Abstract
The third carpal bone (C3) responds to exercise by adaptive modeling of bone and articular calcified cartilage along the dorsal load path. Canals penetrating articular calcified cartilage, thought to contain vascular tissue, are reported in numerous species. Their significance remains unclear. Our objective was to determine if the number of canals was significantly different in strenuously exercised and control young horses and in a site of intermittent high loading compared to sites sustaining lower habitual loads. Volumetric bone mineral density in the radial facet of C3 of strenuously exercised and gently exercised (control) 19-month-old thoroughbred horses (n = 6/group) was determined by peripheral quantitative computed tomography. The hyaline cartilage was corroded to expose the surface of articular calcified cartilage. The number of canals penetrating the articular calcified cartilage surface in en face scanning electron microscopy images was compared in 4 regions. Volumetric bone mineral density of C3 was significantly greater (P = .004) in strenuously exercised horses. There were 2 morphologically distinct groups of canals and significantly fewer (P = .006) large canals in the dorsal than in the palmar aspect of C3 in control but not in exercised horses. Roughly circular depressions in the articular calcified cartilage surface around apparently forming canals were visible in some samples and have not been previously described in the literature. The canals may be evidence of chondroclastic activity reaching the interface of hyaline and calcified cartilage. Further work is needed to elucidate the relationships between presence of canals and the responses to exercise and to joint disease.
Introduction
Despite detailed studies, many questions remain about the microscopic alterations occurring before macroscopic changes appear in hyaline articular cartilage (HAC), articular calcified cartilage (ACC), and subchondral bone (SCB). 4 In the third metacarpal bone of the Thoroughbred racehorse, early changes of osteochondral disease occur in the ACC, 2 and the ACC thickness of the carpal bones alters in response to exercise. 11,27,29 ACC warrants further investigation so its role in growth and development, response to environmental stimuli, and behavior in disease and aging can be better understood.
ACC apparently attenuates shear forces transmitted from the joint surface to the underlying bone. 27,29,30,40 Canals crossing the ACC from the SCB 13 have been reported in humans, horses, rabbits, and dogs. 3,6,23,31 Canals were clustered at the site thought to sustain greatest stress, in larger numbers in joints under greater load, 18 concentrated in sites with imputed lower cartilage surface stresses compared to elsewhere in the joint, 3 or randomly distributed. 20 The evidence for the association between loading and canal occurrence is therefore conflicting.
It has been suggested that canals cross the ACC:SCB interface during early osteochondral pathology 3,32,39 or osteophyte formation 12 in response to cartilage disruption 23,39 or during reparative growth. 21 The canals are related to the topographical interlocking of ACC with the adjacent tissues, HAC and SCB. 29 The findings appear disparate, and the number, size, stimulus, and mechanism for the appearance of canals penetrating the ACC require further study.
These circular openings in the ACC surface, or linear interruptions perpendicular to the osseous-chondral junction, have variably been termed vascular channels, 16,38 vascular canals, 14 cartilage canals, 3 protrusion pores, 20 vascular abutting vessels, 29 cavities, 40 and focal contacts. 25 Elucidation of contents of canals of varying morphological appearances require definition to allow a common terminology to be developed.
The equine third carpal bone (C3) has 2 articular surfaces. The proximal surface, within the intercarpal joint, sustains high forces and has a high range of motion and reduced cartilage contact area during hyperextension at the stance phase of gallop. 10,33 The distal surface, within the carpometacarpal (CMC) joint, has almost no range of motion, and the articular surface is presumed to be fully congruent between the distal surface of C3 and the proximal aspect of the third metacarpal bone. 5 In the dorso-medial half of C3, within the radial facet, cyclic loading leads to osteochondral pathology, 14,26,17,33,35,41 with accompanying osteoarthritis (OA), 5,37 but clinical disease in the CMC joint is rarely described in the literature. 22,34
Trabecular thickening in the dorsal aspect of C3 increases volumetric bone mineral density (BMDv) in exercised horses and despite physiological adaptive responses at higher gait speeds 5,8 –10,41 is associated with increased stiffness of the bone, reduced energy absorption within the articular cartilage, 36,41 and altered HAC composition and thus force transmission through the ACC. 27,28
Relationships between exercise and ACC thickness have been described. 7,11,26,29 ACC thickness in the dorsal aspect of C3 and radial carpal bone sites in strenuously exercised horses was found to be higher than in palmar sites and when compared to gently exercised horses. 27 Kim et al 16 found thickening of ACC at osteochondral injury predisposition sites, but in an osteochondral fragmentation model, thinning of the ACC was seen at the site of highest stress. 29 Exact aetiology for changes in thickness of the ACC are therefore not entirely clear, but it does appear that ACC is metabolically and morphologically a highly active and responsive tissue. We thus used equine carpal tissue to study ACC canals.
