Abstract
A novel form of primary feline hemangiosarcoma and additional cases of plexiform vascularization in the cervical lymph nodes are reported. Sixteen cases of feline lymphadenopathy attributed to abnormal vascular proliferation were identified and evaluated. Most of these lesions were diagnosed histologically as hemangiosarcoma. However, lesions of plexiform vascularization, with and without areas of putative malignant transformation, were also identified. Mean age of the cats was 11 years (range, 3–16 years) with most being domestic shorthair and medium hair (13). Two domestic long hair and 1 Maine Coon were identified. Excisional nodal biopsy was performed in 15 cases and incisional biopsy in 1 case. Six cats were euthanized due to their disease. Survival times ranged from ≤1 month to ≥30 months. We provide a new clinical differential for cervical lymphadenopathy in cats that is not widely recognized. Proper identification of primary nodal vascular lesions in cats will enable further characterization of clinical features and biologic behavior to determine specific therapy.
Vascular transformation of lymph node sinuses has been described in human pathology and a dog 6 and is characterized histologically by conversion of nodal sinuses into capillary-like channels, often accompanied by fibrosis. 8 The underlying mechanism is thought to be associated with venous or lymphatic obstruction. 24 In 1987, Lucke et al 12 described a specific feline lymphadenopathy in which the lesions were characterized by capillary vasoproliferation and lymphoid atrophy, which was designated plexiform vascularization, and it was postulated that it may be related to vascular transformation of lymph node sinuses. In that study, 5 of 9 cases were identified in the ventral cervical region and 4 cases in the inguinal region. 12 In the current study, 16 cases of proliferative vascular lesions involving the cervical lymph nodes of cats are described. These proliferative lesions demonstrate a spectrum of changes that range from benign (plexiform vascularization) to malignant (hemangiosarcoma) based on histomorphologic features. We provide clinical follow-up on 11 cases indicating that histomorphologic features of malignancy correlate to risk for aggressive regional growth and local recurrence after incomplete excision. We propose that benign vascular proliferations in the cervical lymph nodes of cats have the potential to undergo malignant transformation and that lesions with cytologic or histopathologic evidence of vascular proliferation should be completely excised.
Hemangiosarcoma (HSA) in feline patients is uncommon, occurring in 0.5% of cats at necropsy. 26 Median age of occurrence ranges from 8 to 12.2 years. 3,11,13,22 No specific cat breeds have been identified as having a predisposition for HSA development. Most tumors are in domestic shorthairs. 3,11,18 The underlying etiology of feline cutaneous or visceral HSA is largely unknown. Ultraviolet light exposure has been associated with canine cutaneous HSA and suggested in cats, especially in hypopigmented regions. 1,21,22 HSA has been described in cutaneous and visceral locations as well as the heart, thoracic cavity, and nasal cavity. 18 To the authors’ knowledge, no cases of primary nodal HSA have been reported in cats. We describe a novel form of feline HSA that is primary to lymph nodes most often located in the ventral cervical region.
