Abstract
Two elegant crested tinamou chicks (Eudromia elegans), aged 27 and 50 days, respectively, died following acute onset of weakness and neurologic disease. Microscopically, the cerebral hemispheres of both chicks and the optic lobes of 1 chick contained multifocal granulomatous and heterophilic inflammation and necrosis with intralesional pigmented, thin-walled, fungal hyphae. In 1 chick, hyphae extended along the optic nerve into the globe and were associated with severe granulomatous and heterophilic inflammation of the choroid, retina, pecten, and vitreous. In both chicks, polymerase chain reaction amplification of the fungal 28S large subunit ribosomal RNA was positive with 99% sequence identity to Ochroconis gallopava. While a well-characterized fungal infection of domestic poultry, ochroconiasis has rarely been reported in exotic avian species, and this is the first histologic characterization of ocular ochroconiasis in any avian species.
Phaeohyphomycoses caused by dematiaceous fungi of the order Ochroconiales have been reported in humans, domestic small animals, amphibians, reptiles, fish, and birds. 3,8 Ochroconis gallopava (synonyms Dactylaria gallopava and Dactylaria constricta var. gallopava) is arguably the most clinically significant member of the Ochroconiales. A thermostable fungus of the family Ochroconiaceae, O. gallopava is commonly found in warm, acidic environments, including decaying vegetation and excrement. 10 Sporadic and epizootic O. gallopava infections are well characterized in domestic poultry; however, in nondomestic species, reports are limited to sporadic disease in gray-winged trumpeters (Psophia crepitans) and snowy owls (Nyctea scandiaca) and a single outbreak in Japanese quail chicks. 2,6,9 In this report, encephalitis and ophthalmitis resulting from ochroconiasis in 2 elegant crested tinamou (Eudromia elegans) chicks are described.
Case Histories and Gross Necropsy Findings
Two elegant crested tinamou chicks, from a clutch of 4 that hatched on exhibit in a large, warm, arid enclosure, presented with similar clinical signs within a 3-week period. The first chick, a 26-day-old female (tinamou No. 1) had an acute onset of paresis and inability to walk, partially closed eyes, and an obtunded mental state. Physical examination did not identify a clear cause for the clinical signs. The animal was treated for possible head trauma with subcutaneous fluids and dexamethasone but despite treatment was found dead the following day. On postmortem examination, a nondisplaced, linear, sagittal fracture with associated hemorrhage was identified in the occipital bone. No gross abnormalities were noted on external examination of the brain. The second chick (tinamou No. 2) presented similarly at 44 days of age. Physical examination did not identify a cause for the clinical signs, and the chick was treated with subcutaneous fluids, antibiotics, a nonsteroidal anti-inflammatory drug, and vitamin E. Hematology showed heterophilia (11 990/μL) and monocytosis (4200/μL) with hyperglobulinemia (2.7 g/dL). Medical management resulted in an initial improvement in clinical condition. However, cervical paresis developed 3 days following treatment, and 6 days following the initial presentation, the animal re-presented with increased lethargy, a fluffed appearance, and a head tilt. Fluid and vitamin E therapy was repeated and a glucocorticoid was administered. The following day, the animal was found on its back, unable to right itself, and with marked torticollis. Despite continued supportive care, the chick died at 50 days of age. On postmortem examination, the only gross lesions were a focal area of mild hemorrhage and edema of the right carpus and a localized area of hemorrhage in the right lung lobe. No gross abnormalities were noted on external examination of the brain. A complete set of tissues was collected from each chick, fixed in 10% neutral buffered formalin at room temperature for 4 days, and processed routinely. Paraffin sections were cut at 5 µm and stained with hematoxylin and eosin (HE).