The objective of this study was to determine if the number of canals penetrating the calcified cartilage at the ACC:HAC junction differed according to the imputed different loading regimes sustained by various sites in the C3. We hypothesized that the expected BMDv response through the dorsal load path would be seen in the exercised horses following the treadmill training program used, allowing us to draw conclusions on the effect of exercise on canal frequency at the different sites. It was hypothesized that the number of canals in the ACC surface would be significantly higher in the highly loaded dorsal aspect of the radial facet of C3 compared to the putatively lower loaded palmar C3 and CMC sites examined. It was also hypothesized that canal number would be significantly affected by the exercise regime imposed.
Materials and Methods
The experiment from which the tissue was obtained is described in full previously. 1,9,27,28 The use of the horses in this study was approved by the University of Bristol Institutional Review Board. Twelve untrained Thoroughbred fillies aged 19.3 (±0.9) months were paired by bodyweight, with 1 of each pair randomly allocated to group 1, which received intensive treadmill exercise to mimic preparation for racing (n = 6), or to group 2, a control group (n = 6), which were walked for 40 minutes per day (full exercise program detailed in Supplemental Table 1). All were kept in 3 m × 3.5 m stables when they were not being exercised. After 19 weeks, all horses were euthanized and tissue harvested. From the right C3 of each horse, a 6-mm thick slab of bone was sectioned (Supplemental Fig. 1) and used in a previous study. 9 A visual inspection confirmed no macroscopic lesions were present in the HAC.
Peripheral Quantitative Computed Tomography
Each slab was scanned (voxel size 200 microns, XCT2000, Stratec, Pforzheim, Germany) after being oriented and secured in a custom-made polystyrene holder to ensure the beam plane was exactly perpendicular to the proximal and distal articular surfaces. Two 2-mm thick, contiguous scan slices, both entirely within the bone slab were obtained. Analysis using the manufacturer’s software produced BMDv of the entire slab and of 10 contiguous 2 mm × 2 mm volumes of interest (VoI) from the dorsal to palmar aspect of both the proximal and distal surfaces of the C3 slab (Supplemental Fig. 1). One edge of each VoI was beneath the articular surface, and the VoIs coincided with the site of blocks prepared for microscopy as described in the following paragraph; for each VoI, the mean BMDv of the 2 apposing slices was utilized.
Using prior study of pathology and loading site information 11,33,37 and the BMDv values, 6 mm × 4 mm × 4 mm tissue blocks of the VoIs were sectioned using a diamond saw (Supplemental Table 2, Supplemental Fig. 1). Sites selected included a dorsal and a palmar site for each joint surface.
Scanning Electron Microscopy
The 6 mm × 4 mm × 4 mm blocks were prepared for scanning electron microscopy (SEM) (Supplemental Fig. 1) by removing soft cartilage using 27.5% hydrogen peroxide for 20 hours, rinsing in distilled water, ultrasonicating for 15 minutes, and drying following brief immersion in 70% ethanol. Blocks were then critical point dried, gold sputtered, and affixed to specimen mounts with silver paint, with the ACC surface orientated uppermost.
SEM of the ACC surface for each site was conducted (Quanta 200, FEI, Hillsboro, Oregon), initially at 34× magnification and later at 200× to 6000× to elucidate and describe the morphological features of interest.
Image Analysis
The low magnification SEM images were merged so that for each block surface a single image was created and analyzed using XnView for Windows (v1.98). All black areas indicating voids in the ACC surface were examined, and canals were identified, according to the classification described in Table 1, by both size and surrounding morphological features. The diameter of each canal was measured in 1 axis using MbRuler (Markus Bader, MB Software Solutions) on the merged images and immediately colored to prevent duplication of measurements. We divided canals into small and large groups as the morphological features surrounding generally larger canals and generally smaller canals appeared so different. Numbers of large (25-150 microns) and small (6-32 microns) canals on the 6 mm × 4 mm imaged ACC surface of every block were recorded, as was morphological variability in the ACC surface. On the rare occasion when a canal had been sectioned at the edge of a block, the canal was included if its diameter could be measured.
Morphological Features of Large and Small Canals and Depressions in the Articular Calcified Cartilage (ACC) of the Third Carpal Bone.

Data Analysis
Nonparametric data are presented as median and interquartile range (IQR) throughout in Supplemental Tables 3 through 6. The median BMDv of the entire sagittal slab was compared between group 1 and group 2 horses using the nonparametric Kruskal-Wallis test. A t-test for equal variances was used to compare BMDv at the VoIs.
A Kruskal-Wallis test was used to compare group 1 and group 2 with the following analyses: median number of large and small canals in the dorsal (A+C) and palmar (B+D) sites and median number of canals in the intercarpal articular (A+B) and CMC articular surface (C+D) VoIs with significance set at P < .05. Site A (the commonly injured site) was compared to the other 3 VoIs with a post hoc Bonferroni correction applied for multiple comparisons, resulting in a statistical significance level of P ≤ .012. All analysis was conducted in Stata 11.1 (StataCorp Ltd).
