Abstract
The Perdido Key beach mouse (Peromyscus polionotus trissyllepsis) is a critically endangered subspecies of the oldfield mouse. The captive population, currently maintained by 3 Florida zoos, is entirely derived from just 3 wild-caught ancestor mice. Necropsy and histopathology revealed chordoma of the vertebral column in 38 of 88 (43%) mice. The tumors were locally expansile and invasive masses of large physaliferous (vacuolated) cells with small, round, hyperchromatic nuclei, similar to the “classic” form of chordomas described in humans. Primary tumors rarely contained small amounts of bone and cartilaginous matrix, characteristic of the “chondroid” form. Neoplastic cells contained abundant granules positive by the periodic acid–Schiff reaction. Brachyury and cytokeratin AE1/AE3 were detected in neoplastic cells by immunohistochemistry, but uncoupling protein 1 was not identified. Primary tumors occurred along the entire vertebral column—cervical, 5 of 38 (13%); thoracic, 16 (42%); lumbar, 13 (34%); and sacral, 10 (26%)—and 10 (26%) mice had multiple primary masses. Metastases to the lungs were noted in 13 of the 38 (34%) mice. Mice diagnosed with chordomas postmortem ranged from 424 to 2170 days old, with a mean of 1399 days. The prevalence of chordoma was not significantly different between males (n = 23 of 50; 46%) and females (n = 15 of 38; 39%).
Keywords
The Perdido Key beach (PKB) mouse (Peromyscus polionotus trissyllepsis) is 1 of 7 extant coastal subspecies of the oldfield mouse. It is the only subspecies of beach mouse listed as critically endangered on the International Union for Conservation of Nature Red List, 21 and it has been on the federally endangered list since 1985. The PKB mouse has a naturally small range of 9.8 miles of beach dune habitat on the barrier island, Perdido Key, Florida, and it is geographically and genetically isolated from the other subspecies. 35 This small range has been further reduced and severely fragmented due to human development along the island’s coast. In 1986, the natural population was reduced to as few as 30 known individuals inhabiting a single area of the island. 36 In 2004, 8 mice were taken into captivity to rescue the subspecies from potential extinction due to hurricane Ivan. 36 Currently, all the captive PKB mice, housed at 3 zoos in Florida, are derived from just 3 individuals—a male and female from the original 8 captured in 2004 and 1 female captured in 2003. 29 In 2010, captive mice were used to reestablish a wild population on the west end of Perdido Key. 13 Since then, the captive population has been shown to have reduced genetic heterozygosity, and there is general concern regarding the possible loss of genetic diversity within the remaining wild PKB mouse population. 18 To date, however, there have been no reports of negative impacts on either the wild or captive populations due to a loss of genetic diversity.
Necropsy and histopathology of a subset of the captive PKB mouse population recently revealed a high prevalence of chordomas, tumors thought to be of notochord origin that arise from intravertebral notochord remnants. 8,38 Chordomas have been reported in a number of species: humans, in which both sporadic and familial forms have been identified, 26,42 as well as dogs, 25,34,40 cats, 5,6 mink, 14 ferrets, 7,12,16,39 Sprague-Dawley and Fischer 344 rats, 31,44 and zebrafish. 4,11 Typically, chordomas are rare tumors, which have been reported to occur in humans at 0.08 per 100 000 in the United States 22 and among Fischer 344 rats, <0.05%. 31 In ferrets, chordomas are relatively commonly reported; however, neither prevalence nor incidence has been calculated. The purpose of this study was to describe the gross and microscopic pathology, the prevalence, and the biological behavior of a frequent and life-limiting tumor newly discovered among the captive population of the critically endangered PKB mouse.
