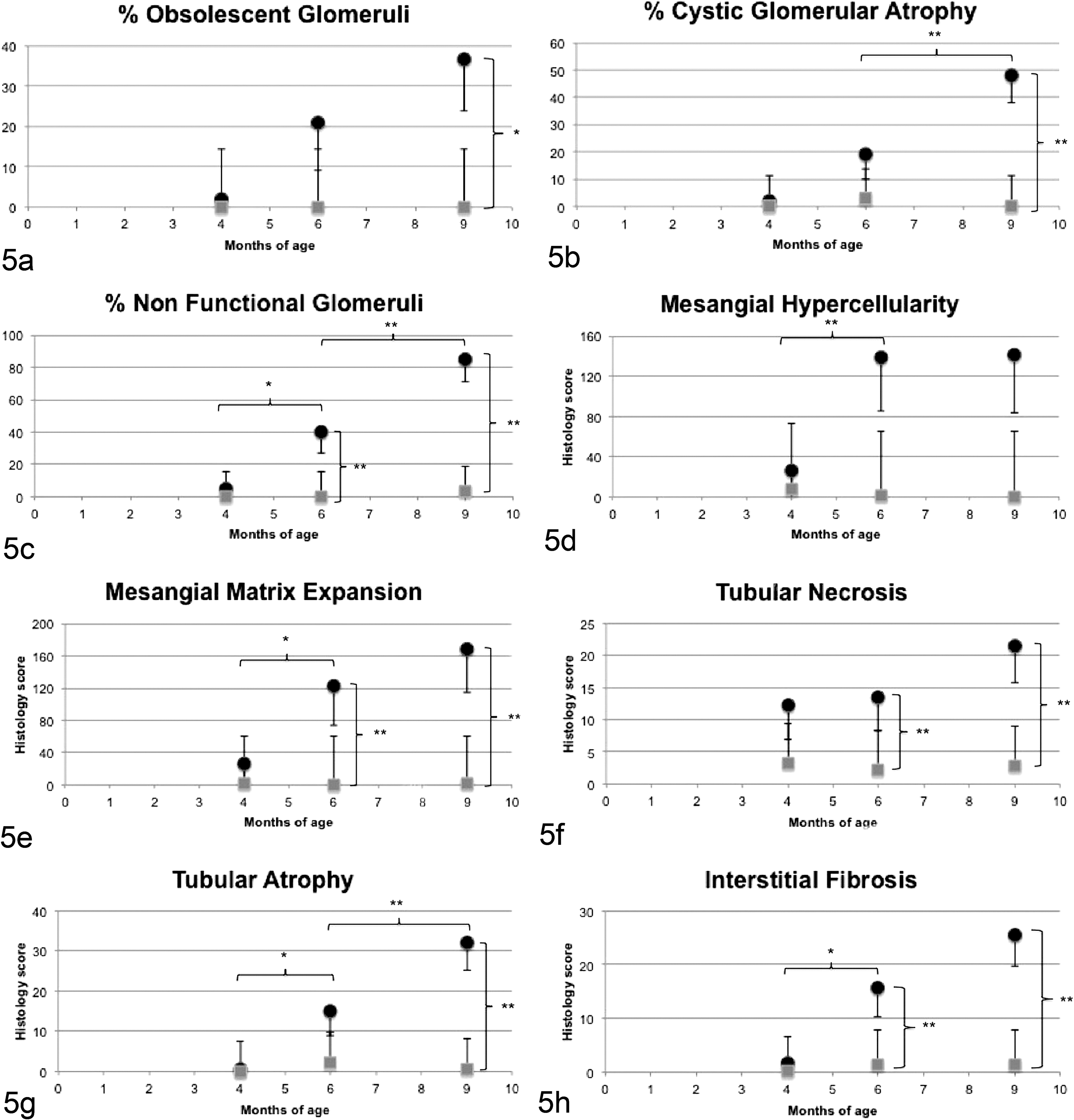
Morphologic parameters with significant differences between dogs affected with X-linked hereditary nephropathy (XLHN; •) and controls (▪). T0, 4 months of age; T1, 6 months of age; T2, 9 months of age. Bonferroni adjusted, * P < .05, ** P < .01. Control dogs were characterized by absence of noteworthy renal histologic lesions at all time points. Dogs with XLHN were characterized by absence of significant lesions at T0. At T1, a significantly increased number of “nonfunctional” glomeruli and mesangial matrix expansion were evident. Significant increases in interstitial fibrosis and tubular necrosis were detected. At T2, glomerular and tubulointerstitial lesions in dogs with XLHN were more severe, with a high number of obsolescent and cystic atrophic glomeruli, increased mesangial matrix expansion, tubular necrosis and atrophy, and interstitial fibrosis.