Results
Peripheral Quantitative Computed Tomography
The median BMDv of the sagittal slab was significantly (P = .004) greater in the group 1 (767 voxels; IQR, 750-829) horses compared to group 2 horses (660 voxels; IQR, 644-692). Mean BMDv were significantly greater in the dorsal (A+C) regions of the joint when compared to the palmar (B+D) regions in the group 1 horses (P < .01) (Fig. 1), but there were no significant differences between sites in group 2.
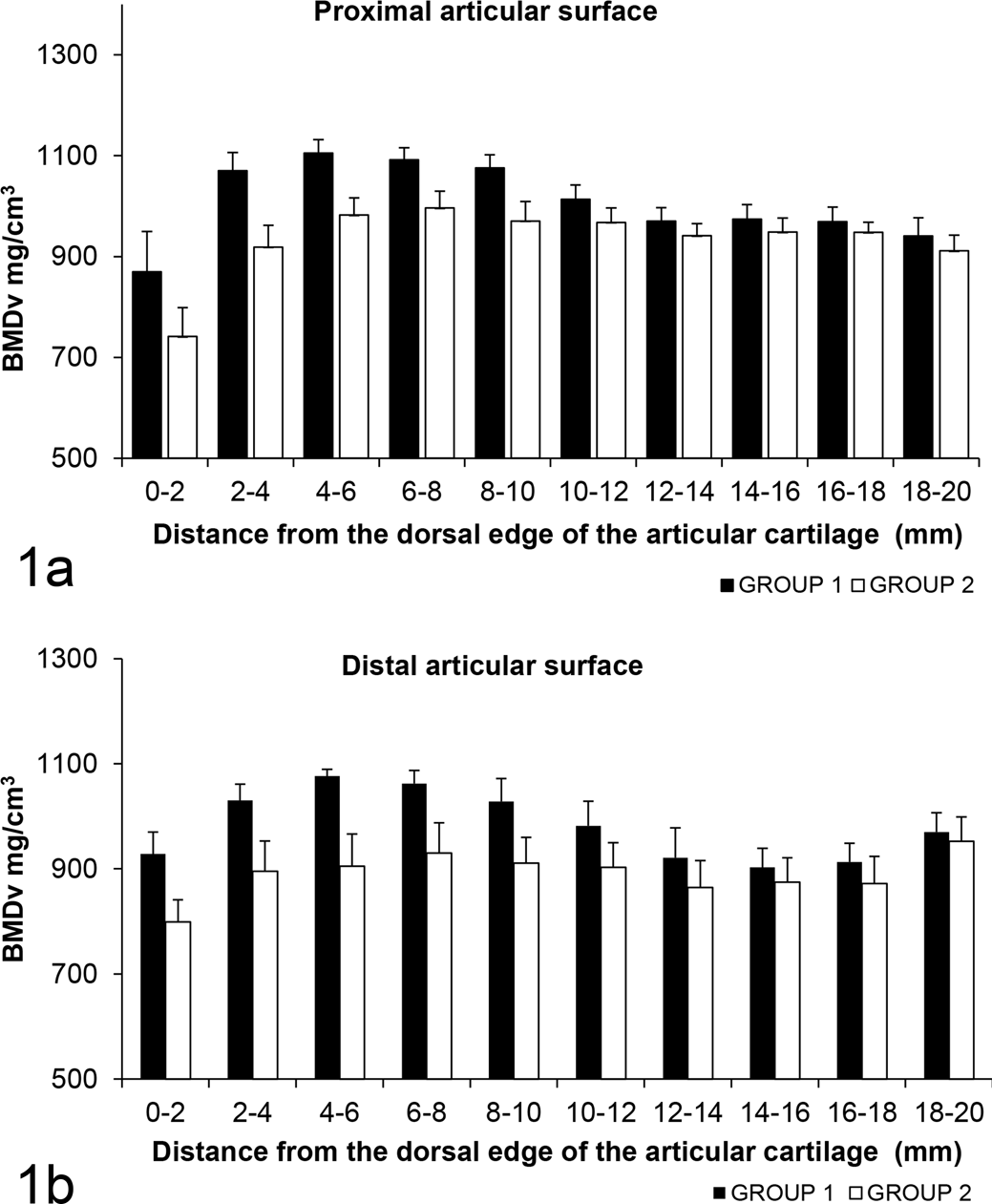
Volumetric bone mineral densities (BMDv) at 10 contiguous sites in the third carpal bone adjacent to the proximal and distal articular surfaces in exercised (group 1) and control (group 2) horses.
SEM Image Analysis
One block was excluded from the study because the surface was eroded after hydrogen peroxide corrosion. Summary data are shown in Supplemental Table 3 for each of the VoIs. The median number of large canals was significantly (P = .006) fewer in the dorsal than the palmar sites in group 2 horses but not in the group 1 horses (Supplemental Table 4). The median number of small canals did not differ significantly between sites in either group 1 (P = .17) or group 2 horses (P = .64). No significant differences were seen between the intercarpal and CMC joint surfaces (Fig. 2, Supplemental Table 5) or between Site A and the other 3 VoIs in either group (Supplemental Table 6).
