Abstract
Bone implants and devices are a rapidly growing field within biomedical research, and implants have the potential to significantly improve human and animal health. Animal models play a key role in initial product development and are important components of nonclinical data included in applications for regulatory approval. Pathologists are increasingly being asked to evaluate these models at the initial developmental and nonclinical biocompatibility testing stages, and it is important to understand the relative merits and deficiencies of various species when evaluating a new material or device. This article summarizes characteristics of the most commonly used species in studies of bone implant materials, including detailed information about the relevance of a particular model to human bone physiology and pathology. Species reviewed include mice, rats, rabbits, guinea pigs, dogs, sheep, goats, and nonhuman primates. Ultimately, a comprehensive understanding of the benefits and limitations of different model species will aid in rigorously evaluating a novel bone implant material or device.
Keywords
Bone and joint implants and devices are a rapidly growing portion of the health care industry, with an estimated $9 billion in Medicare expenditures spent on implantable orthopedic devices in 2009.
17
Considerable research continues toward developing novel devices to improve bone and joint function and health. The implant and device field contains diverse classes, including tissue-engineered constructs, combination products (ie, antimicrobial coatings for devices), and biomaterials (synthetic or natural nondrug substance(s) used as a whole or as a part of a system to treat, augment, or replace any tissue, organ, or function
11
). For the purpose of this review, the term