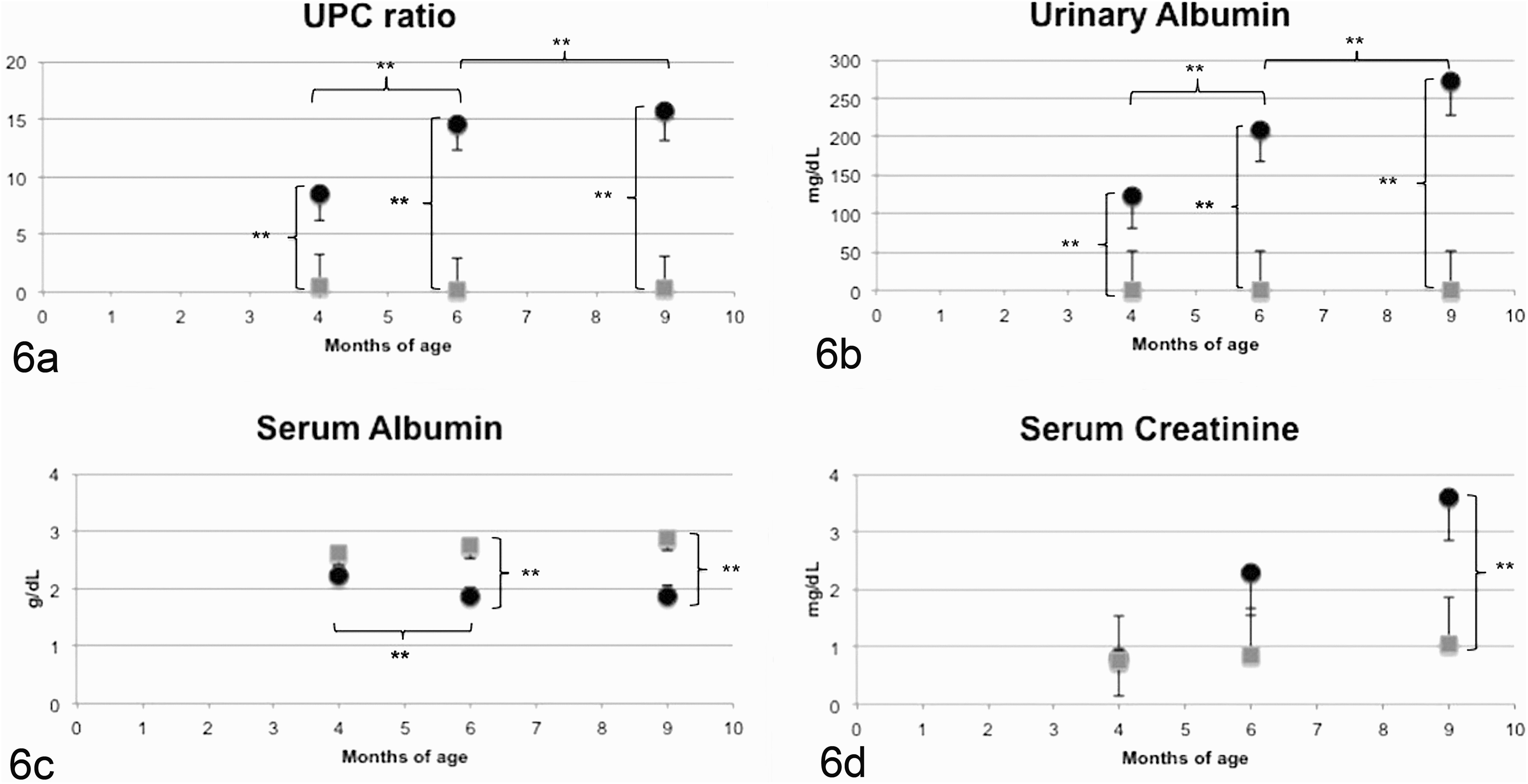
Clinicopathologic parameters with significant differences between dogs affected with X-linked hereditary nephropathy (XLHN; •) and controls (▪). T0, 4 months of age; T1, 6 months of age; T2, 9 months of age. Bonferroni adjusted, * P < .05, ** P < .01. Control dogs were characterized by absence of noteworthy clinicopathologic alterations at all time points. Dogs with XLHN showed a progressive increased in urine protein:creatinine ratio (UPC) and urinary albumin concentration. A significant decrease in serum albumin concentration was progressively observed in XLHN starting at T1. Serum creatinine was significantly increased in XLHN dogs at T2.