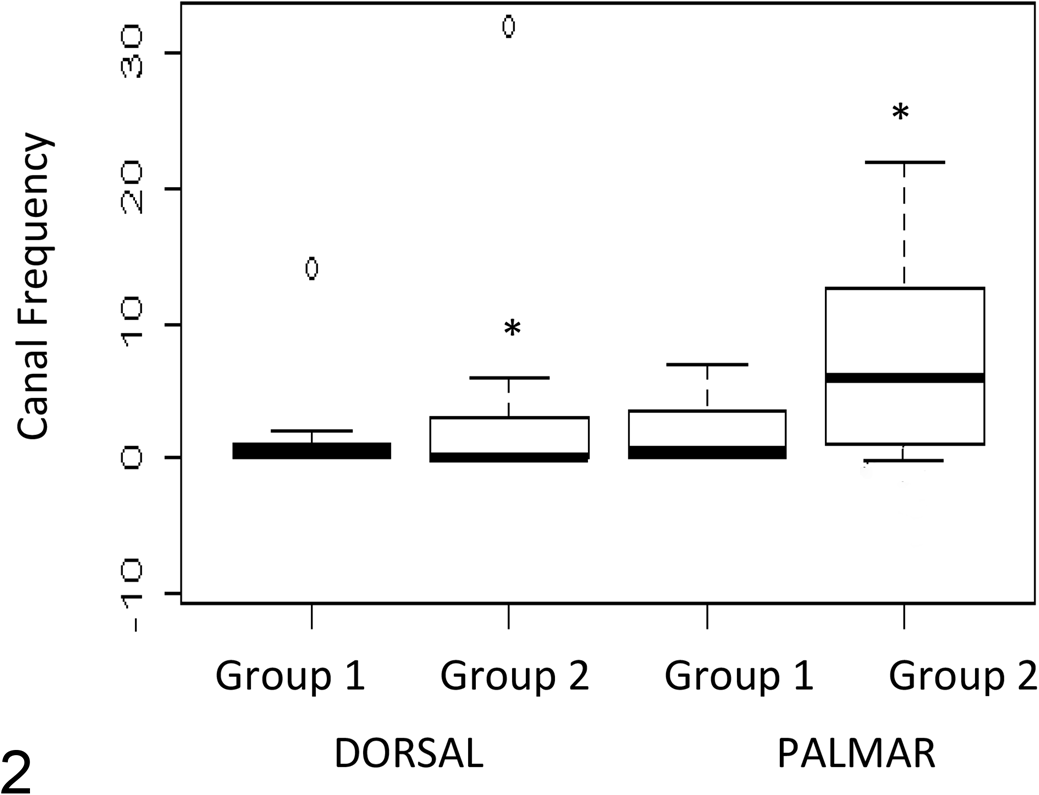
Canal frequency for dorsal (A+C) and palmar (B+D) sites in exercised (group 1) and control (group 2) horses. P < .05.
It was not possible to distinctly separate canals into large and small groups solely based on size, as the extracellular matrix (ECM) surrounding the canals was very different between the 2 types—rough in the large canals and smooth in the small canals. However, large canals with rough surrounding matrix were generally larger than small canals with smooth matrix. In many blocks, no canals were seen at all in both exercised and control horses.
The large canals were well circumscribed and varied in shape from circular to triangular. Many had smooth scalloped edges (Figs. 3 –5). There was no change in the microcalcospheritic appearance of the ECM surrounding the large canals, with remnants of chondrocytic lacunae mostly some distance from the large canals. A border approximately one-third the diameter of the canal with radial cracks in the ECM was seen immediately surrounding the canal.
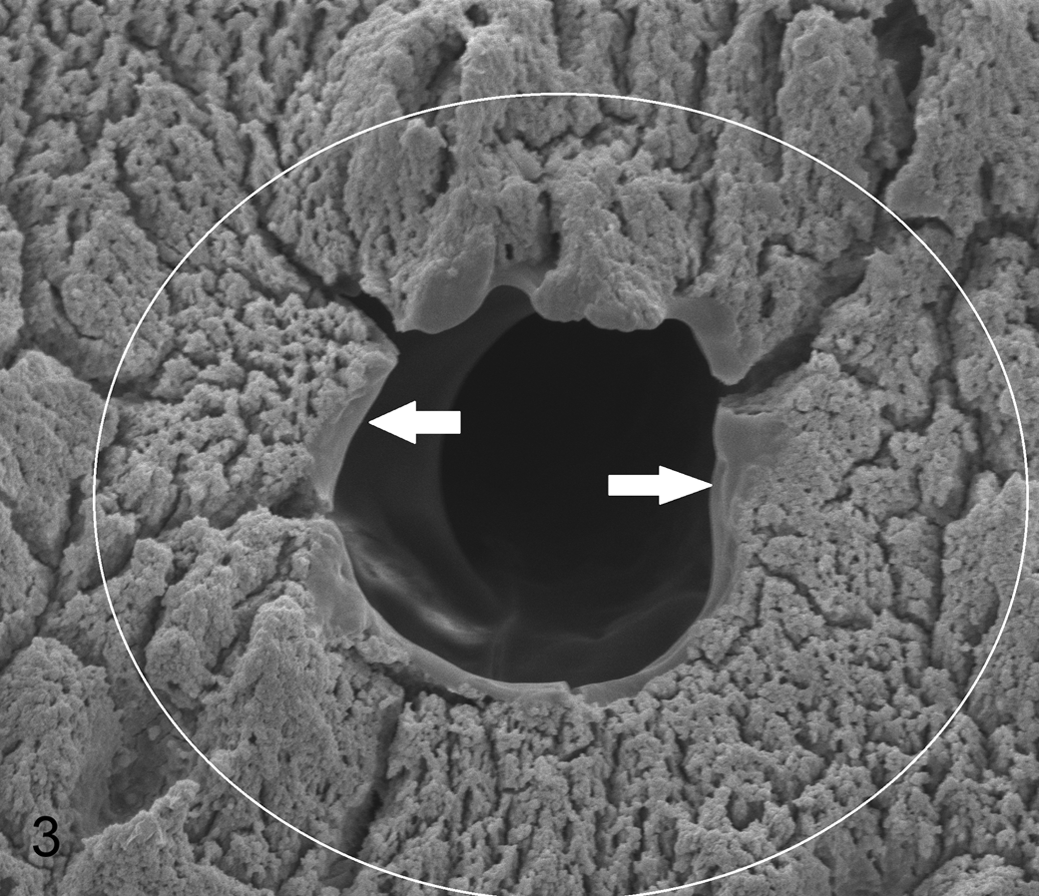
