Abstract
Polyarthritis caused by Erysipelothrix rhusiopathiae is a relatively common infection in lambs characterized by low mortality and high morbidity. E. rhusiopathiae is a ubiquitous Gram-positive bacterium that is both a commensal and a pathogen of vertebrates. The disease was studied during an outbreak in a Norwegian Spæl sheep flock. In the acute phase, 48 of 230 (20%) lambs developed clinical signs and 4 died (1.7%). One acute case was necropsied and E. rhusiopathiae was cultured from all major organs investigated and from joints. There was a fibrinous polyarthritis, increased presence of monocytes in vessels, and necrosis of Purkinje cells. Sixteen of the diseased animals (33%) developed a chronic polyarthritis. Eight of these lambs were necropsied; all had lesions in major limb joints, and 3 of 8 also had lesions in the atlanto-occipital joint. At this stage, E. rhusiopathiae was cultured only from the joints in 7 of 8 (87.5%) lambs, but by real-time polymerase chain reaction, we showed persistence of the bacterium in several organs. Pulsed-field gel electrophoresis typing of the bacterial isolates indicated that the same strain caused the acute and chronic disease. Five of 6 (83%) chronically affected animals had amyloidosis of the spleen, and 6 of 8 (75%) had amyloidosis of the liver. All chronically affected animals had a glomerulonephritis, and 6 of 8 (75%) had sparse degeneration in the brain. Ceruloplasmin and haptoglobin were significantly increased in the chronically diseased lambs. These results show that chronic ovine erysipelas is not restricted to joints but is a multisystemic disease.
Erysipelothrix rhusiopathiae is a ubiquitous Gram-positive rod-shaped bacterium that is both a commensal and a pathogen in a wide range of vertebrates, including birds and mammals, such as humans, pigs, and sheep. 27,32 The bacterium is widespread in nature and able to survive in the environment for at least several weeks. Pigs are the most important reservoir of infection, and 30% to 50% of healthy pigs harbor the bacterium in lymphoid tissues. 21 The bacterium can be excreted in feces, urine, saliva, and nasal secretions by healthy carrier animals as well as sick animals. 16,21 The animal environment, including bedding, soil, water, and food, can be contaminated by carrier animals or wild birds, possibly leading to infection and disease. E. rhusiopathiae is a facultative intracellular pathogen and replicates in phagocytic cells, and the pathogenicity is related to cellular attachment, intracellular invasion, and survival. 27,32 Swine erysipelas is a worldwide prevalent disease that can be controlled by vaccination. In sheep, the most common clinical manifestation is polyarthritis, typically presenting in 2- to 6-month-old lambs. This disease is characterized by low mortality and high morbidity, with development of chronic changes in the joints. 16 Other manifestations include cutaneous infection, endocarditis, pneumonia, and septicemia. 5,12,16,24 Therefore, the disease can have a significant impact on animal welfare, as well as an economic impact since the disease is associated with ill thrift, reduced growth rate, and condemnation at the abattoir. In most countries, there is no registered vaccine for sheep, but in Australia, a vaccine is available also for this species. Possible routes of infection are cutaneous as after mulesing, shearing, or tail docking; 16,23 orally; 16 or via the umbilical cord in neonatal lambs. 16,24 Chronic ovine erysipelas has been described as a disease that is restricted to joints with subchondral bone involvement. 30,32 In sheep, histologic changes are poorly described and are mainly focused on joint pathology. In this article, we study both the acute and chronic phases of ovine erysipelas, identifying and describing lesions in several internal organs in addition to joints. We also show multisystemic persistence of the bacterium in the chronically affected animals by real-time polymerase chain reaction (PCR), and serum analyses of acute phase proteins emphasize that it is an ongoing active infection.
Materials and Methods
Sheep
In a flock of 110 winterfed Spæl (indigenous Norwegian breed) ewes and 230 lambs, 48 lambs aged 5 to 14 days developed a stiff gait, especially in the front legs, and many were recumbent. Three 10- to 11-day-old lambs (Nos. 1–3), recumbent and in poor condition, were blood sampled. Lamb No. 1 was euthanized and necropsied. The farm is situated in the southern part of Norway in the county of Aust-Agder, and the outbreak occurred mid-May. Two other lambs were euthanized and 1 lamb died in the acute phase; these were not included in the study. The diseased lambs were treated with trimethoprim/sulfadiazine tablets (Tribrissen). Most clinically recovered, but approximately 16 lambs developed chronic polyarthritis. Eight of the chronically ill animals (Nos. 4–11) were necropsied late in August when they were approximately 4 months of age. Prior to necropsy, blood samples were taken from these animals in addition to 9 clinically healthy flock mates that were born before the disease outbreak.