Histologic Findings
In the cerebral hemisphere of tinamou No. 1, there was extensive replacement of the neuropil with large numbers of macrophages, heterophils, and multinucleated giant cells (both foreign body and Langhans type) and necrosis characterized by eosinophilic and basophilic (karyorrhectic) debris and clear space (edema) (Fig. 1). Sporadic fibrinoid vascular necrosis of cerebral vessels was also observed. Amid the necrotic neuroparenchyma were narrow, 2- to 2.6-µm, thin-walled, septate, faint brown-yellow, irregularly branching hyphae, consistent with cerebral phaeohyphomycosis (Fig. 2). Grocott’s methenamine silver (GMS) stain highlighted the presence of large numbers of hyphae throughout the affected neuroparenchyma and within the walls and lumina of necrotic vessels (Fig. 2, inset). Scattered necrotic neurons were present amid increased numbers of glial cells and reactive astrocytes. Macrophages, lymphocytes, and heterophils expanded Virchow-Robins space, extended into the lateral ventricles bilaterally, and expanded the leptomeninges. Inflammation also extended along the meninges surrounding both optic nerves, but there was no extension into the globes.
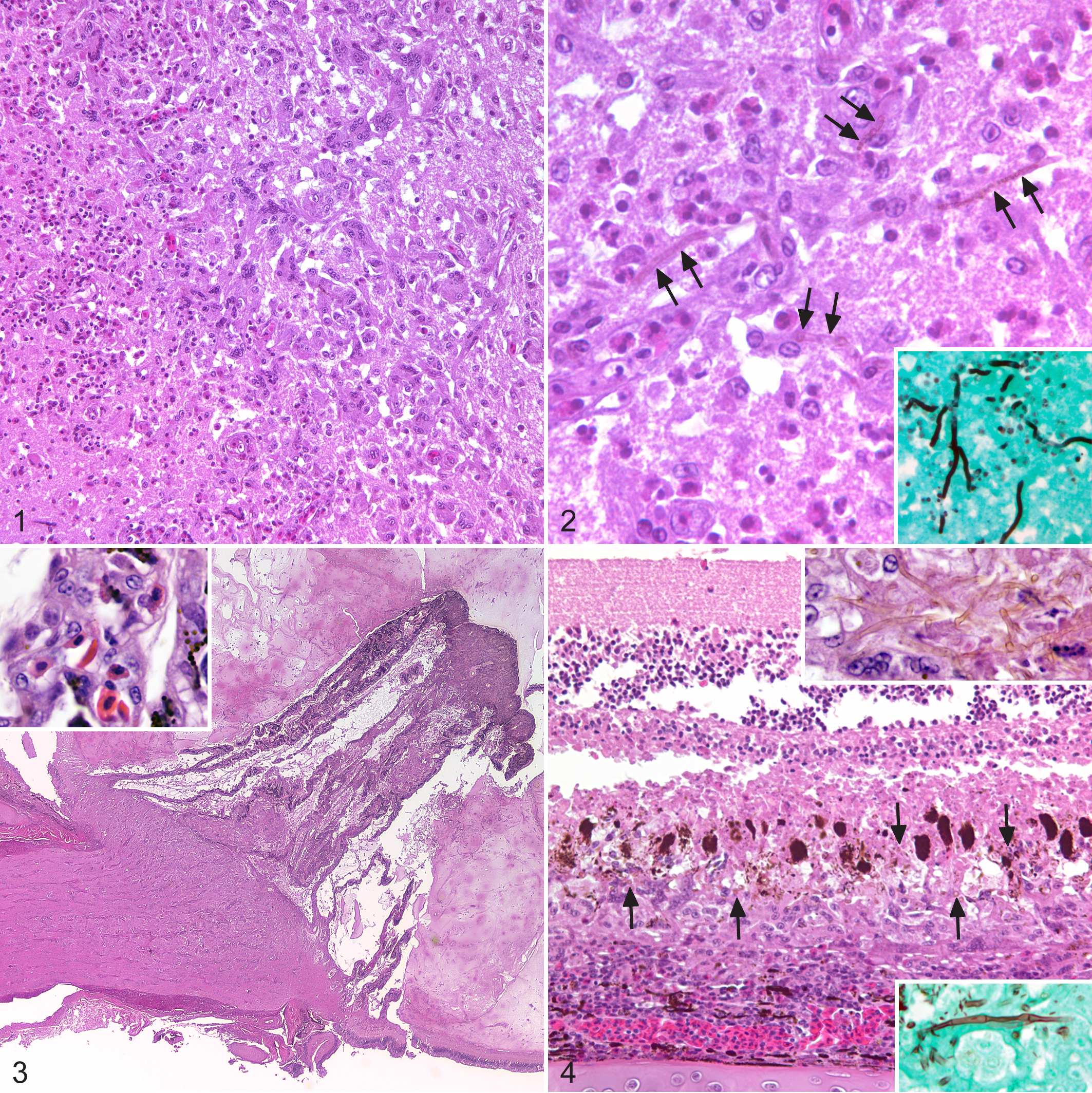
Encephalitis, cerebrum, elegant-crested tinamou, No. 1.