Materials and Methods
Necropsy and histopathologic examinations were performed on 88 PKB mice. Of these, 68 were submitted by the Santa Fe College Teaching Zoo to the University of Florida, College of Veterinary Medicine, Anatomic Pathology service, from November 2007 through June 2014. The remaining 20 mice were submitted by the Palm Beach Zoo and Conservation Society to Northwest ZooPath from February 2008 through August 2014. All retained histology slides were retrospectively examined. Since initial examination revealed metastases only in lung (which was examined for all 88 samples) and vertebral columns were not included in routine histopathology, all retained samples of formalin-fixed lung and vertebral column were processed and examined. Of the mice submitted to the University of Florida, longitudinal or serial cross sections of the vertebral column from atlas to tail base (tails were not evaluated histologically) were examined for 24 mice; partial vertebral columns were similarly examined for 17 mice; and the vertebral column was assessed grossly for only 27 mice. Of the mice submitted to Northwest ZooPath, the vertebral column was either wholly or partially examined histologically for 17 cases and only grossly for 3 cases.
The birth and death dates of mice were obtained from the PKB mouse studbook 29 and supplemented by information provided with the submission. Mouse sex was confirmed at the time of necropsy. Statistical analyses were performed using Excel with XLSTAT add-in version 2013.6.03 (Addinsoft, New York, NY) or InStat version 3.10 (Graphpad Software Inc, La Jolla, CA).
Tissues collected at necropsy were fixed in 10% neutral buffered formalin, paraffin embedded, sectioned at 2 to 5 μm, and stained with hematoxylin and eosin. The oldest tissues were maintained in formalin for 6 years before being embedded. Sections of spinal cord were decalcified in modified Von Ebner’s hydrochloric acid at the University of Florida or in RapidCal (BBC Biochemical Corp, Mount Vernon, WA) at Northwest ZooPath. Selected sections were stained with periodic acid–Schiff and mucicarmine or with oil red O (ORO) on formalin-fixed frozen lung tissue.
Immunohistochemistry was performed on select chordomas to identify brachyury (sc-17743, Santa Cruz Biotechnology Inc, Dallas, TX), uncoupling protein 1 (UCP1; ab23841, Abcam, Cambridge, MA), and cytokeratin (Pan Cytokeratin [AE1/AE3], Biocare Medical, Concord, CA). Lung and spinal column sections from 4 mice were used for brachyury and from 2 mice each for UCP1 and cytokeratin.
For brachyury immunohistochemistry, heat-induced antigen retrieval was performed using citra solution (Biogenex, Fremont, CA). Quenching and blocking were performed using the Avidin/Biotin Blocking Kit (Vector Laboratories Inc, Burlingame, CA) and 10% horse serum prior to incubation with the primary antibody. Brachyury was complexed with biotinylated horse anti-goat IgG and Vectastain ABC Kit, Standard (Vector Laboratories Inc). For UCP1 immunohistochemistry, heat-induced antigen retrieval was performed in EDTA buffer (Thermo Scientific, Waltham, MA). Blocking was performed using Background Sniper (Biocare Medical) prior to incubation with the primary antibody that was complexed with a polyclonal goat anti-rabbit secondary antibody, Mach 2 Rabbit HRP-Polymer (Biocare Medical). Detection for both antibodies was performed using the DAB Chromogen Kit (Biocare Medical) and counterstained using Vector hematoxylin (Vector Laboratories Inc). All products were used as recommended by the manufacturers. Ferret cervical chordoma tissue was used as a positive control for brachyury and a negative control for UCP1. PKB mouse brown fat was used as an internal control.
Cytokeratin staining was performed using Carezyme I: Trypsin Kit (Biocare Medical) for antigen retrieval by digestion. Endogenous peroxidase was quenched using Peroxidaze 1 (Biocare Medical), and Background Punisher (Biocare Medical) was used for blocking nonspecific binding. The primary antibody was complexed with Mouse-on-Canine HRP-Polymer (Biocare Medical) and stained with the Betazoid DAB Chromogen Kit (Biocare Medical). Counterstaining was performed using CAT Hematoxylin (Biocare Medical). All products were used as recommended by the manufacturer. Canine haired skin was used as a positive control.
Results
Of the 88 PKB mice examined, a total of 38 (43%) had histologic evidence of one or more primary vertebral chordomas: cervical, 5 of 38 (13%); thoracic, 16 (42%); lumbar, 13 (34%); and sacral, 10 (26%). Chordomas were significantly less likely to develop in the cervical region compared to the thoracic region in 4-way analysis (P = .04, Kruskal-Wallis test with Dunn multiple comparisons test), but differences among the other regions were not significant. Of 38 mice, 10 (26%) had ≥2 masses, and metastasis to the lungs was detected in 13 (34%) mice. The location of primary tumors and the occurrence of metastasis are listed in Supplemental Table 1.