Animal models are a key component of many nonclinical implant studies, and the ideal species varies by the specific implant and its intended use. This review outlines how bone and joint implants are evaluated in models of human diseases and the advantages and limitations of species commonly used in implant studies.
Evaluating Implants In Vivo: Biological Relevance, Biofunctionality, Biocompatibility/Safety, and Clinical Relevance/Efficacy
Bone and joint implants can have a variety of effects on the host, from providing basic mechanical support to initiating local and systemic host responses. To evaluate both intended and unintended effects, several stages are used to evaluate implants, including biological relevance, biofunctionality, biocompatibility/safety, and clinical relevance/efficacy.
Biological Relevance
Initial laboratory testing is used to demonstrate fundamental biological relevance of an implant’s structure and material composition
53
(
Biofunctionality
A related testing stage that can be separate from or combined with biological relevance is biofunctionality (defined here as the ability of an implant to alter tissue function in a desired way). Unique bone and joint properties often measured include the following:
53,78
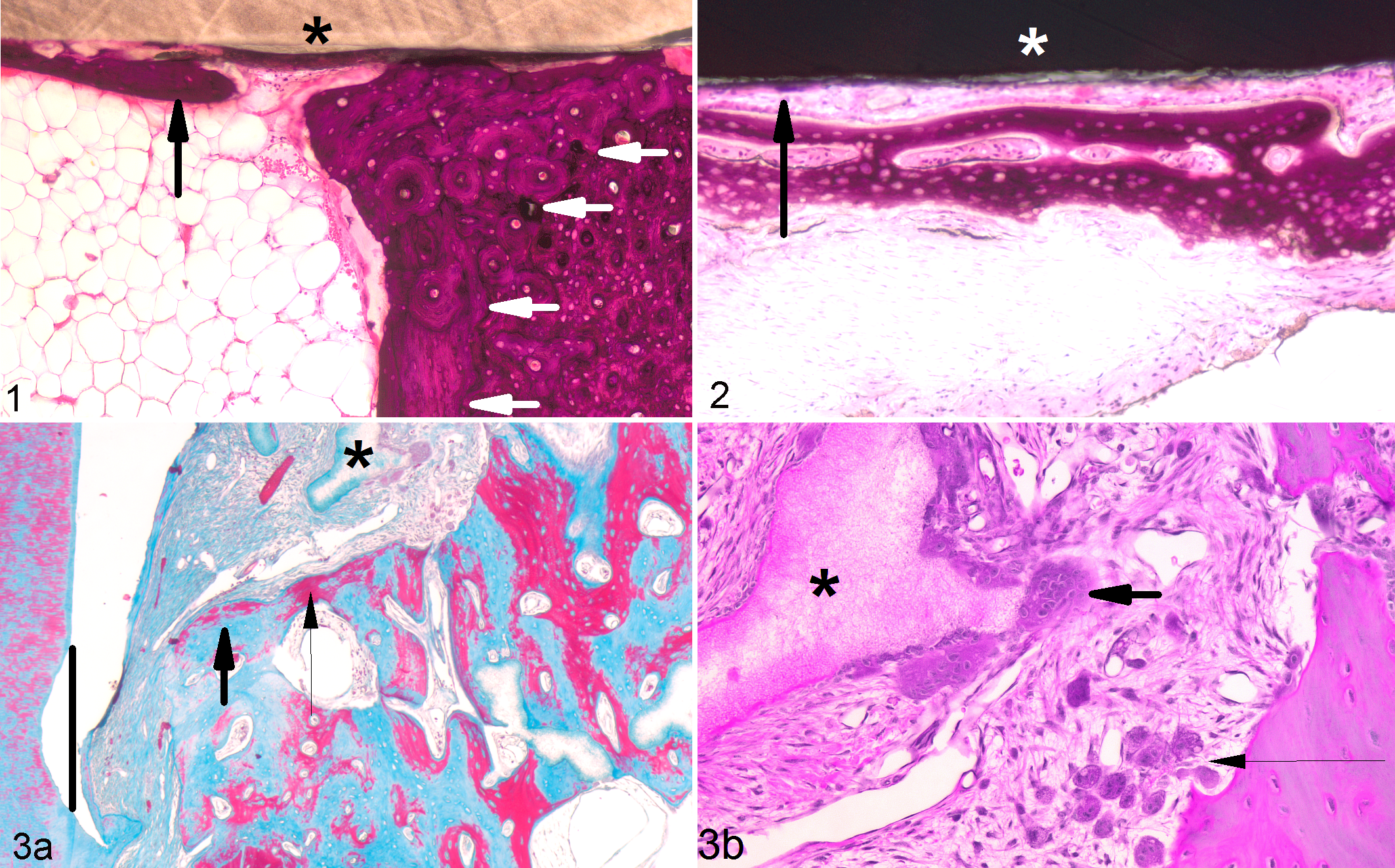
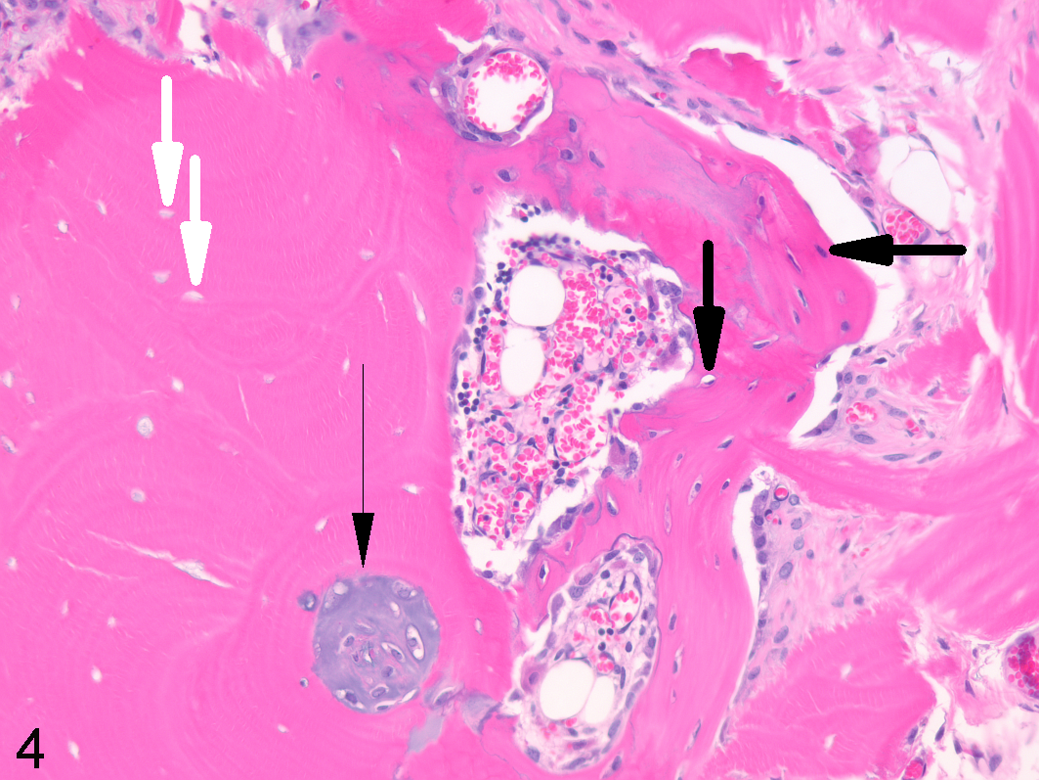
Osteoinduction within xenograft, skeletal muscle, athymic mouse. Demineralized human bone implanted within skeletal muscle for 35 days facilitated production of hyaline cartilage within preexisting osteonal canals (thin black arrow) and appositional woven bone deposition (thick black arrows indicating osteocytes) along the surface of the decellularized implant (white arrows indicating empty lacunae). Decalcified, hematoxylin and eosin.
As with evaluating biological relevance, biofunctionality is often tested in rodents and rabbits if the implant can be scaled to a small size and placed in a bone with similar physiology to that of the intended human site. If small animal models are not appropriate, larger species are used (dogs, pigs, small ruminants).
