Abstract
A 4-year old spayed male domestic shorthair cat was presented with a history of circling and behavioral changes. Neurologic examination showed mild proprioceptive deficits. The lesion was localized in the forebrain, and magnetic resonance imaging revealed the presence of a large midline intracranial mass extending from the frontal lobe to the tentorial region of the brain. Euthanasia was elected due to poor prognosis. Histopathologic evaluation confirmed the presence of a mass composed by sheets and aggregates of large round/polygonal cells and multinucleate cells associated with deposits of cholesterol clefts, scattered hemorrhages and hemosiderin-laden macrophages. Immunohistochemistry showed that the round/polygonal cells and multinucleate cells were strongly positive for major histocompatibility complex class II antigen, variably positive for CD18, and occasionally positive for S100. Subsets of spindle cells showing variable expression of vimentin, S100, and neuron-specific enolase were also present. The final diagnosis was cholesterol granuloma. Differential diagnosis with meningioma is discussed.
Keywords
History
A 4-year-old male neutered domestic shorthair cat was presented for a 3-month history of toileting in inappropriate places and reluctance to move. This progressed to circling, staring, and intermittent ataxia affecting all limbs. General physical examination was unremarkable. On neurologic examination, the only abnormality was a mildly decreased proprioception on the right thoracic and pelvic limbs, with normal gait. Based on the history and neurologic signs, the neuroanatomic localization of the lesion was the forebrain.
Hematology and biochemistry were unremarkable. FIV, FeLV, toxoplasmosis, and coronavirus serologies yielded negative results. Sagittal and/or transverse T1-weighted, T2-weighted, FLAIR, T2*-weighted, and T1–fat saturated magnetic resonance images of the brain were acquired with a 1.5 T system (Magnetom, Siemens, Camberley, UK). A large well-delineated heterogeneous mass was identified in the midline of the brain, between the lateral ventricles, close to the ventral aspect of the falx cerebri, and extending from the caudal aspect of the olfactory lobes to the level of the caudal colliculus. The mass was suspected to be of extraparenchymal and intraventricular origin, leading to compression of the cerebral hemispheres, ventral compression of the thalamus, and caudal displacement of the cerebellum. The lesion was markedly heterogeneous: hypo- to hyperintense compared to gray matter on T2-weighted and FLAIR images, hypo- to isointense on T1-weighted images. Following intravenous administration of gadopentetate dimeglumine (Magnevist, Bayer Schering Pharma AG, Berlin, Germany), the mass showed strong heterogeneous contrast enhancement (Fig. 1). A marked signal void was also noted on T2*-weighted images suggestive of associated hemorrhage and a mild perilesional hyperintensity on FLAIR. There was loss of cerebrospinal fluid signal at the periphery of the brain and signs of subtentorial and cerebellar herniation, suggestive of markedly raised intracranial pressure. Finally, a marked central intramedullary T2-weighted hyperintense signal was observed in the spinal cord, consistent with marked syringohydromyelia, most likely due to cerebrospinal fluid flow obstruction by the intracranial mass and cerebellar herniation.