Gross and Histopathology
Among the 38 PKB mice with chordomas, 5 primary tumors (in 4 mice) were grossly visible. Two chordomas occurred on the ventral aspect of the caudal thoracic vertebrae, and 1 each was on the ventrolateral aspect of the cranial thoracic vertebrae, the thoracolumbar vertebrae of the cranial abdomen (Fig. 1), and the dorsal aspect of the caudal thoracic vertebrae associated with kyphosis and raising the skin on the back. The chordomas were 1 to 12 mm in diameter; white, tan, or translucent; gelatinous; nodular to multinodular; and soft or firm.
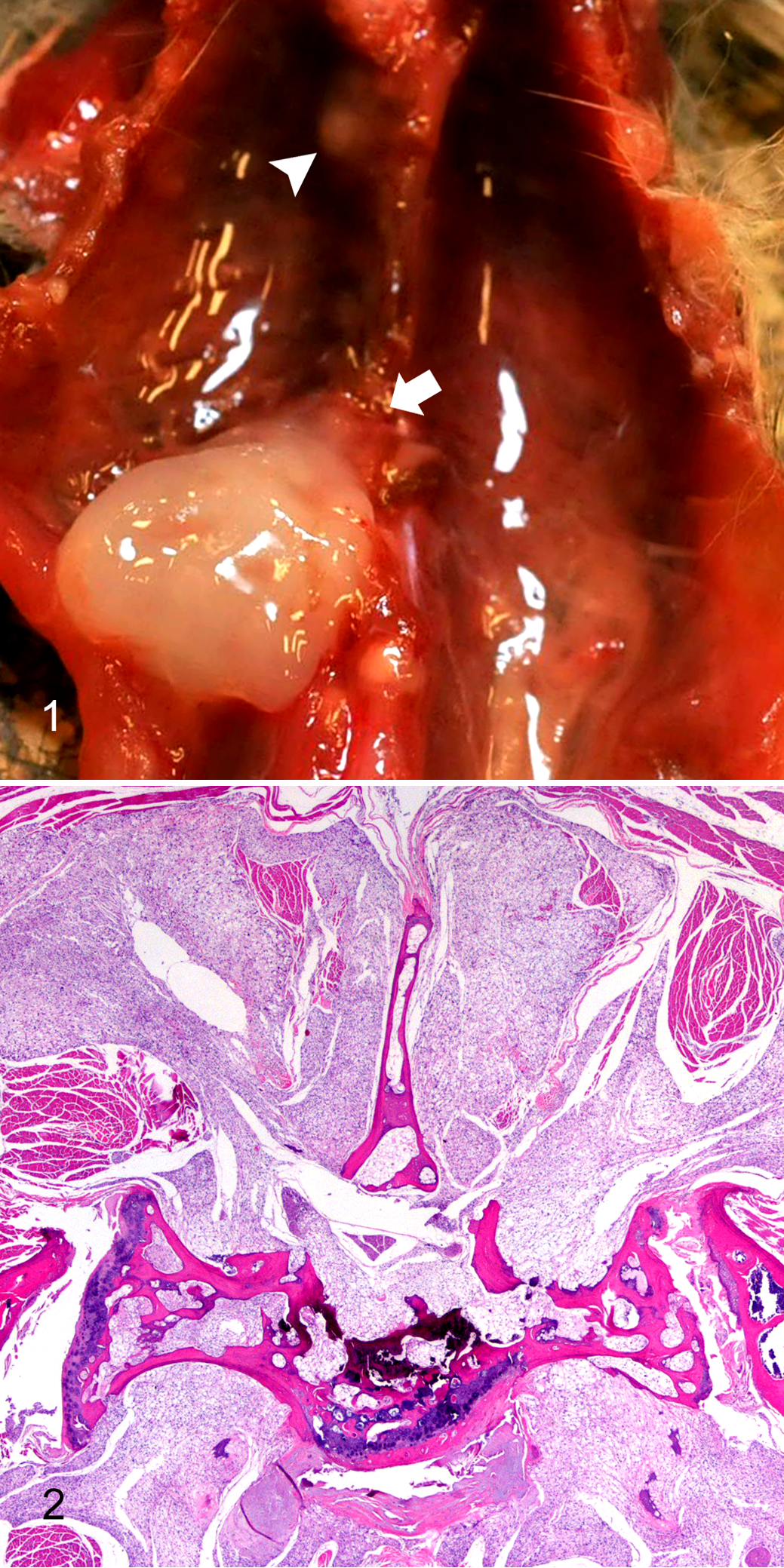
Perdido Key beach mouse, chordoma.