Sampling of Material
Standard necropsies were undertaken with special emphasis on the joints. Samples for bacteriology were taken from 2 or 3 limb joints, brain, lungs, heart, liver, spleen, and kidneys from each animal. Samples for histology were taken from 2 limb joints, lymph nodes (ln. cervicalis superficialis and/or ln. ileofemoralis), brain, lungs, heart, liver, and kidneys from all animals. In addition, samples for histology were taken from diaphragm (8/9) or musculature (m. psoas major and m. quadriceps femoris; lamb No. 1), spleen (8/9), ileum (2/9), and adrenal glands (8/9). Samples for PCR were taken from limb joints (8/9), tonsils (4/9), spleen (8/9), and liver (4/9).
Bacteriology
Samples were inoculated onto sheep blood agar (BA) during the necropsy. The skin was removed over all major limb joints, and those selected for bacteriology were opened with a sterile scalpel. The synovial membrane was scraped with an inoculation loop and immediately spread on BA. All samples were incubated in 5% CO2 atmosphere at 37°C for approximately 20 hours before the initial examination. Agar plates were reevaluated after 48 hours. Bacteria were identified as E. rhusiopathiae based on colonial appearance, Gram-stain, negative catalase test, and hydrogen sulfide gas production in triple sugar iron agar. Isolates assessed as being E. rhusiopathiae after the first seed were replated and reevaluated. Isolates from all cases were further confirmed by real-time PCR.
Histology
The samples for histology were immediately immersion fixed in 4% formaldehyde and subsequently dehydrated in graded ethanols and paraffin embedded. Sections were prepared from all sampled tissues and stained with hematoxylin and eosin (HE). In addition, several tissues were selected for special stains such as Congo red (liver and spleen, lamb Nos. 4–11; kidneys, lamb Nos. 4–6), periodic acid–Schiff (PAS) (liver, spleen, and kidneys, lamb Nos. 1, 4–11), Gram (all tissues, No. 1; joints, all cases; heart and diaphragm, lamb No. 4), and Klüver-Barrera (brain, all cases).
PCR and Sequencing
DNA was isolated from approximately 10 mg bacterial colonies and 200 μl serum (acute phase) or EDTA-blood (chronic phase and healthy animals) by using the DNeasy Tissue Kit (Qiagen, Valencia, CA) and MPLC DNA Isolation Kit III (Roche Applied Science, Mannheim, Germany). The QIAamp DNA Mini Kit (QIAGEN) was used to isolate DNA from 25 mg tissue from the tonsil, liver, joint, and spleen. Bacterial colonies, blood, and tissues were treated according to the manufacturer’s protocol. Primers MO101 and MO102 17 were used. The primer set amplifies a 407–base pair–long DNA sequence coding for Erysipelothrix sp 16S ribosomal RNA (rRNA). The PCR reaction mixture had a total volume of 10 μl containing 1 μl DNA template, 0.2 pMol primers, and PerfeCTa SYBR Green FastMix (Quanta BioSciences, Gaithersburg, MD). The reactions were carried out in a LightCycler LC480 (Roche Applied Science) real-time PCR system with a 1-minute initial denaturation at 95°C and 45 cycles (95°C for 10 seconds, 55°C for 10 seconds, and 72°C for 10 seconds). The maximum level for a positive result was set at threshold cycle (CT ) equal to 38. One PCR product from each animal was sequenced. For enzymatic cleaning, 5 μl PCR product was added to 2 μl ExoSAP-IT (USB Products, Affymetrix, Cleveland, OH), incubated for 15 minutes at 37°C and inactivated for 15 minutes at 80°C, and then cooled to 4°C in PTC 200 (MJ Research, Waltham, MA). Then, 0.5 μl of the cleaned PCR product and 0.2 pMol primer MO102 were added to a total 20-μl reaction volume of an ABI PRISM BigDye TRRM v1.1 Cycle Sequencing Kit (Life Technologies, Applied Biosystems, Foster City, CA). The sequencing PCR was carried out in PTC 200 (MJ Research). The product was precipitated in ethanol, dissolved in 20 μl formamide, and denatured for 5 minutes at 95°C. The sequencing was carried out in an ABI PRISM 310 Genetic Analysator (Life Technologies).
Pulsed-Field Gel Electrophoresis
Bacteria grown from the spleen of lamb No. 1 and the joints from lamb Nos. 4 to 10 were further differentiated by pulsed-field gel electrophoresis (PFGE) using Sma1 as a restriction enzyme. An in-house isolate of E. rhusiopathiae, isolated from swine, was included on the gel for comparison. A λ ladder was used as size marker. The 9 isolates were grown on BA before PFGE was performed as described previously. 8
Protein Measurements
Total protein and albumin concentrations in serum were measured with an ABX Pentra 400 (Horiba ABX Diagnostics, Montpellier, France). Globulin concentration was calculated by subtracting albumin concentration from total protein measurement. Serum ceruloplasmin was measured with COBAS MIRA Plus equipment (Roche Diagnostics, Basel, Switzerland). Serum haptoglobin concentration was measured by the Phase Haptoglobin Assay Kit (Tridelta Development Limited, Kildare, Ireland). The colorimetric reactions were immediately read at 600 nm on a Multiskan* GO Microplate Spectrophotometer using SkanIt Software 3.2 (Thermo Fisher Scientific, Waltham, MA). All samples were run in duplicate, and the mean was used to determine the concentration. Samples with signals higher than the highest calibration standard were diluted and rerun.
