Abstract
Lymphomas are common spontaneous tumors in nonhuman primates but remain poorly characterized in Japanese macaques (Macaca fuscata). This study examined 5 cases of spontaneous malignant lymphoma in Japanese macaques, focusing on the immunophenotypes and presence of simian lymphocryptoviruses, which are Epstein-Barr virus-related herpesviruses in nonhuman primates. The macaques with lymphoma were 5 to 28 years old, indicating that lymphomas develop over a wide age range. The common macroscopic findings were splenomegaly and enlargement of lymph nodes. Histologic and immunohistochemical analyses revealed that all cases were non-Hodgkin type and exhibited a T-cell phenotype, positive for CD3 but negative for CD20 and CD79α. The lymphomas exhibited diverse cellular morphologies and were subdivided into 3 types according to the World Health Organization classification. These included 3 cases of peripheral T-cell lymphoma, not otherwise specified; 1 case of T-cell prolymphocytic leukemia; and 1 case of an unclassifiable T-cell lymphoma. Positive signals were detected by in situ hybridization in 2 of the 4 examined cases using probes for the Epstein-Barr virus-encoded small RNA (EBER). Furthermore, the presence of M. fuscata lymphocryptovirus 2, a macaque homolog of Epstein-Barr virus, was demonstrated in EBER-positive cases by polymerase chain reaction amplification followed by direct sequencing. Immunohistochemistry using antibody to the Epstein-Barr virus-encoded nuclear antigen 2 was negative, even in the EBER-positive cases. The present study suggests that T-cell lymphoma is more common than B-cell lymphoma in Japanese macaques and that M. fuscata lymphocryptovirus 2 is present in some cases.
Lymphoma is a common spontaneous tumor in nonhuman primates, including macaques. 1 Certain types of viruses are associated with the development of lymphomas in nonhuman primates as well as humans. Simian lymphocryptoviruses (LCVs), gammaherpesviruses closely related to Epstein-Barr virus (EBV), have been identified as naturally occurring infections in both Old and New World nonhuman primates. 6 All adult Japanese macaques (Macaca fuscata) are reported to be seropositive for macaque LCVs. 18 As with EBV infection in humans, macaque LCV infection persists for life after primary infection at an early age 18 but is usually subclinical in Japanese macaques. Meanwhile, the oncogenic potential of macaque LCVs has been suspected in prior case reports. Although there are only 2 reports of lymphomas in Japanese macaques, positive signals for EBV-encoded small RNA (EBER) in situ hybridization were detected in the tumor cells of both animals, 16,29 suggesting that lymphoma could be associated with macaque LCV infection. However, it remains unclear how frequently macaque LCV infection is present in lymphomas of Japanese macaques. In addition, while 2 different Japanese macaque LCVs—tentatively named M. fuscata LCV 1 and LCV 2 (MfusLCV1 and MfusLCV2)—have been identified based on the difference in the sequences of the DNA polymerase gene, 8 it is still unclear whether one or both of these viruses are associated with lymphoma.
In situ hybridization for EBER is the most sensitive and widely used method to detect EBV infections, 5,13 including latent infection in EBV-associated tumors. In situ hybridization has also been developed to diagnose simian LCV infection. However, except for rhesus macaques (Macaca mulatta), 25 it has been difficult to detect EBER homolog transcripts of macaque LCVs by in situ hybridization using specific probes due to a lack of available sequence data of EBER homologs. We previously demonstrated that the application of peptide nucleic acid probes for EBER is useful for detecting macaque LCVs in Japanese macaques, 16 which prompted us to further investigate the presence of LCVs in lymphomas in Japanese macaques. Besides in situ hybridization for EBER, immunohistochemistry is used to detect EBV-related antigens, including EBV-encoded nuclear antigen 2 (EBNA2) and latent membrane protein 1 (LMP1). 5 Although a previous study suggested that an LMP1 antibody did not cross-react with macaque LCV antigen, 28 cells reacting with an EBNA2 antibody have been detected in sections of lymphomas in rhesus and cynomolgus macaques (Macaca fascicularis). 2,4,10,20,24,28 Therefore, immunostaining for EBNA2 is a possible option for detecting LCV in Japanese macaques. Finally, a real-time quantitative polymerase chain reaction (qPCR) assay has recently been established to determine LCV viral load in macaque lymphomas. 4 The qPCR assay can quantify LCVs from different macaque species, including rhesus, cynomolgus, and pig-tailed macaques (Macaca nemestrina); 4 therefore, this assay could be applicable to the quantitative detection of Japanese macaque LCVs.
