Abstract
Technical improvements in electron microscopy, both instrumental and preparative, permit increasingly accurate analyses. Digital images for transmission electron microscopy (TEM) can be processed by software programs that automate tasks and create custom tools that allow for image enhancement for brightness, contrast and coloration; for creation of rectangular, ellipsoidal or irregular area selections; and for measurement of mean area and standard deviation. Sample preparation remains a source of error since organelles and spatial arrangements of macromolecules rapidly change after anoxia. Guidelines for maintaining consistency in preparation, examination and interpretation are presented for different electron microscopy (EM) modalities.
Transmission Electron Microscopy
Transmission electron microscopy (TEM) was the major investigation tool in cellular biology from the 1960s to the 1980s. Ultrathin sections of resin-embedded tissues added unique views of the cell and established our modern concepts of organelle morphology. Thereafter, for several decades, the use of TEM declined and with it the number of TEM laboratories and electron microscopists. Pathologists without a working knowledge of TEM failed to include it in their diagnostic and investigative protocols. In publications, it was commonplace to find electron micrographs that had serious artifacts or had been printed at such a low magnification that the reader could not discern in it any component described in the figure legend. Even worse, teachers and textbooks began to use colored cartoons to depict organelle ultrastructure. The end result: an even further detachment from the reality and wonder of cell pathology as revealed by TEM. Despite this, there persisted in veterinary pathology a core of well-trained electron microscopists who continued to produce contributions to our understanding of disease. In the past decade, the wealth of data from new light microscopic technologies has been driving renewed interest in TEM and for new ways to view cell ultrastructure in 3 dimensions at high resolution.
Today, when specimens are properly fixed and embedded and using improvements in existing techniques, both instrumental and preparative, classic TEM continues to reveal striking details of cellular organelles and their alterations in disease. New techniques allow more reproducible data for analysis of cellular structure and function. 15 Digital images can be processed by an amazing choice of software programs that automate tasks and create custom tools. Image enhancement for brightness, contrast, and coloration; for creation of rectangular, ellipsoidal, or irregular area selections; for measurement of mean area and standard deviation; and for automatic creation of changes using a wand tool all improve analyses of data. 3 Some of these programs (such as ImageJ, an image-processing and analysis program written in Java from the National Institutes of Health, Bethesda, MD) are available online without charge. In the discussion of TEM techniques that follows, bolded terms are candidates for online searches in www.pubmed.gov and other search sites.
Despite remarkable new techniques, the greatest source of error in thin sections for TEM still lies in sample preparation. In conventional fixation and embedment in epoxy resins at ambient temperature, organelle structure and spatial arrangements of macromolecular complexes rapidly change after tissue sampling. For example, the cytoskeleton, Golgi complex, and endomembrane system are remodeled within fractions of a second after tissue anoxia. This requires attention to sample collection, especially times of sampling, trimming, and temperature of fixatives and buffers for fixation. With precise preparation, structures bearing nucleic acids and proteins are preserved.
Although the resolution power of electron microscopes is on the atomic scale, artifacts in cell and tissue specimens introduced by sampling, aldehyde fixatives, dehydration, staining, and section thickness reduces it to nanometers. Recommended guidelines for maintaining consistency in glutaraldehyde immersion fixation are as follows:
Fixation
In the standard preparatory protocol, tissue samples are fixed in 2.5% glutaraldehyde at pH 7.2 (at ambient temperature) for 2 to 3 hours and postfixed in 1% OsO4 in 0.1 M cacodylate buffer for 60 minutes (Fig. 1). Fixation with glutaraldehyde alone or osmium tetroxide (OsO4) alone causes artifacts that are substantially avoided when tissue is doubly fixed. Glutaraldehyde fixes proteins well but is harsh on membranes. The reverse is true for OsO4 which enhances membrane contrast but damages proteins; that is, membrane contrast is enhanced by OsO4 fixation.
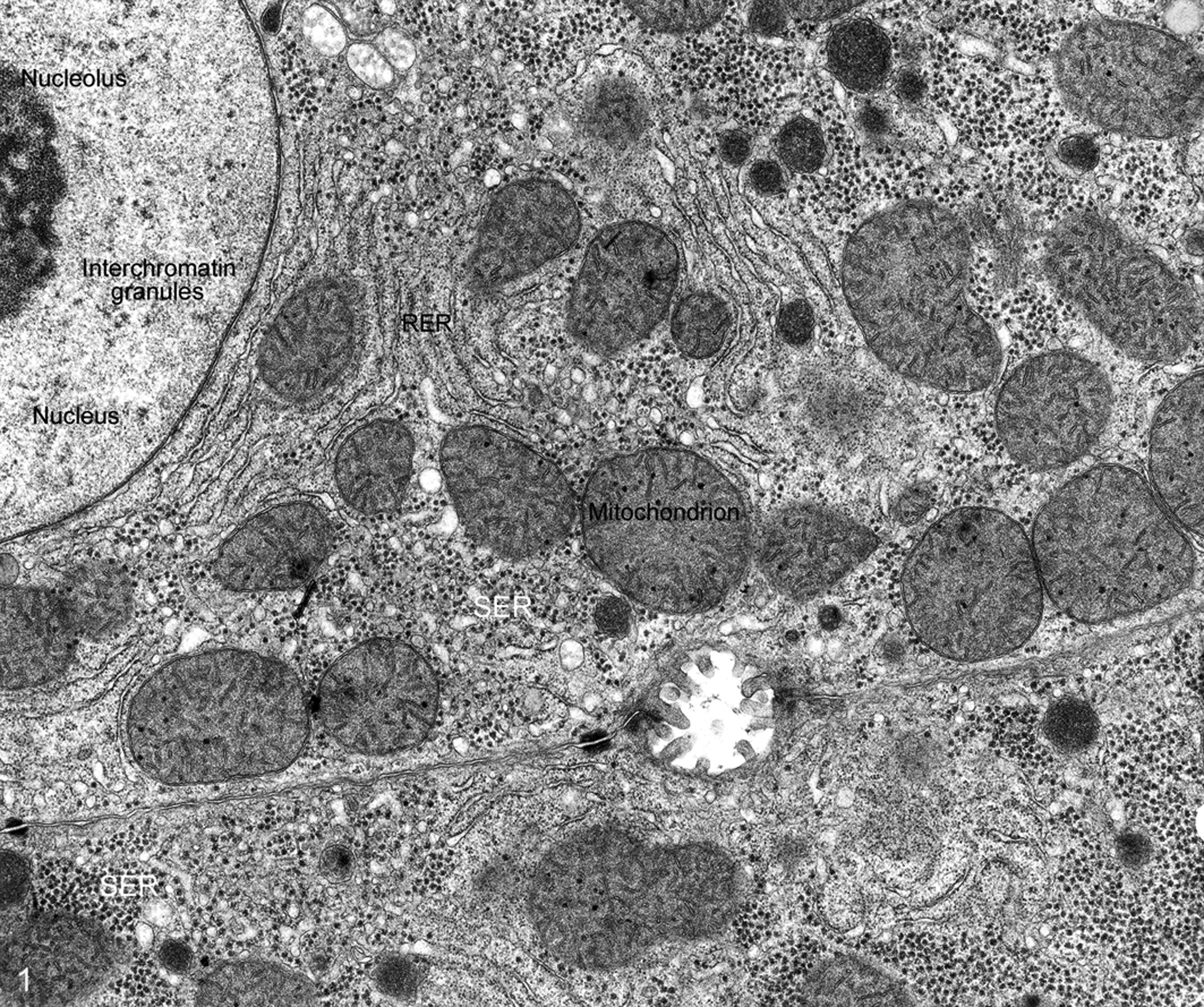
Paraformaldehyde (which produces effects similar to formalin) is used to preserve antigenicity of some proteins in immunolabeling. It is used in combination with glutaraldehyde (referred to as Karnovsky’s solution, which varies from 0.5% to 2% paraformaldehyde with 2% glutaraldehyde).