Biocompatibility/Safety
While biofunctionality tests if the implant has the desired effects (ie, osteoinduction when implanted in muscle), biocompatibility/safety studies can identify undesirable local or systemic effects (ie, immune reaction to osteoinductive material). 30 Biocompatible implants (and any breakdown or leachable derivatives) should be nontoxic, nonthrombogenic, noncarcinogenic, nonantigenic, and nonmutagenic. 30 Safety studies often use rodents and rabbits to detect local tissue damage (often including histiocytic and multinucleated cell infiltrates; Fig. 3b) 47,80 and systemic toxicities (often from breakdown or leachable derivatives). While tissue reactions to specific materials are not discussed here, recent reviews 30,47 provide detailed information about host reactions to different materials.
Clinical Relevance/Efficacy
Finally, while functionality and safety can be established in smaller species, it may be necessary to demonstrate efficacy in larger animals (dogs, small ruminants, pigs, nonhuman primates [NHPs]), where local blood flow and mechanical loading can be critical to implant success. 53,78
Diseases/Conditions Commonly Modeled
Enhancing Osteoinductive and Chondrogenic Potential After Injury
Implants that initiate or enhance bone or cartilage growth are highly desirable for injured tissues, especially those that have limited vascular or marrow supply to support intrinsic repair (ie, membranous craniofacial bone 79 ). Osteoinductive and/or chondrogenic potential is often measured in athymic mice and rats. 65,79 Chondrogenic implants are used in an attempt to repair synovial joints 16,65 and intervertebral disks. Osteoinductive implants are used as interventions to repair vertebral fractures, 10,79 during dental and craniofacial bone surgery, 18,79 and in segmental defects/nonunions (discussed in detail below).