Materials and Methods
Patient Population
The computer records from a veterinary diagnostic histopathology laboratory (Veterinary Diagnostics [VDx], Davis, CA) were searched for feline patients with a diagnosis of vascular proliferation, plexiform vascularization, and/or HSA lymph nodes from July 2007 to July 2012. Selected cases had histologic features of plexiform vascularization or HSA within a lymph node, or described lymph node location, on submission reports. Cases were excluded if the described location was other than a lymph node region. Data regarding signalment, presenting complaint, staging, treatment modalities, and outcome, including diagnostic imaging, therapy, recurrence, survival time, and cause of death, were collected from medical records, laboratory submission forms, and telephone interviews of referring veterinarians and clients. The date of surgical excision was defined as the date of diagnosis. Follow-up time was defined as the time from date of diagnosis to date of last follow-up or death. Local recurrence or persistent HSA was defined as regrowth of a mass at the surgical site. All biopsies were reviewed by 1 pathologist (T. Spangler) and classified as either plexiform vascularization or HSA based on established criteria. 15
Histopathology
All tissues were received as routine biopsy submissions at VDx fixed in 10% neutral buffered formalin. Representative samples were trimmed, placed in tissue cassettes, routinely processed in graded alcohols, cleared in xylene, embedded in paraffin, sectioned at 4 to 6 microns, mounted on glass slides, and stained with hematoxylin and eosin for light microscopic evaluation. The diagnosis of plexiform vascularization was based on characteristic histopathologic features as described by Lucke et al. 12 Affected lymph nodes were markedly expanded due to severe congestion and capillary vasoproliferation. Remaining lymphoid tissue was mostly atrophic, although some hyperplastic lymphoid follicles with well-developed germinal centers were present. There was also fibrous thickening of nodal capsules and supporting trabeculae, features consistent with “vascular transformation of lymph nodes” described in human literature. 20 The diagnosis of HSA was based on the presence of normal tissue effacement by a cellular proliferation forming vascular clefts or channels somewhere in the tumor. The neoplastic cells lining the clefts were typically highly variable and ranged from spindloid to polygonal to ovoid with prominent, bulging, pleomorphic nuclei and were frequently associated with intratumoral necrosis and hemorrhage. 15
Results
Clinical Summary
Signalment, clinical presentation, treatment, and outcome are summarized in Supplemental Table S1. Affected cats ranged from 3 to 16 years. The mean age was 11 years (median, 11.5 years). There were 4 female and 12 male cats. Domestic shorthairs were the majority (12), along with 2 domestic long hairs, 1 domestic medium hair, and 1 Maine Coon cat.
Clinical presentation was similar for 15 cases that presented with single (13) or multiple (2) palpable subcutaneous mass lesions in the submandibular (7), ventral cervical (7), or axillary regions (1). In most patients (14/16), the mass was noted by the owner with no other presenting complaints. Cat No. 4 presented to a referring veterinarian for cross-sectional imaging for chronic otitis, and the veterinarian noted an enlarged right ventral cervical mass. Cat No. 7 had no history or records available.
Clinical Staging
Nine cats had initial cytology of the mass performed, 3 of which were reported as inconclusive or hemodiluted. Two of the 9 cytologies were inconclusive, but a few mesenchymal cells were noted. In 1 cat, a polymorphic population of mesenchymal cells with a population of small lymphocytes was reported, suggestive of a spindle cell sarcoma. Two additional cases were most consistent with lymphoid hyperplasia, and the final cat cytology was identified as a poorly differentiated neoplasm.
Complete blood counts and chemistry panels in 9 cats disclosed no specific abnormalities. Thoracic radiographs were performed in 5 cats preoperatively and 1 patient postexcision; no significant abnormalities were noted. Perioperative abdominal ultrasonography was performed in 4 patients; no abnormalities were noted in 3 cases. Cat No. 10 had mild mesenteric lymphadenopathy, which was aspirated and diagnosed as lymphoid hyperplasia. Cervical ultrasound was performed in 1 patient (cat No. 2) prior to therapy, which confirmed an enlarged lymph node. Three patients (cat Nos. 4, 9, and 11) received advanced imaging prior to therapy. Cat No. 11 underwent nuclear scintigraphy as well as head and neck magnetic resonance imaging. Initial thyroid scintigraphy was inconclusive for a thyroid-associated mass; magnetic resonance imaging confirmed a mass effacing the left medial retropharyngeal lymph node. Head and neck computed tomography in cat No. 9 confirmed an effaced right retropharyngeal lymph node. In cat No. 4, head and neck computed tomography suggested a right ventral cervical mass suspicious for thyroid. No other lesions suggestive of primary or metastatic disease were noted on advanced imaging in these patients.
Cat No. 12 was also evaluated with skull radiographs with no remarkable findings. Thoracic ultrasound was performed in 3 patients (cat Nos. 9, 11, and 14) for staging. Two of these patients had a cardiac murmur and 1 was previously noted to be hyperthyroid. Results demonstrated mild hypertrophic cardiomyopathy, and the other 2 cats had no clinically relevant heart disease. No cardiac masses or pericardial effusion were noted. No additional workup was reported for the remaining 3 patients.
