Abstract
GREMLIN 2 (GREM2)—formerly, protein related to Dan and cerberus (PRDC)—is a potent antagonist of the bone morphogenetic proteins 2 and 4, but little else in known about its functions. We found that Grem2-/- mice developed small deformed mandibular and maxillary incisors, indicating that GREMLIN2 is required for normal tooth morphogenesis. Although DEXA scans suggested that bone mineral density might be increased in Grem2-/- mice, histology did not reveal any evident bone phenotype. Grem2-/- mice did not display any other notable phenotypes evaluated in a high-throughput screening process that encompassed a range of immunologic, metabolic, ophthalmic, and behavioral parameters. Our findings indicate that Grem2 can be added to the growing list of genes that affect tooth development in mice.
The bone morphogenetic proteins (BMPs) compose a group of potent growth factors belonging to the transforming growth factor beta (TGFβ) superfamily that were originally identified according to their ability to induce the formation of bone and cartilage. 32 BMPs are required for skeletal development and maintenance of adult bone homeostasis, and they play a role in fracture healing. Mutations in either BMPs or their antagonists are associated with a number of skeletal disorders, but they can also affect the development of a range of other tissues (see review 33 ). More than 20 BMPs have been identified, and they are known to have a variety of functions in such processes as embryogenesis, skeletal development, hematopoiesis, and neurogenesis. 3 BMPs also play role in the development and progression of cancer. 9,20
The extracellular BMP antagonists are secreted peptides that bind BMPs with high affinity and prevent their interaction with their specific receptors. 7 The 4 subgroups of extracellular BMP antagonists include noggin, the chordin family, twisted gastrulation, and the Dan family. 1,40 GREMLIN 2 (GREM2), formerly protein related to Dan and cerberus (PRDC), represents a member of the DAN (differential screening-selected gene aberrative in neuroblastoma) family, with others including Gremlin 1, Sclerostin, Dan, Ectodin, Cerberus, Caronte, Coco, and Dante. 7
GREM2/PRDC was originally identified in a gene-trap screen for developmentally important genes, 24 but relatively little has been reported about its functions. The protein GREMLIN 2/PRDC is a potent antagonist of BMP2 and BMP4, 37 and recent in vitro studies demonstrated that it participates in the regulation of osteogenesis by blocking BMP2 activity 12 and inhibiting osteogenic differentiation that would otherwise be induced by BMP2. 38 GREMLIN 2 appears to have skeletal actions. Craniofacial development in zebrafish is influenced by GREMLIN 2 expression, as part of a regulatory system involving BMP4, endothelin-1, and jagged 1b. 43 A recent human genomewide association study identified a single-nucleotide polymorphism in GREM2 associated with trabecular but not cortical bone mass and both vertebral and hip fractures. 30
