Abstract
Endocardiosis or myxomatous degeneration of the cardiac valves is a well-described age-related change in humans and dogs. Lesions consist of polypoid nodular proliferations of loose extracellular matrix and valvular interstitial cells, most commonly affecting the mitral valve. This entity has not been previously described in fish. Herein we report the appearance, location, and occurrence of valvular and mural endocardiosis in a retrospective survey of aging laboratory zebrafish. Endocardiosis was present in 59 of 777 fish (7.59%), most commonly affecting the sinoatrial (34 fish; 57.6%) and atrioventricular (33 fish; 55.9%) valves. Lesions were more common in fish raised in recirculating water systems and fed commercial diets (52/230 fish; 22.6%) versus flow-through systems with fish fed semi-purified diets (4/234; 1.71%). Lesions were overrepresented in fish heterozygous for a mutant smoothened allele (34/61 fish, 55.7% vs 17/168, 10.1% wild type). There was no association between endocardiosis and intestinal carcinoids. Valvular endocardiosis is a significant age- and husbandry-related background finding in zebrafish and should be considered in the design and interpretation of research studies.
Introduction
Valvular endocardiosis consists of nodules of cellular and matrix proliferation and matrix degeneration that distort the cardiac valves. Major features include disorganized subendothelial proliferation and activation of valvular interstitial cells (VICs), which take on a myofibroblast phenotype. 1,3,13 Glycosaminoglycans and proteoglycans accumulate in the matrix, with fragmentation of fibrillar collagens and elastin, giving the matrix a basophilic, loose, and myxomatous appearance. The matrix degeneration affects the functional characteristics of the valve, causing it to be less stiff and more distensible (“floppy”). 1 Transforming growth factor beta signaling is critical in both humans and dogs, with matrix remodeling via activation of matrix metalloproteinases. 1 A leukocytic inflammatory component is distinctly absent. 5 As valvular lesions increase in size, they may interfere with valve competency, resulting in regurgitant flow and secondary myocardial compensatory changes. Endocardiosis is a common age-related lesion in several species, including dogs (canine myxomatous mitral valve disease), pigs, and humans. 1,3,7 Among laboratory animals, valvular endocardiosis has also been reported in rabbits, rats, and mice. 9,24 In humans, similar but distinguishable myxoid valvular lesions can also occur with carcinoid tumors and drugs (eg, fenfluramine and phentermine), via activation of serotonergic 5-hydroxytryptamine 2B receptors. 6,8,19
Zebrafish are increasingly utilized as a laboratory animal species in fields such as cardiovascular research. 15,16 A large number of genes are documented to be involved in normal valvulogenesis in zebrafish and when functionally perturbed, will cause abnormal development. 2,18,21,29 While normal and abnormal cardiac development is well documented in zebrafish, 16,18,20,25 there has been limited documentation of age-related lesions in the zebrafish heart. 26,28 To date, valvular endocardiosis has not been reported as an induced or spontaneous phenotype in zebrafish or any other fish species. Herein we report the histologic and demographic features of valvular and mural endocardiosis in aged laboratory zebrafish.
Materials and Methods
Fish cohorts with lesions reported here were from studies of spontaneous neoplasia, including studies of the influences of diet and husbandry. 27 All but 1 of the fish in cohorts with endocardiosis lesions were on an AB background line (http://zfin.org/ZDB-GENO-960809-7); that exception was Cologne (KOLN, http://zfin.org/ZDB-GENO-010725-1). Fish were evaluated at 12 (n = 1), 22 (n = 649), 24 (n = 114), 37 (n = 12), and 41 (n = 1) months of age. Additional fish examined as historical controls came from the archives of the Zebrafish International Resource Center (ZIRC) and from other studies of spontaneous and carcinogen-induced neoplasia; these fish were of a variety of wild-type and mutant lines from a variety of husbandry systems. In addition to the groups of zebrafish evaluated intensively for lesions in heart valves in studies of spontaneous neoplasia, we also examined 2000 fish from 56 separate cohorts, in which most of the fish were raised in recirculating systems with fluidized sand biofilters and fish were fed commercial diets containing fish meal. These fish varied from 6 to 41 months in age. We recorded the incidence and severity of endocardiosis in these fish. Experimental design and procedures conducted at all study sites were approved by each institution’s Institutional Animal Care and Use Committee and were consistent with the most recent Guide for the Care and Use of Laboratory Animals from the Institute of Laboratory Animal Resources, National Research Council.
