Abstract
Norway (Rattus norvegicus) and black rats (Rattus rattus) are common peridomestic species, yet little is known about wild rat ecology, including their natural diseases. We describe gross and histological lesions in the respiratory tract of a sample of 711 wild urban rats. A subset was examined for 19 distinct categories of histological lesions in the respiratory tract. Testing for known respiratory pathogens included serology and polymerase chain reaction (PCR) of lung samples. Grossly evident lesions were rare (8/711; 1%). Upper respiratory tract inflammation was present in 93 of 107 (87%) rats and included rhinitis, submucosal and periglandular lymphoplasmacytic tracheitis, and/or tracheal intraluminal necrotic debris and was significantly associated (P < .05) with the presence of cilia-associated respiratory bacillus (CARB), Mycoplasma pulmonis, and increased body mass (odds ratio [OR] = 1.09; 95% confidence interval [CI] = 1.05–1.14 per 10 g). Within the lungs, peribronchiolar and/or perivascular lymphoplasmacytic cuffs were present in 152 of 199 rats (76%) and were also significantly associated (P ≤ .02) with CARB, M. pulmonis, and increased body mass (OR = 1.20; 95% CI = 1.14–1.27 per 10 g). Rats were frequently coinfected with M. pulmonis and CARB, and lesions associated with these pathogens were histologically indistinguishable. Pneumocystis sp was detected in 48 of 102 (47%) rats using PCR but was not significantly associated with lesions. This description of pathology in the respiratory system of wild rats demonstrates that respiratory disease is common. Although the impact of these lesions on individual and population health remains to be investigated, respiratory disease may be an important contributor to wild rat morbidity and mortality.
Norway and black rats (Rattus norvegicus and Rattus rattus) are cosmopolitan peridomestic species that are well adapted to living in urban habitats. 12 Negative impacts of rats on human society range from economic losses through agricultural crop destruction to zoonotic diseases. 12,13,22 Even though rats have lived in cities with people for centuries, many details of urban rat ecology remain unexplored. Specifically, little is known about the causes of natural morbidity and mortality in rats.
The life span of wild urban rats is considerably shorter than that of laboratory rats, with few individuals living beyond 1 year of age. 12 Conversely, laboratory rats live up to 3 years. 43 Factors contributing to the rapid population turnover among wild rats are unclear. Due to the high fecundity of rats, predation and human rat control activities, including trapping and poisoning, have limited effects on population size. 12 Mortality associated with resource competition does not explain why mature rats die, since they would be expected to outcompete juvenile rats. 12 Natural disease may be a key component to understanding morbidity and mortality in wild rats, but very little research has been done in this area.
Clinical signs compatible with respiratory infections have been observed in wild rats. 6 Previous studies have shown that wild rats have respiratory pathology, including pneumonia, often in association with hyperplasia of bronchus-associated lymphoid tissue (BALT) and laryngitis. 18,24,29,31 Respiratory pathogens identified in wild rats include Pneumocystis carinii, Pneumocystis wakefieldiae, cilia-associated respiratory bacillus (CARB), Mycoplasma pulmonis, Bordetella bronchiseptica, rat coronavirus (syn. sialodacryoadenitis virus), and Sendai virus.* Previous studies were based on small sample sizes or focused on specific pathogens or specific lesions. A recent wild rat study has identified several novel viruses; however, associated lesions were not described. 13 The full range of natural respiratory pathology in rats has not been evaluated.
Understanding natural morbidity and mortality, including respiratory disease, may be a key component to understanding the ecology of these species. Enhanced knowledge of wild rat diseases and urban rat ecology may have implications for ecologically based rat control, 42 zoonotic disease risk mitigation, 22 and laboratory biosecurity.
This study was undertaken to assess respiratory pathology in a large sample of live-captured urban rats, assess the occurrence of respiratory disease, and generate new information about the role of disease in the ecology of wild urban rat populations. The specific objective of this study was to describe, categorize, and determine the cause of respiratory pathology in wild urban Norway and black rats (R. norvegicus and R. rattus) from Vancouver, Canada.
Materials and Methods
Trapping and Rat Collection
Details of rat trapping methods have been described elsewhere. 39 Briefly, outdoor live trapping took place in an inner-city neighborhood of Vancouver, Canada, as part of the Vancouver Rat Project (www.vancouverratproject.com). At the Vancouver international shipping port, rats trapped by a private pest control professional company using snap-type lethal traps were also collected. Over the course of 1 year (September 2011–August 2012), 725 rats were trapped. General anesthesia was induced using isoflurane, followed by blood collection via cardiac puncture and euthanasia by intracardiac injection of pentobarbital. Data collected immediately included sex, species (determined by external morphology), body mass, number of cutaneous bite wounds, and sexual maturity (open vaginal orifice for females and scrotal testes for males). Rats were stored frozen at –30°C prior to autopsy and sample collection at the Animal Health Centre, British Columbia Ministry of Agriculture, Abbotsford, British Columbia. Immediate autopsy of fresh specimens was not feasible given the large sample size and geographic distance between the study site and laboratory. The University of British Columbia’s Animal Care Committee approved this study (A11-0087).