Simian LCVs are associated with B-cell lymphomas in nonhuman primates; this represents an important animal model for studying EBV-associated lymphoma in humans. 4,6,14,20,24,26 However, the previously reported cases of lymphoma in Japanese macaques exhibited a T/NK-cell phenotype with EBER-positive signals, 16,29 suggesting the possibility that Japanese macaque LCVs have tropisms for T cells and/or NK cells. Therefore, further investigations are required to characterize the immunophenotypes of lymphomas and to define the cell tropism of LCVs in Japanese macaques.
In addition to LCVs, a recent study demonstrated that macaque rhadinovirus belonging to the rhadinovirus 2 (RV2) lineage of gammaherpesviruses related to Kaposi sarcoma–associated herpesvirus is also associated with lymphoma in macaques. 4 Interestingly, the RV2 rhadinoviruses were detected in a number of T-cell lymphomas of pig-tailed macaques. 4 Given that the previous cases in Japanese macaques were T/NK-cell lymphomas, there is a possibility that an RV2 rhadinovirus is also associated with lymphoma in Japanese macaques. The present study examined the characteristics of spontaneous lymphomas in Japanese macaques while focusing on the immunophenotypes and presence of macaque LCVs.
Materials and Methods
Animals
Five Japanese macaques were histologically diagnosed with lymphoma at the Laboratory of Veterinary Pathology, Gifu University, between 1995 and 2011. All animals were kept in groups in a zoo or institute in Japan. Necropsies were performed at each facility after death (case Nos. 2–5) or euthanasia (case No. 1). The cases are summarized in Supplemental Table 1. Although no clinical records were available in 2 cases (case Nos. 3 and 5), in all cases the history or necropsy findings suggested no probable cause of death other than lymphoma. Case No. 1 developed severe anemia and was euthanized after a poor response to treatment. Case Nos. 2 and 4 had no clinical signs before death. Case No. 1 has been described in a case report, 16 but additional analysis was performed in the present study.
Histologic and Immunohistochemical Analyses
Tissue samples were excised from the major organs and enlarged lymph nodes during necropsy and fixed in 10% neutral-buffered formalin. For case No. 5, only the spleen and enlarged lymph nodes were submitted for pathologic examination. Samples were processed routinely, and paraffin-embedded tissue sections were stained with HE. Immunostaining for CD3, CD20, CD79α, CD4, CD8, and CD30 were performed with the primary antibodies listed in Supplemental Table 2. The cross-reactivities of the antibodies for CD markers were confirmed on the sections of normal lymph nodes and thymuses of Japanese macaques. Lymphoma tissue from a cynomolgus macaque previously identified to be positive for EBNA2 immunostaining and EBER in situ hybridization in our laboratory was used as a positive control for EBNA2. Positive signals were visualized by a peroxidase-based polymer system (EnVision/HRP, Dako, Tokyo, Japan), and sections were counterstained with hematoxylin. Lymphomas were classified according to the World Health Organization (WHO) Classification of Tumors of Hematopoietic and Lymphoid Tissue (2008 edition). Microscopic examinations were conducted by 2 veterinary pathologists (A.H. and H.S.).
In Situ Hybridization
To examine the presence of simian LCVs in neoplastic cells, in situ hybridization was performed on paraffin-embedded sections using EBER peptide nucleic acid probes (Dako)—which is a mixture of four 15-mer probes complementary to EBER1/2 RNAs—and the peptide nucleic acid ISH Detection Kit (Dako), according to the manufacturer’s instructions. Sections were counterstained with nuclear fast red.