Zebrafish were processed for histology by several different methods, depending on the source or study. For cohorts in which endocardiosis was studied intensively, fish were euthanatized in tricaine methanesulfonate (MS 222; Argent Laboratories, Redmond, WA) pH 7.4 in phosphate buffer. Fish were fixed in buffered zinc formalin for 24 hours and decalcified for 48 hours in Cal X II (formic acid/formalin; Fisher Scientific, Waltham, MA). Most of the fish were processed and embedded whole in paraffin, and 9 whole body step sections were cut between the middle of the lens of the left eye and the middle of the lens of the right eye. Three sections were placed onto each of 3 slides (9 sections total) and stained routinely with hematoxylin and eosin (H&E). For fish larger than 1 g in size (usually large females), fish were bisected sagittally using a razor blade, and both halves of the fish were processed and embedded in paraffin to be sectioned for histology.
For initial historical carcinogenesis studies at Oregon State University, the tail was removed, and an incision was made through the ventral abdominal wall from the heart to the anus to promote internal fixation. Fish were fixed in Bouin’s fixative for 24 hours. Fish were rinsed, dehydrated in a graded series of ethanol solutions, then embedded in paraffin. Sagittal step sections were cut from the fish’s left side. Three 4- to 6-micron sections were saved and placed onto a single glass slide, 1 section through the lens of the left eye, 1 just medial to the left eye, and 1 from midline. Sections were stained with H&E. For diagnostic cases submitted ZIRC at the University of Oregon, fish were routinely fixed in Dietrich’s fixative and decalcified overnight in 5% trichloroacetic acid. Fish were bisected for embedding by cutting sagittally, just to the left of midline, using a razor blade. The 2 halves were placed into a single cassette for paraffin processing and embedding. A single slide was made with a ribbon cut that highlighted structures near midline. Detailed histology protocols are available on the ZIRC website (http://zebrafish.org/zirc/health/diseaseManual.php). All images were obtained with an Olympus BX51 microscope and DP71 digital camera using cellSens Standard 1.6 imaging software (Olympus America, Center Valley, PA). Slides containing representative lesions have been scanned and can be viewed at the Penn State Zebrafish Atlas (http://zfatlas.psu.edu/).
Results
For all fish, individual valves were scored on a 0 to 3 scale for endocardiosis lesions. Normal heart with valves in situ is shown in Figure 1. Normal valves (grade 0) lacked loose or myxomatous matrix. The sinoatrial valve between the sinus venosus and atrium was distinctly muscular (Fig. 2). The atrioventricular (Fig. 3) and bulboventricular (Fig. 4) valves were thin and delicate, with a tapering shape. They had a regular and dense central cellularity with scant loose myxoid matrix (glycosaminoglycans). There was a thin layer of elastin on the atrial or ventricular side, respectively, with dense collagenous matrix on the ventricular or bulbar side. Low grade endocardiosis (grade 1) lesions consisted of small polypoid projections of a loose and myxomatous stippled basophilic matrix with low cellularity, covered by endothelium (Figs. 5, 6). Cells within the nodules were plump and spindle-shaped to stellate. These lesions were generally located at the margins or cusps of the valve leaflets. Intermediate grade lesions (grade 2) were larger, more numerous, and often coalescing (Fig. 7). Cells and stroma occasionally had a distinct whorling pattern. High grade lesions (grade 3) were florid and occasionally wart like (verrucous), with significant distortion or destruction of the valve, and these often affected multiple leaflets (Figs. 8, 9). The matrix occasionally had a more coarse and collagenous appearance.