Autopsy and Tissue Collection
Rats were thawed at 4°C before undergoing a thorough, standardized autopsy and tissue collection protocol. For each rat, the head, larynx, trachea, and representative samples of the lungs were immersed in 10% neutral buffered formalin. Fresh samples of lung and samples or sterile swabs of all grossly observed purulent lesions were collected and stored at –80°C for further analysis.
Histopathology
Tissues from a total of 200 rats were evaluated using light microscopy for histological lesions. These included all rats with gross lesions in any organ system (n = 111) and a random selection of 89 rats with no visible gross lesions (selected using the “sample” function in R; R Development Core Team, Vienna, Austria) to ensure detection of lesions present at ≥3% prevalence (Survey Toolbox; Australian Centre for International Agriculture Research, Canberra, Australia).
Tissue sections prepared for microscopic examination included larynx (longitudinally through the larynx, including the epiglottis and oropharynx), trachea (transverse at the level of the thyroid glands), and lung (1–3 transverse lung lobe sections, including gross changes when present). Following routine processing, these tissues were embedded in paraffin and 5-μm tissue sections were stained using hematoxylin and eosin (HE). The heads from a random selection of 35 of the 200 rats were decalcified and sectioned transversely at the level of the eyes to examine the nasal cavity.
Based on initial microscopic findings of HE-stained slides, additional sections of selected cases were stained with Gram’s, Grocott methenamine silver (GMS), Okajima, and Steiner’s silver stains. Immunohistochemical (IHC) assays were performed on 1 lung tumor using murine monoclonal antibodies against cytokeratin (AE1/AE3; Sigma-Aldrich, Oakville, Canada) and vimentin (BioGenex, Fremont, CA) at 1:200 and 1:100 dilutions, respectively. 5 Samples or sterile swabs of all grossly observed purulent lesions underwent routine aerobic and anaerobic bacterial isolation and identification.
Lesion Categorization
Nasal cavities, sinuses, larynx, trachea, and lung from each rat were initially screened using light microscopy for morphological abnormalities. For some rats, the aforementioned tissues were not adequately preserved (eg, autolysis and freeze-thaw artifact) and/or were not sectioned for examination of certain lesions.
A system for defining and categorizing lesions (Suppl. Tables S1 and S2) was created based on the changes observed in the initial screen, in combination with a review of lesions commonly described in the respiratory tract of laboratory rats. 3,9,32,36,38,43 Inclusion and exclusion criteria were defined for each lesion to enhance objectivity. Stringent binary classification schemata were used to accurately and conservatively evaluate the presence and apparent prevalence of microscopic abnormalities in this sample of rats. Lesion severity or scoring was not assessed. Once the classification scheme was developed, all tissues were reevaluated and categorized by these criteria.
Pathogen Serology and Polymerase Chain Reaction
To identify circulating respiratory pathogens in this population, the heaviest (presumably oldest) 12 male and female rats from city blocks where the majority of rats collected had been analyzed histologically were selected for serological screening (n = 10). Sera were shipped to Research Animal Diagnostic Services, Charles River Laboratories (Wilmington, MA), where a multiplex fluorometric immunoassay (MFIA) against a panel of 17 pathogens of rats, including Sendai virus, murine pneumonia virus, rat coronavirus, M. pulmonis, CARB, and Pneumocystis sp, was performed. A positive result for any respiratory pathogen was confirmed by an indirect fluorescent antibody assay. Only samples that were positive by both methods were recorded as positive in this analysis.
In addition to testing for known rat and murine respiratory viruses, and to screen for possible exposure to novel paramyxoviruses, sera from 139 rats included in the histological screen were tested for antibodies against bovine respiratory syncytial virus (BRSV; a pneumovirus distantly related to murine pneumonia virus) and bovine parainfluenza virus 3 (BPIV-3; a virus related to Sendai virus), using indirect enzyme-linked immunosorbent assay (ELISA) as described, with minor modifications. 10 Briefly, partially purified antigen was prepared from lysed Vero cells infected with the BRSV RB94 isolate or the BPIV-3 isolate, with lysed uninfected Vero cells used as the control. A 1:50 dilution of rat serum or control serum was added to duplicate wells of antigen-coated plates. Horseradish peroxidase–conjugated Rec-protein G (H+L) (Invitrogen, Burlington, Canada) at 1:5000, followed by substrate ABTS (KPL; Mandel Scientific, Guelph, Canada), was used to detect BRSV- and BPIV-3–specific antibodies. Absorbance was read at 405 nm. Optical density (OD) values were converted to OD units by comparing net (antigen – tissue control) sample OD values with the net OD value of a human standard reference serum on each plate.