Real-time qPCR Assays for Detecting Macaque LCV and RV2 Rhadinovirus Lineages
To determine the viral loads of macaque LCV and RV2 rhadinovirus in the lymphoma tissues, DNA was extracted from paraffin-embedded sections and analyzed by real-time qPCR assays as previously described. 3,4 Briefly, DNA was extracted using the QIAamp DNA FFPE Tissue Kit (QIAGEN, Tokyo, Japan), and qPCR assays for quantifying LCVs and RV2 rhadinovirus were performed with the primers and probes listed in Supplemental Table 3, which were designed to detect LCVs and RV2 rhadinovirus from different macaque species. 3,4 To determine viral copy number per cell, qPCR assay targeting oncostatin M (OSM), a single-copy cellular gene, was also performed. 3,4
PCR Amplification and Direct Sequencing of MfusLCV2
The presence of MfusLCV2 was assessed by PCR amplification, followed by direct sequencing based on MfusLCV2-specific primers (Supplemental Table 3). The primers were designed by modifying LCV primers so that they exactly match the genomic sequence of MfusLCV2. PCR was performed with TaKaRa EX taq (TaKaRa Bio, Shiga, Japan) according to the manufacturer’s instructions. After PCR amplification, the PCR products were purified by NucleoSpin Gel and PCR Clean-up Kit (Machery-Nagel, Düren, Germany) and subjected to sequencing analysis using an ABI PRISM 3100 Genetic Analyzer with a BigDye Terminator 3.1 Cycle Sequencing Ready Kit (Applied Biosystems, Foster City, CA).
Results
Subspecies, Age Distribution, and Sex of the Macaques With Lymphoma
The cases of Japanese macaques with lymphoma are summarized in Supplemental Table 1. Spontaneous lymphomas were observed in both subspecies of Japanese macaque: M. fuscata fuscata and M. fuscata yakui (Japanese Yaku macaque). The macaques were 5 to 28 years old (mean ± SD, 16.6 ± 10.2 years), indicating that lymphomas develop not only in aged macaques but in young adult macaques as well. Four macaques were female and the other was male.
Macroscopic Findings
The major necropsy findings are summarized in Supplemental Table 1. At necropsy, splenomegaly and enlargement of lymph nodes, including the superficial and visceral lymph nodes, were observed in all cases of spontaneous lymphoma (Fig. 1a and Supplemental Fig. 1). Ascites and pleural effusions were recorded in 4 of 5 cases (Supplemental Fig. 1).
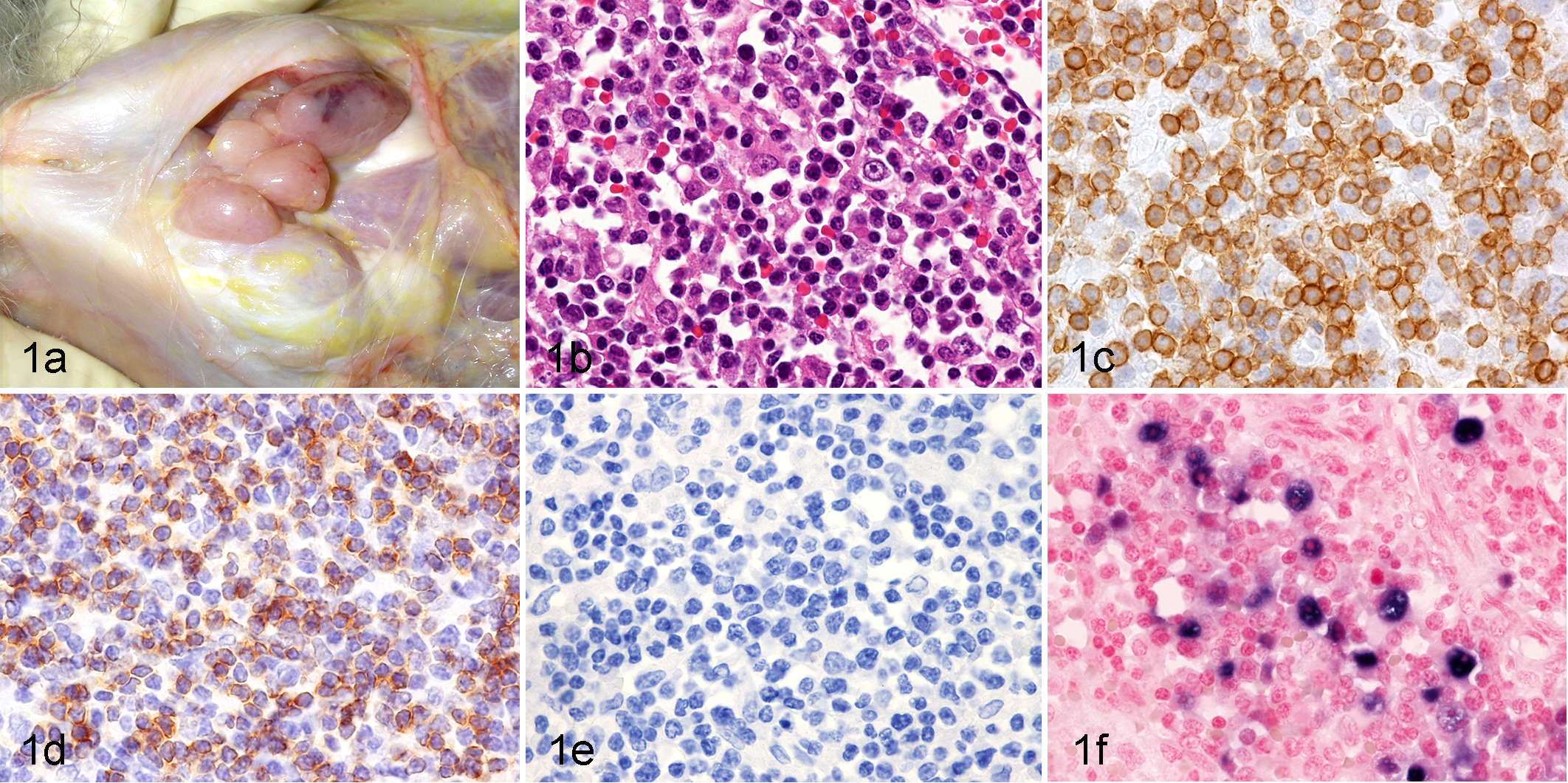