Clinical Outcome
All cats had excisional biopsy (15), incisional biopsy (1), or both (1) of the mass lesions. The primary treatment modality in 15 of 16 patients was excisional biopsy. Incisional biopsy was consistent with malignant hemangiosarcoma in cat No. 14; however, no subsequent therapy was administered. A histologic diagnosis of HSA partially or completely effacing the lymph nodes was made in 12 of 16 cats. Two of 16 biopsy submissions were diagnosed as plexiform vascularization of the lymph node. Another 2 were diagnosed as plexiform vascularization of the lymph node with features of early capsular invasion and/or malignant transformation. A single patient (cat No. 12) received postoperative doxorubicin chemotherapy.
Two of the masses had complete margins. Complete margins were defined by an intact capsule of dense connective tissue with ≥1 mm separating the closest tumor cells from the tissue edge. Three patients had incomplete margins or tumor extending to tissue margins. The remaining 10 patients had marginal excisions (narrowly around the capsule) with <1 mm separating the neoplastic cells from the tissue edge. The incisional biopsy margins were not evaluated for completeness. Of those diagnosed as HSA, 3 of 12 cases were incompletely excised and the other 9 were marginal.
Four patients with HSA that developed palpable postoperative masses after regional lymph node excision were considered to have local recurrence or progressive disease. Time to recurrence or progressive residual disease ranged from 2 to 10 months (mean, 7.4 months; median, 7 months) postdiagnosis. Surgical margin evaluations in these 4 cases were considered inadequate, and these patients were euthanized due to suspected progressive disease. No further information was available.
Two patients (cat Nos. 1 and 2) with plexiform vascularization of cervical nodes were alive with no recurrence at 19 and 8 months, respectively. Two patients (cat Nos. 3 and 4) with transition from plexiform vascularization to vascular neoplasia were alive at 30 and 13 months, respectively. Surgical margins around the lesion in these 2 patients were reported to be less than 1 mm. Cat No. 6 had surgery for 2 masses in the right superficial cervical region; both had a similar appearance of plexiform vascularization with features of HSA. This patient developed a mass effect at the surgery site, which was presumed recurrence or residual disease, approximately 10 days postoperatively along with new hemorrhagic cutaneous nodules on the left and right medial thighs. This patient was euthanized 14 days later for anorexia and further progression of cutaneous nodules and associated limb edema. Cutaneous metastasis was suspected but not confirmed.
Histologic evaluation of excised lymph node material from cat No. 11 indicated HSA with invasion of adjacent tissues. Local recurrence was clinically apparent 10 months postoperatively, and the patient was euthanized with no further workup.
Only 1 cat (No. 12) received adjuvant chemotherapy. Doxorubicin was delivered once every 21 days for 2 cycles. Therapy was discontinued due to vomiting and mild anorexia. This patient had complete surgical margins and was alive 25 months later. Cat No. 10 received prednisone prior to surgical removal with progressive disease noted within 5 months. Cat No. 14 had an incisional biopsy and died at home 3 weeks from diagnosis. This patient was described by the owner as anorectic with progressive enlargement of the mass postbiopsy. No additional diagnostics were performed to confirm progression.
Histology confirmed the excised tissue was a lymph node in 11 patients. Of the remaining cases, 2 patients had lesions considered of likely lymph node origin based on the presence of dense, peripheral, nodular clusters of lymphocytes. Of these cats, 1 had surgical removal of a thyroid carcinoma (cat No. 16) at the time of mass excision in the same vicinity. Of the remaining 3 patients, 2 lesions were diagnosed clinically as a lymph node based on head and neck computed tomography (cat No. 9) or cervical ultrasound (cat No. 14). The remaining patient had surgical excision of both a normal salivary gland and adjacent neoplastic structure supporting cervical lymph node as the mass origin. The tumor from the patient with the incisional biopsy (cat No. 14) was also suspected of being lymph node in origin based on physical examination but could not be confirmed by histologic examination. Due to the retrospective nature of this study, limited follow-up was available for all cats, and some areas are incomplete.