Based on serology results, frozen lung tissues from 102 rats were sent to Charles River Laboratories for further analysis. Polymerase chain reaction (PCR) for respiratory pathogens identified by serology was performed using TaqMan techniques as previously reported. 21 Controls included 100 copies of an algal gene sequence to monitor for sample-mediated inhibition of PCR amplification. All assays fulfilled standard qualification criteria, including a limit of detection of 1 to 10 target copies. All tests were performed in duplicate and included positive (100–1000 copies for each agent) and negative (Chinese hamster ovary DNA) controls.
Statistical Analysis
Bivariate associations between each inflammatory microscopic change in the respiratory tract and pathogen PCR results were assessed using simple logistical regression. Rats with equivocal PCR results were considered positive in this analysis. Lesions with relationships that were statistically significant (P ≤ .05) were then combined to create new composite variables. Any rat that was positive on one or more of the component variables was considered positive on the composite variable. Agreement among histopathology detection of CARB and CARB PCR was evaluated using Cohen’s κ statistic.
Respiratory tract lesions were used independently as the outcome in logistic regression analyses investigating the relationship between the aforementioned morphological changes and a variety of explanatory variables. These variables included season (September–November = fall; December–February = winter; March–May = spring; June–August = summer), sex, sexual maturity (immature vs mature), body condition as assessed by volume of internal fat stores (subjective score of 0–2), body mass (considered in 10-g increments), species, presence or absence of cutaneous bite wounds, and number of bite wounds.
Variables that were significantly associated with the outcome of interest at an α level of ≤.10 on bivariate analysis were considered for inclusion in a multivariate generalized linear mixed model (GLMM) controlling for clustering by city block of origin. The goal of the model-building strategy was to identify the most parsimonious set of explanatory variables that predicted the outcome. Individuals for which data were missing for one or more of the variables under study were excluded. To evaluate variations in demographic characteristics among rats with and without gross lesions, the relationships between mass and sex, as well as the presence of gross lesions (vs no gross lesions), were evaluated using a 2-sided t test and χ2 test, respectively. All statistical analyses were conducted using R.
Results
Gross and Histopathological Findings