Unclassifiable T-cell lymphoma, Japanese macaque (Macaca fuscata fuscata), case No. 1. (a) Mandibular lymph nodes. Enlarged lymph nodes were apparent. (b) Spleen. The lesions are composed of small lymphoid cells with scattered medium-sized lymphoid cells. HE. (c) Spleen. Most neoplastic cells stain positively for CD3. Immunohistochemistry (IHC) for CD3. (d) Liver. Neoplastic cells are positive for CD4. IHC for CD4. (e) Spleen. Neoplastic cells are negative for CD20. IHC for CD20. (f) Spleen. EBER-positive signals are found mainly in the larger neoplastic cells. In situ hybridization for EBER transcripts. EBER, Epstein-Barr virus-encoded small RNA.
Histologic Findings
The histologic distribution of neoplastic lymphoid cells is summarized in Supplemental Table 4. Consistent with the macroscopic findings, neoplastic lymphoid cells were microscopically observed in the spleen and lymph nodes in all cases. In the spleen, there was diffuse infiltration of neoplastic cells in the white and red pulp in all 5 cases; extensive necrosis was found in 4 cases. In the superficial and visceral lymph nodes, the neoplastic cells proliferated diffusely, replacing the normal architecture, and extensive necrosis was occasionally observed. Furthermore, although general necropsy was not performed for case No. 5, histologic examination revealed a similar pattern of dissemination in all other cases, in which the neoplastic cells infiltrated the lungs, livers, and kidneys (Supplemental Table 4). In the livers, there were periportal and sinusoidal infiltrations of neoplastic cells. In the kidneys, neoplastic cells infiltrated the interstitium in all cases and formed macroscopically visible nodules in case No. 4. Neoplastic cells also infiltrated the lamina propria of the gastrointestinal tract and myocardial interstitium. In addition, circulating neoplastic cells were frequently observed in the blood vessels of various organs in all cases, indicating the hematogenous spread of neoplastic cells.
Immunophenotype and Cellular Morphology
The results of the immunohistochemical analysis are summarized in Supplemental Table 5. Although the neoplastic cells exhibited diverse cellular morphologies as described in detail below, they were positive for CD3 and negative for CD20 and/or CD79α in all cases (Figs. 1, 2). This demonstrates that all examined lymphomas were of T-cell origin. Two cases exhibited a CD4-positive T-helper phenotype (Figs. 1, 2).
