Abstract
Multiple small sessile raised lesions were detected on the ventral surface of the tongue in two 13-year-old domestic cats. The lesions were incidental in both cats. Lesions from both cats appeared histologically as well-demarcated foci of markedly thickened folded epithelium that formed keratin-filled shallow cuplike structures. Large keratinocytes that contained a swollen nucleus surrounded by a clear cytoplasmic halo (koilocytes) were common, suggesting a diagnosis of a papillomavirus-induced papillomas, and papillomavirus antigen was demonstrated by immunohistochemistry. The papillomas exhibited diffuse intense cytoplasmic and nuclear immunoreactivity against cyclin-dependent kinase inhibitor 2A protein (also known as p16 or INK4a protein). Felis catus papillomavirus type 1 DNA sequences were amplified from both papillomas. The papillomas resolved in 1 cat within 3 months of diagnosis, while the papillomas were still visible 4 months after diagnosis in the other cat. This is the first evidence that these papillomas are caused by F. catus papillomavirus type 1.
Papillomaviruses (PVs) have been detected in all species that have been studied, with most species infected by multiple PV types. Most PV infections do not result in the development of clinically detectable lesions; however, depending on both PV type and host factors, PVs can cause a range of hyperplastic and neoplastic lesions. 1 Currently, 4 PV types have been fully classified from domestic cats, including Felis catus PV-1 (FcaPV-1) from a cutaneous viral plaque, FcaPV-2 and FcaPV-3 from cutaneous bowenoid in situ carcinomas, and FcaPV-4 from a sample of gingivitis. 2,10 PV infection is also thought to cause feline sarcoids (fibropapillomas), although current evidence suggests that these lesions are caused by a bovine PV. The PVs of domestic cats have been referred to as Felis domesticus PVs.
In contrast to those of other species, PV-induced papillomas (warts) in domestic cats are rare, with only 2 oral papillomas and 2 cutaneous papillomas previously reported. 2,9 While a cutaneous papilloma was found to contain DNA sequences from a human PV type, the PV type present in oral papillomas in domestic cats has not been previously investigated. Herein, 2 cats with oral papillomas are described. Papillomas in both cats developed on the ventral surface of the tongue and contained FcaPV-1 PV DNA sequences.
Cat No. 1 was a 13-year-old neutered male domestic shorthair cat that was observed during a routine dental procedure to have numerous <3-mm diameter pale raised lesions on the ventral surface of the tongue (Fig. 1). Similar lesions on the ventral surface of the tongue were also observed in a 13-year-old male neutered Abyssinian cat (cat No. 2) during surgical drainage of a ranula. Samples were taken from both cats and submitted for histology.
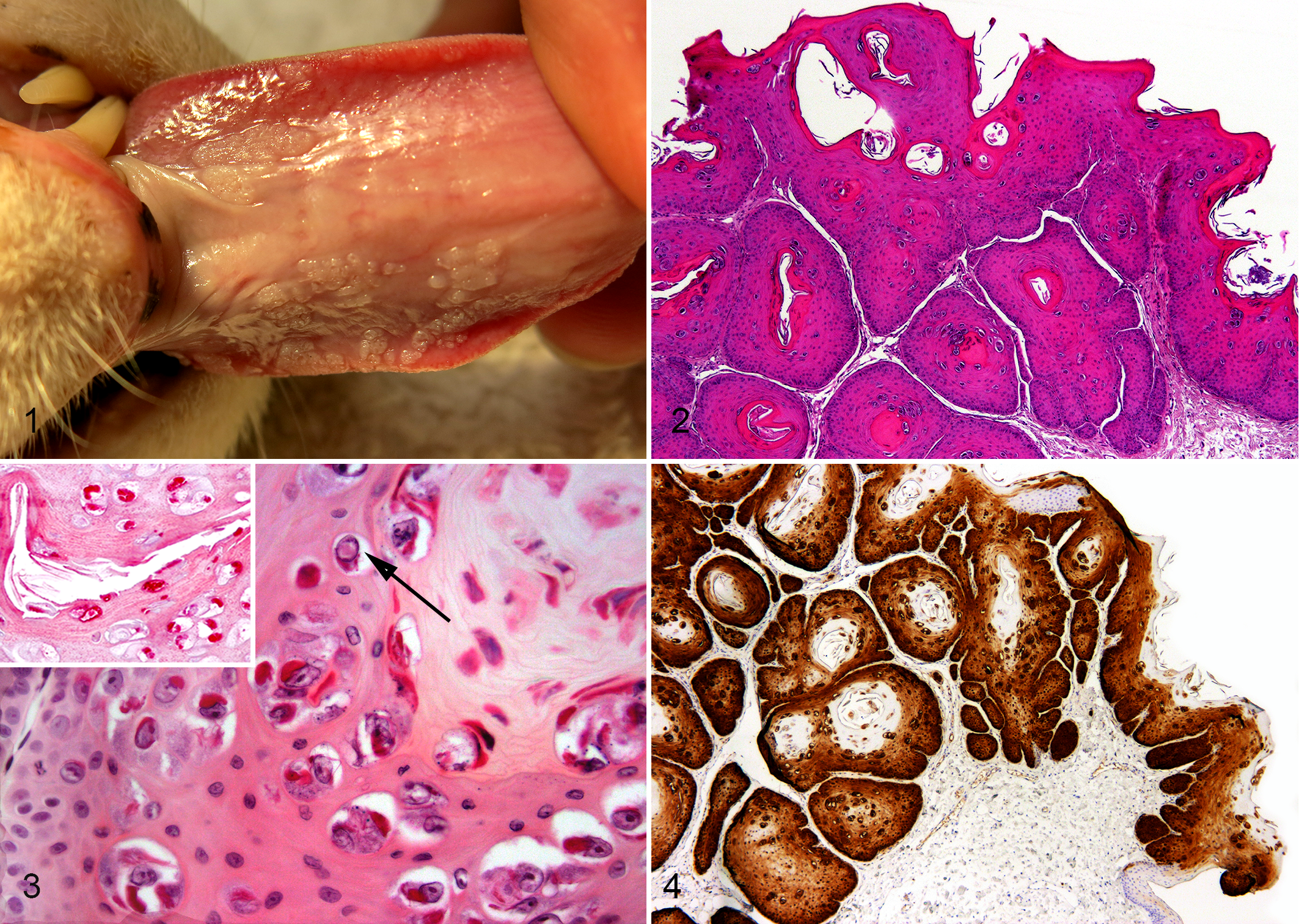
Oral papillomas, cat, tongue.