αSMA-positive cells consistent with myofibroblasts were observed surrounding tubules in affected and control dogs. In control dogs (at all time points) and XLHN dogs at T0, the renal tissue was mostly characterized by the presence of a monolayer of myofibroblasts surrounding <50% of the tubules. In T1 and T2 affected dogs, >50% of the tubules showed a myofibroblast lining, frequently in multilayer and/or in aggregates in areas where the interstitium was expanded and tubular architecture distorted. The αSMA immunohistochemical scores were positively correlated with the extent of fibrosis (r = 0.841; P < .01).
Gene Expression
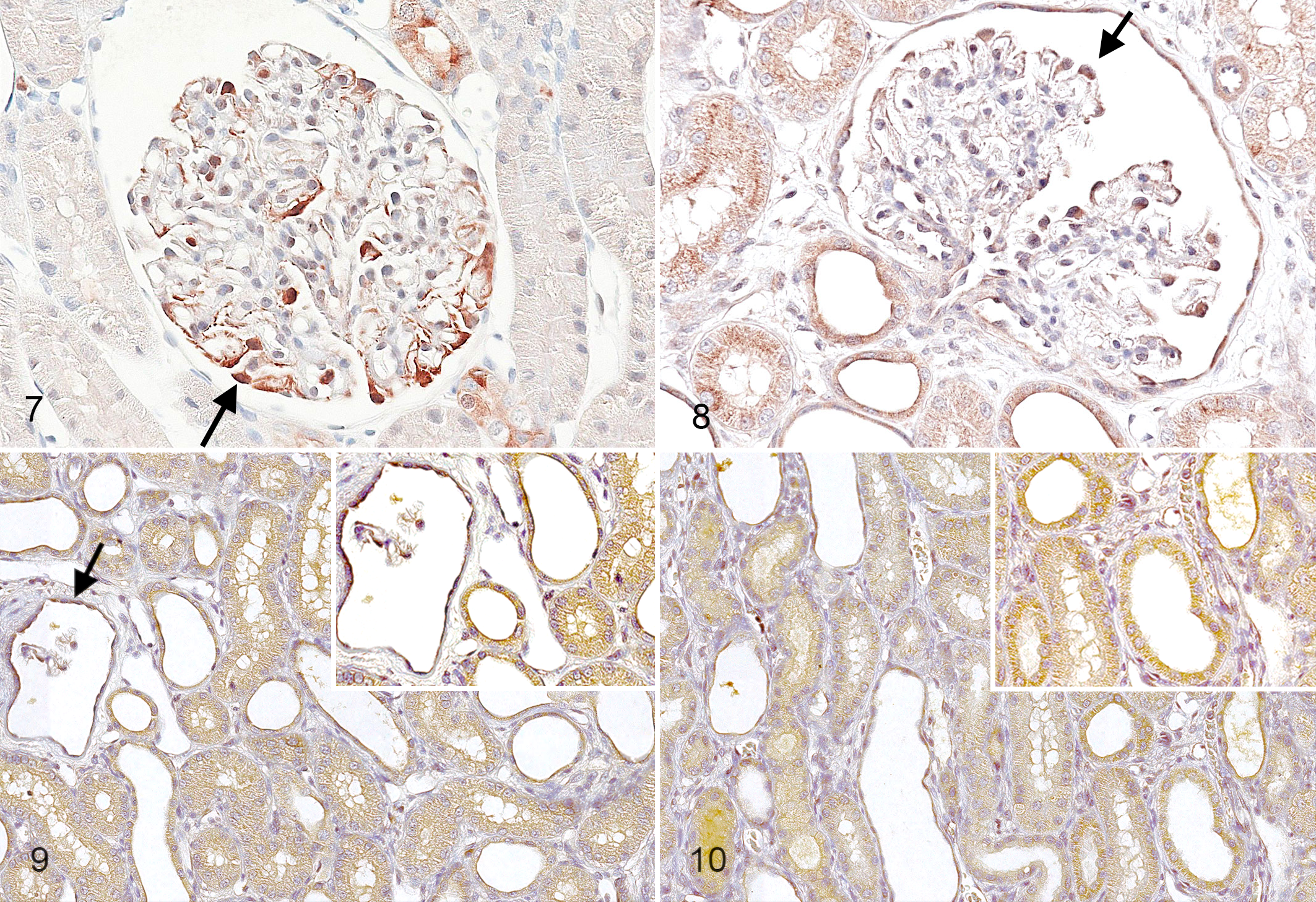
Based on relative quantification values of all target genes from all time points, the HCL tree and PCA differentiated XHLN from control dogs (Suppl. Fig. 1). Even when based on gene expression data from only T0, the HCL tree and PCA differentiated XHLN from controls dogs (Suppl. Fig. 2). The first 3 components of the PCA analysis accounted for a substantial fraction of the total variance in both instances (86% and 82%, respectively). When all target genes from all time points were analyzed, there were 3 biopsies (all from T0) where the PCA procedure resulted in incorrect grouping. These cases were adjacent to the border between the 2 groups and included 1 control dog biopsy that clustered with affected dogs and 2 affected dog biopsies that clustered with controls (Fig. 7). The first one, although from a control animal, was characterized by minimal multifocal TI changes, including epithelial cell degeneration and mild, focal fibrosis and inflammatory cell infiltration. In the kidney from the 2 affected dogs, TI lesions were absent, resembling normal renal tissue. Moreover, these affected dogs exhibited relatively slow progression of disease, reaching the study end point at 10 months of age.