When fixative takes 1 minute to reach the cell, organelles suffer from autolysis. Even when fixative penetrates in less than 1 second, cross-linking reactions are selective (eg, glutaraldehyde will only react with certain amino acid residues on proteins and will not effectively cross-link nucleic acids and carbohydrate molecules). The result is extraction of unlinked molecules from the cell during rinsing and dehydration. 44
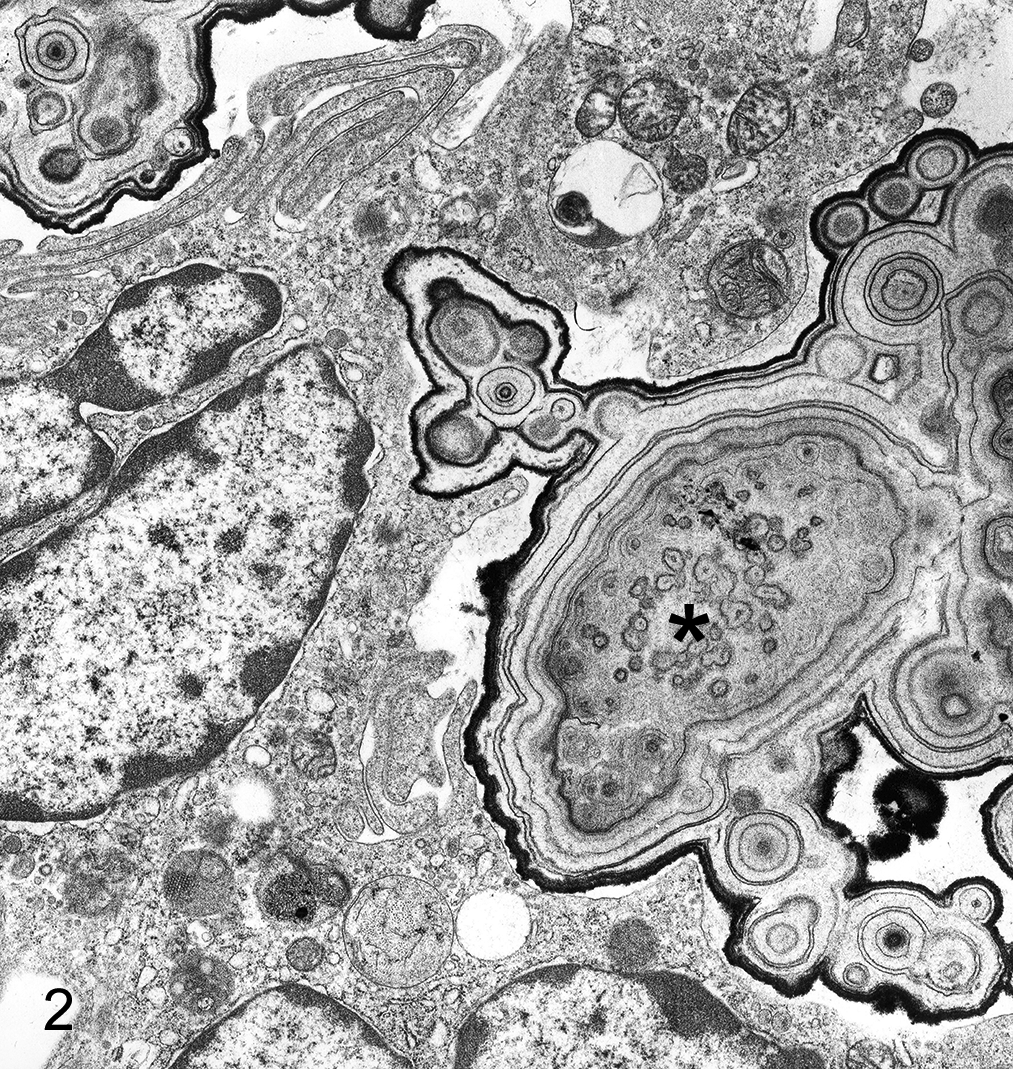
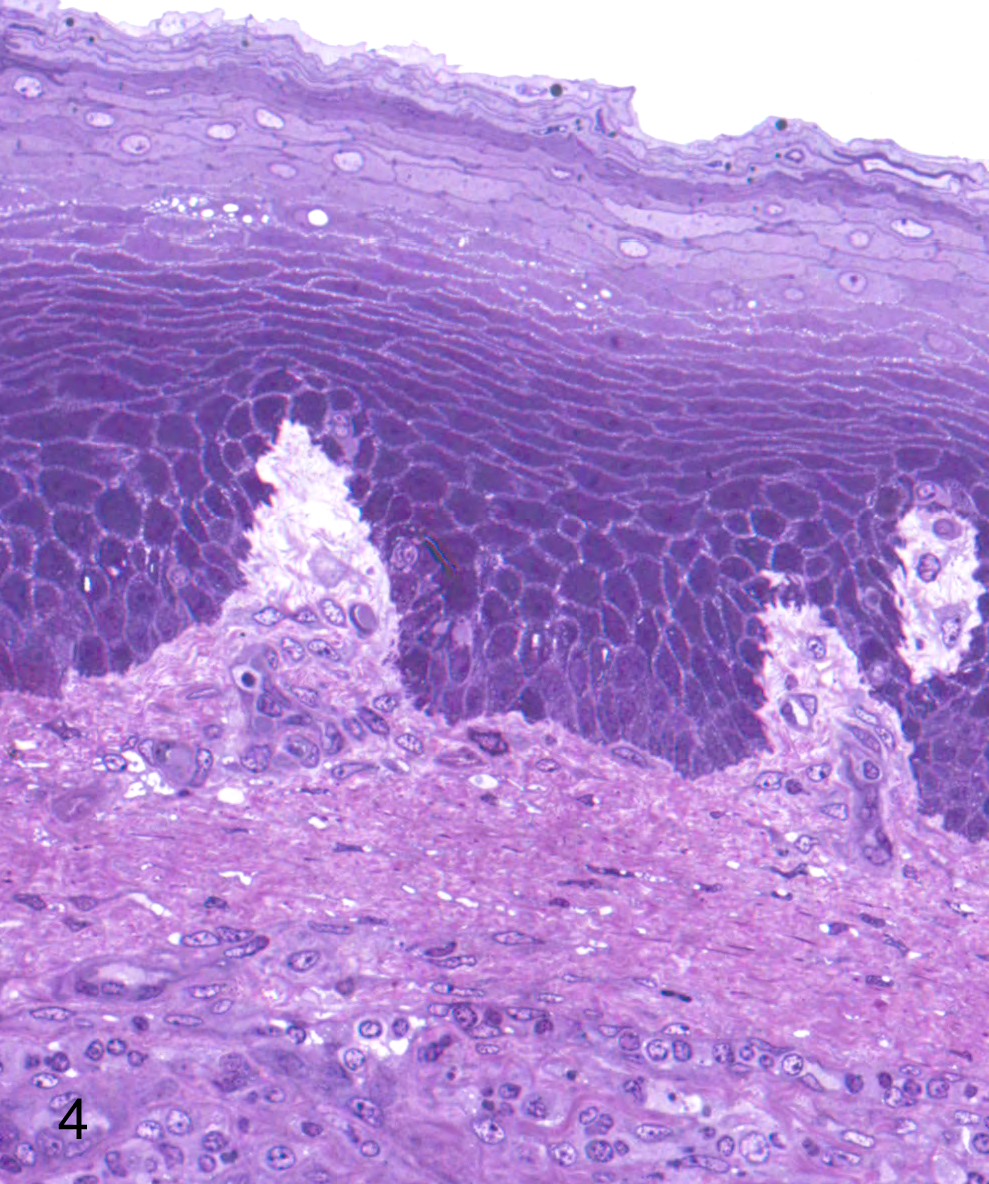
Cadodylate and phosphate buffers predominate in fixatives for electron microscopy, but be aware that high concentrations of phosphate damage mitochondria. In cases where phosphate buffers are undesirable, organic buffers such as 1,4 piperazine bis(2-ethanolsulfonic acid) (PIPES) might be considered since it has fewer detrimental effects on cells (Table 1). It is especially important that staining and buffers be appropriate in fixation of calcified soft tissues since redistribution of calcium salts during fixation can lead to misinterpretation. There is some decalcification with OsO4 fixation, and increasing the time of immersion may give striking results in the differential degrees of calcification in lesions of interstitial tissues (Fig. 2).