Pathology
Pathology findings are summarized in Supplemental Table S2. Grossly, the lesions ranged from 1 to 4 cm in diameter and were heterogeneous on the sectioned surface. Lymph nodes with plexiform vascularization were enlarged or prominent, with severe congestion and characteristic pale follicular structures distributed along the cortex. HSAs had solid, white, tan, or variably congested tissue intermixed with cavitated areas of hemorrhage and/or necrosis. Occasionally, remnants of compressed lymph node tissue were present around the edges. Most tumors had well-demarcated, smooth margins composed of a compressed pseudocapsule and surrounding fat and were narrowly or marginally excised.
Cat Nos. 1 and 2 were diagnosed with plexiform vascularization and had nodal expansion due to severe congestion and capillary proliferation. The remaining lymphoid tissue was mostly atrophic, although some hyperplastic lymphoid follicles were present. In addition, in cat No.2 (Figs. 1, 2), the perinodal vasculature was also more prominent than normal, markedly congested, and partially occluded with fibrinocellular thrombi. Two cases (cat Nos. 3 and 4) had features of plexiform vascularization and areas of increased cellular density and proliferative vascular channels that disrupted the preexisting lymph node capsule or extended into the perinodal fat. Figures 3, 4, and 5 represent cat No. 3 with characteristics of both plexiform vascularization and HSA. These lesions were thought to demonstrate early malignant transformation of plexiform vascularization into HSA.
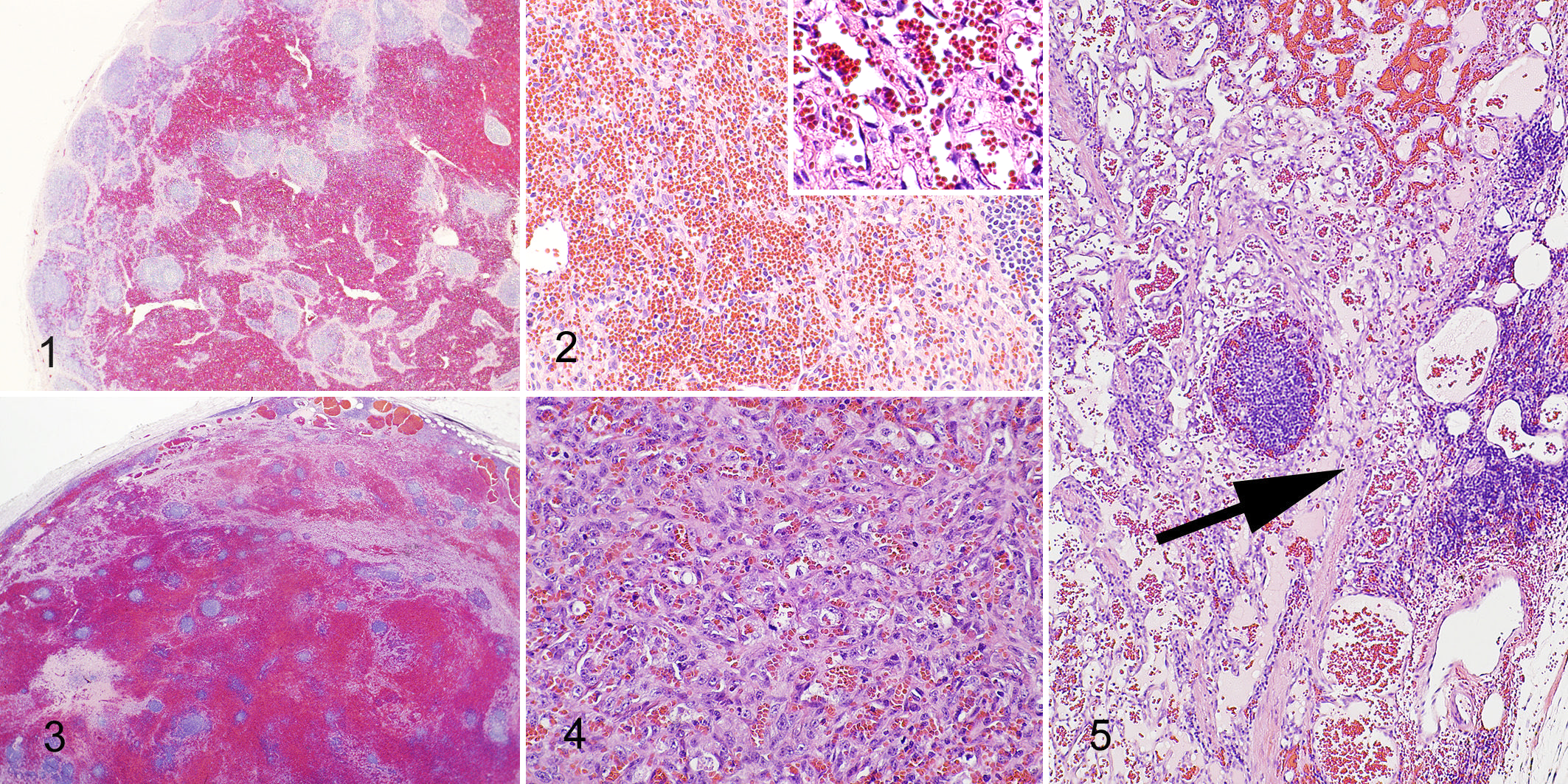
Neoplastic lesions were readily identified based on examination at low magnification due to the presence of disorganized, high cellular density intermixed with foci of necrosis and hemorrhage that effaced normal lymph node architecture (Fig. 6). Remnant lymph node architecture was present in 7 cases of HSA. In 3 cases, definitive, histologic confirmation that the neoplasm was infiltrating a preexisting lymph