Articular calicified cartilage, third carpal bone, horse. Typical appearance of a large canal, diameter 38 μm, with a narrow smooth border to the edge (arrows) within a border of uninterrupted rough mineralizing front radiating out from the canal edge (within white circle). Scanning electron micrograph (SEM).
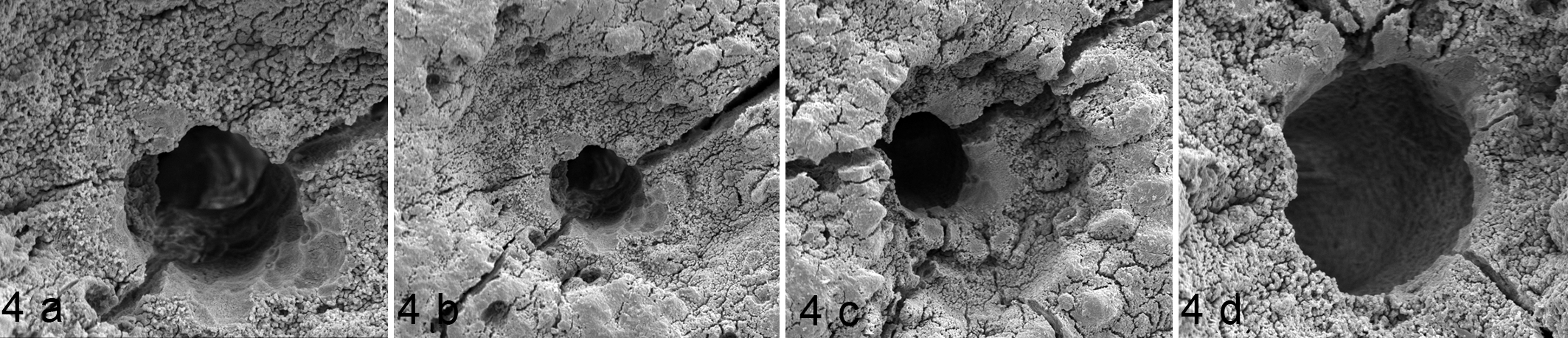
Articular calicified cartilage, third carpal bone, control horse 8, site D. Scanning electron micrographs showing randomly distributed large canals within depressions. (a) 43 μm canal. Scalloping is visible at 3 to 6 o’clock. The extracellular matrix is rough, and this roughness can be seen within the canal. (b) Same canal as 4a, demonstrating the rough calcified surface within the depressed region around the large canal. The scalloping and smooth regions in this canal are suggestive of chondroclastic activity. (c) A large (48 μm) canal within a region of depressed matrix. There is a smooth scalloped edge from 11 to 4 o’clock surrounded by rough matrix. The small holes within the depression around the canals (Figs. 4b and 4c) could be consistent with the processes of a chondrocyte. (d) A large (55 μm) canal with a smooth edge from 10 to 6 o’clock surrounded by a depression and rough matrix. The canal edge is irregular, consistent with previous scalloping, and this is likely to be a more mature canal than the others shown.