Preparatory Techniques in Transmission Electron Microscopy.
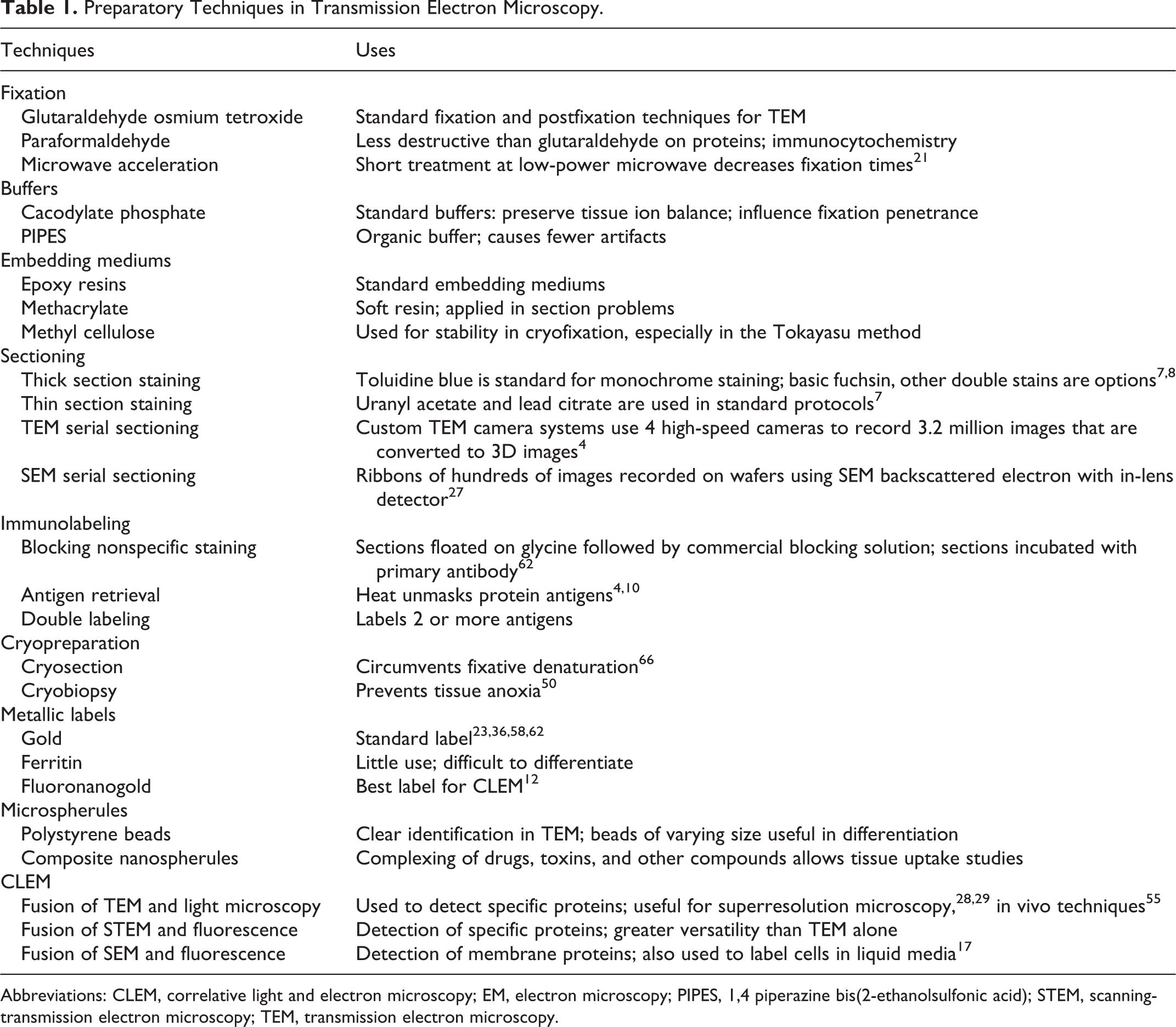
Abbreviations: CLEM, correlative light and electron microscopy; EM, electron microscopy; PIPES, 1,4 piperazine bis(2-ethanolsulfonic acid); STEM, scanning-transmission electron microscopy; TEM, transmission electron microscopy.