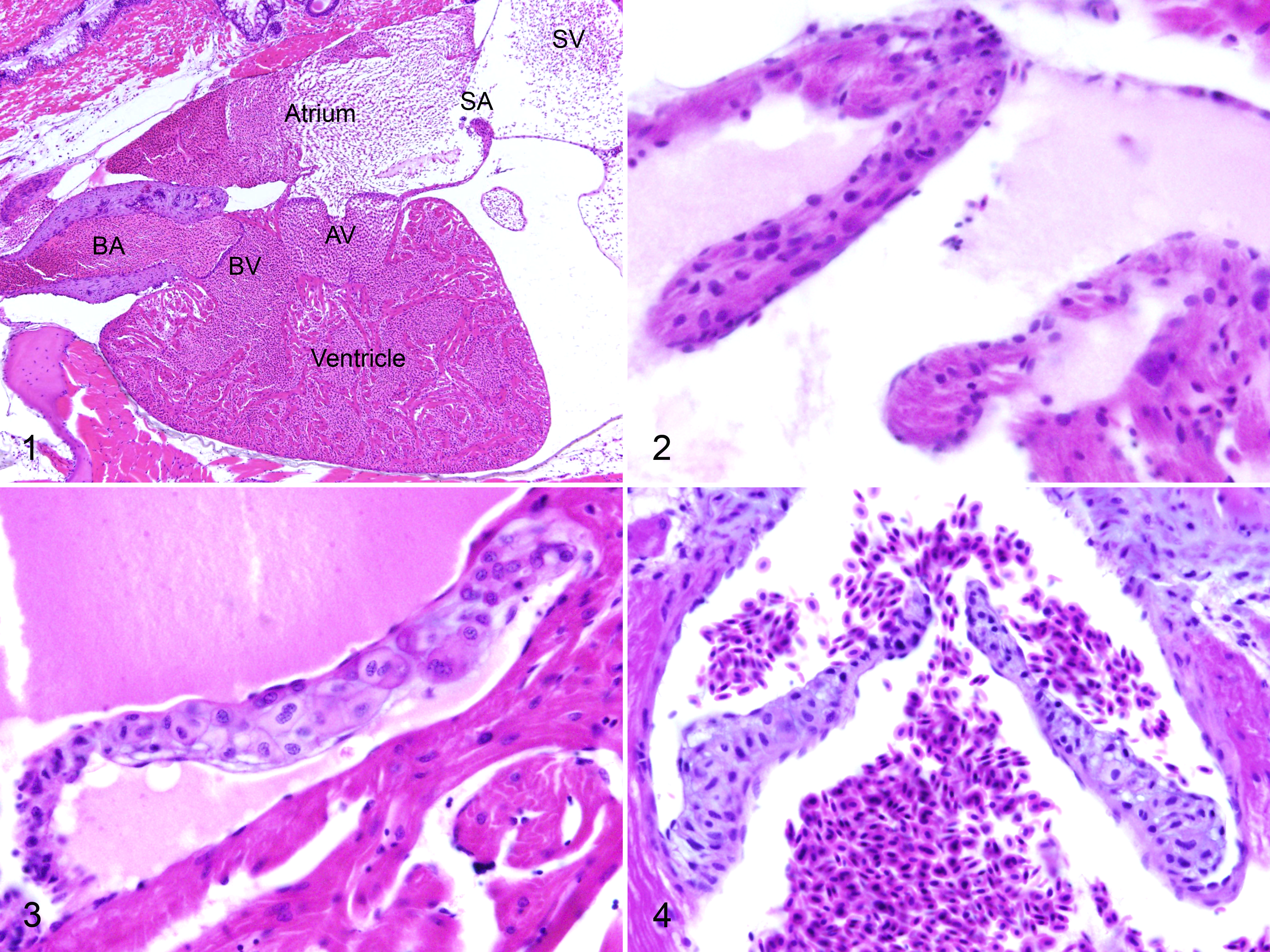
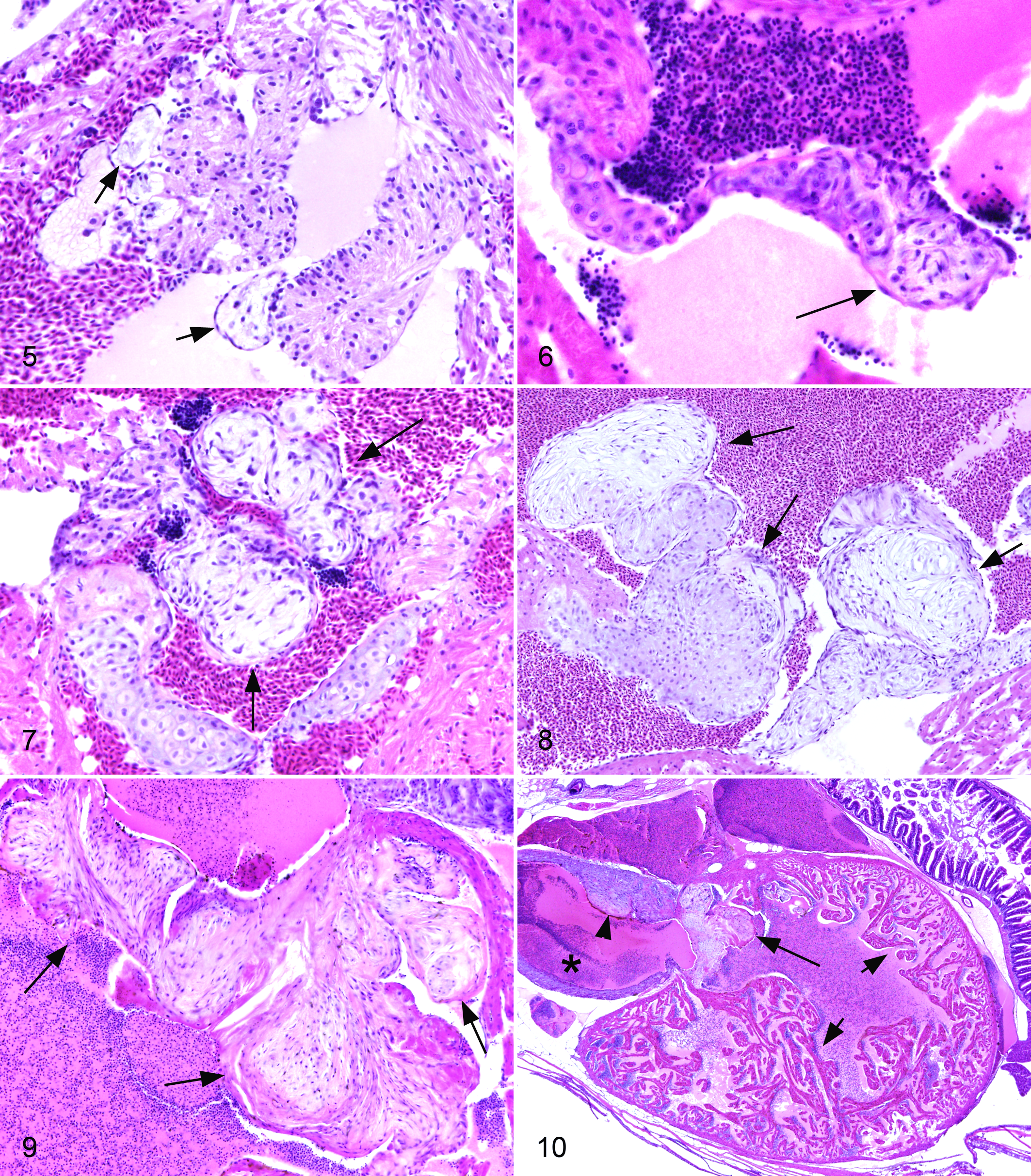
Of 777 fish evaluated in detail for this study, valvular endocardiosis was present in 59 (7.59%, Suppl. Table 1). There was endocardiosis of the sinoatrial valve in 34 fish (57.6%), with an average severity score of 1.21 (SD, 0.47). Endocardiosis affected the atrioventricular valve in 33 fish (55.9%), with an average score of 1.36 (SD, 0.64), and the bulboventricular valve in 11 fish (18.6%), average score 1.36 (SD, 0.64). Thirty-seven fish had endocardiosis of a single valve (62.7%, usually sinoatrial or atrioventricular), and 20 fish had lesions of 2 valves (33.9%). Only 1 fish (1.69%) had lesions of all 3 valves. Non-valvular mural endocardiosis lesions were present in 13 fish (22.0%), with an average severity of 1.23 (SD, 0.42). Lesions were present in the atrium in 3 fish, ventricle in 6, and bulbus arteriosus of 3, with lesions in both atrium and ventricle of 1 additional fish. All but 3 of those fish also had endocardiosis in an adjacent valve. Pericardial edema (hydropericardium) was present in 77 fish (9.90%), with an average severity score of 1.67. There was no consistent relationship between valvular endocardiosis and the presence or severity of pericardial edema (Suppl. Fig. S1), as the latter occurred at nearly equal incidence in fish with or without endocardiosis. Although there was no consistent association between valvular endocardiosis and cardiac atrial or ventricular dilatation (Suppl. Fig. S2) or hypertrophy (dilated or hypertrophic cardiomyopathy), secondary dilatation of the cardiac chambers was often evident in fish with severe endocardiosis (Fig. 10 and Suppl. Fig. S3). Fish with pericardial edema generally (but not invariably) also had hypertrophy and hyperplasia of the outer compact layer of the ventricular myocardium (Suppl. Fig. S1). The incidence of mural (non-valvular) fibrin thrombi in the heart (Suppl. Fig. S4) was the same in fish with and without endocardiosis.