Of the 725 rats trapped, 14 were excluded from analysis because of incomplete data or autolysis. Grossly evident lung lesions were observed in 8 of 711 rats (1%). Lesions, bacterial culture results, and individual demographic characteristics of affected rats are listed in Supplemental Table S3. No gross lesions were identified in the external nares, nasal cavity, pharynx, larynx, or trachea.
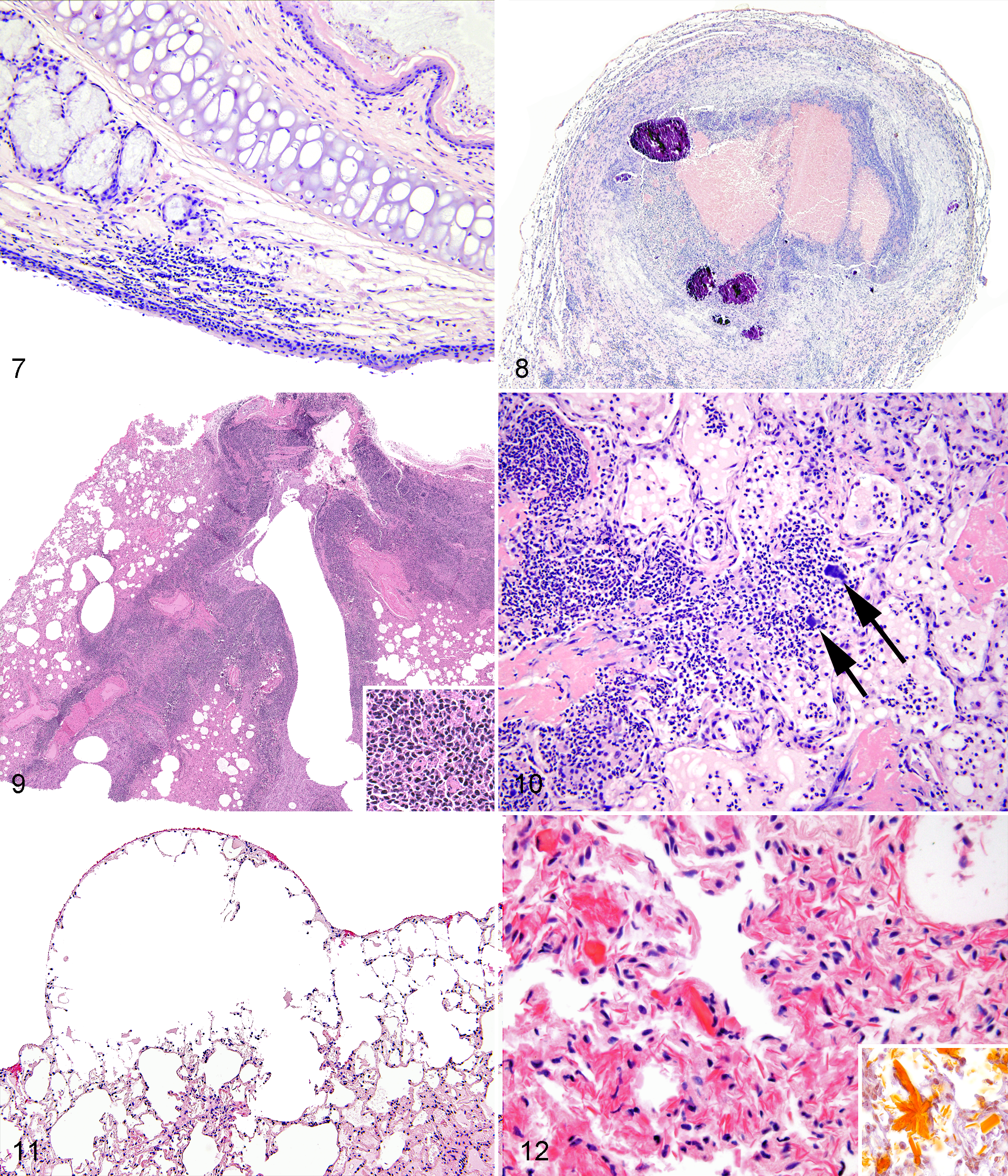
Among the grossly evident lesions, purulent bronchiectasis or intraparenchymal abscesses were observed in the lungs of 4 rats (rat Nos. 1–4). Distribution of these lesions ranged from a small, focal (5-mm3) area to a lesion encompassing the entire right lung lobe (Suppl. Figs. S1, S2).
In 1 rat, a large (2 × 2–cm) abscess occupied approximately 60% of the thorax and compressed adjacent lung tissue (rat No. 5; Suppl. Fig. S3). A metal airgun pellet was found within this abscess. Presumably from the pellet entrance wound, there were 2 small (3 × 3 mm) abscesses on the medial aspect of the right scapula. Staphylococcus aureus was cultured from all 3 abscesses. In rat No. 6, approximately 80% of the left lung lobe was severely congested, atelectatic, and consolidated while, elsewhere, the lung was hyperinflated and failed to collapse. Histologically, these areas corresponded to large, coalescing areas of hemorrhage within the pulmonary parenchyma with multifocal clusters of hemosiderophages (confirmed with Prussian blue stain; Suppl. Fig. S4). This rat also had locally extensive areas of subcutaneous hemorrhage consistent with anticoagulant rodenticide poisoning.
Two rats had masses in the lung parenchyma. The right cranial lung lobe of rat No. 7 contained a 2-mm, firm, spherical mass with a central depression on its pleural surface (Suppl. Fig. S5). Histologically, this mass was consistent with a focus of mucous cell metaplasia with severe, granulomatous inflammation. 38 Rat No. 8 had multiple neoplastic masses (1–2 cm3) arising from the right cranial and middle lung lobes (Suppl. Fig. S6). Histologically, this mass was consistent with a round cell tumor, perhaps of histiocytes. 40 Immunohistochemistry was negative for cytokeratin and weakly positive for vimentin.
Histological lesions identified in this study are described and quantified in Supplemental Tables S1 and S2. The most common upper respiratory tract lesions included tracheal necrotic debris (22/179 [12%]; Fig. 1), lymphoplasmacytic submucosal tracheitis (82/175 [47%]; Fig. 2), tracheal submucosal gland adenitis (20/168 [26%]; Fig. 3), tracheal submucosal gland ectasia (43/168 [12%]; Fig. 4), rhinitis (11/36 [31%]; Fig. 5), and laryngitis (24/112 [21%]; Fig. 7). Using light microscopy, CARB was visualized in 42 of 113 rats (37%; Fig. 6). Note that not all upper respiratory tract tissues were available from all rats.