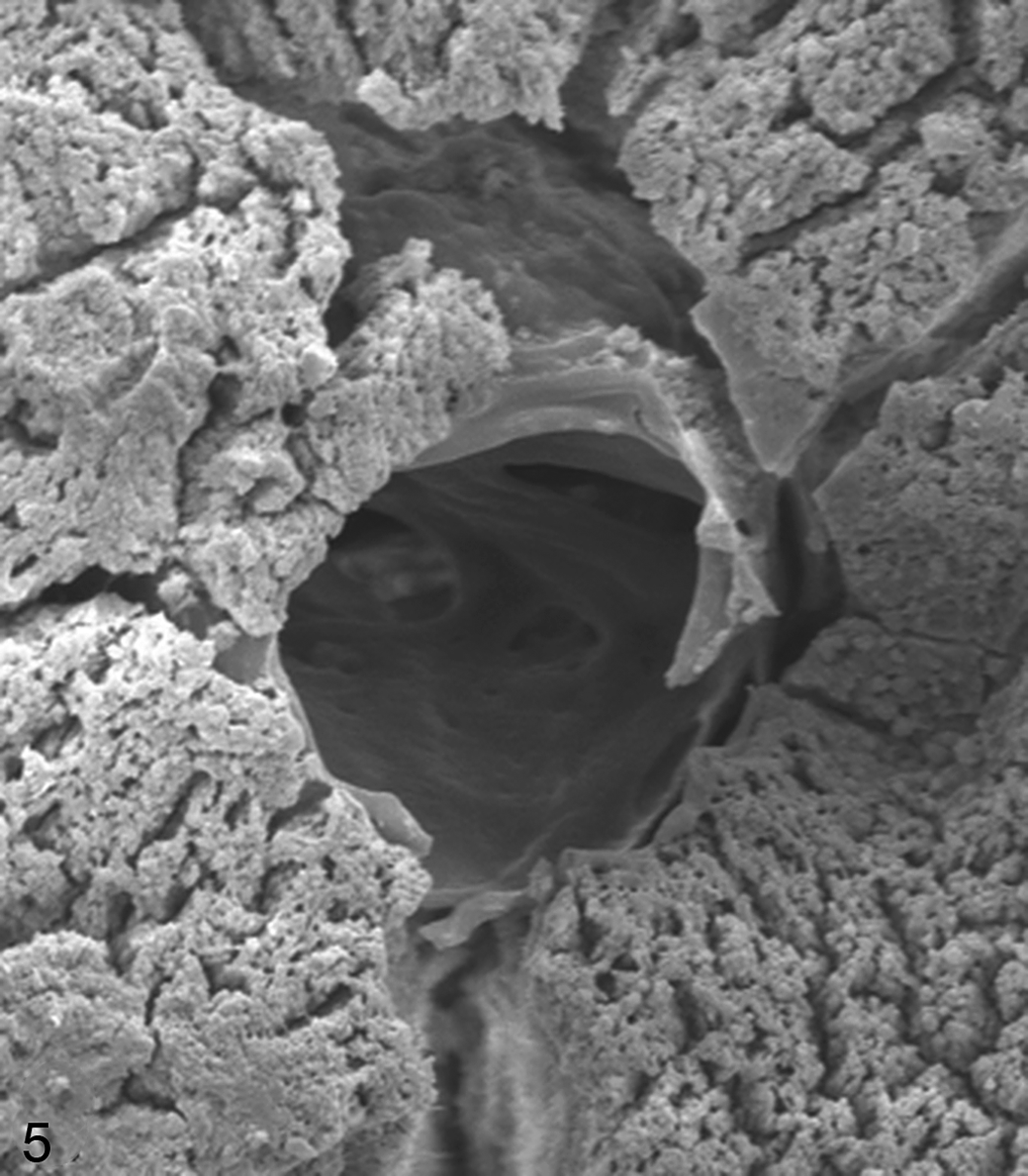
Articular calicified cartilage, third carpal bone, control horse 6, site B. A 35 μm canal with a smooth round edge and scalloping from 3 to 5 o’clock. At 12 o’clock, there is (likely artifactual) loss of surface matrix displaying a bulge around the crater, probably the wall of the canal. The tissue surrounding the canal appears smooth. Branching canals of various sizes can be seen within the canal. Very small foramina consistent with those made by osteoclast or chondroclast podia can be seen. Scanning electron micrograph (SEM).
Small canals were seen centrally or eccentrically within smooth depressed areas of ECM (Fig. 6, Supplemental Figs. 2, 3) and were often surrounded by remnants indicative of chondrocytic lacunae. One or many small canals could be seen within each depressed region of the mineralising front.