Statistical Analysis
One-way analysis of variance in protein measurements between the diseased animals and healthy flock mates was done by the statistical program JMP 10 (SAS Institute, Cary, NC).
Results
Gross Pathology—Acute Case
Lamb No. 1, a 10-day-old ram lamb, weighed 4.3 kg and was of poor body condition. There were no changes in the umbilical cord. There were increased amounts of fluid with fibrin clots bilaterally in carpal and hock joints. The synovial membranes were hyperemic. Regional lymph nodes were slightly increased in size. There was milk in the abomasum but little content in the intestines and some meconium in the descending colon.
Histological Changes—Acute Case
There was a large amount of fibrinous exudate and attenuation and necrosis of synovial cells in the hock joint (Fig. 1). In some areas, there was subsynovial edema with only a mild infiltration of neutrophils, lymphocytes, and plasma cells (Figs. 1, 2). Gram stain showed multifocal scattered colonies composed of thin, slightly curved Gram-positive rods in the necrotic synovium and in the fibrinous exudate. Many of the bacteria were intracellular in synoviocytes. There were few bacteria in vessels in the joint capsule. In all organs investigated, increased amounts of sequestered monocytes were found in vessels (Fig. 3). The monocytes were filled with Gram-positive bacteria (Fig. 4), and there were also free bacteria in the vasculature. In the kidneys, there were a moderate number of bacterial emboli in glomeruli (Fig. 5) and in vessels in the medulla. In the cerebellum, there were multifocal areas with necrosis of Purkinje cells (Fig. 6).
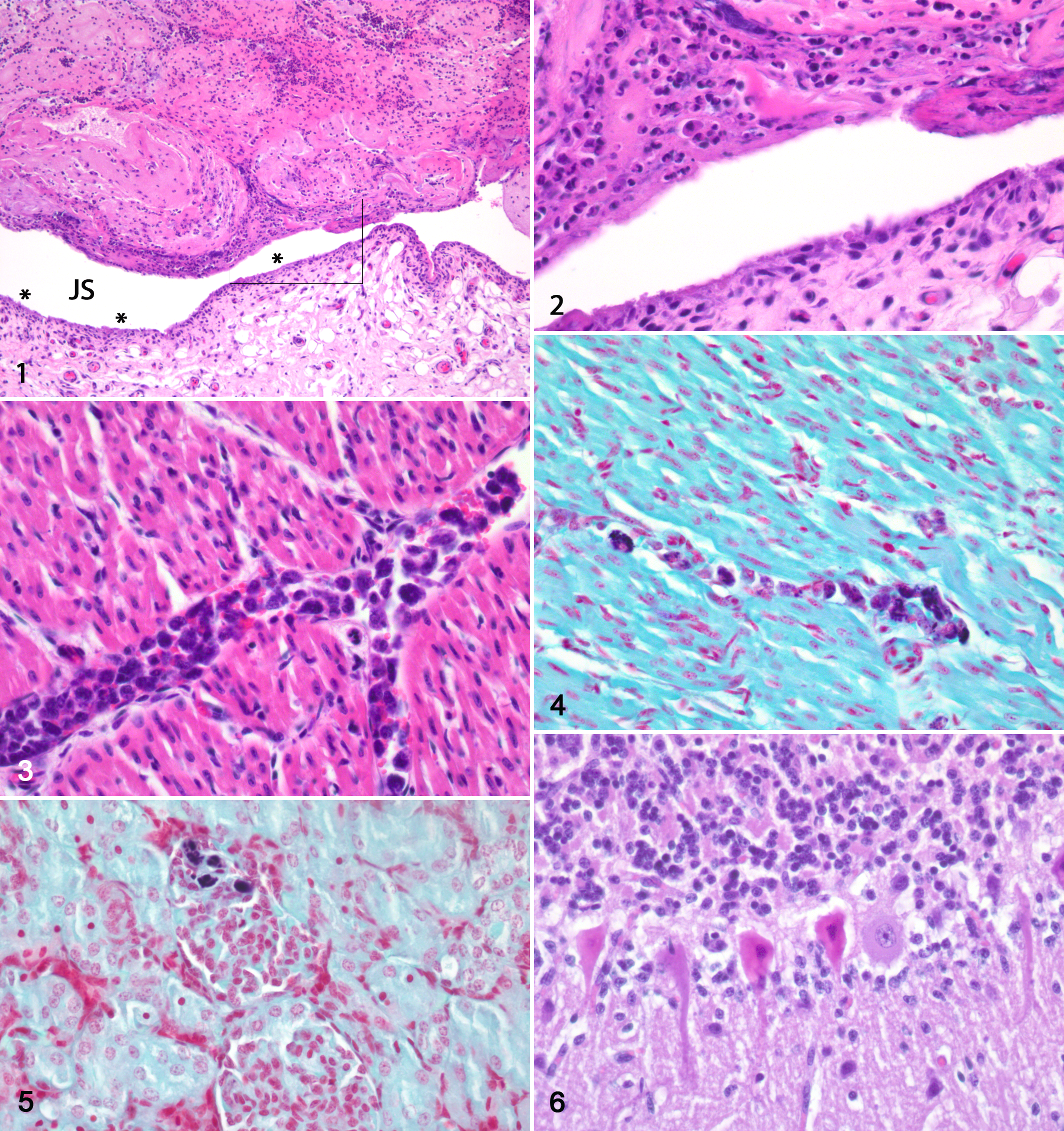
Arthritis, fibrinous, hock joint, lamb No. 1. Figure 1. There is a large amount of fibrinous exudate in the joint space (JS). In several areas of the synovial lining (asterisks), there are attenuation and necrosis of synovial cells. There is a mild cellular infiltrate below the synovial lining. Hematoxylin and eosin (HE). Figure 2. Higher magnification of the marked area of Fig. 1 showing in detail the necrosis and loss of the synovial lining and the exudate in the joint space. HE.