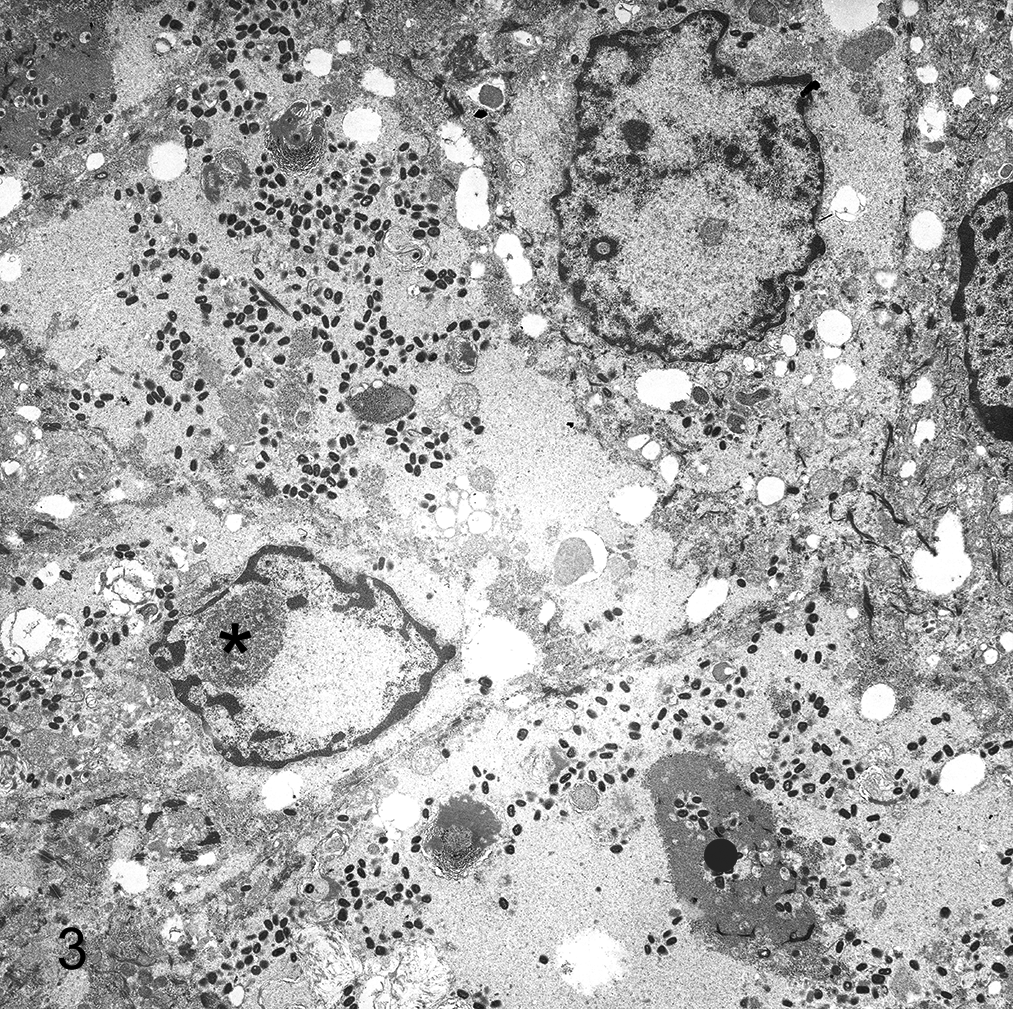
Embedding
For standard embedment for TEM, samples are dehydrated through an ethanol series; transferred to several changes of a transitional solvent, propylene oxide; and embedded in epoxy resin. The epoxide resin is intolerant of water and will fail to polymerize properly if the tissue is not totally dehydrated; this results in rubbery blocks that are impossible to cut and tissues that are retrievable to correct the issue. Furthermore, water remaining in the specimen during dehydration will not allow embedding media to completely infiltrate the specimen.
It is critical that embedding mixture ingredients be thoroughly mixed prior to use. Improper mixing of ingredients will result in tissue blocks not hardening sufficiently. Wrong proportions or insufficient time for mixing, as well as defective or insufficient accelerators, may prevent proper infiltration and polymerization. Excessive vigor in mixing for this introduces air bubbles into the resin, which makes cutting difficult and will make samples unstable in the electron beam. 6
Sectioning
Preparatory cutting of
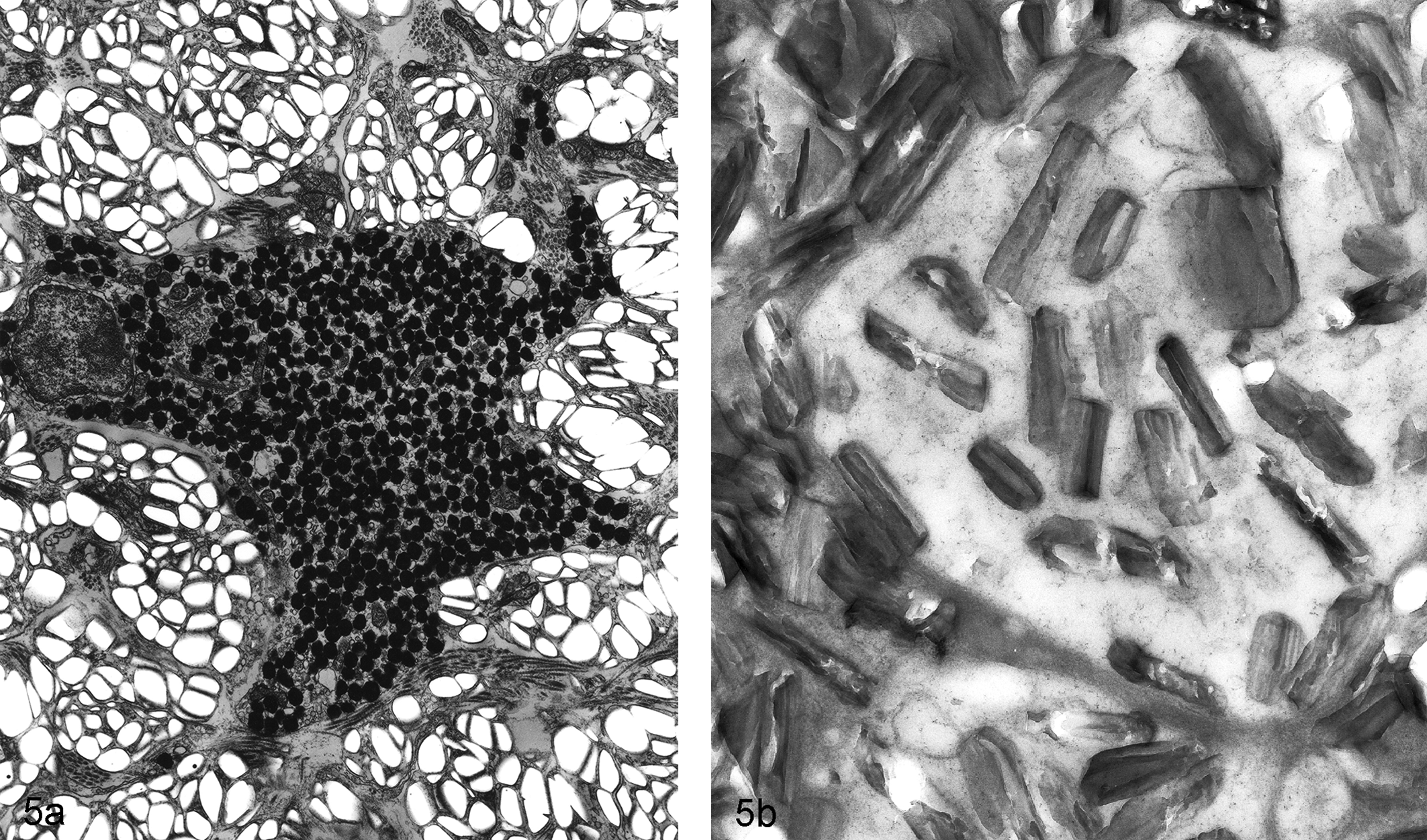
Sectioning of crystals, foreign bodies, and mineralized lesions may require sections that are greater than 70 nm. When these lesions are destroyed by the electron beam after multiple attempts, a resin section overlay procedure may be useful (Fig. 5). New digital technology provides for a less intense beam so destruction of thin section is rarely an issue.