X-linked hereditary nephropathy, kidney, dog No. 2, 4 months of age.
Among 21 candidate genes, mRNA levels of 8 genes were overall significantly higher in XLHN dogs compared with controls: TGFβ, CTGF, PDGFD, N-CADHERIN, CLUST, MMP2, MMP9, and TIMP1 (P < .05). In contrast, EGFR was lower in XLHN dogs (P < .01). The gene expression results for affected and control dogs are shown in Supplemental Tables 5 and 6.
Subsequently, genes were analyzed individually according to the different time points. TGFβ, CTGF, and PDGFRα were significantly overexpressed at T0 in affected dogs compared with controls (P < .05). TGFβ, TIMP1, and CLUST were significantly overexpressed at T1 and CLUST at T2 in affected dogs compared with controls (P < .05). Significant correlations among genes at different time points are shown in Supplemental Table 7.
Gene expression data and morphologic and clinicopathologic findings showing differences in XLHN dogs compared with controls were analyzed together to associate the expression of gene(s) with renal lesions and function at the different time points. The most interesting findings involved CLUST and TIMP1 expression. The two genes were positively correlated with glomerular mesangial hypercellularity and matrix expansion at all time points (0.56 < r < 0.69 for TIMP1 and 0.65 < r < 0.87 for CLUST; P < .05). Regarding TI changes, TIMP1 expression was positively correlated at all time points with the degree of fibrosis (0.66 < r < 0.74; P < .05) and tubular dilation (0.58 < r < 0.68; P < .05). CLUST expression was correlated with tubular necrosis at all time points (0.71 < r < 0.76; P < .05). At T2, CLUST and TIMP1 expression positively correlated with all histologic parameters (0.58 < r < 0.81; P < .05). Additionally, TIMP1 and CLUST expression positively correlated with UPC and urinary albumin at all time points (0.65 < r < 0.80; P < .05) and with serum creatinine at T1 and T2 (0.69–0.77; P < .05).