Gross Pathology—Chronic Cases
All 8 chronically affected lambs (Nos. 4–11) were in poor body condition, and several major limb joints were grossly distended. Typically, there was an increased amount of flocculent synovial fluid, the joint capsule was thickened, and the synovial membrane was hyperemic with villous proliferation (Fig. 7). In the most severely affected joints, there were erosions/ulcerations in the synovial cartilage. All lambs had changes in carpal and hock joints; most were affected bilaterally. The stifle was the third most affected joint. Lamb No. 11 was the least severely diseased animal, with bilateral lesions in carpal and hock joints. Three lambs had similar changes in the atlanto-occipital joint as in the limb joints. Three lambs had petechial hemorrhages in the kidneys (Fig. 8) (lamb Nos. 4, 6, 7), lamb No. 4 had radiating 1-mm-thick pale stripes in the kidney cortex, and 3 lambs had pale kidneys (lamb Nos. 4, 8, 10). Lamb No. 4 had a fibrous pericarditis.
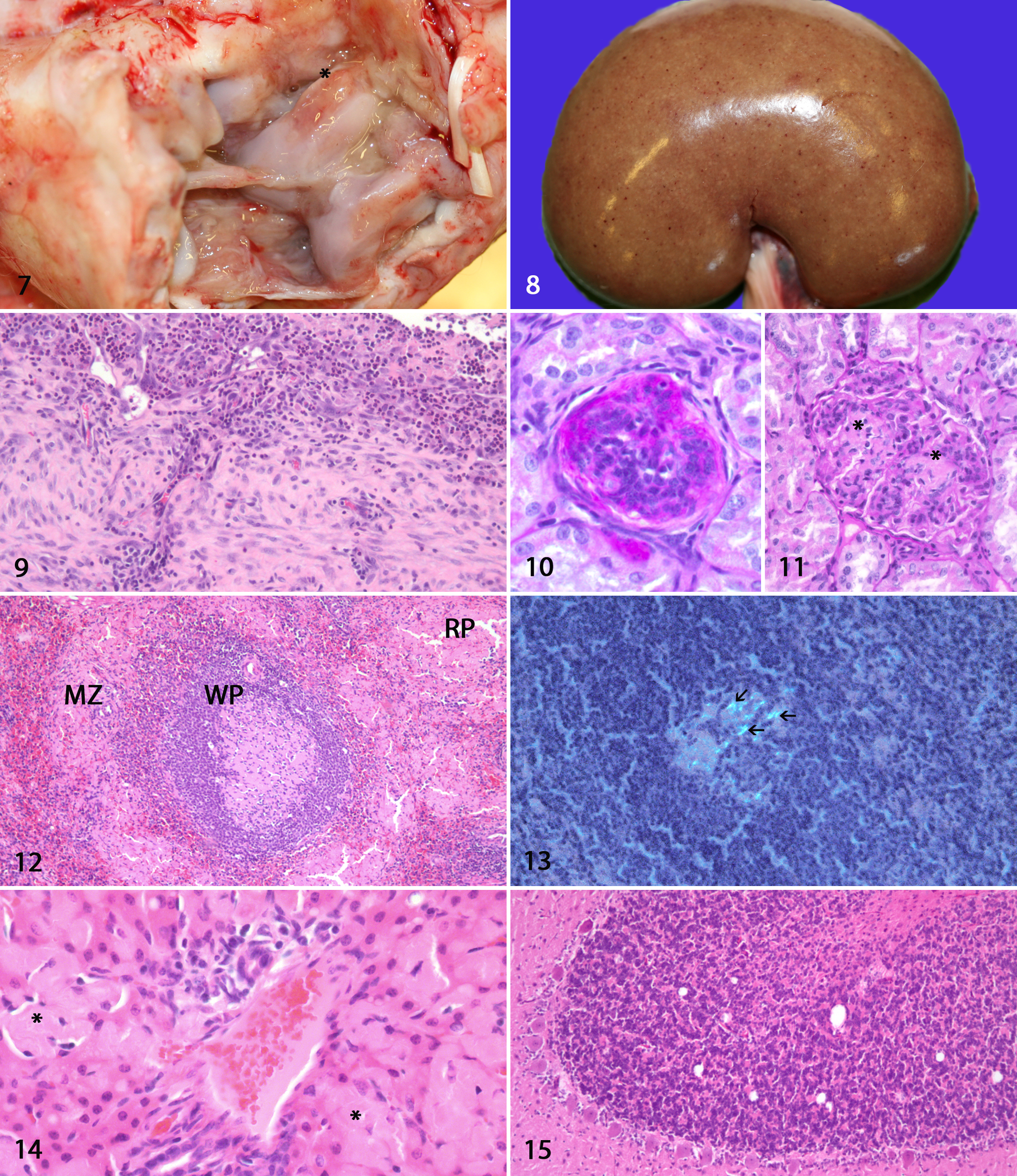
Arthritis, fibrinopurulent, hock joint, lamb No. 11. In the joint space, there is a large amount of fibrinopurulent exudate. There is proliferation and hyperemia of the synovial membrane. The joint capsule is thickened. There is an erosion on the trochlea (asterisk).