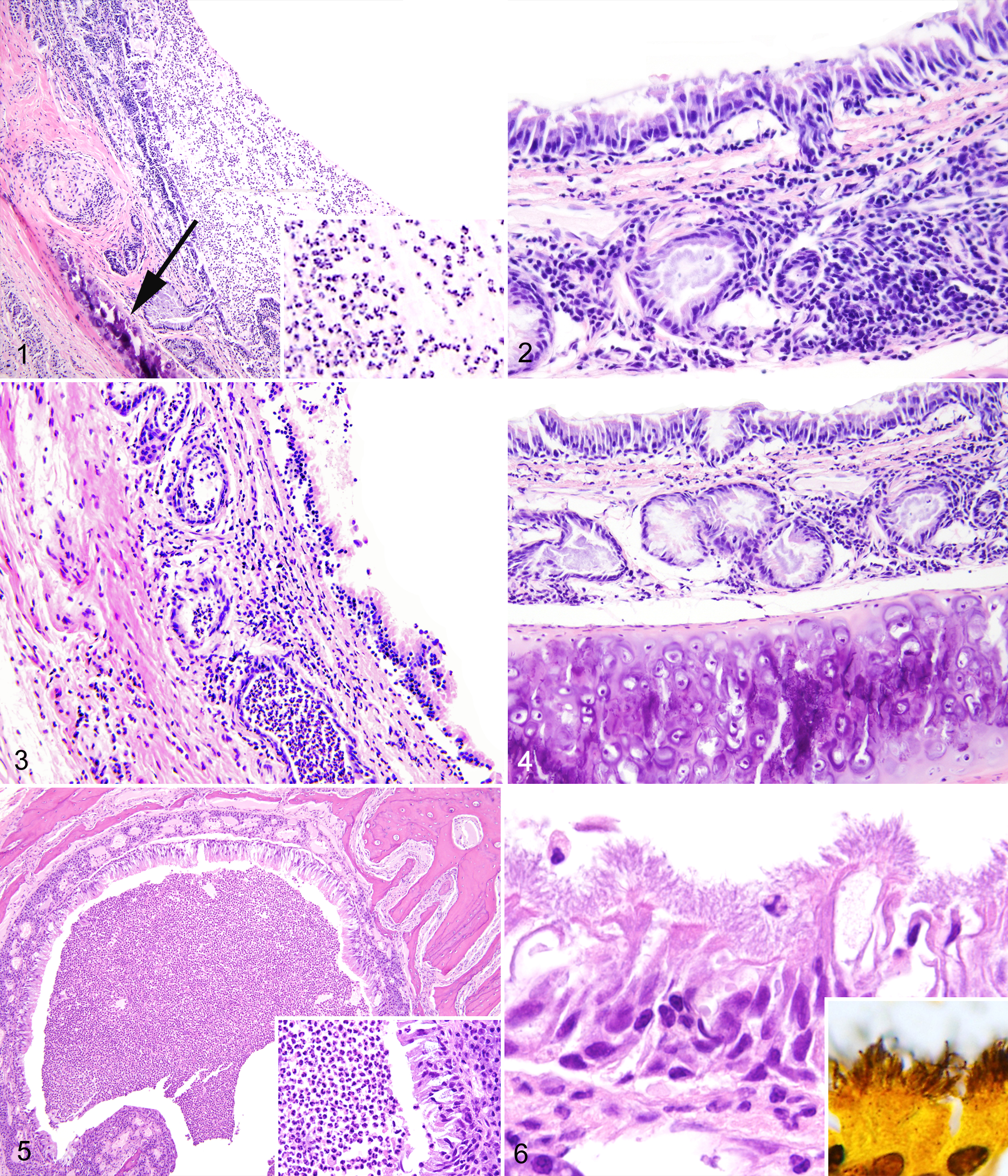
Upper respiratory tract inflammation, Norway rats (Rattus norvegicus). Hematoxylin and eosin (HE).
Laryngitis, larynx, Norway rat. Lymphocytes and plasma cells infiltrate the connective tissue adjacent to the laryngeal cartilage. Hematoxylin and eosin (HE).
Lower respiratory tract inflammatory lesions included abscesses (3/199 [1%]; Fig. 8), hyperplasia of BALT (4/9 [44%]; Fig. 9), and bronchopneumonia (5/199 [3%]; Fig. 10). Frequent noninflammatory changes included emphysema (144/199 [72%]; Fig. 11) and eosinophilic crystals (96/199 [48%]; Fig. 12). Lesions consistent with inducible BALT (iBALT) included peribronchiolar lymphoplasmacytic cuffs (143/199 [72%]; Fig. 13), perivascular lymphoplasmacytic cuffs (102/199 [51%]; Fig. 14), and perivascular mixed inflammatory cuffs (54/199 [27%]). 37
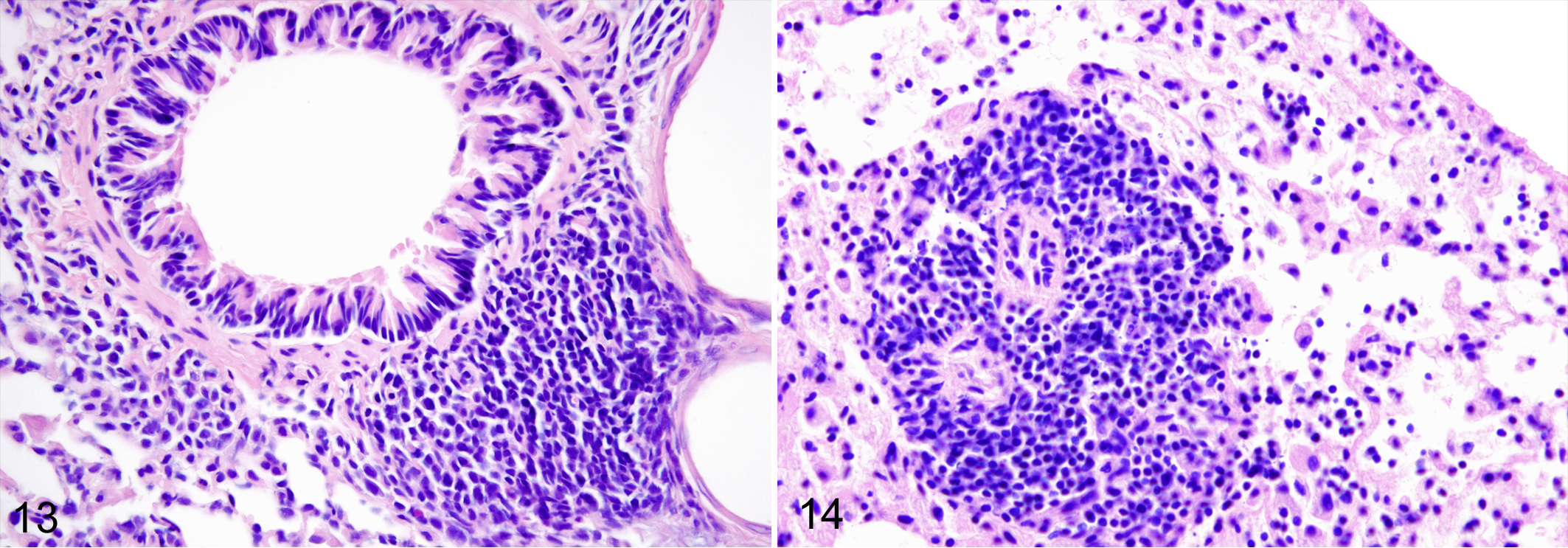
Lymphoplasmacytic cuffs, lung, Norway rats. Hematoxylin and eosin (HE). These changes were significantly associated (P ≤ .02) with cilia-associated respiratory bacillus and Mycoplasma pulmonis.
Several other histological changes identified were not included in the lesion categorization scheme. These lesions were either rare or could not be definitively diagnosed due to artifacts of tissue preservation. These included increased number of alveolar histiocytes, atelectasis, edema, and interstitial pneumonia (alveolar septa thickened by increased number of inflammatory cells and sometimes accompanied by hyaline membranes). The lung of 1 rat contained a circular, intra-alveolar, birefringent foreign particle with minimal inflammatory response. Multinucleated giant cells, macrophages, lymphocytes, and plasma cells filled the alveoli in 2 rats, one of which also had mucous cell metaplasia (rat No. 16 described above). Resolving pulmonary hemorrhage with hemosiderin-laden macrophages (confirmed with Prussian blue stain) was found in 2 rats (rat No. 4 described above). Ectopic bone and interstitial mineralization were identified in 2 rats. Pneumocystis sp organisms were not identified in any of the tissues examined that were stained with HE and GMS. Changes in the pulmonary blood vessels have been described elsewhere. 39
Microbiology Findings