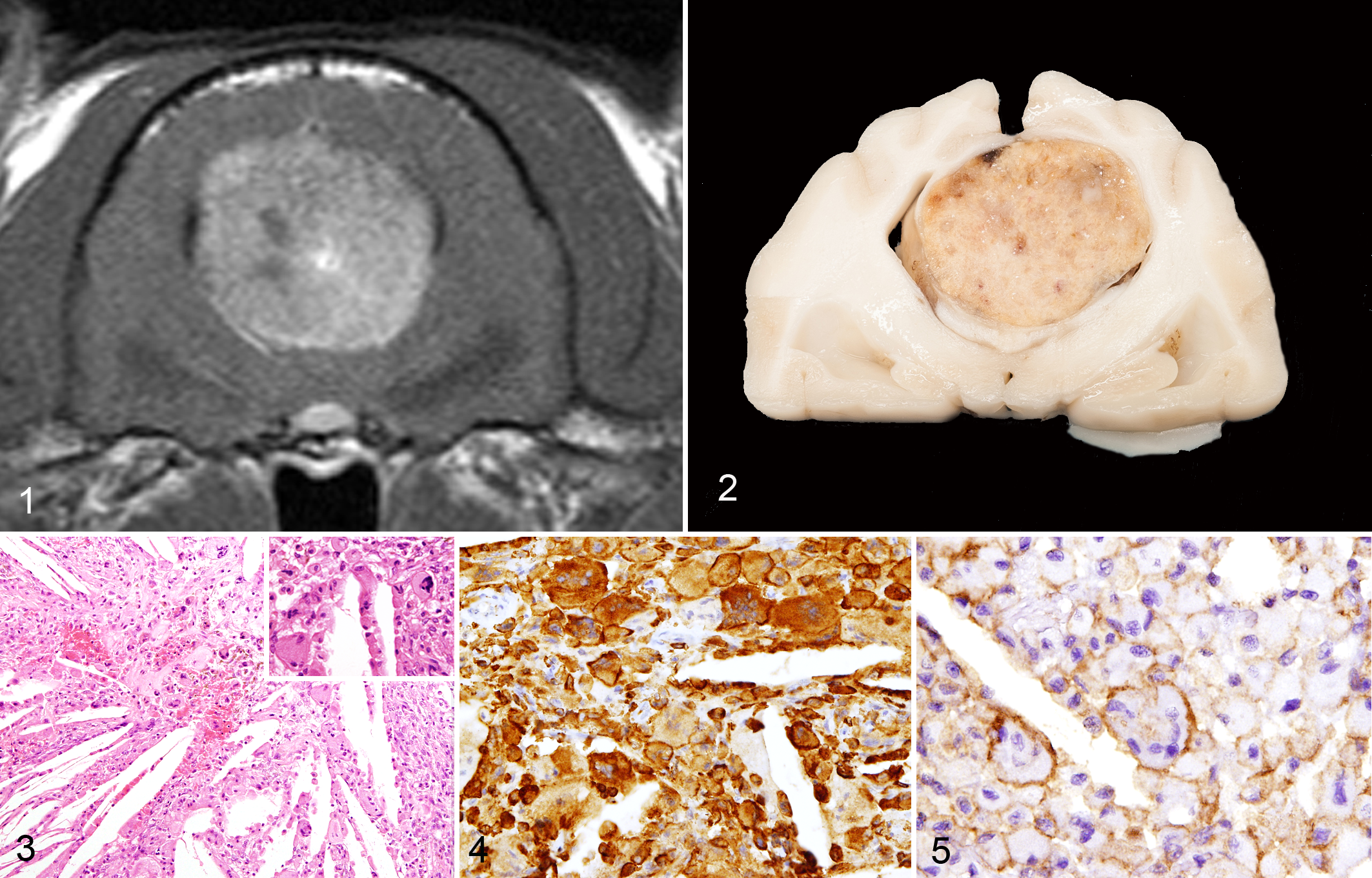
Cholesterol granuloma, brain, cat. The mass is heterogenous but overall shows marked contrast enhancement. Transverse T1-weighted postgadolinium magnetic resonance image.
Differential Diagnoses
The main clinical differential diagnosis included a choroid plexus neoplasm, a meningioma arising from the falx cerebri, or an ependymoma. A granuloma associated with hemorrhage was considered less likely. Due to the poor prognosis, suggested by the markedly raised intracranial pressure, the owner elected for euthanasia.
Necropsy Findings
Macroscopically, the brain showed widening of the midline fissure with mild separation of the cerebral hemispheres. Beneath the cerebral hemispheres, extending from the frontal lobes to the tentorial region, a well-demarcated mass with a mottled yellowish-reddish discoloration of the surface was noted (Fig. 2). Focal herniation of the ventral portion of the cerebellar vermis was also observed. The medulla oblongata and the proximal portion of the cervical spinal cord appeared slightly enlarged.
Histopathology and Immunohistochemistry
Multiple transverse sections of the mass were processed for histologic evaluation. Sections were stained with hematoxylin and eosin, periodic acid–Schiff (PAS), Perls’ Prussian blue, and Masson’s trichrome. Immunohistochemistry for MHC-II (TAL.1B5, Dako), CD18 (FE3.9F2, UC Davis), cytokeratin (MNF116, Dako), glial fibrillary acidic protein (GFAP; polyclonal, Dako), vimentin (V9, Dako), S100 (Polyclonal, Dako), neuron-specific enolase (NSE; BBS/NC/VI-H14, Dako), lysozyme (EC 3.2.1.17, Dako), Mac387 (MAC387, Dako) was performed on selected additional sections. Anti-rabbit or anti-mouse Envision (Dako) was used as visualization system.