Histological Changes—Chronic Cases
In the joints, there was villous proliferation of the synovial membrane with multiple lymphoplasmacytic foci below the synoviocytes and perivascularly. Moderate to large amounts of neutrophils were diffusely spread in and below the synovial membrane (Fig. 9). Multifocally, the synovial membrane was attenuated with necrosis of the synoviocytes. In most animals, focal areas of granulation tissue replaced the synovial membrane. Fibrinous exudate of varying amount was found in the joint space. The joint capsule was thickened with fibrous tissue in all animals. In several animals, there were focal hemorrhages and erythrophagocytosis in the joint capsule. Microscopic lesions in other organs are summarized in Supplemental Table S1. All animals had a segmental thickened PAS-positive basal membrane in some glomeruli (Fig. 10). In each animal, a moderate number of glomeruli had increased cellularity (Fig. 10), and there were synechiae and a thickened Bowman’s capsule. Few glomeruli were shrunken. Lamb Nos. 4 and 8 had focal necrosis with cellular debris, fibrillar hyaline, PAS-negative material, and mild neutrophilic infiltrate within glomerular tufts (Fig. 11). In 5 of 6 lambs, there were large amounts of extracellular hyaline amorphous material, showing birefringence using Congo red stain, in germinal centers and marginal zones and less in red pulp of the spleen (Figs. 12, 13). In the liver, 6 of 8 lambs had varying amounts of similar birefringent material in the periportal space of Disse, and there was atrophy of hepatic cords in these areas (Fig. 14). In the lungs, there were mild to moderate hyperplasia of bronchus-associated lymphoid tissue and perivascular accumulation of lymphocytes and plasma cells. Six lambs had sparse to moderate numbers of vacuoles in the cerebellar granular cell layer (Fig. 15). In several cases, there appeared to be segmental loss of Purkinje cells. Four lambs had sparse degeneration of the white matter in the medulla oblongata and cerebellum. Lamb No. 8 had some neurons with central chromatolysis in the medulla oblongata and mesencephalon. There was no evidence of demyelination as judged by Klüver-Barrera stain. No lesions were observed in the adrenal glands, ileum, or diaphragm.
Bacteriology and PCR
Summarized results of bacteriological investigations and PCR are found in Supplemental Table S2. From lamb No. 1, E. rhusiopathiae was isolated from all investigated organs. From all chronic cases, except lamb No. 11, E. rhusiopathiae was isolated from 1 or 2 joints. All other organs were negative in the chronic phase of the disease. The bacterium, in both the acute and chronic phases, was resistant to trimethoprim/sulfamethoxazole, slightly sensitive to streptomycin, and sensitive to penicillin and tetracycline. All bacterial cultures were confirmed as E. rhusiopathiae by PCR and sequencing of the 16S rRNA PCR product from several isolates. The maximum level for a positive PCR result was set at a threshold cycle (CT ) equal to 38; samples with a higher cycle number were categorized as negative. Serum from lamb Nos. 2 and 3 with acute infection were PCR positive. In contrast to the bacteriological results, tonsil, spleen, and liver from several chronically infected lambs were positive by PCR (Suppl. Table S2). Blood from the chronically affected animals and healthy flock mates was PCR negative.
Pulsed-Field Gel Electrophoresis
All bacterial isolates were investigated by PFGE. The isolates produced banding patterns with 9 to 11 bands between approximately 45 and 300 kb (Fig. 16). Three fragments of similar size between 97 and 150 kb were present in all isolates. The corresponding bands were somewhat difficult to distinguish; therefore, the PFGE analysis was repeated twice. The 3 fragments were finally interpreted as identical between the isolates. Hence, all but one of the isolates from the chronic cases had the same banding pattern as the isolate from the acute case No. 1. The banding pattern of an isolate from lamb No. 10 differed by 2 bands.