During high-throughput mutagenesis and phenotyping of more than 4600 knockout mouse lines, 41,42 we discovered phenotypes that have proven useful in elucidating fundamental processes in biology. Although GREMLIN 2–deficient mice (Grem2 tm1Lex is abbreviated as Grem2-/- in this report) were essentially normal in almost all phenotyping assays, they displayed defects in the upper and lower incisors. Given these observations and our preliminary data showing elevated bone mineral density in Grem2 knockout mice, additional studies are required to characterize the effects of Grem2 disruption on bone mass and architecture in mice. These findings in otherwise apparently normal mice provide another example of the value of including histologic evaluation of teeth as a part of high-throughput phenotyping systems. 34
Methods
Mouse Production
The Grem2 targeting vector was derived through long-range polymerase chain reaction (PCR) to generate the 5′ and 3′ arms of homology through 129S5 ES cell DNA as a template. The 3813-bp 5′ arm was generated through primers Grem2-5 (5′-TACTA
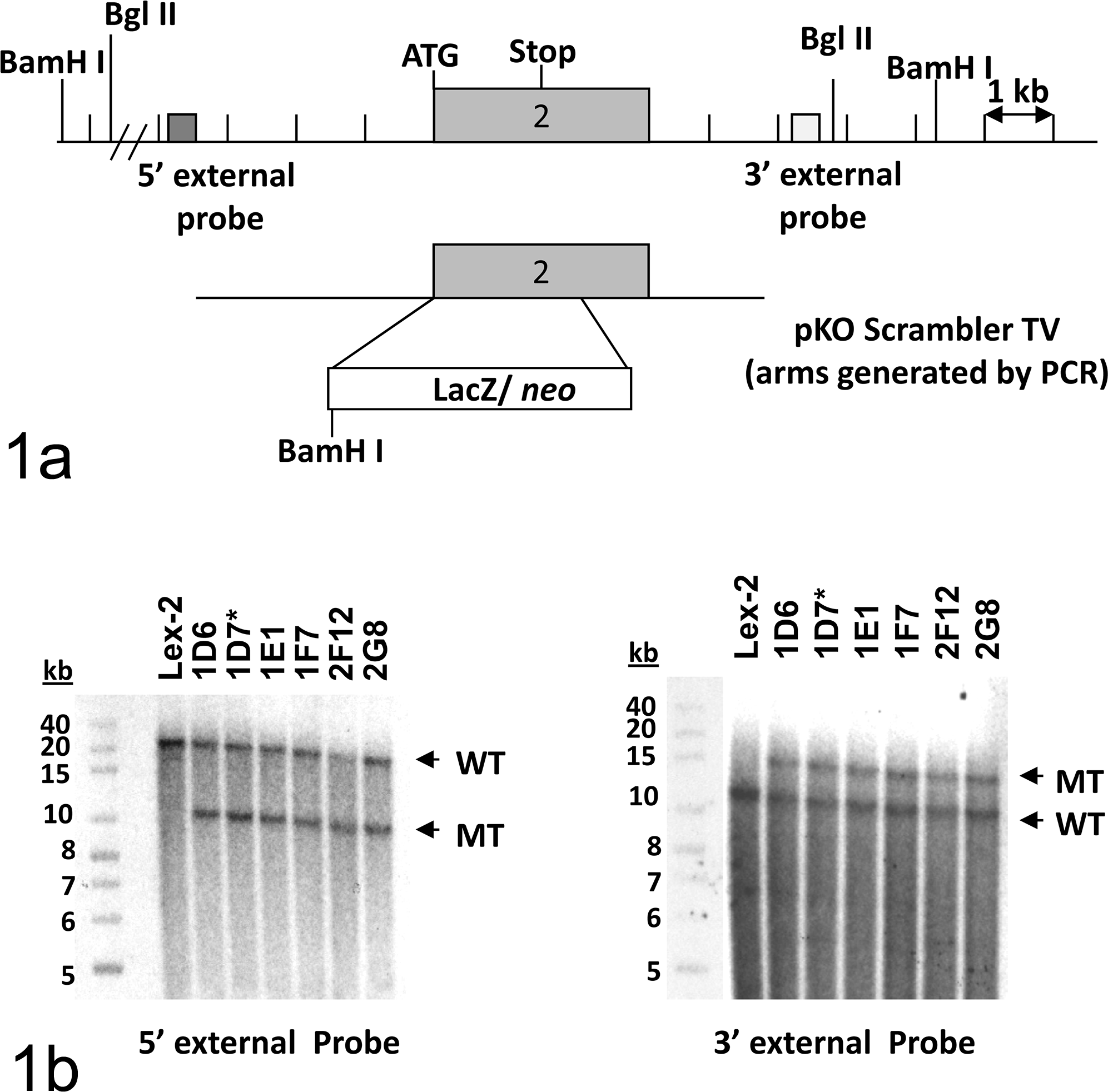
Targeting strategy and Southern blot confirmation of the Grem2 gene mutation. (a) Homologous recombination between the pKO Scrambler targeting vector (TV) and the Grem2 gene resulted in the replacement of the coding portion of exon 2 with the selection cassette. Restriction enzyme recognition sites are indicated by BamH I and Bgl II. ATG represents the translation start site. (b) Southern hybridization indicating Grem2 gene targeting in the embryonic stem cell clones. Clones 1D6, 1D7, 1E1, 1F7, 2F12, and 2G8 were analyzed by Southern blot analysis through BamH I–digested genomic DNA with a 5′ external probe and Bgl II–digested genomic DNA with a 3′ external probe. Two targeted clones were selected for blastocyst injections. An asterisk (*) indicates the clone that was transmitted through the germ line. WT indicates the band from the wild-type allele. MT indicates the band from the mutant allele. Band sizes are indicated in kilobases (kb) using a labeled size marker. Lex2 represents untransfected embryonic stem cell DNA.
Mouse Husbandry and Testing
Mice were housed in microisolator cages within a barrier facility at 24°C on a fixed 12-hour-light and 12-hour-dark cycle and provided acidified water and Purina rodent chow (No. 5001, Purina, St Louis, MO) ad libitum. All procedures involving use of live mice were conducted in conformance with Lexicon’s Institutional Animal Care and Use Committee guidelines, which are in compliance with the state and federal laws and the standards outlined in the National Research Council’s Guide for the Care and Use of Laboratory Animals. Quarterly sentinel surveillance showed no evidence of pathogenic rodent viruses and Mycoplasma or Helicobacter species in the Lexicon Pharmaceutical source colonies.