Protein Expression
TGFβ and CTGF immunostaining was detected diffusely in the cytoplasm of podocytes at all time points (Figs. 7, 8), with no difference observed between control and affected dogs. However, in cystic and obsolescent glomeruli, positive-staining podocytes were rarely lining the glomerular tuft but were commonly free within the urinary space. In the tubular compartment, TGFβ and CTGF were multifocally detected in the cytoplasm of TECs in affected and control kidneys at all time points (Figs. 7, 9).
For both CLUST and TIMP1, staining was absent in normal dogs at all time points and at T0 in XLHN dogs (Suppl. Figs. 3–6). However, at T1 and T2, affected dog kidneys were characterized by diffuse, mild positive cytoplasmic staining in TECs and parietal epithelial cells of the glomeruli (Figs. 9, 10).
Discussion
Naturally occurring inherited type IV collagen defects that cause progressive renal injury leading to end-stage renal disease have been described in humans (Alport syndrome) and several canine families. 20,27,28,41 NAV dogs represent a potential model in which to study the progression of canine juvenile nephropathies. 30 In this study, the availability of serial samples obtained in the early stages of disease permitted visualization of the sequence of events occurring in progressive XLHN.
Based on our findings, proteinuria is the earliest detectable change in XLHN and is the only parameter discriminating affected dogs from controls at 4 months of age. Histologic lesions (via light microscopy) are minimal at that age and are not discriminating until later in the disease. XLHN dogs eventually develop a mesangioproliferative glomerulopathy characterized by progressive loss of glomeruli that are mostly undergoing cystic glomerular atrophy. This primary and persistent glomerular injury is associated with secondary progressive tubular loss, interstitial fibrosis, inflammation, and myofibroblast activation/proliferation. With respect to the clinicopathologic data, the UPC as well as urinary albumin and SCr concentrations increased in parallel with severity of tissue injury. These are common features of chronic TID that progress independently from the primary cause of the renal damage. Thus, this study confirms that XLHN represents a good model to study chronic renal disease in dogs.
Cystic glomerular atrophy is frequently observed in canine juvenile nephropathies 20,27,41 and emerged as a peculiar feature of XLHN. The cystic dilation of glomeruli is the consequence of tubular obstruction, followed by increased intratubular pressure and compression of the glomerular tuft. However, the factor primarily responsible for tubular obstruction in XLHN is not known. Interstitial fibrosis likely contributes to this phenomenon, but cystic glomerular atrophy is not seen exclusively in association with renal fibrosis. Recent studies demonstrated that many renal disorders are characterized by the formation of atubular glomeruli. 7,8,10 According to these studies, the formation of atubular glomeruli would be a consequence of damage occurring at the glomerulotubular junction and might represent an essential step in the progression of pediatric nephropathies. 7,10,13 It was beyond the purposes of this study to examine the possible role of atubular glomeruli in canine XHLN, but the large number of cystic and atrophic glomeruli could indicate the presence of atubular glomeruli and thus identify a potential key feature in the pathogenesis of hereditary nephropathies in general.
The expression of a number of transcripts potentially involved in the pathogenesis of XLHN was investigated in this study. The entire data set obtained by gene expression analysis was used for preliminary HCL and PCA procedures that identified 2 separate well-defined groups: control and affected dogs. Progression of renal disease in NAV dogs is variable, with the standardized study end point occurring between 6 and 15 months of age. It would be of great interest to identify factors predisposing to or predicting faster progression of XLHN, but the number of dogs in the present study was too small to statistically explore an association between the mRNA expression profile and progression rate of XLHN. However, no difference was observed upon cursory examination of the gene expression data related to dog survival. Interestingly, upon examination of only samples at T0 by PCA and HCL, control and affected dogs were separated into 2 well-defined groups. This observation illustrates that gene expression is altered well before renal lesions become significant by light microscopy.