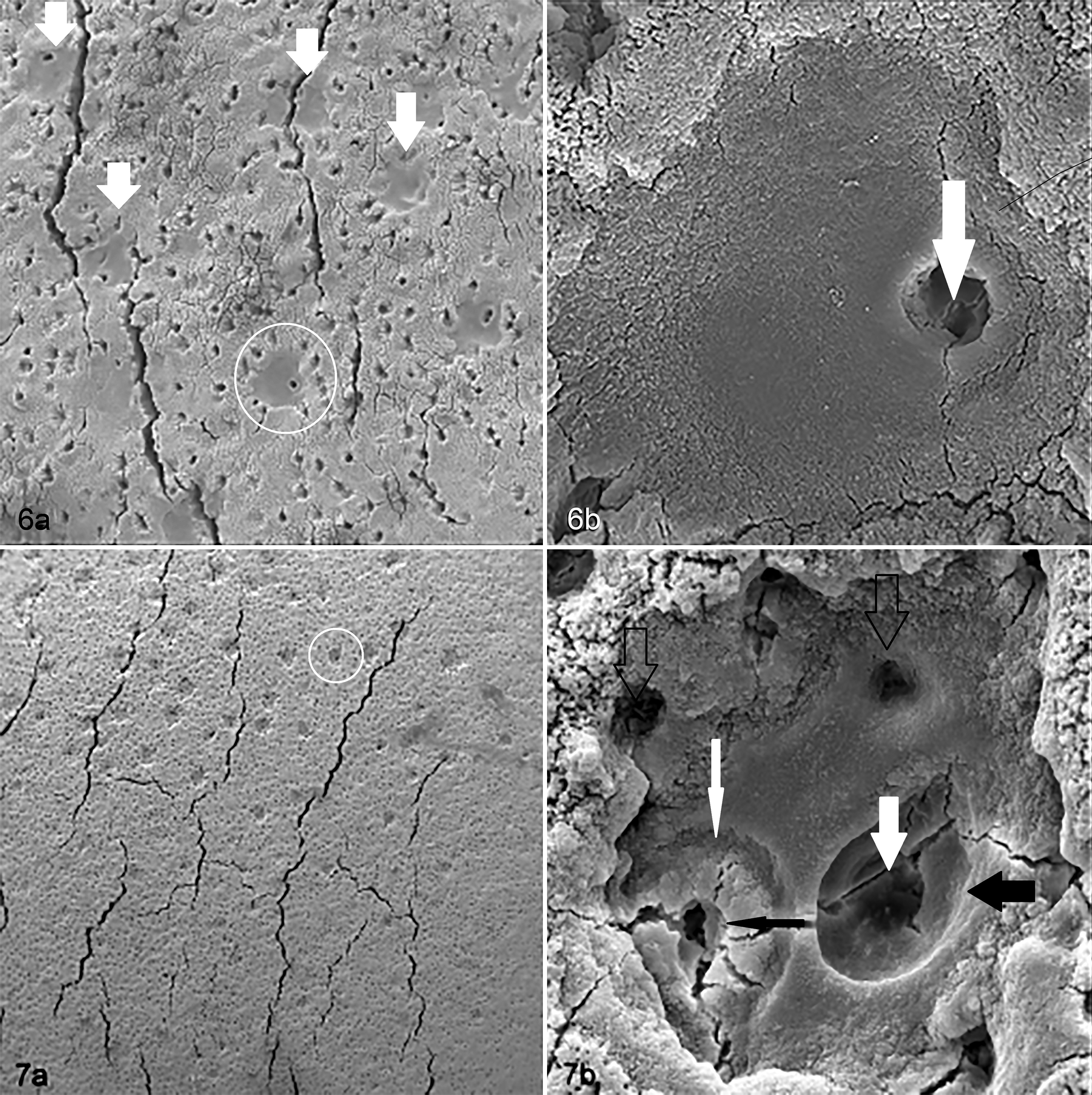
Articular calcified cartilage, third carpal bone. Scanning electron micrograph (SEM).
Regions with regularly spaced depressions in the mineralizing front (Fig. 7, Supplemental Fig. 2) were seen in 11 of the samples. The depressions measured 60 to 90 microns diameter and were of similar frequency among sites and between exercise groups. There did seem to be a within-horse effect, with 11 sites affected in 5 horses (1 horse with 1 site, 2 with 2 sites, and 2 with 3 sites affected). Where depressions occurred, they followed a regular distribution (Fig. 7, Supplemental Fig. 2) and appeared shallow, suggesting no direct communication between the ACC surface and underlying SCB. Higher magnification revealed no depressions with an entirely smooth internal surface. Small holes or cracks were always present, either centrally or eccentrically.
One horse exhibited single large canals within a circular depressed region of surrounding ACC surface (Fig. 4). Two horses in group 2 had a large number of small canals at site A (Supplemental Fig. 2). One canal (Fig. 5) was seen with some loss of surface, exposing smooth tissue forming the wall of the canal with the mineralized matrix appearing very thin at this site. These insights into the 3-dimensional structure of these canals shows their variation and complexity.
Discussion
The adaptive modeling response of equine carpal bones to intensive exercise has been well described 9,27,41 and is thought to be related to the pattern of loading in this bone. The BMDv pattern seen in this study, with a significant difference between the exercise and control groups in the dorsal aspect of C3 but not in the palmar aspect, was therefore as expected, and this hypothesis was accepted. No significant differences were seen between the number of canals in site A (the highly loaded, injury site) and other sites examined in either group. We therefore rejected this hypothesis. A significantly lower number of large canals were seen in the dorsal sites compared to the palmar sites in the group 2 but not group 1 horses, so it does appear that exercise and loading have some significant effects on canal frequency.