node could not be made. However, the presence of multiple, small, dense clusters of lymphocytes around the peripheral edge of these masses was interpreted as lymph node parenchyma. In cases of HSA, the histopathologic findings were typical and characterized by a “classic” appearance of proliferative, pleomorphic, spindle-shaped cells lining various, abnormal vascular channels (Figs. 7–9).
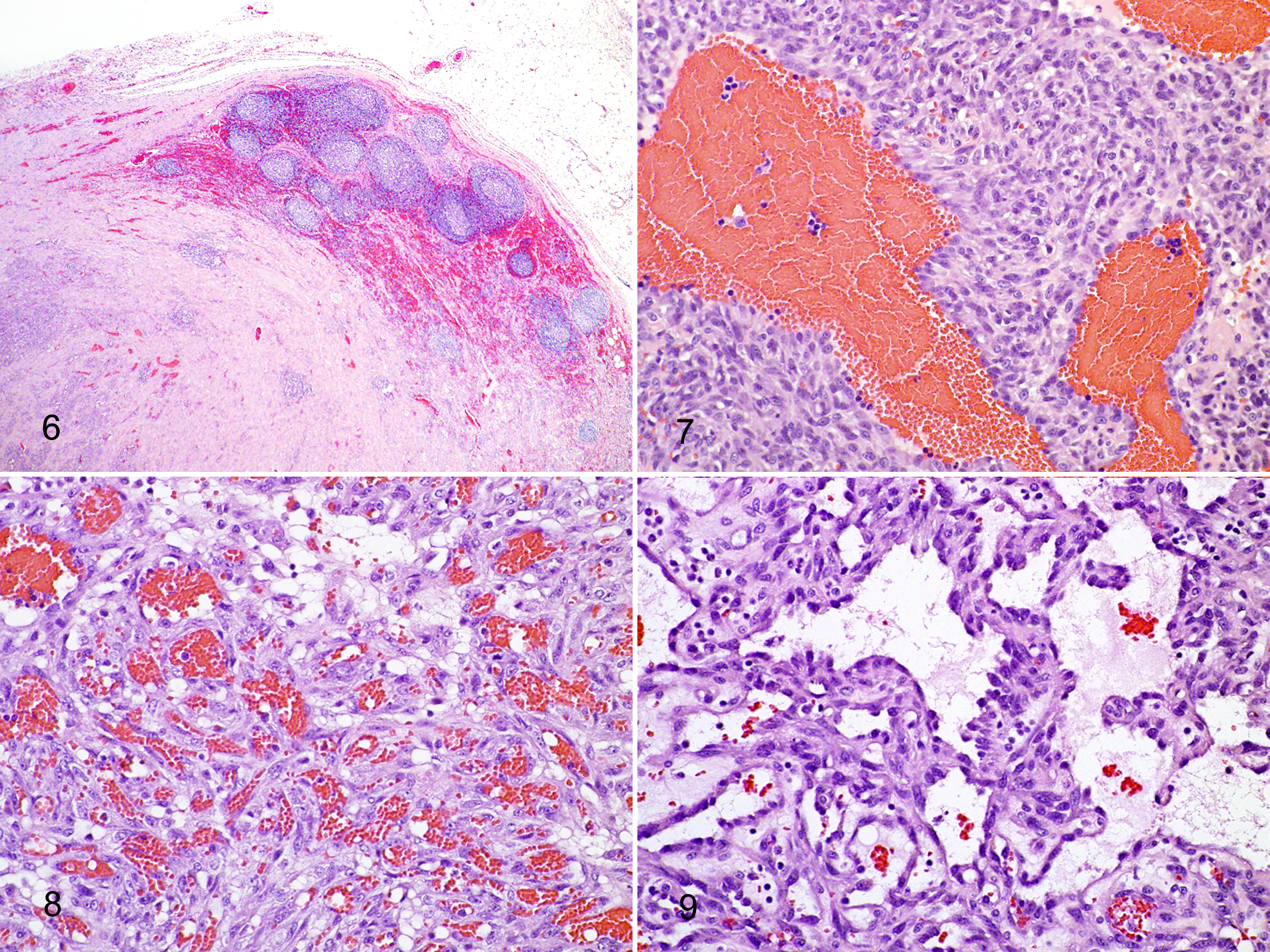
Neoplastic endothelial cells were often aligned on delicate, interconnecting collagen trabeculae that formed redundant, blood-filled channels indicative of trabecular or capillary HSAs. Other histologic patterns consistent with HSA—including areas of solid, dense spindle cell proliferation; proliferation of a tangled cellular meshwork; and cells separated by small clefts containing a few red blood cells and also multiple large, blood-filled cavernous sinuses—often coexisted in the same lesion.
Multifocal to coalescing areas of necrosis and variably sized lakes of hemorrhage were observed in most tumors. The percentage of lesion necrosis ranged between 0% and 95%. The growth pattern of the neoplastic endothelial cells most commonly effaced normal architecture. Around the edges, the masses formed a broad front and an expansile surface that invaded the surrounding adipose tissue. These edges were mostly smooth and well demarcated, although small areas of capsular invasion were present. Individual cells were pleomorphic but mostly spindloid. Nuclei were typically large and oval with 0 to 2 nucleoli, stippled chromatin, and occasional karyomegaly. The mitotic index ranged from ≤1 to ≥25 per 10 high-magnification fields.
Discussion
Plexiform vascularization of lymph nodes in cats has been described by Lucke et al 12 and is characterized by capillary vasoproliferation with lymphoid atrophy, which has been described in humans as “vascular transformation of lymph node sinuses.” 17 In 1971, Haferkamp et al 8 initially described a combination of focal lymphoid atrophy and proliferation of capillary-sized blood vessels described as vascular transformation in humans. 