Perdido Key beach mouse, chordoma.
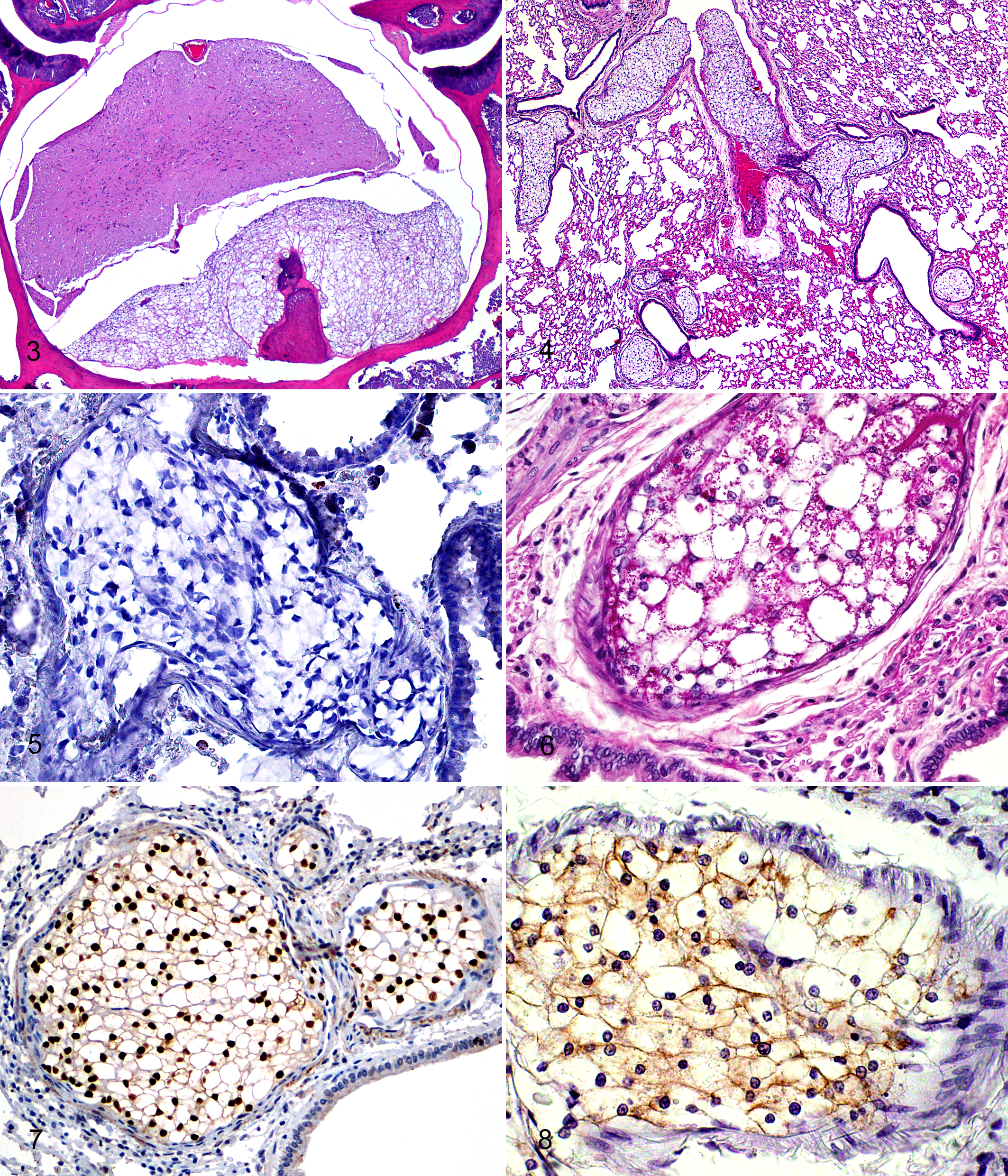
Histologically, primary chordomas were locally invasive masses in and around vertebral bodies and intervertebral discs, invading and effacing or compressing local skeletal muscle and adipose tissue, as well as compressing and effacing spinal cord, vertebral bone, and nerves (Figs. 2, 3). Some masses were contiguous with vertebral articular cartilage or bone. The masses were arranged in lobules surrounded by a thin layer of connective tissue and composed of 5-to 30-µm diameter, monomorphic, markedly vacuolated polygonal cells (physaliferous cells) with small, round, randomly located, hyperchromatic nuclei. Mitotic figures were not seen. Lobules rarely contained small to moderate amounts of bony and cartilaginous matrix (Fig. 3).