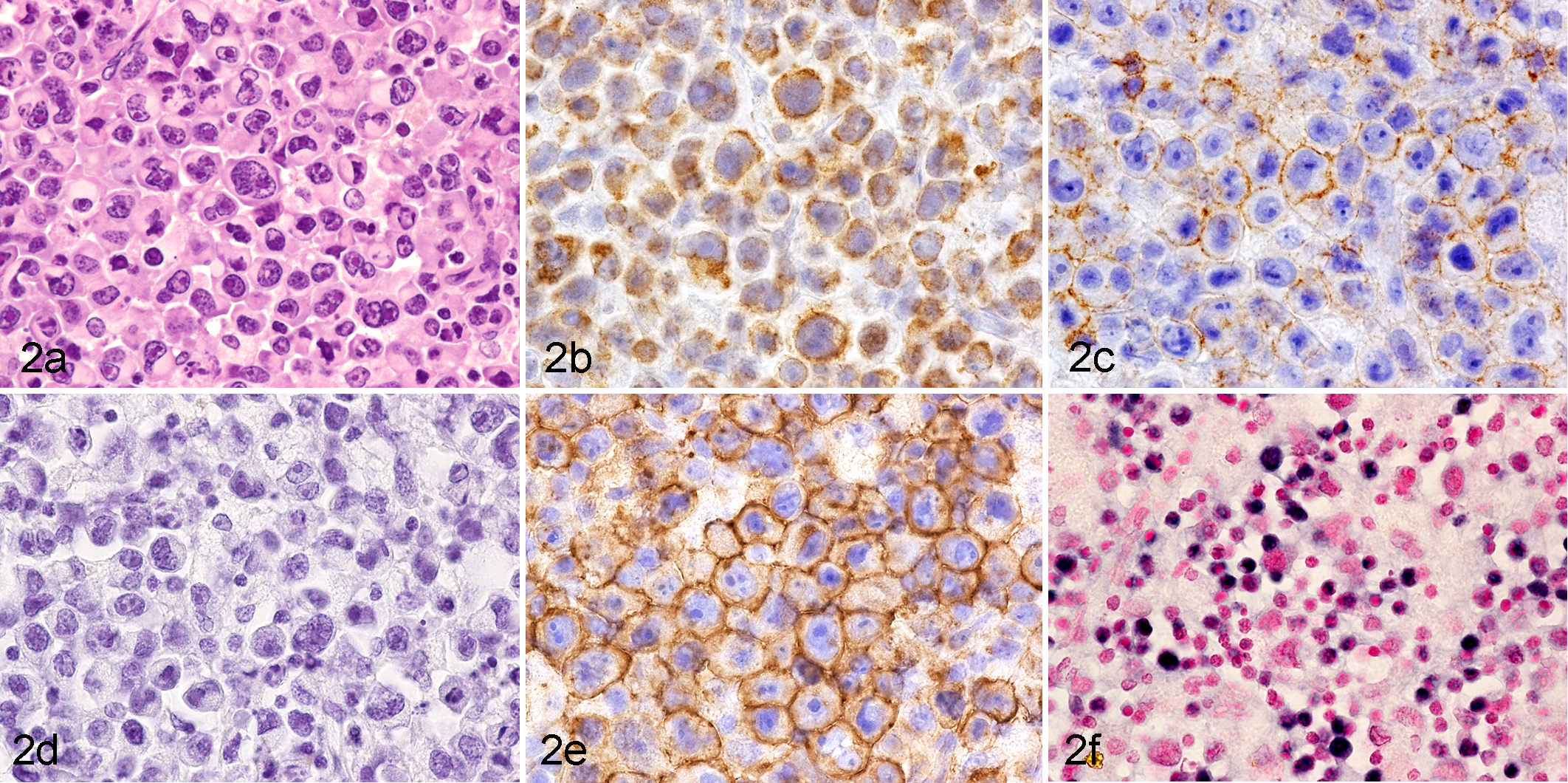
Peripheral T-cell lymphoma, not otherwise specified (NOS), lymph node, Japanese macaque (Macaca fuscata yakui), case No. 5. (a) The lesions are composed of large lymphoid cells with abundant cytoplasm and pleomorphic nuclei. HE. (b) Neoplastic cells are positive for CD3. Immunohistochemistry (IHC) for CD3. (c) Neoplastic cells are positive for CD4 on the cell membrane. IHC for CD4. (d) Neoplastic cells are negative for CD20. IHC for CD20. (e) Neoplastic cells are strongly positive for CD30 on the cell membrane and in the Golgi region. IHC for CD30. (f) Positive signals can be observed in neoplastic cells. In situ hybridization for EBER transcripts.