Staining of Ultrathin Sections
Grids bearing thin sections are processed by positive staining with heavy metals to increase tissue contrast. Staining en bloc (before embedding) can also be done to increase contrast. The most commonly used stains for TEM are uranyl acetate and lead citrate. Uranyl acetate can be used for pre- or postembedding staining. It is typically a 2% aqueous solution, although some resins need an alcoholic (either ethanol or methanol) uranyl acetate to penetrate the resin. The most commonly used lead citrate stain is Reynolds’s lead citrate. Although time-consuming to prepare, Reynolds’s lead is stable as long as it is protected from carbon dioxide and light. Care must be taken during storage and staining since lead citrate will react with atmospheric carbon dioxide to form a fine precipitate of lead carbonate. 7
Focusing and Photographing the Specimen in the TEM
Careful focusing is one of the most important functions required for successful TEM use. To obtain the best photograph during examination of the stained ultrathin section, one must improve contrast, a task that requires the objectives and condensers of the electron microscope to be in alignment. The TEM uses a high-voltage electron beam emitted by an electron gun to create an image. The beam is accelerated by an anode, focused by electrostatic and electromagnetic lenses, and carried through the specimen, which is transparent to electrons or scatters them out of the beam.
The basic rule of focusing is that the greater the magnification, the more accurate the focusing must be and the closer it should be to the true or near focus setting. True or near focus is where the specimen is exactly in the conjugate plane to the image plane of the objective lens. Optimum results should be obtained from a focused micrograph, but a certain degree of underfocusing is and can be generally favored. Since focusing of the image is affected by variations in the objective lens current, it is best to under- and overfocus to ensure that features in the specimen are genuine.
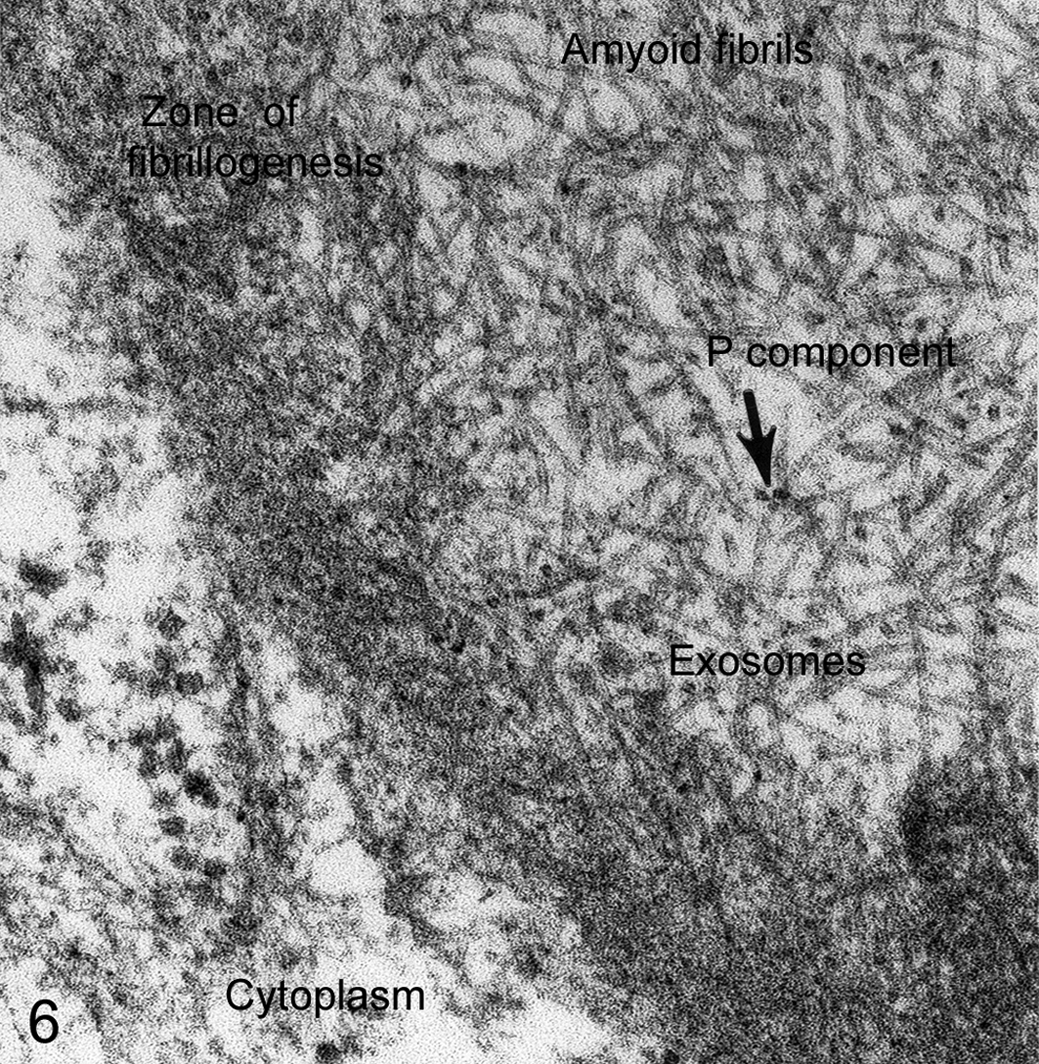
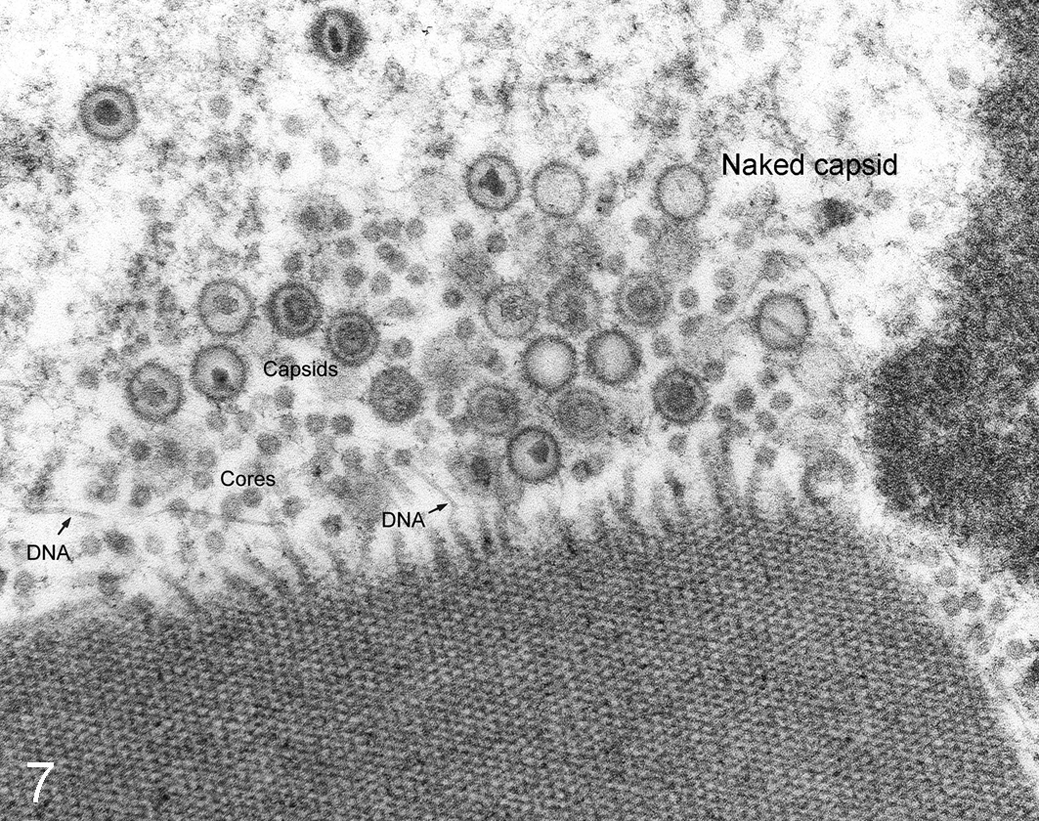
In producing electron micrographs, the goal is always the generation of valid data, not of electron micrographs of a particular quality. In publishing, electron microscopists face hurdles by reviewers with TEM experience but little understanding of the nature of electron beam focus or deflection and the depth of focus within the ultrathin section. Never “balance the light” to create a uniform background in the micrograph; that manipulation almost always destroys data, especially of tiny vesicles and protein fragments that provide important data for the interpretation of pathologic tissues (Figs. 6, 7).