Endocardiosis lesions were more frequent in fish fed commercial diets and housed in recirculating water systems, occurring in 52 of 230 fish (22.6%) versus 4 of 234 (1.71%) fish fed a purified diet with flow-through water. Only a single fish out of 219 fed a commercial diet but housed in flow-through water systems developed endocardiosis (0.46%), while 2 of 94 fish fed a purified diet in a recirculating system were affected (2.13%). All fish with multiple valves affected and/or mural lesions were in recirculating water systems; all but 1 was fed a commercial diet.
Endocardiosis lesions were especially frequent in heterozygous smoothened mutant fish (smob641/+ , ZFIN ID: ZDB-ALT-000821-2), occurring in 34 of 61 fish (55.7%). These fish were fed a commercial diet and maintained with recirculating water. In wild type (wt) fish of the same age and background maintained under similar conditions, only 17 of 168 were affected (10.1%). There was no association between endocardiosis and small cell carcinoma (carcinoid) of the intestine, despite this tumor also occurring more frequently in smoothened heterozygotes (10/61, 16.4% vs 10/168, 5.95% for age-matched wt AB fish under similar husbandry conditions).
In the 56 cohorts of retired broodstock that were studied less intensively for heart valve lesions, we documented endocardiosis in just 9 of these cohorts. Lesions were present in any or all 3 valves, most commonly the sinoatrial, with severity of lesions grade 1 to 2. Fish with endocardiosis were 12 to 41 months old. While in most affected cohorts the incidence of endocardiosis was 10% or less, in certain cohorts, there was a higher incidence of valve lesions. Ungenotyped offspring of 2 crosses of heterozygous tbx16b104/+ (Df (LG08); spadetail; sptb104 , ZFIN ID: ZDB-ALT-980224-16) fish on an AB background showed valve lesions in 5 of 8 (63%) or 1 of 4 (25%), and genotyped heterozygotes had an incidence of endocardiosis of 1 of 2 (50%). A single 12-month-old genotyped cdh2m117/+ (glass onion glo; ncad, ZFIN ID: ZDB-ALT-980203-450) fish in TU/AB background had congestive cardiomyopathy with grade 2 endocardiosis of the sinoatrial valve. We did not observe endocardiosis in our carcinogenesis studies with zebrafish of a variety of wt and mutant lines, although the fish were typically sampled for histology at relatively young ages (6–12 months) and were raised in a flow-through system and fed a semi-purified diet.
Discussion
Similar to the described effects on the occurrence of neoplasia, diet and husbandry appear to play a significant role in the incidence of endocardiosis lesions. 27 In particular, lesions are more common in fish fed commercial diets and housed in a recirculating system with fluidized sand biofilters than in fish fed a semipurified diet with flow-through circulation. Excessive nitrite has been reported to cause abnormal valve development in zebrafish via nitric oxide (NO) generation. 17 NO has also been hypothesized to play a role in myxomatous mitral valve disease (endocardiosis) of dogs. 22 Nitrite (and nitrate) can accumulate due to husbandry and water quality fluctuations due to biofilter overloading or suboptimal function and could potentially explain some of the differences observed between fish raised in recirculating versus flow-through water treatment systems. However, prolonged adverse water quality parameters that are routinely monitored, like nitrate or nitrite, are unlikely in the systems studied because these were carefully designed, highly sophisticated systems managed and monitored by highly trained staff. Significantly, analysis of tumor data suggests the possibility that episodic spikes of toxicants occur in recirculating water systems, and the same or similar spikes of natural toxicants could contribute to the cardiac valve and pericardial edema lesions in fish from these systems. 27 We do not yet know the nature of these toxicants.