Histologic evaluation confirmed that the mass originated within the subarachnoid space of the leptomeninges in the midline, dorsal to the corpus callosum. The mass did not arise within the lateral ventricles and did not originate from the choroid plexus of the lateral or fourth ventricles. It was composed of numerous variably sized deposits of elongated cholesterol clefts admixed with sheets and aggregates of round/polygonal cells, 15 to 30 μm in diameter, with abundant pale eosinophilic granular or finely vacuolated cytoplasm. Numerous multinucleate giant cells, up to 80 μm in diameter, with pale eosinophilic homogeneous to finely granular or wispy cytoplasm and containing from 4 to more than 20 nuclei were detectable scattered throughout the mass. Multifocal hemorrhages were noted in association with the cholesterol clefts, infiltrates of round/polygonal cells, and multinucleate cells (Fig. 3). There were variable numbers of spindle cells interspersed among the aggregates of round/polygonal cells and multinucleate cells. Small- to medium-sized blood vessels, interpreted as subarachnoid vascular structures deriving from the leptomeninges, were noted in the central portion of the mass. The cells composing the mass were PAS negative. Fine to coarse granular intracytoplasmic hemosiderin deposits were observed within round/polygonal cells and, to a lesser extent, within multinucleate cells, particularly adjacent to hemorrhagic areas. Masson’s trichrome staining highlighted the presence of collagen bundles admixed with the cellular infiltrates and the cholesterol clefts. No mitotic figures were detectable in 10 high-power fields (×400). Scattered perivascular infiltrates of lymphocytes were noted in the compressed neuroparenchyma adjacent to the mass. The results of immunohistochemical investigations showed heterogeneous staining patterns in the different cell populations. All round/polygonal cells and multinucleate cells showed strong cytoplasmic and membranous staining for MHC-II (Fig. 4), and in a subset of these cells, membranous immunoreactivity for CD18 was also observed (Fig. 5). Moderate numbers of medium-sized cells scattered throughout the mass were characterized by strong cytoplasmic immunoreactivity for Mac387 and were interpreted as recently recruited monocytes/macrophages. The larger round/polygonal and multinucleate giant cells associated with the cholesterol clefts were negative for Mac387. All cells within the mass were negative for lysozyme, cytokeratin, and GFAP. Few spindle cells revealed moderate cytoplasmic immunoreactivity for vimentin, and few showed weak cytoplasmic positivity for NSE, whereas large round/polygonal cells and multinucleate cells were negative. Variable numbers of spindle cells, round/polygonal cells, and multinucleate cells showed moderate cytoplasmic immunoreactivity for S100, particularly in the peripheral areas of the mass. Nuclear immunoreactivity for S100 was also noted in scattered round/polygonal or spindle cells.
Diagnosis
The final diagnosis was cholesterol granuloma of the leptomeninges of the brain, with marked compression of the corpus callosum, thalamus, and brainstem.