In tinamou No. 2, similar, but less extensive, areas of necrosis and inflammation were present in the cerebral hemispheres and optic lobes. As in tinamou No. 1, inflammation and necrosis were associated with intralesional, pigmented hyphae. An additional finding was extension of severe heterophilic and granulomatous inflammation along one optic nerve into the globe. Optic nerve infiltrates included heterophils, macrophages, lymphocytes, and multinucleated giant cells that were associated with axonal degeneration, digestion chambers, and increased glial cells (Fig. 3). GMS stain highlighted small numbers of thin-walled, septate hyphae within the nerve. Heterophils and macrophages infiltrated the choroid, pecten, and the outer layers of the retina (Figs. 3, 4). Immediately surrounding the optic nerve head, inflammatory cells elevated the retina from the choroid. The vitreous body contained large numbers of intact and degenerate heterophils. Many thin-walled, pigmented hyphae were present within the choroid, outer layers of the retina, and the vitreous (Fig. 4). Mild heterophilic inflammation was noted in the posterior and anterior chambers, iris, and ciliary bodies. This chick also had a locally extensive area of granulomatous pneumonia in the right lung that was associated with hemorrhage and erythrophagocytosis; no fungal or bacterial organisms were identified with HE, GMS, or Gram staining.
Molecular Diagnostics
Nucleic acids were extracted from scrolls of paraffin-embedded brain (tinamou No.1) and eye (tinamou No. 2) by using a commercially available kit (RecoverAll Total Nucleic Acid Isolation Kit; Life Technologies, Grand Island, NY), and fungal DNA was amplified by polymerase chain reaction (PCR) by using universal primers that target a variable region within the large 28S subunit. 7 Amplicons of 224 and 228 base pairs, respectively, were amplified from the 2 tinamous. Both strands of the amplicons were sequenced using an automated capillary sequencer (3730XL; Applied Biosystems, Foster City, CA) at the University of Chicago Cancer Sequencing Facility and compared for homology with sequences in the GenBank genetic sequence database. Consensus sequences from both cases were 100% identical to each other and were 99% homologous (1 base difference) to multiple strains of O. gallopava (GenBank accession numbers KF282655-7 and AF125280-1). Sequences were 93% similar to Ochroconis calidifluminalis, the next closest isolate.
Discussion
O. gallopava appears to have a distinct tropism for tissues of the central nervous system, causing cerebral ochroconiasis in birds. Inhalation is suspected to be a common exposure route, and experimental inoculation of the fungus via the respiratory tract has successfully reproduced the disease in chickens and turkeys. 1,4 Avian cerebral ochroconiasis also appears to be primarily a disease of young birds, with reports limited to young domestic turkeys (<5 weeks of age) and chickens (<7 weeks), Japanese quail (7–21 days), gray-winged trumpeters (17 and 18 days), and a snowy owl (28 days). 1,2,4–6,9 Common clinical presentations in affected birds include ataxia, torticollis, limb paralysis, and/or opisthotonos.