Large Bone Defects and Nonunions
Large bone defects occur secondary to a pathologic event (ie, trauma, inflammation, surgical resection 64 ) that causes extensive bone loss. In human clinical settings, critical-size defects are defined as those that do not heal within ∼6 months postinjury in otherwise healthy adults. 12 Affected sites (ie, anteromedial tibia 20 ) often have limited adjacent muscle that could provide physical coverage and an auxiliary blood supply. Critical-size defects in animal models are defined as “the smallest size intraosseous wound in a particular bone and species of animal that will not heal spontaneously during the lifetime of the animal” 27,67 or a defect with <10% bone regeneration over the animal’s lifetime. 27 Surgically created critical defects are a gap in length >2.25 times the diameter of the affected bone 26,46 or large cylindrical defects in femoral condyles or tibial metaphysis. Rats, rabbits, and increasingly small ruminants are used in defect models. 59 One advantage of small ruminants is that their repair rate is more comparable to humans than rodents (a large tibial defect can fully repair within 12 weeks in rats but may take 32–52 weeks in goats or sheep 32 ).
Implant-Related Complications
Finally, while implants have an overall positive impact on human health, individuals may have implant-related complications (usually modeled in rodents and rabbits). Negative responses to implants can include reactions to wear debris, osteolytic bone resorption, anatomic malalignment, implant instability/breakage, heterotopic ossification, pain, and immunologic reactions/hypersensitivity.
80
Over 100 000 cases of implant-associated infection and osteomyelitis are reported annually in the United States.
55
On average, 2% to 5% of internal fixation devices become infected
19,74
and may require surgical removal to eliminate the infection. Up to 70% of organisms isolated in prosthetic joint infections are Gram-positive agents, including
Human Bone: Terminology, Organization, and Development
Terminology
Understanding how human bone is organized and develops is important when selecting an appropriate model species. Since there are multiple anatomic and histologic terms for some bone structures, in this review, a single term is used for each feature, with alternate terms listed in parentheses. Microscopically, bone is generally organized into woven (type I collagen fibers arranged haphazardly) or lamellar (collagen in parallel layers or concentric rings) patterns. Additionally, plexiform (or laminar or fibrolamellar) bone is produced within the periosteum when significant bone strength and rapid deposition are required. Plexiform bone is lamellar bone deposited on brick-like cores of nonlamellar bone (Fig. 2). 60 Plexiform, woven, primary lamellar, and primary osteons are forms of primary bone (formed during initial bone modeling on a substrate of bone or cartilage without resorption of preexisting bone). Secondary bone includes secondary osteons (Haversian systems) that replace primary bone through remodeling (coordinated deposition of new bone with resorption of preexisting bone). Macroscopically, bone is generally organized into cortical (or compact, referring to filling in spaces in plexiform bone) and cancellous bone (or trabecular or spongy or medullary). Structural differences between the 2 forms include bone density (cortical > cancellous), vascular access (cancellous > cortical), and presence of full osteons (cortical) or hemiosteons (cancellous).
Organization
All normal adult human cortical bone is lamellar with extensive secondary osteonal remodeling. 28,36 Plexiform bone is rarely present in humans, and in adults, woven bone is only formed during rapid bone formation (ie, fracture callus, hyperparathyroidism). 45,64 Macroscopically, approximately 80% of the adult human skeleton comprises cortical bone (by weight), and the remaining 20% is trabecular. 24,34 Load-bearing long bones are 80% to 100% cortical bone, while vertebrae are 80% cancellous and 20% cortical. 69 Subchondral plate bone adjacent to articular cartilage is generally thin 36 with minimal regenerative capabilities.