In the gene expression analysis, an unexpected observation was the variable expression in control dogs of some genes that were initially expected to be constant over time. However postnatal renal maturation must be considered, especially given that many of the studied molecules (ie, TGFβ, CTGF, EGFR) are reported to be involved in renal development in several animal species. 14,17,21,22,35
In the present study, increased expression of TGFβ and CTGF was detected by 4 months of age in dogs affected with XLHN and preceded light microscopic damage. TGFβ and CTGF protein expression was detected in podocytes, parietal epithelium, and TECs. Recently, podocytes have been reported as the first cellular elements that detect altered ultrafiltration, reacting with conformational changes and producing proinflammatory mediators with autocrine and paracrine effects. 12 A previous study in a mouse model of Alport syndrome described increased expression of TGFβ in podocytes. 40 In that study, the authors hypothesized an effect of TGFβ in the development of glomerular changes by stimulating mesangial cell proliferation and matrix deposition. In our study, the overexpression of TGFβ mRNA, possibly by podocytes, and the presence of mesangial cell proliferation occurring in initial stages of XLHN lend support to this hypothesis. TGFβ and CTGF are known as profibrotic factors. Surprisingly, the present study found their mRNA expression not positively correlated to fibrosis, although the lack of significant overexpression of TGFβ in late stages of XHLN is consistent with data by Greer and colleagues. 15 In late stages of XLHN, tubular atrophy and the marked loss of glomeruli are responsible for an overall reduction in the number of viable cells (ie, TECs and glomerular epithelial cells) that might explain the reduction in TGFβ and CTGF expression observed in this study.
An upregulation of TIMP1, in RNA and protein expression, was observed in XLHN dogs, and its expression was positively correlated with progressive glomerular loss and TI lesions. A profibrotic activity for this enzyme has been proposed in the progression of renal fibrosis, since its inhibitory activity on MMPs would lead to the accumulation of ECM. 1 In this study, the positive correlation of TIMP1 expression at all time points with the degree of fibrosis and with MMP2 expression (0.62 < r < 0.81; P < 0.05) is consistent with a profibrotic role of this enzyme in the progression of chronic TID. Similarly, studies in humans and dogs with chronic liver disease associated TIMP1 overexpression with marked fibrosis. 25,42 The authors hypothesized that profibrotic activity of TIMP1 is not limited to MMP inhibition, but TIMP1 could also promote ECM accumulation through an antiapoptotic activity on hepatic stellate cells. 42 By translating that information into renal pathology, the mechanisms through which TIMP1 could enhance fibrosis might include inhibition of MMP activity and reduced apoptosis of interstitial (myo)fibroblasts.
In the present study, RNA and protein expression of CLUST was higher in canine XLHN at later stages of disease compared with controls, which is in agreement with the results reported by Greer and colleagues. 15 The increased expression of CLUST in affected dogs was progressive and positively correlated with worsening renal damage. CLUST is constitutively expressed in almost all mammalian organs and interacts with a wide range of molecules. 23 Increased expression of CLUST was found in several renal disease models, suggesting that it might have an important role in the pathogenesis of renal injury. Nevertheless, the role of CLUST in the progression of TID is still unclear, and some authors have even hypothesized a nephroprotective activity of this molecule via attenuation of interstitial fibrosis. 24
Among the examined PDGF and PDGFR isoforms, higher PDGFRα expression was detected in XLHN dogs compared with controls only at 4 months of age. Furthermore, the positive correlation between PDGFRα and the profibrotic CTGF (0.63 < r < 0.76; P < .05) suggests crosstalk between the 2 molecules in the progression of interstitial fibrosis. Given these findings, we can speculate that PDGFRα expression and activation play a prominent role in the beginning of the fibrotic process, recruiting fibrocytes as seen in pulmonary and hepatic fibrosis, 2,19 but more data are needed to explain the role of this receptor in the pathogenesis of XLHN.
Conclusions
In this study, we characterized morphologic and clinicopathologic changes occurring in XLHN of NAV dogs, a naturally occurring model of juvenile nephropathy.
TGFβ, CTGF, and PDGFRα were identified as potential key players in the initial events of XLHN. CLUST and TIMP1 appeared to be more involved in the progression of chronic TI damage. A limitation of this study is that gene expression was performed on homogenized cortical renal tissue and thus included multiple cell types. The immunohistochemical staining was helpful in identifying the cells expressing the different genes, but further investigations are needed to better clarify the role of these molecules in the pathogenesis of XLHN and juvenile nephropathies. However, the opportunity to study an established and reproducible animal model offers certain important advantages. One of these is the availability of samples from early stages of disease that are typically not possible to collect in routine diagnostic settings. Furthermore, examination of serial samples permits analysis of the evolution of renal disease over time.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: S.L.B. received a PhD grant from the University of Padova, Italy.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