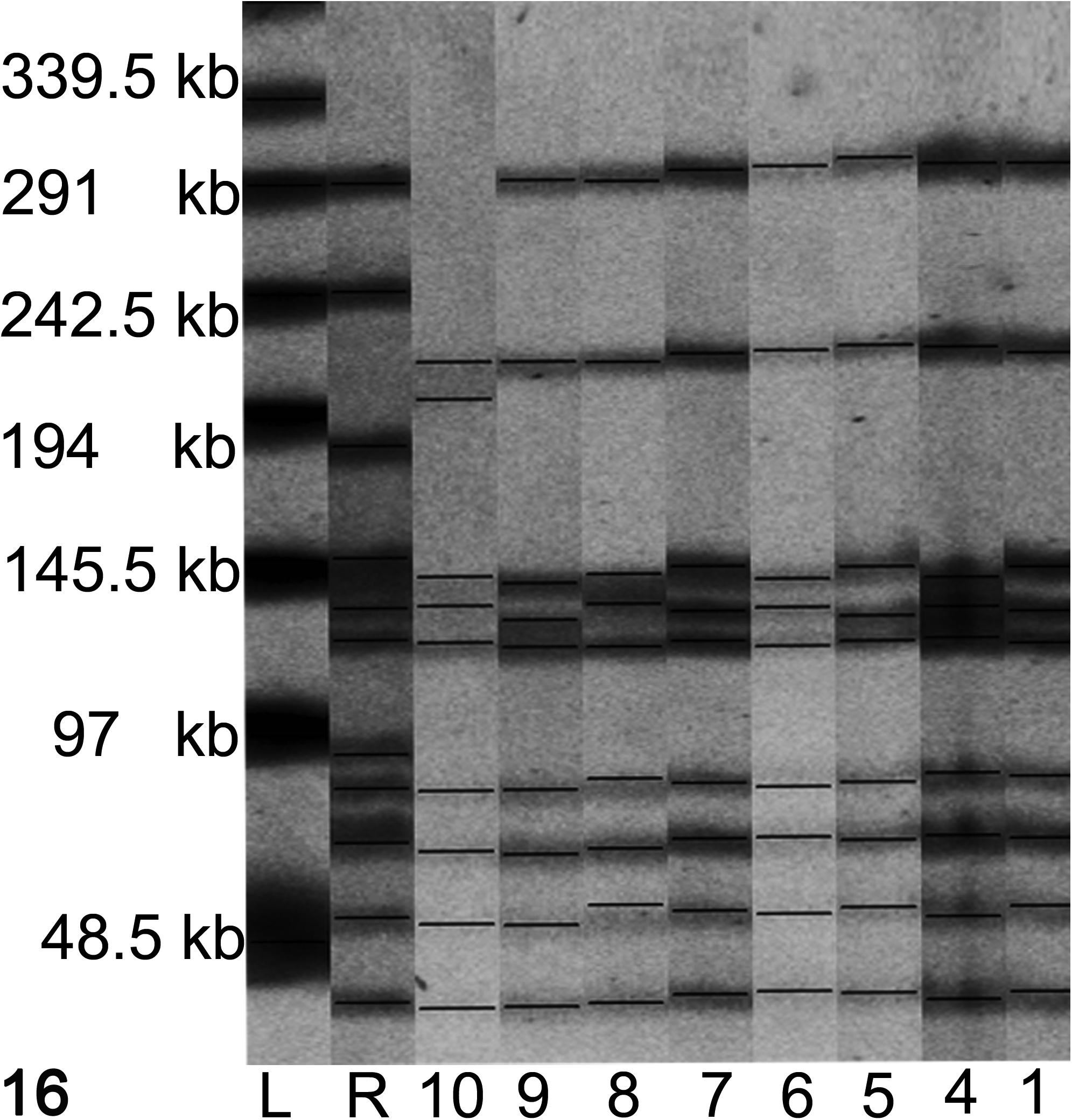
Pulsed-field gel electrophoresis profiles of Sma1-restricted Erysipelothrix rhusiopathiae isolated from 7 lambs included in the study. All profiles are similar except for lamb No. 10 An in-house strain of E. rhusiopathiae from swine was included as a reference strain (R). A λ ladder (L) was used as a size marker.