Discussion
Histopathologic evaluation confirmed that the mass arose from the leptomeninges dorsal to the corpus callosum in the midline fissure, and it ruled out origin from or direct involvement of the choroid plexi. The microscopic features of the cells composing the mass, along with the lack of cytokeratin and GFAP expression, ruled out choroid plexus neoplasm and ependymoma. 12,13 Expression of MHC-II and CD18 in the large round/polygonal cells and multinucleate cells partially supported their identification as phagocytic elements, although these cells lacked expression of Mac37 and lysozyme. Strong positivity for Mac387 was observed in macrophages associated with the mass. The presence of cholesterol clefts is a well-recognized feature of feline meningiomas; 12 however, they have also been documented in a case of feline meningeal granular cell neoplasia. 6 After the initial histopathologic assessment, meningeal granular cell neoplasia was also considered as a differential diagnosis. In the case reported by Mandara et al, 6 the granular cells were characterized by the presence of PAS-positive cytoplasmic granules inconsistently positive for S100 and were diffusely immunoreactive for vimentin and occasionally positive for progesterone receptor. The meningeal granular cell neoplasm in that report also demonstrated a distinct and heterogeneous component of “interstitial cells,” variably positive for NSE, lysozyme, and S100 and inconsistently expressing GFAP. 6 In our case, the microscopic features (including lack of distinct PAS-positive cytoplasmic granules) and immunostaining pattern of the lesion did not match those described for meningeal granular cell tumor. However, expression of vimentin, S100, and NSE in variable numbers of spindle cells within the mass suggested the presence of a component likely derived from the arachnoid elements of the leptomeninges. Expression of vimentin and S100 has been well documented in feline meningiomas. 10
Sporadic cases of feline cerebral cholesterol granuloma have been reported. 3,9,11 The first reported case offered a detailed clinical description, with pathologic characterization limited to the microscopic features in hematoxylin and eosin–stained sections, 3 whereas a more detailed histochemical and immunohistochemical characterization was reported by Ricci et al, 11 demonstrating HAM56-positive macrophages as the dominant cell population. Recently, coexistence of intraventricular meningioma and cholesterol granuloma of the choroid plexus was reported in a cat. 9 The occurrence of cholesterol granulomas in the brain has been described in several other species, including human, 4 horse, 7 dog, 5 and meerkat. 1 Equine cholesterol granuloma of the choroid plexus is a well-known age-related condition affecting up to 20% of old horses. 7 The most widely accepted theory for the pathogenesis of equine choroid plexus cholesterol granuloma is recurrent congestion, edema, and microhemorrhage with subsequent membrane breakdown of damaged erythrocytes resulting in accumulation of cholesterol crystals that elicit a foreign body granulomatous reaction. 7 In other species, including the cat, the pathogenesis is not completely understood. Local recurrent microhemorrhages are again considered as a plausible underlying mechanism, but the possible role of dyslipidemia and hypercholesterolemia has also been speculated. 1,3,4,11 The overall results of the immunohistochemical evaluation—including expression of MHC-II and CD18 in the large round/polygonal and multinucleate cells associated with the cholesterol clefts, along with the presence of a spindle cell component characterized by expression of vimentin, S100, and NSE—may support interesting speculations on the histogenesis of cholesterol cleft–rich masses in the feline brain. Recruitment and proliferation of MHC-II- and CD18-positive dendritic cells associated with variable numbers of arachnoid cells from the leptomeninges might be hypothesized as a mechanism underlying a local response to the accumulation of cholesterol crystals. Resident macrophages and dendritic cells have been identified in the meninges of mice and rats. 2,8 The present case further highlights the importance of considering cholesterol granuloma in the differential diagnosis for intracranial masses in young adult cats presenting with severe neurologic signs.
Footnotes
Acknowledgments
The authors are gratefully indebted to Mr. Richard Irvine for his help with photographic documentation of the gross pathology findings, and to Lynn Stevenson and Lynn Oxford (Veterinary Diagnostic Services, School of Veterinary Medicine, University of Glasgow) for their outstanding technical support with histopathology and immunohistochemistry.
Author Contribution
K. Faller contributed to conception and design; contributed to acquisition, analysis, and interpretation; drafted manuscript; critically revised manuscript; gave final approval; and agrees to be accountable for all aspects of work, ensuring integrity and accuracy. J. Leach contributed to interpretation, critically revised manuscript, gave final approval, and agrees to be accountable for all aspects of work, ensuring integrity and accuracy. R. Gutierrez-Quintana contributed to interpretation, critically revised manuscript, gave final approval, and agrees to be accountable for all aspects of work, ensuring integrity and accuracy. M. Finck contributed to acquisition and analysis, critically revised manuscript, gave final approval, and agrees to be accountable for all aspects of work, ensuring integrity and accuracy. G. Hammond contributed to acquisition and analysis, critically revised manuscript, gave final approval, and agrees to be accountable for all aspects of work, ensuring integrity and accuracy. J. Penderis contributed to interpretation, critically revised manuscript, gave final approval, and agrees to be accountable for all aspects of work, ensuring integrity and accuracy. F. Marchesi contributed to conception and design; contributed to acquisition, analysis, and interpretation; drafted manuscript; critically revised manuscript; gave final approval; and agrees to be accountable for all aspects of work, ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