All (10/10) rat sera had antibodies against Pneumocystis sp (100%), and 5 of 10 had antibodies against M. pulmonis and CARB (50%). No rats were seropositive for Sendai virus, murine pneumonia virus, or rat coronavirus. Given the sample size, there is 95% confidence that the seroprevalence of these viruses was 30% or less. 19 None of 139 rats tested by ELISA had IgG antibodies in serum that were reactive with either BRSV or BPIV-3. Given the sample size, there is 95% confidence that the seroprevalence of novel paramyxoviruses was 2% or less. 19 PCR for M. pulmonis, CARB, and Pneumocystis sp was performed on previously frozen lung tissues from 102 rats. Twenty-five (25%), 83 (81%), and 48 (47%) were infected with M. pulmonis, CARB, and Pneumocystis sp, respectively.
Statistical Evaluation
Demographic characteristics of the study population have been previously described. 39 Among rats included for histological examination, 184 were Norway rats and 16 were black rats. There was no significant difference in the number of male and female rats with and without gross lesions. However, rats with gross lesions were more likely to be heavier (P < .001) than rats without gross lesions.
Perivascular mixed inflammatory cuffing was not associated with any pathogens tested by PCR (P ≥ .15), and other lower respiratory tract lesions (abscess, BALT hyperplasia, bronchiolectasis, bronchopneumonia, and pleuritis) were too infrequent for meaningful comparisons, and therefore these lesions were excluded from the composite variable.
Peribronchiolar and perivascular lymphoplasmacytic cuffs were independently strongly associated with infection with CARB and M. pulmonis (P ≤ .02 for all comparisons) but not with Pneumocystis sp (P ≥ .37). These 2 lesion categories were combined to create a new composite variable called iBALT. iBALT was present in 152 of 200 (76%) rats examined.
There was a strong association between infections with CARB and M. pulmonis. Of the 53 rats infected with either pathogen, 20 (38%) were infected with both. Rats that were infected with CARB had increased odds of being infected with M. pulmonis (odds ratio [OR] = 7.00; 95% confidence interval [CI] = 2.52–22.92). Cohen’s κ statistic found moderate agreement between histological detection of CARB and PCR results (κ = 0.515; 95% CI = 0.316–0.714; P < .001).