Metastases to the lungs were characterized by lobules of physaliferous cells as described above, occluding and expanding arteries and arterioles (Fig. 4). Occasionally, metastases were associated with vessel rupture with minimal to moderate hemorrhage and infiltration of neoplastic cells into the surrounding interstitium and alveoli.
Histochemistry and Immunohistochemistry
ORO did not stain physaliferous cells (Fig. 5). Large numbers of granules positive for periodic acid–Schiff were within physaliferous vacuoles (Fig. 6). Mucicarmine stain failed to demonstrate mucinous matrix within tumors. Immunohistochemistry for brachyury exhibited strong nuclear immunoreactivity in essentially all neoplastic cells within intravascular pulmonary metastases (Fig. 7). In decalcified sections, 30% to 60% of the neoplastic cells exhibited moderate nuclear immunoreactivity to brachyury, and all neoplastic cells exhibited moderate cytoplasmic immunoreactivity. Immunohistochemistry for cytokeratin exhibited mild to moderate cytoplasmic immunoreactivity, which often appeared membranous, in approximately 60% of neoplastic cells (Fig. 8). Neoplastic cells did not exhibit immunoreactivity to UCP1, which is typically seen in tumors of brown fat (hibernoma).
Demographics
Chordomas were found in dead or euthanized mice ranging from 424 to 2170 days old, with a mean age of 1399 ± 354 days. Mice exhibited a statistically significant increase in prevalence of chordomas with increasing age: <1 year, 0 of 3 (0%); 1 year, 2 of 17 (12%); 2 years, 5 of 11 (45%); 3 years, 15 of 28 (54%); 4 years, 11 of 22 (50%); and 5 years, 5 of 7 (71%; P = .001, Cochran-Armitage trend test; P = .002, logistic regression). There was no significant difference in prevalence of chordomas between male and female mice: 23 of 50 (46%) and 15 of 38 (39%), respectively (P = .66, Fisher exact test).
Clinical Findings and Mortality
Chordoma was considered to be the most important lesion diagnosed after spontaneous death or euthanasia of 7 mice (case Nos. 25, 48, 49, 67, 81, 86, 87). Three mice (case Nos. 49, 86, 87) were euthanized for ambulatory difficulty, difficulty using hind legs, or ataxia and unilateral hind limb weakness, respectively. One mouse (case No. 81) was euthanized for inability to use its hind legs (paraparesis) and for radiographic evidence of a soft tissue paravertebral mass with an associated pathologic fracture. The clinical signs in these 4 cases were attributed to effacement of portions of the spinal column by chordomas. Three mice (case Nos. 25, 48, 67) were found dead in their enclosures. In 2 of those mice (case Nos. 25, 48), primary vertebral chordoma and numerous lung metastases were the most notable lesions found at necropsy and histopathology. For 1 mouse (case No. 67), primary vertebral chordoma with spinal compression and intervertebral disc degeneration was the most notable lesion.