Histologically, the lymphomas were all non-Hodgkin type and subdivided according to the WHO classification. Case No. 1 was composed of predominantly small lymphoid cells with hyperchromatic round nuclei and scant cytoplasm with scattered medium-sized lymphoid cells (Fig. 1), which did not fit into any WHO classification category of human lymphomas. Case Nos. 2, 4, and 5 were diagnosed as peripheral T-cell lymphoma, not otherwise specified. Case Nos. 2 and 4 consisted of medium to large lymphoid cells with pleomorphic nuclei (Supplemental Figs. 2, 4). Many mitotic figures were observed in Case No. 2. Case No. 5 was characterized by the predominant proliferation of large lymphoid cells with strong CD30 expression (Fig. 2). The anaplastic morphology and uniformly strong expression of CD30 in case No. 5 suggested that it could be anaplastic large cell lymphoma. However, this case showed EBER positivity, as described below. As the WHO classification stated that anaplastic large cell lymphoma is consistently negative for EBV, case No. 5 was also diagnosed as peripheral T-cell lymphoma, not otherwise specified, rather than anaplastic large cell lymphoma. Case No. 3 showed monomorphous proliferation of small lymphoid cells with round nuclei and was diagnosed as T-cell prolymphocytic leukemia (Supplemental Fig. 3).
Expressions of EBER Homologs and EBNA2
The results of the histologic analysis for macaque LCVs are summarized in Supplemental Table 5. Positive signals for EBER in situ hybridization were detected in 2 of the 4 examined cases, case Nos. 1 and 5 (Fig. 1f, 2f), indicating the presence of macaque LCVs in these spontaneous lymphomas. In case No. 1, the positive signals were detected much more frequently in the medium-sized lymphoid cells than the small lymphoid cells. The 2 EBER-positive cases come from different subspecies, suggesting that LCV infects M. fuscata yakui as well as M. fuscata fuscata. In addition, EBER-positive lymphomas were found in both young and old Japanese macaques (5 and 28 years old). The cellular morphology also differed substantially among the EBER homolog-positive cases (Figs. 1, 2). Furthermore, EBNA2 expression in the EBER-positive cases was examined by immunohistochemistry. Positive nuclear staining was observed in the cynomolgus macaque that served as a positive control but in neither of the Japanese macaques.
PCR Analysis for Macaque LCV and RV2 Rhadinovirus
DNA was successfully extracted from case Nos. 1 and 5 and used for the subsequent molecular analyses, but we failed to obtain sufficient DNA from the other cases. The macaque LCV viral loads in the lymphomas were first examined by the established real-time qPCR assay that can detect LCVs from different macaque species. 4 Unexpectedly, the qPCR assays were negative in case Nos. 1 and 5, although EBER-positive signals were detected by in situ hybridization in both these cases. The primers and probe for this LCV qPCR assay were designed based on the consensus sequences in the DNA polymerase gene of LCVs from a number of macaque species. Since these primers and probe match the MfusLCV1 sequences exactly (GenBank accession No. AF534224), 8 our results indicate the absence of MfusLCV1 in the examined cases. However, this qPCR assay is not specific for MfusLCV2, since there are several nucleotide mismatches between the MfusLCV2 sequence (GenBank accession No. AY172954) and the forward and reverse primers. 8 To test for possible MfusLCV2 infection in Japanese macaque lymphomas, PCR amplification was performed with primers modified to completely match the MfusLCV2 sequences (Supplemental Table 3), and this gave amplified products of the expected size (188 bp) from both case Nos. 1 and 5 (Fig. 3). Specific amplification was also confirmed by direct sequencing of the PCR products (Fig. 4), demonstrating the presence of MfusLCV2 in the lymphomas of Japanese macaques.