Development
Ossification in humans begins early in the neonatal period and continues throughout early adulthood, with fusion of the proximal and distal femoral epiphyses completed by 19 to 20 years in males and 2 to 3 years earlier in females. 15,53,84 Human remodeling rates range from 10% to 55% (average whole-body trabecular bone turnover per year 64 ), with age-associated cortical bone loss occurring primarily at the endocortical endosteum. 10 Bone adjacent to red marrow generally has higher rates of remodeling (eg, 10%–15% per year in ilium trabecular bone) compared with sites adjacent to yellow (fatty) marrow. 63
Factors to Consider When Selecting a Model
Certain characteristics are desirable when selecting any animal model, including low intra- and interanimal variation, the availability of sensitive methods to assess a biological parameter, and the use of treatment conditions that most closely match human clinical disease. 53 Additional factors should be considered in bone and joint models (Table 1). First, since implants may be intended for use in either cortical or cancellous bone, it is important to identify which sites and at what age a model species’ macro- and microstructure best matches human bone. Second, blood supplies that support bone healing can differ markedly between species 84 and by macroscopic structure (cancellous > cortical). Third, control groups or sites (ie, sham-operated, autologous cancellous bone graft 78 ) can aid in differentiating implant effects on bone repair from intrinsic bone-healing abilities (which can be significant in rodents and rabbits). Next, repair responses in skeletally immature animals can be more rapid than in mature adults. 65 For example, femoral fractures have been shown to heal within 4 weeks in 6-week-old rats but may take up to 26 weeks in 52-week-old rats. 51 These age-related changes can be further complicated by sexual dimorphism, which has been well documented in rodents and is influenced by sex hormones. 40 Finally, implants that cannot be scaled down (ie, joint replacement) require a large animal model. 53
Species Factors to Consider When Selecting a Bone/Joint Implant Model.

The following sections outline common model uses and comparative bone pathology for the most common preclinical species (summarized in Table 2).
Summary of Human and Animal Bone Properties.

NA, not applicable; NHP, nonhuman primate.
Rodents
Model Uses
Knockout and transgenic rodent models (ie, senescence accelerated mice) are a key resource in the biological relevance stage of implant studies. 12,48 Athymic nude mice and rats are also commonly used to test osteoinductive and cartilage regenerative potential of allogenic and xenogeneic materials. 16 Rats are used to model bone infection 55 and extraoral surgical approaches. 18 While rodents have limited cortical remodeling or secondary osteon formation, they can model cancellous bone responses in lumbar vertebral bodies and the distal femoral and proximal tibial metaphyses. 4,28,63
Relevance to Human Bone and Joint Conditions
There are significant limitations to consider when extrapolating implant findings from rodents to humans, including differences in bone micro- and macrostructure, rates of growth, and size. Unlike the secondary bone present in the adult human long bone cortices, rodent long bone cortices are predominantly primary bone and have minimal cancellous bone available to harvest for autografts. 53 Bone growth in rats continues much longer after sexual maturity than in humans. 39 Bone-healing capacity is higher in rodents than in humans at sites such as craniofacial bones, 71 and the retention of open physes in adults may enhance intrinsic articular cartilage healing potential. 16 Finally, some implants cannot be scaled down to a rodent size to test clinical equivalency. 12
Guinea Pig
While guinea pigs are not a common model for bone implants, strains that develop spontaneous degenerative joint disease, like the Dunkin-Hartley, are used to evaluate cartilage regeneration and joint support implants. Dunkin-Hartley male guinea pigs develop histologic lesions by 3 months of age, which progress within 6 months to lesions that closely resemble human joint changes. 38,76
Rabbit
Model Uses
Rabbit hindlimbs are commonly used for cortical and cancellous bone implant studies. The tibia is easily accessible due to minimal associated soft tissue, and the femur has sufficient medullary space for many internal fixation studies.
14
Mature rabbit condyles can support 3- to 4-mm defects to test biocompatibility and osteoinduction in cancellous bone.