Cryo-electron Microscopy
Ischemic and anoxic artifacts are inevitable due to immersion fixation. The introduction of
Negative Staining in TEM
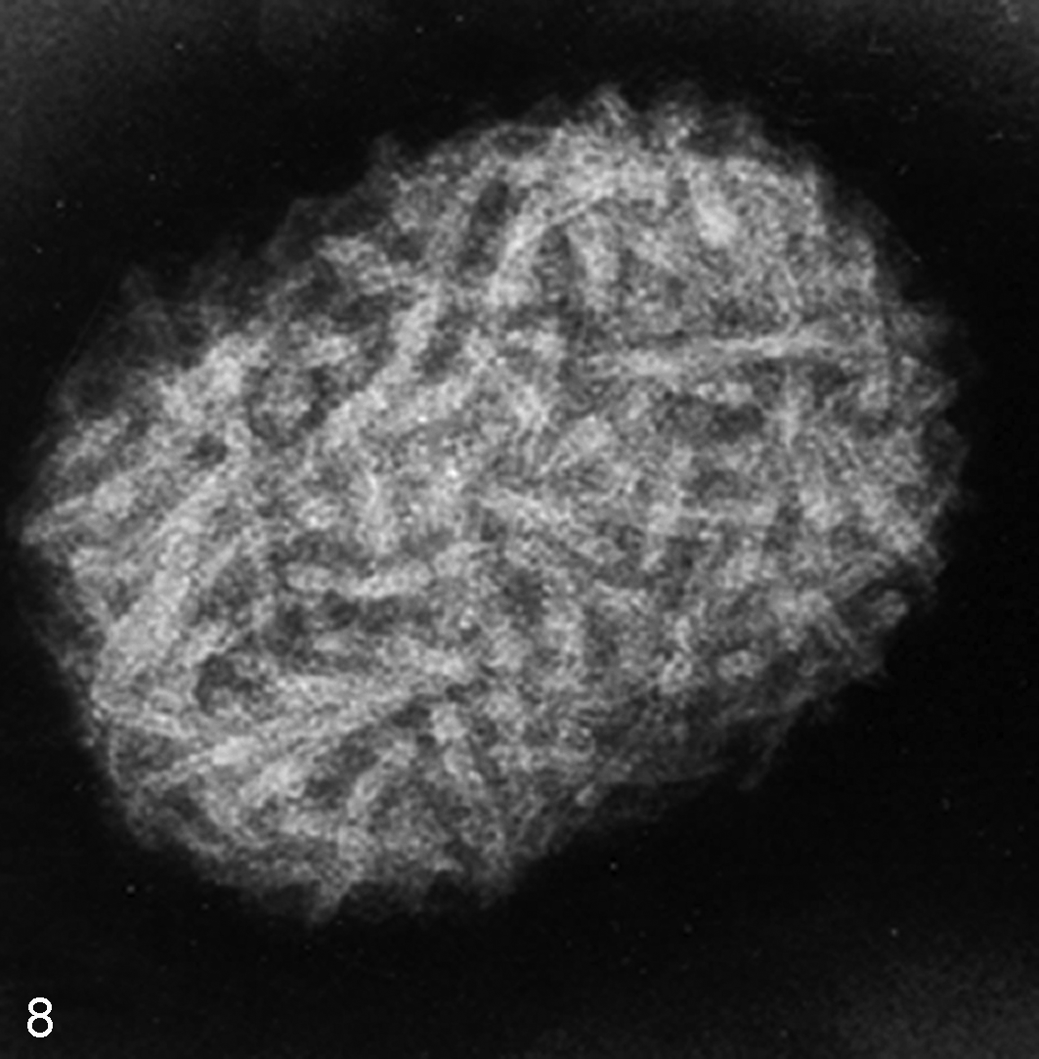
Scanning Electron Microscopy
Scanning electron microscopy (SEM) is a surface imaging technique that provides high depth of field with a 3-dimensional appearance (Fig. 9). SEM produces images by probing the specimen with a focused electron beam that is scanned across the surface; the beam in an SEM does not carry an image. In contrast, TEM uses a high-voltage electron beam emitted by an electron gun to create an image.