Lexicon Pharmaceutical’s phenotyping program included assays to evaluate a range of behavioral and physiologic parameters intended to identify novel drug targets. In this program, screening assays were normally completed in mice ranging in age from 8 to 16 weeks, and knockout lines lacking potentially useful phenotypes were generally terminated after the initial screening process was completed. Clinical chemistry values, including serum alkaline phosphatase, calcium, and phosphorus, were measured with COBAS Integra 400 Analyzer (Roche Diagnostics, Indianapolis, IN). MicroCT scans were obtained with a Scanco Medical μCT40 (Brüttisellen, Switzerland) with voxel dimensions of 6 μm (teeth), 16 μm (fifth lumbar vertebra), and 20 μm (femurs). DEXA (dual-energy X-ray absorptiometry) was performed on anesthetized mice (8 Grem2-/- and 4 Grem2+/+ , divided equally between males and females) with a Lunar PIXImus scanner (GE-Lunar, Madison, WI). Statistical comparisons were made with the 2-tail Student t test, based on inspection of group data and the assumption that the group data show a normal distribution.
Histopathology
For histology, a pair of knockout mice (1 male and 1 female) and a pair of age-matched wild-type littermate control mice (2 male and 2 female) were euthanized at each of 3 time points (11, 14, and 20 weeks of age) and fixed by cardiac perfusionwith 10% neutral buffered formalin. Tissues were collected and immersed in 10% neutral buffered formalin for an additional 48 hours except for eyes, which were removed and fixed by immersion in Davidson’s fixative (Poly Scientific, New York, NY) overnight at room temperature. After overnight fixation, bones and heads were transferred into a decalcifying/fixation solution consisting of a mixture of formic acid and formaldehyde (Cal-Rite, Thermo Fisher Scientific, Waltham, MA), which allows simultaneous decalcification and fixation. All tissues were embedded in paraffin, sectioned at 4 μm, mounted on positively charged glass slides (Superfrost Plus, Thermo Fisher Scientific), and stained with hematoxylin and eosin for standard histopathologic examination.
Results