Fish heterozygous for a mutant allele of smoothened (smob641/+ ) were overrepresented in this study. When the signaling molecule hedgehog binds to its cognate receptor patched, the complex then interacts with smoothened, a G-protein coupled receptor. Although homozygous smoothened mutant mice and zebrafish have abnormal cardiac development, including outflow tracts and valves, 11,21 cardiac defects are undescribed in heterozygotes, and no evidence for altered hedgehog signaling pathways has been reported for endocardiosis. Although relatively few individuals were evaluated, the spadetail (tbx16b104/+ ) mutant line also seemed at higher risk for developing endocardiosis lesions than the other lines studied. T-box transcription factors, including tbx16 and brachyury (T), are involved in normal heart and limb development but have not been previously implicated in endocardiosis. 12 There is no direct mammalian orthologue of tbx16 identified, a not uncommon consequence of the whole genome duplication of teleost fish.
Activation of serotonergic 5-hydroxytryptamine 2B receptors in carcinoid syndrome can result in myxomatous valve disease in humans. 6,8,19 Therefore, we specifically looked for any association between valvular endocardiosis and the occurrence of small cell tumors (carcinoids) of the intestine. 23 No association was found in this study. However, the potential to take advantage of the favorable characteristics of zebrafish (small size, bath exposures, high stocking density) to screen drugs for valvulopathy during preclinical testing remains intriguing.
Because of the high-throughput nature of these studies, combined with the fact that valvular pathology was not the primary expected phenotypic outcome, there are several limitations to this study. Not all valves could be visualized in every fish, although all 3 were identified in the majority of fish examined. Also, because the paraffin blocks containing the fish were step sectioned beyond the level of the heart, no unstained sections or unsectioned blocks were saved for analysis of special histochemical stains (eg, Alcian blue) or immunohistochemistry. Future studies to specifically characterize the nature of these lesions would be valuable. Further, fish were only evaluated at certain specific endpoints, so the evolution of these lesions could not be charted.
There was no consistent relationship between endocardiosis lesions and cardiac dilatation, although the latter did often occur with high-grade valvular lesions. In comparison to mammals with dilated cardiomyopathy, zebrafish hearts with severe cardiac dilatation show remarkably little histologic evidence of degenerative change in the myocardium. We did not observe increased connective tissue (fibrosis) in the cardiac interstitium, nor did we see degenerative changes of cardiac myocytes. 10,14 The fatty replacement of cardiac cells (steatosis), which often occurs in dilated cardiomyopathy of humans, did not occur in zebrafish. This relative lack of cardiac degeneration in zebrafish with dilated cardiomyopathy may be attributed to the remarkable regenerative ability of zebrafish cardiac tissue in comparison to cardiac myocytes of mammals. 4,30
Cardiovascular circulatory dynamics differ significantly in zebrafish versus mammals. In cases of congestive cardiomyopathy in zebrafish, we have never observed chronic passive congestion of liver, as occurs in mammals. When we have observed chronic passive congestion of a visceral organ in zebrafish, typically the affected organ is the spleen, which can become greatly congested and may show multifocal cystic degeneration in older zebrafish that have ultimobranchial neoplasms. 23
Complete characterization of laboratory animals used to model disease should include documentation of background and age-related lesions, including influences of husbandry conditions. Herein we have described myxomatous degeneration of the valves and mural endocardium of aging zebrafish, including the influence of diet and water system.
Footnotes
Acknowledgements
We thank Jean Copper, David Mandrell, Belinda Giardine, and Dr Keith Cheng (Penn State Zebrafish Atlas, NIH grant R24 OD011152) for scanning and hosting digital slides.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Parts of this work were funded by the US Public Health Service, National Institutes of Environmental Health Sciences (grants R01ES011587, R21ES013124, P30ESO3850, and P30ES00210), the National Center for Research Resources (grant 3P40RR12546 and its supplement 03S1), and the US Army (contract DAMD 17-91Z1043). The Zebrafish International Resource Center is supported by the National Institutes of Health Office of Research Infrastructure Programs.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