Protein Measurements
Total protein, albumin, globulin, haptoglobin, and ceruloplasmin in serum of both chronically diseased lambs and clinically healthy flock mates were measured. The mean values for the diseased lambs were total protein, 88 mg/ml (95% confidence interval [CI], 82.5–93.5); albumin, 19 mg/ml (95% CI, 16.2–21.8); globulin, 69.3 mg/ml (95% CI, 61.6–76.9); haptoglobin, 5.6 mg/ml (95% CI, 3.7–7.8); and ceruloplasmin, 33.9 mg/dl (95% CI, 31.1–36.7). In the healthy flock mates, the mean total protein level was lower than diseased lambs, 67 mg/ml (95% CI, 64–70); albumin level was higher, 28 mg/ml (95% CI, 27.4–28.6); globulin level was much lower, 39 mg/ml (95% CI, 36–42); haptoglobin level was much lower, 0.3 mg/ml (95% CI, 0.2–0.4); and ceruloplasmin level was lower, 18.5 mg/dl (95% CI, 15.3–21.7). All mean values differed significantly with nonoverlapping CIs between diseased and health animals (P < .05, one-way analysis of variance).
Discussion
In this article, we describe both acute and chronic lesions in systemic E. rhusiopathiae infection in lambs and complement it with real-time PCR investigations, PFGE of bacterial isolates, and serum protein analyses. E. rhusiopathiae infection in lambs is a well-known disease, but to our knowledge, a thorough report of the disease in sheep, including histologic description of all involved organs, has been missing until now.
The acute case (lamb No. 1) had a fibrinous polyarthritis affecting carpal and hock joints. Interestingly, from this case, we isolated the bacterium from not only the joints but also all the major organs investigated. In the affected carpal and hock joints, there was necrosis of the synoviocytes and both extracellular and intracellular bacteria in synoviocytes. It has been shown that E. rhusiopathiae can both survive and replicate within phagocytic cells, 27 and it is possible that maintenance of infection and survival of the bacteria also involve the synoviocytes. In humans, the obligate intracellular Gram-negative bacteria Chlamydia pneumonia and Chlamydia trachomatis both have the ability to persistently infect synovial tissues, although distant to the initial site of infection. 4 Another Gram-negative facultative intracellular bacterium, Brucella abortus, is also able to infect and survive in synoviocytes. 26 Common characteristics of infection by these 3 bacterial genera are that they involve phagocytic cells for survival and transportation, and infection can involve different organ systems as well as joints with detection of the bacteria in synovial tissues.
The most common form of E. rhusiopathiae infection in sheep is chronic polyarthritis in 2- to 6-month-old lambs. 16 This manifestation is most likely to be a sequela of an earlier bacteremia or septicemia, and this view is supported by the present findings. The acute disease is described as having a morbidity ranging from 7% to 28% and low mortality. 16 In the reported sheep flock, the morbidity was 20% and the mortality was 1.7%. Of the diseased animals, approximately 33% developed a chronic disorder with polyarthritis. A similar outbreak of erysipelas in neonatal lambs has previously been reported with a morbidity of 50%, although no chronic sequelae were described, and the outbreak resolved after treatment by enrofloxacin. 24 By contrast the lambs in this study had been treated with trimethoprim/sulfadiazine. In our laboratory, the isolates from both the acute and chronic phases were resistant to the latter antibiotics. E. rhusiopathiae is generally considered resistant to sulfonamides, while susceptibility is variable to streptomycin and tetracyclines. 22,31 The drug of choice in Erysipelothrix infections is penicillin.
Several studies report that it is difficult to recover bacteria from chronically affected joints in both pigs and sheep, and it has been proposed that the ongoing inflammation in some instances could be due to persistence of the Erysipelothrix antigen rather than live bacteria. 16,30 Others describe detection of bacteria in various compartments of chronically affected joints. For example, the number of bacteria decreased from 2 to 5 months postinoculation in a study of erysipelas in pigs in which the bacteria were directly injected into the joint. 10 There were fewer bacteria in the synovial fluid compared with the synovial membrane and chondrocytes at all time points, 10 while in an oral infection study, the bacterium was isolated only from the synovial fluid from 1 of 2 joints in 2 pigs 4 weeks after inoculation. 13 A high number of bacteria were found in the tonsil and a lower number in blood. 13 In the present study, we paid particular attention to the joint sampling. The seeding was done in the necropsy room and involved scraping the synovial membrane with the inoculation loop. Despite this, there were only 1 or 2 colonies growing on the agar from 4 lambs with chronic disease. Most likely, there are few E. rhusiopathiae bacteria in the joints of chronically infected animals, and this underlines the importance of a thorough sampling technique for bacterial culturing.