19 This usually represented an incidental finding. 8 Although the underlying mechanism is poorly understood, it is thought to be associated with venous obstruction and histologically characterized by nodal sinuses forming complex, anastomosing vascular channels. 10,20
The precise nature and pathogenesis of our cases is uncertain, but the plexiform vascularization of feline lymph nodes described by Lucke et al 12 in 1987 and in this study resembles vascular transformation lesions in humans. In this study, a total of 4 lymph nodes were diagnosed as plexiform vascularization and had histologic changes consistent with that previously described (cat Nos. 1–4). 25 Two of the 4 also had areas of abnormal proliferations that extended beyond the lymph node capsule (cat Nos. 3 and 4). The remaining 12 cases were diagnosed with intranodal hemangiosarcoma based on established criteria that include trabecular capillary proliferation, endothelial cell nuclear pleomorphism and atypia, intralesional necrosis, sinusoidal cavitation, and thrombosis (cat Nos. 5–16). 12
Malignant transformation from benign lesions has been described for various feline neoplasms. 15 An association among chronic inflammation, fibroplasia, and subsequent development of sarcomas has been identified in cats with injection site sarcomas. 2,4,7,9,27 This phenomenon is also illustrated in cats that develop intraocular sarcomas after trauma or chronic uveitis. 9 The specific pathogenesis of malignant transformation in these lesions has not been elucidated. 5,14 Inflammatory reactions in cats are theorized to lead to uncontrolled fibroblast and myofibroblast proliferation and eventual malignant transformation. 23 On the basis of the propensity of feline patients for demonstrating malignant transformation of previously benign lesions, the authors speculate a similar phenomenon may occur in cases of primary nodal plexiform vascularization and HSA.