Grem2-/- mice showed no notable phenotypes in a comprehensive high-throughput screen examining a range of immunologic, metabolic, ophthalmic, and behavioral parameters. DEXA bone mineral density values were elevated in knockout mice for body (4%, P = .03), spine (13%, P = .05), and femur (12%, P < .001). However, no changes were observed in microCT scans (4 knockout and 2 wild-type males) of LV5 vertebral body trabecular bone or midshaft femur cortical bone. No skeletal abnormalities were observed during histopathology examination. Given the small number of mice examined in this screen, studies on additional mice will be required to determine if disruption of Grem2 reliably produces a skeletal phenotype.
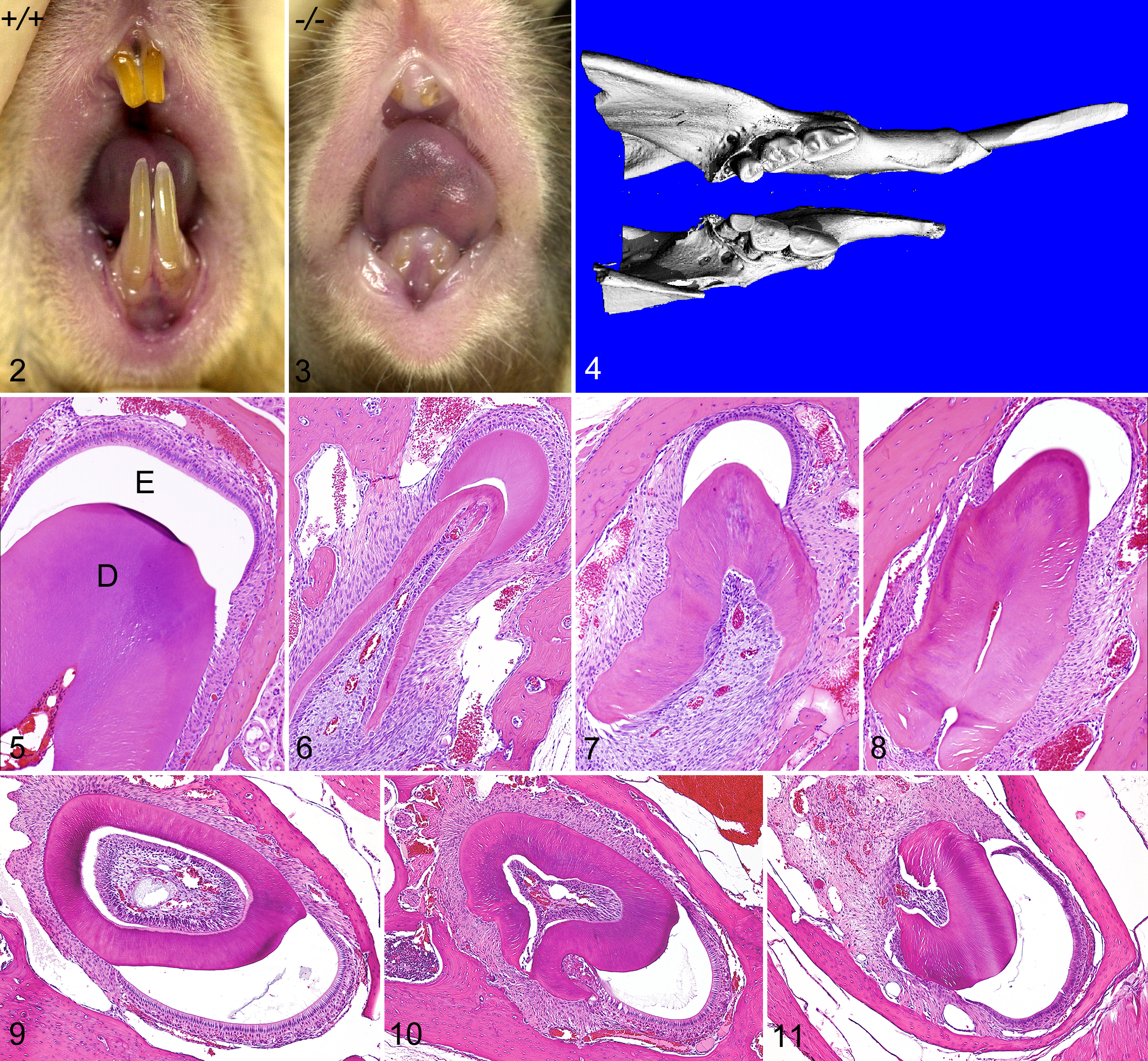
On gross examination, both upper and lower incisor teeth of all Grem2-/- mice had markedly reduced breadth and depth, giving them a needle-like appearance. The upper incisors appeared to be more severely affected than the lower, but both sets were prone to excessive wear and breakage. In contrast to the long incisors seen in normal mice (Fig. 2), both pairs of incisors were worn down to the gum line in most Grem2-/- mice by 20 weeks of age (Fig. 3). MicroCT scans revealed that the length and diameter of the mandible was reduced in proportion to the reduced incisor size; they also suggested that cusp development in molar teeth was reduced or that cusps were effaced by accelerated wear (Fig. 4). In histologic sections, normal maxillary and mandibular incisors have a chisel-like appearance, characterized by broad, flat enamel caps squared on both sides covering the top edge of an elongated thick dentin layer that completely encircles the dental pulp (Fig. 5). In contrast, the maxillary incisors in Grem2-/- mice were much smaller than normal, and their dentin layer was narrow and irregular in appearance and did not completely encircle the dental pulp. The irregular dentin layer in maxillary incisors was open posteriorly (was not circumpulpal as in normal teeth) at all stages of tooth maturation and covered anteriorly by a crescent-shaped enamel cap (Figs. 6–8). Although the severity of malformations and the reduction in size of mandibular incisors in Grem2-/- mice in comparison to normal (Fig. 9) were not as severe as in maxillary incisors, the defects in dentin (malformed and incomplete surrounding of pulp) and enamel (small narrow caps) were similar (Figs. 10, 11).