16
Rabbits are also used to model vertebral fracture repair, methods to control common implant-associated pathogens (ie,
Relevance to Human Bone and Joint Conditions
Rabbits offer advantages over large animal models by reaching skeletal maturity at a relatively early age and advantages over rodents by undergoing more secondary osteonal remodeling. 53 Despite these positive characteristics, there are significant differences between rabbit and human bone. The rate of cortical bone remodeling is rapid in the young rabbits often used in studies, which can be less representative of the healing response in adult humans. 78 Rabbits also have relatively fatty bone marrow, which is not ideal for autogenous bone grafts. 53 Rabbit femoral condyle cartilage is relatively thin, 5,65 and articular cartilage in young, 3- to 9-month-old rabbits has rapid healing potential that can be a confounding factor in long-term studies. 16,82
Dog
Model Uses
Dogs are commonly used in dental studies 60 and are a valuable model of peri-implantitis (inflammation of tissue adjacent to dental implants), due to their susceptibility to biofilm accumulation and spontaneous periodontitis. 18 Large-breed dogs can often support human-sized implants, 60 and their large synovial joints, such as the stifle, can be monitored via arthroscopy. 16 Additionally, sufficient material for autogenous bone grafts can be harvested from the proximal humerus. 53 Finally, dogs are also amenable to studies that evaluate how rehabilitation activities affect implant function. 16
Relevance to Human Bone and Joint Conditions
Canine and human bone have many similarities, including formation of secondary osteons, epiphyseal fusion after maturity, comparable intracortical remodeling activity, 28 and age-associated bone loss. 10 The 2 species also have similar bone weight, density, and composition of bone organic, inorganic, and water fractions. 1,64 Humans and dogs also both lack substantial intrinsic healing ability for cartilage defects. 16 Key differences include the retention of plexiform bone on canine cortical periosteal and endosteal surfaces 45,81 and thinner articular cartilage in dogs. 16 Dogs have higher annual total-body trabecular bone turnover rates (averaging 94%–101% in male and female canine trabecular bone compared with an estimated 10%–55% in adult humans 41,60,64 ), which may partially explain the high rate of solid bony fusion and low rates of nonunion in canine models. 64 One important limitation of using dogs is that remodeling and apposition rates can vary significantly both within and between dogs. 25 For example, individual ribs in a single-purpose-bred Beagle may have statistically different bone remodeling parameters. 6
Pig (Conventional and Minipig)
Model Uses
Conventional pigs and minipigs are used to evaluate bone osteogenic material in craniomaxillary facial surgery models, 12 extra- and intraoral surgical approaches for dental implants, 18 interventions for femoral head osteonecrosis, and repair of cartilage and bone fractures. 60 Sufficient material for autogenous cancellous bone grafts can be harvested from the proximal humerus. 53 Skeletally mature conventional pig articular cartilage is also thick enough (∼1.5 mm) to create both full- and partial-thickness cartilage defects that can be monitored via arthroscopy. 16
Relevance to Human Bone and Joint Conditions
Bone mineral density, anatomy, morphology, remodeling rate, and healing of pigs are similar to humans. 64 Pigs have both growing and adult skeletal phases 5 with well-developed Haversian systems in mature bone. 10 Swine intra-articular cartilage and associated ligaments in synovial joints are similar to the conformation in human joints. 75 Disadvantages include high growth rates, excessive body weight, and difficulty in being handled compared with other species. 64 Pig tibial and femoral length is also relatively short, which can prevent direct testing of human-sized implants 64 or creation of stable, critical-size defects. 53
Sheep and Goats
Model Uses
Use of small ruminant orthopedic models is increasing, with reports of sheep and goat models growing from ∼5% of bone papers in the 1980s to 11% to 15% in the 2000s. 50,59 Small ruminants are used to model defect repair in long bones, bone filler materials in cranial defects, fracture repair devices, tissue response to wear debris, extraoral surgical approaches, and intervertebral disk replacements. 12,18 The relatively vertical alignment of the cervical vertebrae in small ruminants undergoes some of the same axial compression and rotation forces as the human spine. 53
Relevance to Human Bone and Joint Conditions
Small ruminants and humans have a comparable body weight, bone size, and bone-healing potential, as well as tibial blood supply (young sheep) and bone composition and remodeling rates (goats). 64 Additionally, the ratio of cartilage to subchondral bone, subchondral bone consistency, joint size, accessibility for arthroscopy, and limited intrinsic healing capacity in goats are similar to human joints. 2,16,33 Autogenous cancellous grafts can also be performed in small ruminants. 53 Despite these similarities, there are several important differences between small ruminant and human bone. Sheep cortical bone remains predominantly plexiform through 7 to 9 years of age. 64,66 Sheep undergo seasonal bone loss, and if a study occurs during winter (when bone formation is depressed), control animals may spontaneously lose and regain bone. 10 While in mature sheep, trabecular bone is more dense and stronger than human bone, immature sheep have weaker, low-density trabecular bone that is highly flexible (due to higher collagen content 54 ).