Previous analysis of the bones 9 using dual-energy X-ray absorptiometry, which has lower resolution than pQCT, did not allow BMDv near the edge of the SCB to be determined because of the projection issues associated with this imaging modality. The tomographic technique gave an accurate indication of high and low density in the VoIs and optimal choice of VoI sites for comparison of canal frequency in dorsal and palmar sites.
In the control group (group 2), there were fewer large canals in the dorsal load path site compared to the palmar sites, which apparently sustain lower but more habitual loads. This supports the findings of Boyde and Firth, 3 who found greater numbers of canals in a site on the sagittal ridge of the third metacarpal bone that wasn’t directly congruent with the proximal phalanx during stance; this site had lower bone density and was thus presumed to undergo lower loading than other regions of the metacarpophalangeal joint. Potential reasons for the reduced numbers of canals could include a change in loading due to increased stiffening at the cartilage:subchondral bone interface, 9 excessive compressive loading resulting in chondrocyte death in a similar fashion to that reported in hyaline cartilage, 19 or a decreased rate of mineralization in highly loaded sites 7 requiring less vascularization due to lower cell turnover. It appears that loading is an important factor in loss of canals; however, examination at different stages of training would be required to determine the exact sequence of events that lead to canal loss and whether these are adaptive or pathological.
We had expected more canals at the common site of pathology (Site A) and other sites studied. This did not prove to be the case. Norrdin et al 29 also found no difference between the numbers of vessels/mm crossing the ACC:SCB interface on the intercarpal aspect of the C3 between previously fragmented and unfragmented osteochondral sites similar or close to our Site A. Regions with carpal osteochondral changes in exercised horses showed a higher area of vascular canals in the radial facet of C3 when HAC lesions were present compared to regions with normal HAC appearance. 15 In human and small mammal studies, vascular invasion into the ACC from the SCB was demonstrated in models of osteoarthritis. 23,38 Our findings suggest that either few such canals penetrate the full depth of the ACC at the earliest stage of osteochondral pathology or that in the exercised horses in our study, which had no visible lesions present on the HAC surface of C3, no pathology was yet present.
To our knowledge, this is the first study of ACC in the CMC joint. The latter is seldom studied because it is rarely affected by clinical disease in Thoroughbreds and difficult to disarticulate for examination. We considered that the very low relative movement of the opposing surfaces of this joint would provide each horse with its own control to enable inferences to be drawn on the influence of a very high range of motion on the loading of the proximal surface of C3. However, no significant differences were seen between the response of the intercarpal and the CMC joints to exercise. Gross pathology is rarely seen in the CMC joint of Thoroughbreds 5,22,34 so further study of this site may help differentiate significant pathological change in the ACC from the adaptive response to exercise and aging.
The shallow depressions on the ACC surface, described apparently for the first time, warrant further study. These depressions could be evidence of chondroclastic resorption on the ACC surface, 7 the endpoint of infilling of canals that has occurred from SCB toward the articular surface, 4 the result of immaturity and consequent thin ACC, 24 or an early pathological change. These potentially chondroclastic depressions may also signify the start of endochondral ossification. It is unclear if there is any relationship with the reduced density around cartilage canals observed previously, 3 but it is highly interesting that both are circular in shape. Further studies are required to evaluate these depressions, in particular evaluating their presence at different ages and different stages of training. Confocal microscopy of similar blocks of tissues could help determine any local changes in HAC or SCB relevant to these depressions. En face histological sections could also provide information but would be challenging to obtain.
The literature shows considerable disparity regarding size of canals at various locations. For instance, there is mention of numerous empty lacunae in third metacarpal condyle of horses aged less than 3 years, 24 canals between the ACC and the HAC generally being smaller than those at the junction of ACC and SCB, 20 and vascular channels in the subchondral plate being of 2 types with some being small. 6 These variable findings coincide with our having found small canals as well as the previously described large canals. 3 The variability of techniques and findings shows that further detailed study is required if progress in this area is to be made.
A limitation of this study was the lack of HAC histological examination to confirm absence of pathological changes in the samples used as this would have been more sensitive than the gross examination we performed.
This work describes the distribution of cartilage canals in the ACC of young horses without pathology that were strenuously or gently exercised for 5 months. Canal distribution was negatively correlated with loading through the dorsal load path of the third carpal bone in the control horses (in contrast to our suspicion that these changes would be seen in exercised horses only), and this change was the same in both articular surfaces of the C3, with no significant difference between joint sites. The depressions seen on the ACC mineralizing front surface have not been discussed previously to the authors’ knowledge. These may relate to the effects of chondroclastic activity, which appear to be central to the formation of the canals but may also affect the ACC tissue adjacent to the canals. Further information on this feature could be important in understanding cartilage disease.
Footnotes
Acknowledgements
We would like to acknowledge Doug Hopcroft of Manawatu Microscopy and Imaging Centre for performing the scanning electron microscopy, Charlotte Bolwell and Chris Rogers of Massey Equine for statistical assistance, and Joe Mayhew of Massey University Veterinary Teaching Hospital for help with writing the grant proposal and support throughout the project.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The project was funded by New Zealand Racing Board as part of the Equine Partnership for Excellence.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