PFGE typing showed that in all but 1 case, the bacterium had a similar banding pattern, including both 1 acute case and 7 chronic cases. The 3 fragments of similar size between 97 and 150 kb were interpreted as identical between the isolates, although slight fragment shifts may have occurred in some of the isolates. This indicates that 1 single, highly infectious strain caused the described disease in both the acute and chronic phases. The isolate from lamb No. 10 differed by 2 bands from the other isolates, indicating that a mutation, insertion, or deletion occurred during the course of the outbreak. Genetic changes in the infecting bacterial strain can be observed during the course of an outbreak, and isolates with up to 3 band differences can still be considered closely related. 29 The pathologic changes of this case did not differ compared with the other cases. Genetic characterization of E. rhusiopathiae from sheep by PFGE has not been reported but is described as a useful tool to distinguish between strains found in other species. 7,20
In the present study, we used a primer set directed against Erysipelothrix sp 16S rRNA for the real-time PCR. 17 Serum from 2 acute cases was PCR positive, suggesting that this is a potential diagnostic tool in the acute stage of the disease. One joint from each chronic case was investigated by PCR, and all were positive. In addition, 3 of 4 tonsils, 2 of 8 spleens, and 1 of 4 livers, all from chronic cases, were positive. This is in agreement with previous research in swine that demonstrated that the tonsil is a site of persistent E. rhusiopathiae infection. 13 In our study, 2 animals that had positive tonsil samples also had positive spleen (lamb No. 4) or liver (lamb No. 10) results, showing dissemination of the bacterium in the chronic phase of the disease.
In the chronically affected lambs, there was evidence of an active ongoing infection in both joints and kidneys. In addition to characteristic proliferative synovial changes and lymphoplasmacytic infiltrates, there was ongoing destruction of the synovial membrane, fibrinous exudate, and infiltration of neutrophils in the superficial layers of the affected joints. In glomeruli of 2 chronically affected animals, there was focal fibrinoid necrosis of the tufts, which is a characteristic lesion of acute erysipelas in swine. 21,30 All except the least chronically affected lamb (No. 11) had amyloid deposits, identified by Congo red stain, in the spleen and/or liver. Deposition of amyloid correlated with globulin level, and the animals with the most severe changes had the highest levels. In amyloidosis, the precursor protein undergoes a conformational change that leads to a high content of β-sheets that aggregates as extracellular fibrillar material. 3 Amyloidosis associated with chronic infections is caused by deposition of fibril-forming fragments of serum amyloid A (SAA) and is the most frequent form of amyloidosis in ruminants. 19,36 Amyloidosis has been associated with E. rhusiopathiae but has not been reported in sheep, although amyloidosis affecting the liver, kidneys, and gut has been described in goat kids with chronic polyarthritis and a bronchopneumonia. 34 No organisms were isolated from the joints, but Mannheimia haemolytica was grown from the lungs. The 3 kids had high serum titers for E. rhusiopathiae, and it was therefore postulated that the amyloidosis could be a consequence of an Erysipelothrix septicemia. In an older study, 2 of 61 pigs experimentally infected with E. rhusiopathiae were found to have amyloidosis affecting the spleen and liver. 35 In the present study, the serum protein profiles of the chronically diseased lamb were typically that of a chronic infection with a high globulin fraction and a low albumin fraction. Albumin is a negative acute phase protein (APP), and in chronic infections, there will be a decrease in the serum concentration as a result of reduced synthesis in the liver. 6 The globulin fraction in serum comprises APPs such as haptoglobin, ceruloplasmin, and SAA and immunoglobulins. Haptoglobin and ceruloplasmin were both increased in the chronically diseased animals, indicating an ongoing inflammation and tissue destruction. For these parameters, there were nonoverlapping confidence intervals of the mean values of the diseased and healthy animals. These APPs are described to be increased in different infections in sheep such as scab, 33 blue tongue, 25 and scrapie. 18
In the acute case (lamb No. 1), there was evident necrosis of cerebellar Purkinje cells, but there were no accompanying inflammatory lesions in the brain. Necrosis and loss of Purkinje cells have been described in other infections such as louping ill in calves 2 and enterohemorrhagic Escherichia coli infection in mice. 28 Such changes have also been described in other noninfectious disorders of ruminants such as Marsdenia megalantha poisoning, 11 head traumas, 9 and ovine pregnancy toxemia. 14 Purkinje cells are vulnerable to both cerebral ischemia 15 and hypoglycemia, 1 and these abnormalities might be contributory factors to the multifocal necrosis seen in lamb No. 1. Unfortunately, we were not able to measure serum glucose in this lamb. In several of the chronic cases, it seemed to be regional loss of Purkinje cells, but no morphometric analyses were done. This could be a sequela to Purkinje cell necrosis in the acute phase of the disease, as noticed in lamb No. 1. Several of the chronic cases also had a sparse degeneration of white matter in the medulla oblongata and cerebellum, indicating mild chronic brain involvement.
In conclusion, Erysipelothrix rhusiopathiae infection in lambs is a multisystemic disease affecting many organs and is not limited to the major limb joints. This may explain the general wasting that can be seen in lambs with this infection.
Footnotes
Acknowledgement
We thank Eli Brundtland, Siri B. Hamre, and Bjørg Kvitle for skillful technical assistance. We also thank Dr Clare Joan Phytian for her critical reading of the manuscript.
Author Contribution
Conception or design: CE, KIL. Data acquisition, analysis, or interpretation: CE, HJJ, KIL. Drafting the manuscript: CE, HJJ, KIL. All authors participated in critically revising the manuscript, gave final approval, and agree to be accountable for all aspects of work to ensure integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