Adequate sample preparation used in modern SEMs can provide resolution comparable to that achieved with TEMs—down to ∼3 nm for biological material. Standard SEM equipment uses an electron beam (detected in a raster scan pattern) that interacts with electrons on the sample surface. The beam’s position is combined with the detected signal to produce an image. For nonbiological materials, SEMs can achieve resolution better than 1 nm. The type of signal produced by an SEM can also include back-scatter electrons, characteristic x-rays, and cathodoluminescence. 12
Special Investigative Techniques
EM of Nanoemulsions
Electron microscopy of
Microspherules
Electron-Dense Markers for TEM
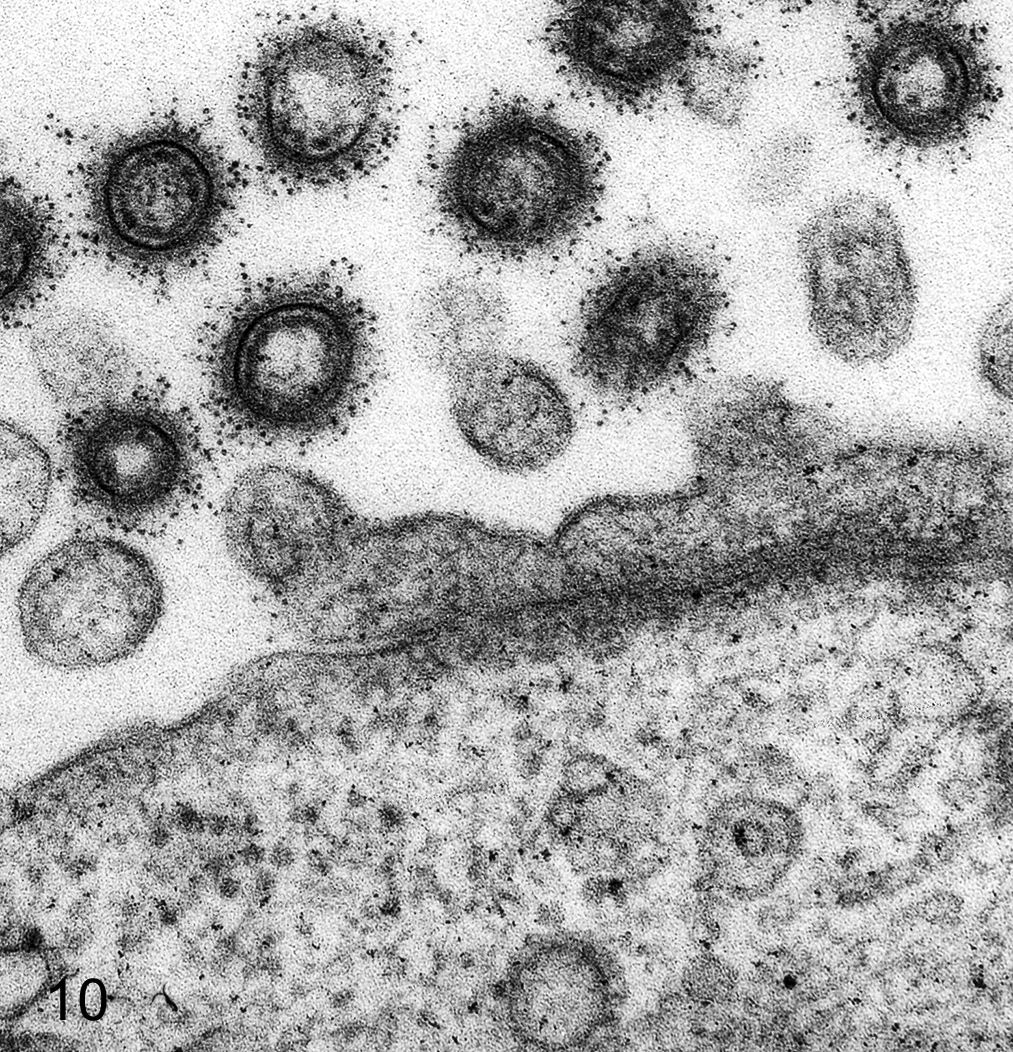
Ferritin, colloidal gold, and other heavy metal–based nanoparticles with sufficient contrast for high-resolution TEM have been used for in vivo, in ovo, and in vitro investigations to determine particulate trafficking among organelles. If particles can be counted, their spatial distributions can be subjected to statistical analysis. 40,42,72 One approach is the detection and section volume localization for particles injected into the animal prior to sampling (ie, particles that are within the cell prior to fixation and embedding). Various sizes of electron-dense particles of colloidal gold are used to study particle transport, especially in blood-tissue, air-tissue, and urine-tissue pathways. For example, when instilled in the trachea of mice, 20-nm diameter colloidal gold particles are found in pulmonary alveolar macrophages within 15 minutes and slightly later are internalized by alveolar epithelium using caveolae.
Immunoelectron Microscopy
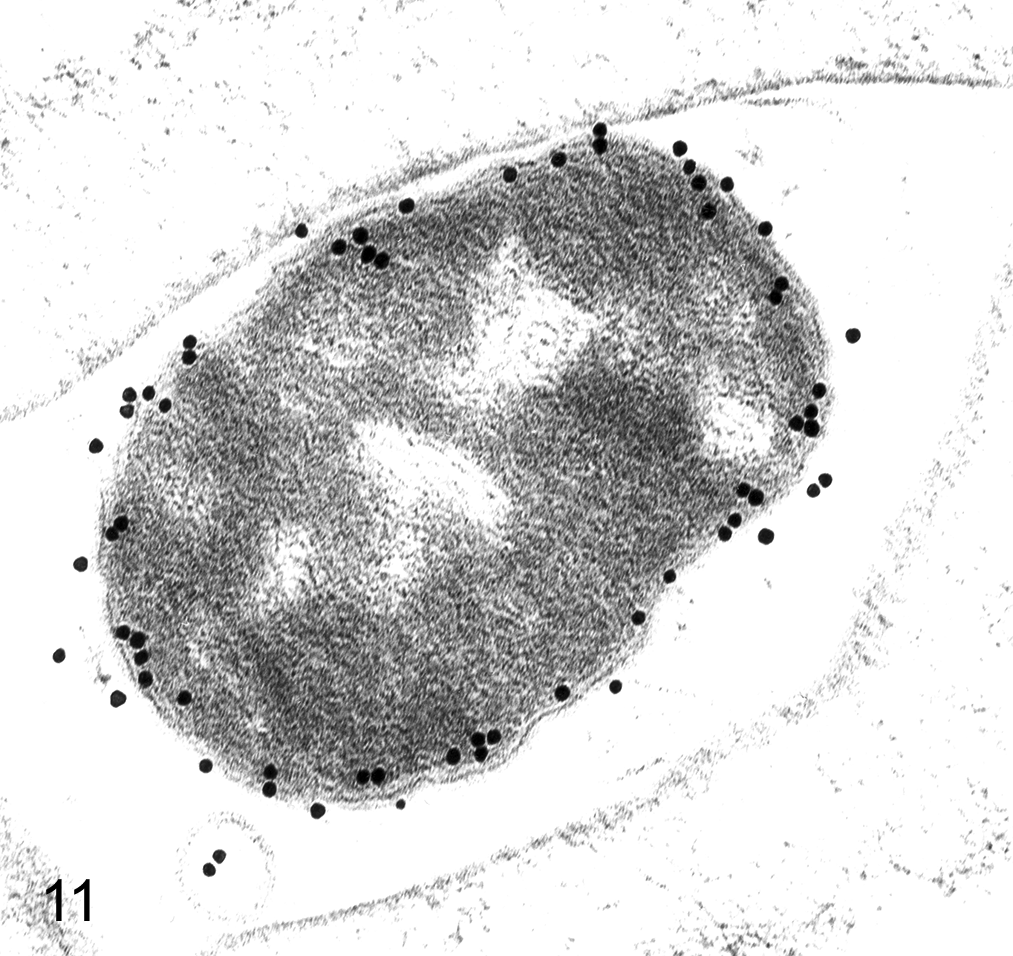
Immunoelectron microscopy using heavy metal labels for staining of specific proteins expanded the use of morphology in cellular pathology, and the quantitative analysis of metallic labels in intracellular compartments added precision. Colloidal gold of varying sizes is superior. Ferritin, although useful, suffers from uncontrolled particle size and a confusing similarity to natural ferritin (Fig. 10). Particulate gold labeling on ultrathin sections for antigen localization at the TEM level has had widespread use for 2 decades; most studies have used simple, rapid, and unbiased methods for assessing immunogold labels distributed over different cellular compartments (Fig. 11). 36,37 Sampling approaches have been developed for stereology in which a regular array of EM fields or linear scans is positioned randomly on labeled sections. Quantitative methods allow estimation of both density and distribution of gold labels in organelles. Spatial mapping, especially important in polarized cells, can be done using a stereologic approach referred to as the rotator. 41
Morphometry and Stereology in TEM
Precise and accurate quantification of cellular changes in electron micrographs has traditionally used morphometric tools to measure numbers of organelles as well as the surfaces, lengths, and volumes. Development of new models for TEM stereology and computational software for data analysis provide exciting modalities in scientific investigation. 2 Electron microscopic tomography, freeze-fracture electron microscopy, and immunolabeling techniques have added new dimensions to TEM. Analytical techniques such as electron energy loss spectroscopy or energy-dispersive x-ray spectroscopy, as well as atomic force microscopy, are additional assets to determine elemental composition of materials. Due to advances in crystallography and magnetic resonance imaging (MRI), almost any protein can be modeled at atomic resolution, yet TEM is still required to place the protein in context of the cell.