In this report, the clinical follow-up information suggests that plexiform vascularization of lymph nodes has a benign course when completely excised. The 3 animals in this study with follow-up were alive at the end of the study (cat Nos. 1, 3, and 4) without reported recurrence or progression of disease. This is similar to the data reported by Lucke et al, 12 in which 6 of 9 cats had no local recurrence, hematologic abnormalities, or additional lymphadenopathy in a follow-up period ranging from 3 to 35 months, as well as a case report of plexiform vascularization of a retropharyngeal lymph node in a 4.5-year-old male neutered domestic shorthair treated with surgical extirpation alone and followed for 3 months postoperatively. 26 Surgery alone may be curative for this early stage disease complex.
We described a novel form of feline HSA and report additional cases of plexiform vascularization that are primary to the ventral cervical lymph nodes. Surgical excision in this study was a means of diagnosis and putative treatment. In a report of 18 cats with nonvisceral HSA, surgery was associated with longer median survival times than in those cats that did not receive surgery. 13 Surgery was also the primary treatment in 47 of 53 cats with HSA in a 2007 study by Johannes et al. 11 Seventeen of the cats had subcutaneous HSA compared with 24 cutaneous, 10 visceral, and 2 gingival lesions. Completeness of excised tumors was associated with longer survival times compared with incompletely excised tumors. In addition, subcutaneous HSAs were associated with a high rate of local recurrence (6/12 with follow-up) compared with cutaneous lesions. 11 In the current study, limited data precluded assessment of surgical margins and recurrence, but in 5 patients in whom local recurrence was suspected postoperatively, these margins were deemed narrow in 4 and incomplete in 1 cat. All of these patients were euthanized for suspected recurrent disease at the surgical site. Wide surgical margins are indicated for removal of any subcutaneous mass or lymph node suspicious for HSA.
In a cat with an enlarged cervical lymph node, various differentials for diagnosis such as distinctive peripheral lymph node hyperplasia of young cats, 24 feline Hodgkin-like and non- Hodgkin lymphoma, 16 lymphoid hyperplasia, plexiform vascularization, and nodal hemangiosarcoma should be considered. Cytology may be inconclusive, making biopsy necessary for definitive diagnosis. Cytology in a suspected lymph node or ventral cervical mass yielding blood should prompt differentials of plexiform change and nodal HSA. If surgical extirpation is attempted, wide margins are suggested. Other diagnostic studies including angiograms, cervical ultrasound, and magnetic resonance imaging or computed tomography to further define the extent of disease and guide treatment may be indicated. Plexiform vascularization and intranodal HSA are differentials for a single, enlarged, ventral, subcutaneous mass in a felid.
There are limitations to retrospective analyses, and incomplete follow-up in these cases precludes many therapeutic recommendations. Since postmortem and cytologic or histologic confirmations were not available for any suspected recurrence, this information is speculative; hence, survival data were not calculated for this study. More information is needed to further characterize feline nodal HSA. Prospective studies to understand biologic behavior are necessary as well as continued research in etiopathogenesis of vascular transformation.
Footnotes
Acknowledgements
We thank Dr John Peauroi for laboratory support and guidance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