Lymphoplasmacytic tracheitis, intraluminal necrotic debris, and rhinitis were also associated with CARB and M. pulmonis (P ≤ .05 for all comparisons) but not with Pneumocystis sp (P ≥ .47). These lesion categories were combined to create a new composite variable, “upper respiratory tract inflammation” (URTI). Other upper respiratory tract inflammatory lesions were not significantly associated with any pathogens tested by PCR, and therefore these were excluded from the composite variable (data not shown). The composite variable URTI was present in 93 of 107 (87%) rats examined.
Demographic Associations
Results of bivariate analysis are presented in Supplemental Tables S4 and S5. For both the URTI and iBALT variables, the final GLMM included only body mass. The odds of an individual rat having either upper respiratory tract inflammation or iBALT increased with increased body mass (for URTI: OR = 1.09 [95% CI = 1.05–1.14 per 10 g]; for iBALT, OR = 1.20 [95% CI = 1.14–1.27] per 10 g).
Discussion
Macroscopic respiratory tract lesions were rare in this population of rats, but the few identified were severe. The true prevalence of macroscopic lesions likely was underestimated, since live trapping may bias toward rats that are healthy enough to be actively foraging. Conversely, microscopic lesions were very common, affecting substantial proportions of the study population. The results are in broad agreement with Brogden et al, 4 who found that 20 of 28 (71%) wild Norway rats had histological evidence of chronic respiratory disease, and with Laurain, 28 who briefly described pulmonary abscesses, but they differ from Kakrada et al, 24 who found only 2 of 27 wild rats affected by lung lesions. The inflammatory lesions observed in our study are similar to those described in pet rats and in early literature regarding laboratory rat pathology when laboratory animal pathogens were poorly controlled. 16,17,26
There were strong statistical associations between the composite changes we defined as “iBALT” (perivascular and peribronchiolar cuffing) and “URTI” and infection with CARB and M. pulmonis. CARB is an unclassified, filamentous, Gram-negative bacterium that intermingles with cilia of large airways and is transmitted through direct contact. 2 It has been identified at high apparent prevalence (52%–86%) in wild rats from Maryland, Texas, Iowa, and New Zealand. 4,11,24,31 The lower prevalence in this study (37%) may reflect our use of light microscopy for screening rather than more sensitive PCR tests. There was significant agreement between PCR and light microscopy results, and thus, light microscopy remains a useful and inexpensive method for detection of this bacterium. In other studies of laboratory and wild rats, CARB has been associated with chronic bronchiolitis, perivascular and peribronchiolar cuffing, rhinitis, and tracheitis, 4,31 which is in agreement with this study. The severity of peribronchial and peribronchiolar cuffing induced by CARB is reported to increase with time in laboratory rats, and coinfection with M. pulmonis can lead to more severe lesions. 2,15
M. pulmonis is an important respiratory pathogen of laboratory rats and is transmitted both vertically and through aerosols. 2 In this study, M. pulmonis was associated with rhinitis, lymphoplasmacytic tracheitis, and peribronchiolar and perivascular lymphoplasmacytic cuffing. Coinfection with M. pulmonis and CARB was common in this study, and the lesions associated with these pathogens were histologically indistinguishable. Body mass was the only demographic characteristic associated with the lesions and pathogens of iBALT and URTI, as defined in this study. If body mass is considered a proxy for age, 12 this association likely reflects increased exposure to CARB and M. pulmonis over time, chronic infection, and/or pathogen transmission resulting from social interactions among large rats.
Peribronchiolar and perivascular lymphocytic cuffs may not actually be inflammatory lesions but rather iBALT, which functions as tertiary lymphoid structures in the lungs. 14,37 These lymphoid aggregates develop in response to chronic immune stimulation, which, in this case, may have been a response to M. pulmonis, a recognized B- and T-cell mitogen in rats, 8,33 or to antigens of the more commonly identified CARB, for which the effects on BALT have not, to our knowledge, been specifically examined in rats.
Pneumocystis sp was identified in 48 of 102 (47%) rats in this study using PCR and all rats tested with serology (10/10 [100%]), but no lesions were statistically associated with this organism. Furthermore, no organisms were identified using histology, which may be due to poor sensitivity of this technique in immunocompetent rats, low numbers, or artifactual changes in the tissues examined. The apparent prevalence was lower than that reported by Palmer et al, 35 who found Pneumocystis sp in 75% of wild Norway rats, also with PCR. It is possible that rats in the current study were proportionately older and had cleared the infection. Based on histology, apparent prevalence has ranged from 2% to 100% in wild Norway rats from Europe, Brazil, Venezuela, and Japan. 41 The lack of distinguishing lesions associated with Pneumocystis sp in our study population contrasts with studies of infection in immunocompetent laboratory rats, in which perivascular lymphocyte cuffs and interstitial pneumonia were described, and the severity of inflammation was correlated with the concentration of P. carinii DNA. 20,30 It is unclear why rats in our sample had no lesions statistically associated with Pneumocystis sp, despite the presence of previously described perivascular lymphocyte cuffs that are associated with this infection. There are several possible explanations for this discrepancy. Rats in this sample may have developed adaptive immune responses that cleared infection and subsequently led to lesion resolution. Host-pathogen adaptation may be better among wild rats. Finally, the high prevalence of M. pulmonis and CARB and their associated lesions may have masked lesions associated with Pneumocystis sp.