NHPs
NHPs and humans share significant genetic homology, have comparable osteonal remodeling in skeletally mature individuals, and develop similar diseases, including osteoarthritis and age-associated bone loss. 37 Comparable bone biopsy sites are also available in both species, such as vertebral bodies, the cortex of the seventh rib, proximal femur, and ilium. 4,63 NHPs are generally used in bone implant studies when other large animal models do not adequately represent human bone biomechanics and loading, including dental implants and vertebral repair products. The tooth and jaw structure of NHPs provide relevant information about stress responses to mechanical loading of dental implants. 18 Baboons are also used to evaluate specialized intervertebral artificial disks. 12 While NHPs have key advantages as a model for these specialized implant sites, implants for other sites such as long bones are sometimes better suited for other large animal models. NHP long bones can be too small to support adult human implants. For example, a mature male rhesus has a ∼16.5-cm-long femur with a 1.25-cm mid-shaft diameter while mature hounds can have a 20-cm-long femur with a 2.5- to 3-cm mid-shaft diameter. 53
Study Design, Nomenclature, and Scoring Criteria for Bone/Joint Implants
While many factors used to design effective toxicology studies of small and large molecules are also applicable to bone implant studies, there are several additional criteria to consider. Reactions to implants are generally time, not dose, dependent. 72 Implants designed to promote bone growth may require studies lasting longer than 6 months to allow sufficient time for bone regeneration, remodeling, and detection of implant absorption or degradation. 64 Also, implant placement almost always incites a significant nonspecific localized tissue reaction, secondary to surgical/implantation trauma. 72 It is therefore important to include sham surgery and/or control implants to differentiate reactions that are implant induced from those that are primarily implantation induced.