Lesions from both cats consisted of well-demarcated foci of markedly thickened folded epithelium that projected as thick tongues into the underlying submucosa (Fig. 2). Cornification of the folded epithelium resulted in the development of shallow keratin-filled cuplike structures. The epithelium retained orderly maturation, and no dysplasia was visible. Numerous swollen keratinocytes containing an enlarged nucleus surrounded by a clear cytoplasmic halo (koilocytes) were visible within the superficial half of the thickened epidermis. Prominent eosinophilic bodies were visible within the cytoplasm of many of the cells with cytoplasmic clearing (Fig. 3). Rare koilocytes contained nuclei that had marginated chromatin and a central eosinophilic body suggestive of a PV inclusion. Cells were infrequently visible with enlarged nuclei surrounded by slightly granular faintly basophilic cytoplasm. Mildly increased cornification was present overlying the foci of thickened epithelium. Intranuclear PV antigen was infrequently detected in cells within the superficial epithelium of the hyperplastic lesions according to immunohistochemistry (IHC) with an antibody that has been shown to cross-react with PVs from multiple human and nonhuman PV types (Lifespan Biosciences, Seattle, WA; inset, Fig. 3). Immunostaining based on antibodies against human cyclin-dependent kinase inhibitor 2A (CDKN2A) protein (also known as p16 or INK4a protein) revealed intense cytoplasmic and nuclear immunostaining throughout the hyperplastic epithelium (BD Biosciences, San Jose, CA; Fig. 4).
The histologic and immunohistochemical features suggested a diagnosis of PV-induced oral papilloma. To investigate the causative PV type, total DNA was extracted from formalin-fixed paraffin-embedded tissue scrolls, 4 and the FAP59/64, MY09/11, and CP4/5 consensus primers were used to try to amplify PV DNA. Positive controls were DNA extracted from a bovine papilloma for the MY09/11 primers and DNA extracted from a feline bowenoid in situ carcinoma for the FAP59/64 and CP4/5 primers, while no template DNA was added to the negative controls. The same reaction conditions previously described for the FAP59/64 primers 4 were used for all primer sets. DNA was amplified from all positive controls as expected, but PV DNA was only amplified from the feline oral papillomas using the CP4/5 primers. Both amplicons were sequenced as previously described, 4 and both were identical to published sequences of FcaPV-1.
Neither cat received any specific treatment for the oral papillomas. When cat No. 1 was examined 4 months after the papillomas had been first observed, the size and number of the papillomas were judged to be unchanged. In contrast, no papillomas were visible in cat No. 2 when the cat was examined 3 months after the papillomas had been first observed. Neither cat showed any clinical signs that were attributable to the presence of the oral papillomas. Antibodies against feline immunodeficiency virus (FIV) or feline leukemia virus (FeLV) antigen (SNAP FIV/FeLV Combo Test, IDEXX Laboratories, Inc. Westbrook, ME) were not detected in blood from cat No. 1, taken 4 months after the papillomas had been initially detected. Cat No. 2 had never been tested for either disease before it died of traumatic injury shortly after the papillomas had been observed to resolve.