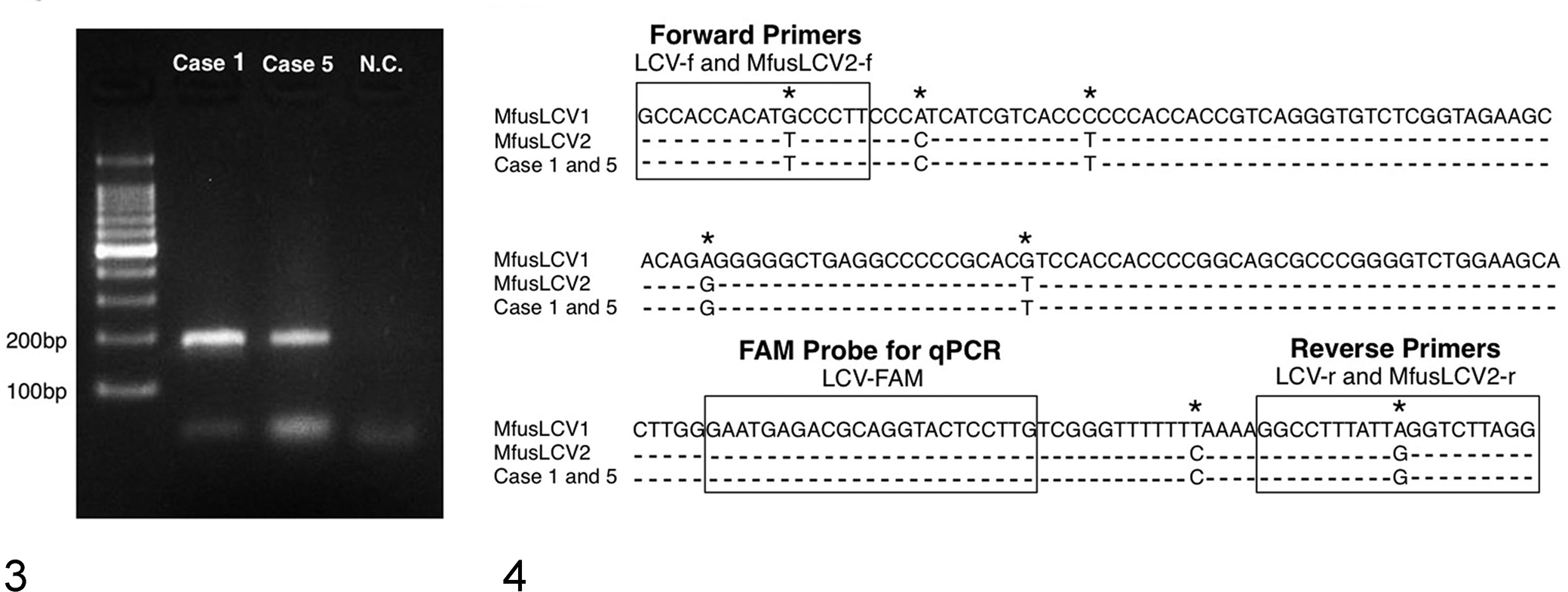
Agarose gel electrophoresis of polymerase chain reaction (PCR) products amplified with MfusLCV2-specific primers from case Nos. 1 and 5. PCR products migrate as 188-bp bands. MfusLCV2, Macaca fuscata lymphocryptovirus 2; NC, negative control (no DNA template).
In addition to macaque LCVs, the presence of a Japanese macaque RV2 lineage rhadinovirus was quantitatively evaluated by an established qPCR assay that can sensitively detect RV2 rhadinoviruses from different macaque species based on consensus sequences in the ORF60 and ORF59/60 intergenic region of rhadinoviruses. 3,4 While the primers and probe of the RV2 qPCR assay matched the genomic sequences of Japanese macaque RV2 rhadinovirus (GenBank accession no. AY528864), 3 no viral DNA was detected in case Nos. 1 and 5, indicating the absence of RV2 rhadinovirus infection.
Discussion
Interestingly, all the spontaneous lymphomas in Japanese macaques in our study exhibited a T-cell immunophenotype. To date, B-cell lymphomas have not been reported in Japanese macaques. While T-cell lymphomas have also been reported in pig-tailed macaques, 3,27 they appear to be much less common than B-cell lymphomas in cynomolgus and rhesus macaques. Although there are many reports on B-cell lymphomas in cynomolgus and rhesus macaques, 4,11,14,20 –24,28 T-cell lymphomas have not been reported in these macaque species, except for 1 case in a rhesus macaque diagnosed with CD8-positive leukemia by flow cytometry. 12
Identifying the cause of lymphoma in each macaque would help in understanding the differences in the tumor phenotype. We therefore examined the Japanese macaque lymphomas for the presence of macaque LCV and RV2 rhadinoviruses, which have been etiologically associated with lymphomas in other macaque species. While EBER in situ hybridization is considered to be the gold standard for identifying tumor-associated EBV in humans, 13 specific probes are not available to detect EBER homologs in most macaque LCVs due to the lack of available sequence data. However, in a previous study, it was found that EBER genes are relatively conserved between EBV and rhesus macaque LCVs and contain highly conserved regions. 25 Therefore, EBER probes for human EBV have been used to detect macaque LCVs in lymphomas of different macaque species, including rhesus, 2,14,24 cynomolgus, 14,26 pig-tailed, 27 and Japanese macaques. 16 Some studies have used PCR amplification of macaque LCV-specific DNA fragments to provide additional specificity data supporting the use of EBER probes for the detection of simian LCVs. 14,26,27 In the present study, we used the EBV EBER probes to detect LCVs by in situ hybridization and found that 2 of the Japanese macaque lymphomas were positive. Using a PCR assay that we developed to detect MfusLCV2, we identified MfusLCV2 sequences in both EBER-positive lymphoma samples. In case No. 1, the EBER signals were found mainly in the larger neoplastic cells. The presence of LCV in a subpopulation of tumor cells suggests that the viral infection is the consequence of a secondary molecular event, as discussed in human peripheral T-cell lymphomas. 7 Further studies are necessary to determine whether there is an etiologic association in these lymphomas with LCV.