Histologic processing of bone and joint implant samples can be uniquely challenging, and variations in implants are too numerous to be addressed here. Readers interested in processing specific implant materials and common bone-staining methods are referred to recent publications. 4,70 Additional analytic techniques (macroscopic evaluation, radiographs, computed tomography [CT], micro-CT 8 ) can also provide key information on 3D structure and device surface-tissue interactions, factors that can be difficult to appreciate with histology alone.
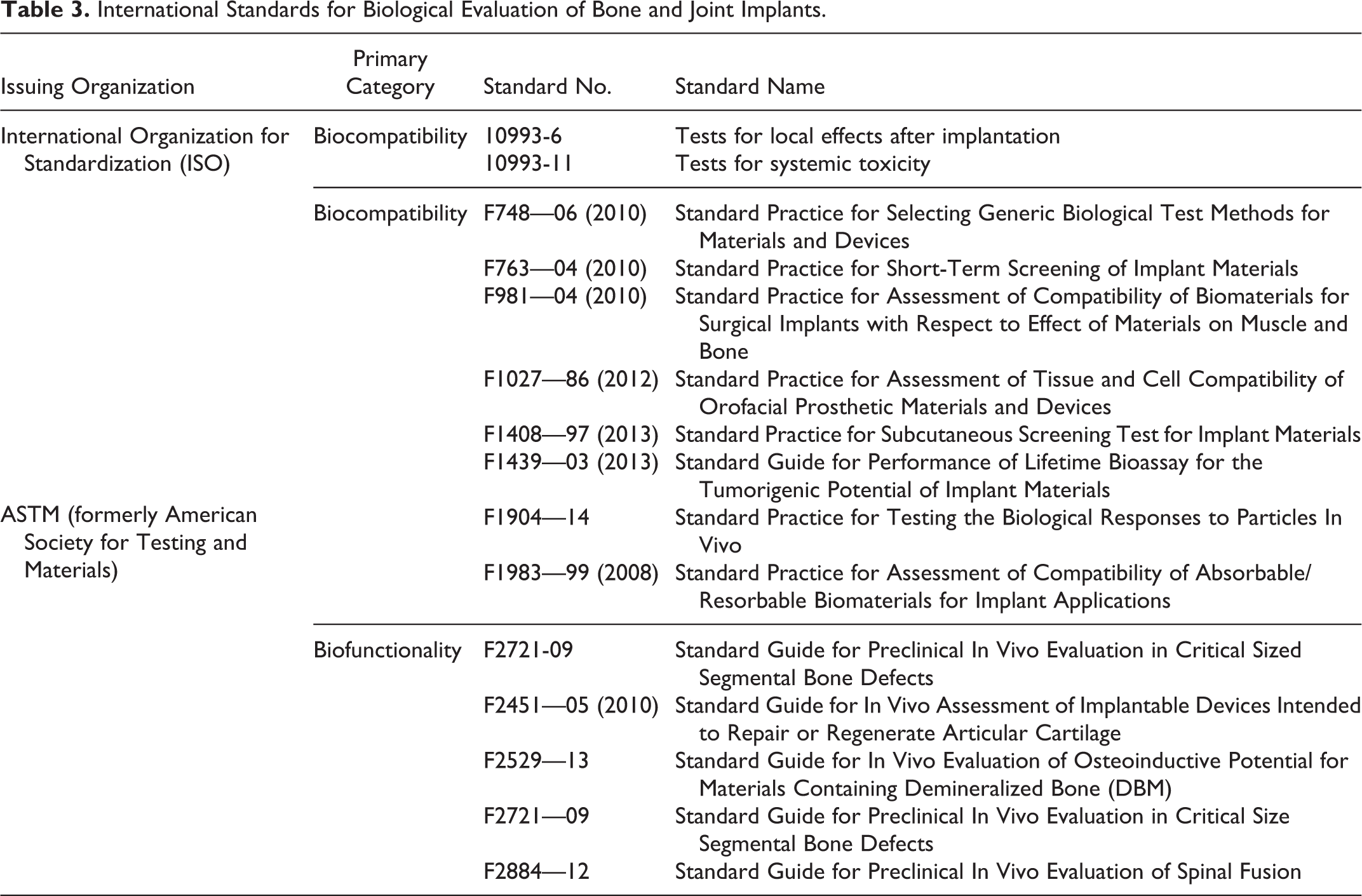
Excellent resources are available for both the design and interpretation of in vivo studies, including guidances that are the basis for implant regulations in several countries (Table 3). Clinical and basic bone researchers have developed standardized histomorphometry terms and evaluation methods to provide consistency across studies. 21,63 Cartilage scoring criteria are recommended by the International Cartilage Repair Society (ICRS, preferred for implant studies 31,49 ) and the Osteoarthritis Research Society International Histopathology Initiative (OARSI, preferred for drug-related effects 42,43,61 ). Finally, standard nomenclature and image resources for spontaneous bone and joint lesions are available in the forthcoming bone, cartilage, and tooth INHAND manuscript (http://goreni.org/index.php) and National Toxicology Program Non-neoplastic Lesion Atlas (http://ntp.niehs.nih.gov/nnl/guide/index.htm).
International Standards for Biological Evaluation of Bone and Joint Implants.
Concluding Thoughts
As implant development and clinical use continues to expand, it is important for pathologists evaluating bone and joint implant studies to understand the basics of bone structure and the advantages and disadvantages of different animal models. While no single animal model completely recapitulates human bone and joint development and repair, various species can provide key support when developing implants.
Footnotes
Acknowledgements
Dr David H. Johnson provided assistance with reference formatting and figure preparation.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