While 2 oral papillomas in domestic cats were included in a review of feline PVs, 9 there is currently scant information regarding the clinical significance and behavior of these lesions. The oral papillomas in the present 2 cats were found incidentally, and it appears most likely that these lesions rarely cause clinical disease in domestic cats. Spontaneous resolution was observed within 3 months of initial detection in 1 cat. Although the papillomas persisted for 4 months in the other cat, the papillomas remained small and confined to the ventral surface of the tongue. Evidence from other species indicates that PV-induced papillomas almost always resolve, although some papillomas can persist for longer than a year. 1 While oral papillomas appear to be rare in domestic cats, it is possible that some cats develop papillomas that subsequently resolve without being detected.
Four feline PVs have been fully classified. In the present study, the only PV sequences amplified from the papillomas were from FcaPV-1. As DNA from FcaPV-2, FcaPV-3, and FcaPV-4 have been previously amplified by the consensus primers used in this study, it can be confirmed that FcaPV-1 was the only currently recognized feline PV present within the papillomas. While the possibility that the papillomas were caused by an undetected novel feline PV type cannot be definitively excluded, the detection of FcaPV-1 in both papillomas is consistent with this PV being the etiologic agent of oral papillomas in domestic cats.
FcaPV-1 is classified in the Lambdapapillomavirus genus and is closely related to the lambdapapillomaviruses that are reported to cause oral papillomas in exotic cats, including snow leopards, lions, bobcats, and cougars. 6 As observed in the 2 domestic cats, oral papillomas in the exotic felids typically appear as multiple small raised plaques on the ventral surface of the tongue. The oral papillomas of exotic felids are also histologically similar to those of domestic cats and, in contrast to most nonfeline species, typically contain prominent PV cytopathic changes. 7,9 As PVs within a genus tend to have the same tissue tropism and cause the same lesions, the similarities between the lambdapapillomavirus-induced oral papillomas of exotic felids and the oral papillomas in domestic cats provide further evidence that FcaPV-1 is the etiology of these lesions.
FcaPV-1 was initially detected in a feline cutaneous viral plaque, suggesting that it could also cause these lesions. 10 However, whereas oral papillomas are exophytic with marked thickening and folding of the epithelium, viral plaques are sessile with only modest epithelial thickening. 9 Furthermore, oral papillomas, but not viral plaques, demonstrate prominent koilocytosis with condensation of the cytoplasm into brightly eosinophilic bodies, and an immunohistochemical study found that the PV present within an oral papilloma reacted to different monoclonal antibodies than did the PV present within a cutaneous plaque. 9 Thus, viral plaques and oral papillomas may be caused by different PV types. FcaPV-2 was consistently detected in a series of viral plaques, 5 and it appears most likely that oral papillomas are predominantly caused by FcaPV-1 and cutaneous viral plaques by FcaPV-2.
Canine oral papillomas typically develop in young dogs, presumably reflecting the age at first infection. In contrast, both cats with oral papillomas were 13 years old at presentation. The older age of the affected cats could indicate that infection occurred late in life or that oral PVs in cats are better adapted than oral PVs in dogs and do not cause lesions unless there are other contributing host factors. Supporting the importance of host factors is the observation that lions infected by the lion-adapted strain of FIV more frequently develop oral papillomas than do uninfected lions. 7 In the present case, FIV infection could be excluded only in cat No. 1, although neither cat showed other evidence of immunosuppression.
As most PV infections do not result in lesion development, simply detecting a PV within a lesion does not prove causality. 1 In the presently described cats, the histologic appearance of the oral papillomas and the presence of PV cytopathology were consistent with these lesions being caused by PV infection. However, to provide additional evidence that the papillomas had a PV etiology, the presence of CDKN2A within the lesions was determined through IHC. Most PVs influence cell growth and replication by inhibition of the retinoblastoma tumor suppressor protein (pRB) by the PV E7 protein, and loss of pRB results in an immunohistochemically detectable increase in cell CDKN2A. The PV-induced changes to pRB and CDKN2A are so consistent that CDKN2A IHC is used in some human neoplasm types to predict a PV etiology. 8 In cats, increased CDKN2A was observed in a series of cutaneous viral plaques, 3 suggesting that feline PVs also increase cell CDKN2A. The CDKN2A immunostaining present within the feline oral papillomas is therefore consistent with a causal role of FcaPV-1.
In conclusion, as only FcaPV-1 was amplified from both papillomas, this is the most likely cause of the feline oral papillomas. Therefore, as with other felids, oral papillomas in domestic cats most frequently develop as multiple plaques on the ventral surface of the tongue and are likely to be caused by a lambdapapillomavirus. The observations from the present cases suggest that either persistence for at least 4 months or spontaneous resolution can occur.
Footnotes
Author Contribution
Conception or design: JM. Data acquisition, analysis, or interpretation: JM, RF, HM, MK, BV. Drafting the manuscript: JM. All authors participated in critically revising the manuscript, gave final approval, and agree to be accountable for all aspects of work to ensure integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