Electron Microscopic Tomography
Computational steps are necessary for interpretation—such as noise reduction, segmentation, and analysis of subvolumes. Computation demands are large in some systems, and high-performance computing (HPC) techniques are useful. 19 EMT software designed to develop cellular representations provides the ability to see the ultrastructural architecture of organelles and fibers (as well as viruses, bacteria, and other microbes 50 ), as well as the clarity of structural relationships. EMT constructs of attachment, entry, and uncoating of herpes viruses have documented the process of viral infection at neuronal synapses. 39
EMT studies using
Correlative Light and Electron Microscopy
Correlative light and electron microscopy (CLEM) is a fusion of imaging techniques—the combination of methods of different microscopes that use photons or electrons for imaging. 47,63,65 Modern CLEM techniques focus on the systematized matching of cellular areas between microscope modalities. CLEM means imaging the same structure with 2 or more imaging modalities, typically combining fluorescence and TEM using cells in culture. High-resolution light microscopic images and relocation of the same object at the ultrastructural level using a TEM demand improved ultramicrotomy techniques. 31 In most CLEM studies, a 3-dimensional construct of a macromolecular assembly is computed from 2-dimensional TEM images. Single-particle analysis (SPA) requires a large number of images or randomly oriented individual complexes and correction of the control transfer function (CTF) of the microscope. 16 CLEM is technically challenging and time-consuming, and it is designed for and applied to special approaches to a specific problem.
CLEM expanded with the advent of new
The SEM is widely used in CLEM studies. One dedicated instrument, the JOEL ClairScope (Tokyo, Japan), is a correlative scanning electron and optical microscope based on an inverted SEM column that allows images of wet samples to be obtained in ambient conditions in a culture dish via a silicon nitride field under the base. A standard inverted optical microscope above the dish holder can take reflected light and epifluorescence images of the same sample. 46
Freeze-Fracture Electron Microscopy
The cryofixation replica technique provides a unique view of biological membranes. Plasma membranes are rapidly frozen to the temperature of liquid nitrogen (rapid freezing prevents formation of ice crystals). In this state, the membrane, when given a gentle tap, falls apart along the plane of least resistance, the central plane of the lipid bilayer (ie, the membrane comes apart between the lipid tails). This exposes the 2 half-membranes, each containing half the bilayer. One side of each half-membrane is the true membrane surface; the other is a view of the interior of the membrane, termed the fracture face. The background lipid is smooth, so the large protein molecules stand out as particles or depressions.
Since FFEM provides a planar view of membranes, a major emphasis in its use is to identify integral membrane proteins. In freeze-fracture studies, membrane proteins are seen to be missing or abnormally aggregated in response to several types of injury. In splitting the frozen lipid bilayer membrane into 2 halves, FFEM immobilizes membrane proteins and lipids by the vacuum evaporation of platinum and carbon. After treatment by sodium dodecyl sulfate (SDS) to remove extra-membrane materials, the specimen can be stained using immunogold labeling techniques to provide a 2-dimensional distribution of membrane proteins. 51,64,69
Autoradiography and X-ray Microanalysis
The 2 physical methods to detect and quantify intracellular elements in organelles are
Atomic Force Microscopy
Atomic force microscopy (AFM) imaging is a high precision type of scanning probe microscopy with resolutions in fractions of a nanometer. Introduced for biological use in the 1990s, AFM provides surface detail of molecular aggregates and domains, membrane-associated structures, receptors, coated pits, and intercellular junctions. AFM also offers nano-manipulation possibilities, strengths and elasticity measurements, force interactions, affinity measurements, cell stiffness, and other physical aspects of membranes and cytoskeleton. Protocols for tissue section AFM techniques are available 25 and have been valuable in studies on acute injury (eg, shape measurements for the quantitative analysis of mitochondrial swelling in myocardial injury). In studies of thin endothelial cells of liver, AFM has been used to characterize the filtration of lipid-carrying particles during transport from blood to hepatocyte. 9 In AFM, data are acquired by “feeling” the surface of the section with a mechanical probe—a cantilever with a sharp silicon tip for scanning. Forces between tip and sample deflect the cantilever and are measured using a laser spot reflected from the tip surface of the cantilever into an array of photodiodes. The astonishing precision comes from forces that deflect the cantilever: chemical bonding, van der Waals forces, electrostatic forces, and others.
The Future of Electron Microscopy
The renewed interest for TEM in medical and veterinary pathology arises from several changes over the past decade: (1) requirements that scientific proof of a cellular biochemical change revealed by the new light microscopic techniques ultimately requires evidence of ultrastructural change—the drawing of simple colored cartoons at this level, although instructive, simply doesn’t cut it; (2) improved electron microscopes with instrumentation that includes digitized systems for photography, analytical software programs that solve the problems of sampling, and specimen holders that allow tilting the specimen to a range of angles for unique diffraction angles; and (3) improvements in medical and veterinary education. Bioscientists who shy away from TEM because of the small sampling size, an issue that leads to problems of experimental design, difficulties in interpretation, and defenseless positions in the discussion of results, can now use new technology to prove their point.
Nowadays, a real problem is that electron microscopists are rarely lead scientists in research. Thirty years ago, research project leaders had an intimate knowledge of TEM and actually examined the tissues themselves—the changes that were discovered readily led to new TEM approaches. Now, instead, electron microscopists are asked to contribute after the project design has been completed and to provide data that will support the hypotheses. If the TEM results point in another direction, chances are that the TEM part of the project will be abandoned. Recently, I was asked to consult on an academic TEM study of livers of mice given drugs that disrupt the unfolded protein response. The resulting electron micrographs, despite having striking evidence of a new pathway of dysfunctional lipid metabolism, did not fit with the project’s hypothesis. The TEM part of the study was dropped with no interest in pursuing the astonishing possibilities of what was going on in lipid pathways of treated hepatocytes.
Part of this problem arises from the disturbing move of veterinary education away from critical thinking in basic science over the past decade. Fifty years ago, there was a much-needed push to move clinical material into the basic science curriculum, a push that has continued over the half-century. Today the need is for the reverse—the movement of basic science into the clinical curriculum. Over the long haul, the loss of understanding of abnormal biology is much more important than spending precious educational years teaching future practitioners how to conduct their future marketing and business skills. Changing how students think about cellular and molecular pathology will ultimately lead to thinking about electron microscopy to solve a critical problem.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