Discussion
We report and describe for the first time chordomas in the PKB mouse, a species of conservation interest. Chordomas occurred in the vertebral column of 43% of captive mice, and metastases were noted in 34%. The primary tumors and metastases were predominantly composed of physaliferous cells, comparable to the “classic” form of chordomas described in humans. Chordomas were considered an important cause of captive PKB mortality, and the high prevalence of this tumor is suggestive of a germline mutation, which raises concerns for conservation efforts. Furthermore, the biological behavior of chordomas in PKB mice may be more similar to that of human chordomas than that reported for other mammalian species, making them of interest for comparative pathology.
Immunohistochemistry panels used for identifying chordomas have mainly included cytokeratin, vimentin, S-100, and neuron-specific enolase. 7,12,25,31 Of these, cytokeratin is the most consistently positive and is used primarily to rule out chondrosarcoma. Recent research, however, has shown the transcription factor brachyury to be a highly specific and reliably sensitive marker for identification of chordoma in humans, particularly when used in conjunction with cytokeratin. 19,20,38 In the current study, the neoplasms in PKB mice and the positive-control ferret chordoma exhibited strong nuclear immunoreactivity for brachyury and cytoplasmic immunoreactivity for cytokeratin, supporting the diagnosis of chordoma and suggesting that brachyury is a viable marker for chordomas in PKB mice. It was not clear why decalcified primary masses exhibited cytoplasmic immunoreactivity to brachyury. Notably, strong nuclear immunoreactivity for brachyury was also noted among rare cells visible within the nucleus pulposus of intact intervertebral discs in PKB mice, supporting the notion that these tumors are of notochord origin.
An important differential diagnosis for chordoma is liposarcoma. Traditionally, this has been identified using histochemical stains for lipid, such as ORO or Sudan black performed on frozen sections, or by electron microscopy. In these cases, the tumors did not stain with ORO, while lipid droplets did. In the PKB mice, based on similar biological behavior, as well as histologic similarities and frequent contact of the masses with brown fat, hibernoma was considered a differential diagnosis. 1,3,9,32 Hibernomas are identified by immunohistochemistry for UCP1. 3,43 Brown fat from PKB mice used as a positive internal control exhibited intense cytoplasmic immunoreactivity, but metastases of chordomas in the lungs of PKB mice exhibited no UCP1 immunoreactivity. Unexpectedly, the ferret chordoma used as a negative control exhibited moderate cytoplasmic immunoreactivity for UCP1 around vacuoles; the reason for this is not clear.
Primary chordomas in PKB mice are predominantly composed of physaliferous cells, corresponding to the classic type of chordoma in humans. Only rare cases exhibited cartilage or bone formation, characteristic of the “chondroid” type. Histologic findings in these mice are consistent with those reported in chordomas of rats, where cartilage and bone are rarely present. 31,44 In contrast, ferret chordomas are typically of the chondroid type, producing large amounts of cartilage, bone, and “mucinous” matrix. The classic type is thought to have a more malignant clinical course and a worse prognosis in humans. 15 In accordance with this, metastasis does not usually occur in cases of ferret chordoma, which are generally considered to have a favorable prognosis. 12,24
The distribution of vertebral chordomas in PKB mice is similar to that in humans, where cervical, spinal, and sacral chordomas each account for approximately 1/3 of the tumors. 22 In humans, however, the prevalence of cervical chordomas is inflated by a disproportionately high occurrence in younger individuals (age, <35 years). 22 In PKB mice, the cervical region accounted for a significantly smaller proportion of chordomas compared to the thoracic region, which may be a reflection of the short length of the cervical region or the absence of the factors that lead to chordomas in younger individuals. Ferret chordomas most often form as grossly visible masses caudal to the last vertebra, at the tip of the tail, or rarely in the cervical region. 12,39 There is only 1 report of a ferret developing a chordoma in the thoracic region. 27 Although tails were not evaluated histologically in this study, tail masses were not noted grossly. Among Fischer 344 rats, chordomas are most frequent in the lumbosacral region, but a chordoma in the thoracic region has been reported in a Sprague-Dawley rat. 31,44 Multiple primary chordomas in a single individual, as seen in PKB mice, have not been reported in mammalian species other than humans. 10