Exposure to rat coronavirus, Sendai virus, and murine pneumonia virus was not detected in this study using serology. These negative findings may reflect the small sample size tested by serology (limited by budget) and a prevalence less than 30%, which is the highest prevalence that could reliably be detected in a sample of 10 animals. 19 However, the lack of antibodies to BRSV and BPIV-3 in a larger sample of rats may be a further indication that rats in this population were not exposed to respiratory paramyxoviruses that may induce cross-reactive antibodies to these and related viruses. 25 A recent study of 114 wild rats from New York City also did not identify paramyxoviruses. 13 In a previous study, the seroprevalence to rat coronavirus in wild rats was 92% but was only 4% to Sendai virus. 11 The dynamics of respiratory paramyxovirus infections in wild rat populations beg further investigation.
The eosinophilic crystals noted histologically in 48% of rats are considered an artifact of freezing, intracardiac puncture, and barbiturate euthanasia. They were found only in euthanized rats and not in rats found dead or killed in snap-traps. The crystals were extracellular, were not accompanied by inflammation, and bore no resemblance to eosinophilic crystalline pneumonia. 1,23 Given the shape and positive staining with Okajima, these are likely crystalized hemoglobin. 38
There are several potential limitations to the current study. Trapping seldom samples an animal population uniformly and might oversample or undersample certain segments, including rats with more and less respiratory pathology. Macroscopic and microscopic tissue changes associated with blood collection, euthanasia, and freezing may have masked subtle lesions. For example, alveolar histiocytosis, atelectasis, edema, and interstitial pneumonia could not be assessed in all rats due to tissue artifacts and were therefore excluded from the screening criteria. The inclusion/exclusion criteria were designed to err on the side of exclusion with only the most obvious and severe lesions meeting the inclusion criteria. The presence of these tissue artifacts likely contributed to an underestimation of the apparent prevalence and extent of respiratory lesions among these rats. An alternative euthanasia method to intracardiac pentobarbital, immediate autopsy and tissue collection could improve tissue preservation and lesion assessment in future studies. Few black rats were included in this study (n = 16), and it is not known whether the proportion of black rats in our sample represents the true ratio of Norway and black rats in the study area.
Strengths of the current study include the large sample size, robust statistical analysis, and systematic lesion categorization. Previous studies of natural respiratory tract pathology in wild rats have focused on a specific lesion or pathogen in relatively small samples. † The inclusion of autopsy and histopathology in our study permits broad assessment of the impact of pathogens and associated lesions on the health of individual animals.
Although respiratory pathology was common in this population of wild rats, the effects of these lesions on survival, reproduction, and longevity have not been evaluated. The high prevalence of lesions and associated infections may reduce the general fitness of affected individuals, predator evasion, adaptation to changing environments, and population parameters. These results also highlight the high rates of M. pulmonis, CARB, and Pneumocystis sp infection in wild rat populations and thus serve as a reminder of the critical importance to exclude wild rats from laboratory animal facilities.
Conclusion
This study describes pathology in the respiratory tract in a large sample of wild rats. Overall, gross lesions were uncommon. Microscopic respiratory lesions were common and strongly associated with CARB and M. pulmonis. The prevalence and severity of respiratory lesions found in this study suggest that disease may be a factor contributing to the short life span of wild urban rats. It is important to understand natural disease in wild rats to better characterize their ecology, design ecology-based rodent control strategies, and mitigate disease risks.
Footnotes
Acknowledgements
This study was made possible by the efforts of K. Parsons, A. Feng, V. Chang, D. Rempel, T. Urness, and H. Anholt. I. Shirley, C. Stewart, K. Brown, and C. Rhodes provided invaluable technical assistance. This study was undertaken as part of a Master’s of Veterinary Science research project, and appreciation is given to the following committee members: S. Gomis, T. Bollinger, and B. Kidney. An abstract of this study was presented at the 2013 Annual Meeting of the American College of Veterinary Pathologists in Montreal, Canada, and the 2014 Annual Meeting of the American Association of Veterinary Laboratory Diagnosticians in Kansas City, MO.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Canadian Institutes of Health Research (MOP–119530), the Western College of Veterinary Medicine University of Saskatchewan Interprovincial Graduate Student Fellowship, and Charles River. Serology for paramyxoviruses was supported in part by discretionary funds (Ellis) to study viral immunology.
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