The histologic changes noted in the brains of the 2 tinamou chicks of this report were similar to those reported in other species, although well-defined granulomas with central necrotic debris, as reported in quail and turkeys, were absent. In addition, cerebellar involvement, as described in Japanese quail, turkeys, and chickens, was not present in the tinamous. In tinamou No. 2, focal granulomatous inflammation in 1 lung lobe was associated with hemorrhage and erythrophagocytosis; no hyphae were identified on HE-, GMS-, or Gram-stained sections. While the macrophage infiltrates were likely secondary to localized hemorrhage, a relationship between the cerebral and pulmonary lesions, such as concurrent or previous pulmonary ochroconiasis, remains possible. Concurrent granulomatous pulmonary ochroconiasis has been reported in turkey poults with cerebral ochroconiasis. 1
In both tinamous, inflammation extended along the optic nerves, and in tinamou No. 2, it extended into the globe. This report is the first to provide histologic and molecular characterization of ocular ochroconiasis in an avian species. In 1 outbreak of ochroconiasis in turkey poults, some affected older birds (5 weeks), but not younger birds (2-4 week olds), were reported to have grossly apparent ocular opacities, and O. gallopava was isolated from affected eyes. 1 However the observed ocular lesions were not characterized microscopically or with molecular diagnostics. Given the presence of pigmented hyphae within the optic nerve and perineurial inflammation of the optic nerve, extension from the brain to the globe is most likely.
Tinamous, native to Mexico and Central and South America, are the sole members of the order Tinamiformes. Including this report, O. gallopava has been reported in 6 species of birds from 4 diverse orders (Galliformes, Gruiformes, Strigiformes, and Tinamiformes), suggesting broad susceptibility of birds to this fungus. Domestic or exotic birds housed in environments with warm, moist, acidic conditions that favor fungal growth are likely at highest risk for developing opportunistic infection and disease; however, as these chicks were being raised in a large, arid, and generally dry enclosure, suspicion of ochroconiasis should not be limited to animals housed only in moist environments. Furthermore, other unidentified factors likely predispose birds to infection as outbreaks in large avian populations generally only involve a fraction of the exposed population (up to 20%). 4,5 Cases in wild, captive birds appear to be sporadic, although infection and disease may be unrecognized and/or underreported. 2,6,9 The two surviving tinamou chicks from the affected clutch did not exhibit any clinical signs consistent with ochroconiasis and matured into healthy adults.
Footnotes
Acknowledgements
We thank the Wildlife Conservation Society's curatorial and clinical medical staffs for their exceptional care, Alfred Ngbokoli and Daniel Friedman for slide preparation, Julia Rodriguez-Ramos Fernandez for assistance with ocular pathology, and Nadia Ahmed for assistance with molecular diagnostics.
Author Contribution
R. J. Ossiboff contributed to conception and design; contributed to acquisition, analysis, and interpretation; drafted the manuscript; critically revised the manuscript; gave final approval; and agrees to be accountable for all aspects of work ensuring integrity and accuracy. M. M. Clancy contributed to analysis and interpretation, drafted the manuscript, critically revised the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring integrity and accuracy. K. A. Terio contributed to design; contributed to acquisition, analysis, and interpretation; drafted the manuscript; critically revised the manuscript; gave final approval; and agrees to be accountable for all aspects of work ensuring integrity and accuracy. K. J. Conley contributed to conception and design; contributed to acquisition, analysis, and interpretation; critically revised the manuscript; gave final approval; and agrees to be accountable for all aspects of work ensuring integrity and accuracy; D. McAloose contributed to conception and design; contributed to acquisition, analysis, and interpretation; critically revised the manuscript; gave final approval; and agrees to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