Although subjective, chordomas in PKB mice histologically appeared to arise from within intervertebral discs and were frequently associated with disc herniation into the vertebral canal. Moreover, as mentioned above, cells within the nucleus pulposus exhibited strong nuclear immunoreactivity for brachyury, similar to that seen within the tumors. Human chordomas are thought to arise from intraosseous notochord remnants within vertebrae. 8,38 However, since the intervertebral discs of mice are derived exclusively from the embryonic notochord, it is conceivable that chordomas in PKB mice originate from the discs. 8
Metastasis to the lungs occurred in 13 of 38 (34%) mice with chordomas. This is comparable to the proportion of human chordoma cases reported to metastasize: 20% to 43%. 2,17,37 Among Fischer 344 rats, metastasis to the lungs has been reported in 75% of cases. 31 Ferret chordomas typically do not metastasize. 12,24
The prevalence of chordomas (43%) in this captive population of PKB mice is far higher than what is reported in any other species, with the exception of transgenic zebrafish. 4 While an oncogenic virus or a carcinogen could contribute to the unusually high prevalence, a genetic predisposition is a more likely explanation. This is particularly true in light of the known bottlenecks and inbreeding that have occurred in these captive PKB mice. 29,36 Familial chordomas have been repeatedly reported in humans. 23,33,41 Furthermore, genetic studies have suggested correlation between chordoma formation and single-nucleotide polymorphisms in the brachyury gene, as well as DNA copy losses on chromosomal arms 3p and 1p and gains on 7q. 26,28,30,41 Duplication of the brachyury gene has been shown to be a predisposing factor for familial chordoma. 42 It is conceivable, therefore, that the captive population of PKB mice may have a similar or alternative germline mutation that predisposes to chordoma. Given that chordoma prevalence is 50% and 71% in mice 4 and 5 years old, respectively, it is likely that the entire captive population is at significant risk of developing this neoplasm.
In at least 7 cases, chordoma was thought to be the most likely cause of death or euthanasia. Additionally, 10 mice had metastases to the lungs, which were likely to compromise health. Thus, a minimum of 8% to 19% of the entire population succumbed to chordoma. The youngest of these 17 mice was euthanized at 1052 days (2 years) for unilateral hind limb paresis. The youngest PKB mouse with a chordoma unrelated to the cause of death died at 424 days (1 year). In the wild, the average life span of PKB mice is 9 months, with only 4% to 8% of the population surviving more than a year after initial capture. 36 Therefore, although chordoma is a major factor in captive mouse mortality, it is unlikely that chordomas would ever be a limiting factor for survival or fecundity of wild PKB mice.
If subsequent research indicates that the high prevalence of chordomas is a result of mutation and founder effect, it may suggest that other deleterious mutations exist within this population, as well as the population reestablished on the west end of Perdido Key in 2010. 13 However, it is not yet known whether the predisposition to forming chordomas is also present in the wild population.
This is the first report of chordomas in the captive population of the critically endangered PKB mouse, as well as the genus Peromyscus. Chordomas in this species are of interest from a conservation standpoint and for the similarity of their biological behavior to human chordomas. Chordomas are an important life-limiting malignant tumor that occurs at a high prevalence (43%) in the captive population of the PKB mouse, which raises concern that the captive and wild populations may have developed deleterious germline mutations due to repeated bottlenecks. Furthermore, localization, malignancy, and metastasis of chordomas in PKB mice may be more similar to those seen in humans than any other species for which chordomas have been reported, making chordomas in PKB mice of interest for comparative pathology.
Footnotes
Acknowledgements
We thank the histopathology laboratory at the University of Florida College of Veterinary Medicine and the Molecular Pathology Core at the University of Florida for their assistance in slide preparation, histochemistry, and immunohistochemical staining. We also thank Roy Brown of Histology Consulting Service for Northwest ZooPath slide preparation and the staff at Northwest ZooPath for data retrieval. Furthermore, we thank the Santa Fe College Teaching Zoo and the Palm Beach Zoo for their continued use of our anatomic pathology services, as well as the Zoological Medicine Service, University of Florida College of Veterinary Medicine, for antemortem assessment of some of the mice.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a University of Florida College of Veterinary Medicine Resident Research Grant.