Discussion
Although the shape of teeth is highly variable, the fundamental steps of tooth morphogenesis are similar and involve the same pathways during tooth formation in all vertebrates. 16 The TGFβ, fibroblast growth factor, sonic hedgehog, tumor necrosis factor, and Wnt signaling pathways are all required repeatedly during tooth development, and disruption of any of these pathways can result in developmental arrest of teeth. These different pathways are integrated at several stages of tooth development, and the overall regulatory network is highly conserved in evolution. 6,15,16 The continuous growth of open-rooted incisors of rodents permits the study of the complex intercellular interactions involved in the morphogenesis of teeth and the formation of dentin and enamel. As a result, dental findings in mice have proven valuable in elucidating processes involved in tooth development in humans. 28,36
Tooth development involves the processes of morphogenesis, epithelial histogenesis, and cell differentiation (reviewed by Lesot and Brook 23 ). In the mouse, localized thickenings of the oral epithelium (dental lamina) are first visible at around E11.5 (embryonic day 11.5), and these areas express key signaling molecules such as Shh that promote cell proliferation at the sites of tooth development. 8 The proliferating oral epithelium soon invaginates into the underlying neural crest–derived mesenchyme and forms a clearly defined tooth bud by E13.5. The tooth bud is surrounded by mesenchyme that expresses signaling molecules and transcription factors such as Bmp4, Msx1, and Pax9. By the E14.5 cap stage, signals from the dental mesenchyme have induced the formation of the enamel knot at the tip of the tooth bud. The enamel knot is a specialized nonproliferative structure that expresses numerous signaling molecules, including Shh, Fgf4, Bmp4, and Wnt10b. 4,14
Although all cells in Grem2-/- mice were deficient for GREMLIN 2, detectable phenotypic changes were restricted to the teeth and possibly bones. The small deformed incisors indicate that this protein is required for normal morphogenesis of teeth, while the possible minor elevation of bone mineral density suggests a milder effect on bone structure. The processes involved in control of tooth development and morphogenesis are complex and involve the combined activities of several different BMPs, BMP receptors, and BMP antagonists. Moreover, tooth development involves the reiterative activation and patterning of the nonproliferative epithelial enamel knots. 14,15 The failure of circumpulpal dentin formation in Grem2-/- mice is suggestive of a severe defect at early stages of incisor development.
The primary enamel knot determines the size and overall shape of the tooth, while the generation of cusps is believed to be driven by the formation of additional secondary enamel knots. Defects in the size or shape of the primary enamel knot can disrupt the arrangement of the secondary enamel knots in molars and lead to cusp defects. Expression of the genes Edar and Edaradd during tooth development has been linked to the formation of primary enamel knots. For example, the enamel knot is abnormal, and molar tooth cusps are rounded and reduced in height in tabby, downless, and crinkled mice. 22,29,31,35,39 These mouse strains all have mutations in proteins involved in the same pathway: tabby mice have a mutation in ectodysplasin (Eda); downless mice have mutations in the receptor for EDA (Edar); and crinkled mice have mutations in the EDAR adaptor protein known as EDARADD. 11,19 However, the incisors in these 3 mutant lines appear largely unaffected. Humans with mutations in the syntenic genes EDA, EDAR, and EDARADD develop hypohidrotic ectodermal dysplasia and have peglike molar teeth with reduced cusps. 11,19,25 The enamel knot–derived signaling molecules induce the adjoining dental epithelial and mesenchymal tissues to differentiate and pattern the shape of the tooth. 14 After the formation of the cusp pattern, the tooth grows to its final size, and mesenchymal odontoblasts and epithelial ameloblasts differentiate at the epithelial-mesenchymal interface to form dentin and enamel, respectively. 16
Our discovery that GREMLIN 2 is involved in the morphogenesis of incisor teeth is not entirely surprising, since GREMLIN 2/PRDC is a potent antagonist of both BMP2 and BMP4 37 and both these BMPs have been localized to structures in developing teeth. 26 The relationship between GREMLIN 2 and BMP4 is particularly intriguing because Wnts and BMP4 appear to be the key mediators of odontogenic epithelial-mesenchymal interactions, 27 and Wnt/beta-catenin signaling has been shown to regulate the expression of GREMLIN 2/PRDC. 13 The importance of BMP4 in tooth development is further documented by the arrested development of teeth before the induction of primary enamel knots in mice lacking Bmp4 expression. 5 More recently, BMP4 signaling was shown to suppress the inhibitors of tooth development in the tooth mesenchyme and to synergize with MSX1 in inducing tooth morphogenesis and formation. 17 An additional example of the roles that BMP4 plays in the morphogenesis of teeth is provided by ectodin-deficient mice, which develop enlarged enamel knots, crown abnormalities, and extra teeth. 18,21 Ectodin is expressed in developing ectodermal organs, including teeth and hair follicles, and like Gremlin 2, ectodin is a member of the Dan/Cerberus family of secreted BMP/Wnt antagonists that antagonizes BMP4. 1,21 Our findings indicate that Grem2 can be added to the growing list of genes that affect tooth development in mice. 2,10
Footnotes
Acknowledgements
For their invaluable necropsy and histology support, we wish to thank Kathy Henze and June Wingert of Lexicon Pharmaceuticals.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by and all authors employed by Lexicon Pharmaceuticals Inc., The Woodlands, TX 77381.