Macaque LCVs are associated with B-cell lymphomas in cynomolgus and rhesus macaques. 2,4,6,14,20,21,24,26,28 Therefore, it is particularly noteworthy that both cases of LCV-positive lymphomas in our study exhibited a T-cell phenotype in Japanese macaques exhibiting cell tropism different from those in cynomolgus and rhesus macaques. In addition, the LCV-positive macaque lymphomas differed substantially with respect to age and cellular morphology. Further studies are required to determine the age of peak incidence and characteristic morphologic type of LCV-positive lymphoma in Japanese macaques. It is worth mentioning that LCVs were found in the lymphomas of both Japanese macaque subspecies. To our knowledge, this is the first case of LCV-positive lymphoma in M. fuscata yakui. Although no serologic study was conducted, this case suggests that simian LCV infects M. fuscata yakui as well as M. fuscata fuscata.
In addition to EBER homologs, the expressions of EBV-related antigens were immunohistochemically examined. While EBNA2 expression has been detected in lymphomas from cynomolgus and rhesus macaques, 2,10,20,24 no EBNA2 expression was detected in the Japanese macaque lymphomas. Although a previous study suggests that LMP1 antibody does not cross-react with simian LCV antigen, 28 we performed LMP1 immunostaining on sections of Japanese macaque lymphomas but did not observe membranous staining for LMP1 (data not shown). At present, EBER in situ hybridization is the first choice for the histologic detection of simian LCVs in Japanese macaques.
A recent study found high levels of the RV2 lineage of rhadinovirus in pig-tailed macaque T-cell lymphomas, suggesting an etiologic role in its development. 4 While Japanese macaque lymphomas consistently exhibited a T-cell phenotype and while rhadinovirus is found in Japanese macaques, 9 RV2 rhadinovirus was not detected in the 2 cases that we examined. However, although high levels of RV2 rhadinovirus were found in simian LCV-negative lymphomas in pig-tailed macaques, 4 we could not examine the viral levels in the EBER-negative cases. Therefore, further study is required to determine whether RV2 rhadinovirus is associated with Japanese macaque lymphomas. Since EBER-negative cases also exhibited a T-cell phenotype, simian T-cell lymphotropic virus could be another potential candidate. Although no information about the serologic status of the Japanese macaques used in the present study was available, about 25% to 30% of Japanese macaques have anti-T-cell lymphotropic virus antibody. 15,19
In conclusion, the present study suggests that T-cell lymphoma is more common than B-cell lymphoma in Japanese macaques and that MfusLCV2 is detected in some cases. In Japan, a project is in progress to establish Japanese macaques as laboratory animals. 17 Therefore, Japanese macaques may serve as an animal model of T-cell lymphomas in humans.
Footnotes
Acknowledgements
We thank Ms Chikako Iriyama at Laboratory of Veterinary Pathology, Gifu University, for her expert technical assistance and the Division of Genomics Research, Life Science Research Center, Gifu University, for its support in molecular analysis. This study was supported by the Cooperation Research Program of the Primate Research Institute of Kyoto University and a Grant-in-Aid from the Ministry of Education, Culture, Sports, Science, and Technology of Japan.
Author Contribution
Hirata A. contributed to conception and design of study and acquisition, analysis, and interpretation of data and drafted manuscript. Hashimoto K. contributed to acquisition of data. Katoh Y. contributed to acquisition of data. Sakai H. contributed to acquisition of data. Rose T. contributed to conception and design of study and acquisition, analysis, and interpretation of data and critically revised manuscript. Bruce G. contributed to acquisition of data. Kaneko A. contributed to acquisition of data. Suzuki J. contributed to acquisition of data. Nikami H. contributed to acquisition of data. Yanai T. contributed to conception of study and acquisition of data.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
