Abstract
The ferret has emerged as an excellent animal model to characterize several physiologic and pathologic conditions. The distribution and characterization of different types of immune system cells were studied in healthy ferret tissues. Eight primary antibodies were tested for immunohistochemistry in formalin-fixed tissues: anti-CD3, anti-CD79α, anti-CD20, anti-HLA-DR, anti-lysozyme, anti-CD163, anti-SWC3, and anti-Mac387. The anti-CD3 antibody labeled T cells mainly in interfollicular and paracortical areas of lymph nodes, cortex and thymic medulla, and periarteriolar lymphoid sheaths in the spleen. The anti-CD79α and anti-CD20 antibodies immunolabeled B cells located in lymphoid follicles at lymph nodes, spleen, and Peyer patches. The CD79α and CD20 antibodies also labeled cells with nonlymphoid morphology in atypical B-cell locations. The anti-HLA-DR antibody labeled macrophages, some populations of B and T lymphocytes, and different populations of dendritic cells in lymph nodes, Peyer patches, spleen, and thymus. The anti-lysozyme antibody immunolabeled macrophages in the liver, lymph nodes, spleen, and thymus. The Mac-387, CD163, and SWC3 antibodies did not show any positive reaction in formalin-fixed or frozen tissues. To elucidate the origin of the uncommon CD79α/CD20 positive cells, a double immunohistochemistry was carried out using the anti-HLA-DR + the anti-CD79α, the anti-HLA-DR + the anti-CD20, and the anti-lysozyme + the anti-CD79α antibodies. Double labeling was mainly observed when the anti-HLA-DR + the anti-CD79α antibodies were combined. The immunohistologic characterization and distribution of these immune system cells in healthy ferret tissues should be of value in future comparative studies of diseases in ferrets.
In recent years, the ferret (Mustela putorius furo) has emerged as a useful animal model to characterize several physiologic and pathologic conditions, such as neuronal migration, 40 reproductive physiology and endocrinology, 26 cystic fibrosis, 42 canine distemper, 48 and influenza virus infection. 4,21,23,31,38,46
Recent studies have expanded the range of methods and reagents suited to the study of immunologic parameters in ferrets. Methods such as the detection of ferret interferon-gamma by immunoassays, 33 the description of cross-reacting monoclonal antibodies against leucocytes and cytokines for flow cytometry, 27,39 and quantitative reverse transcription polymerase chain reaction to assess inflammatory cytokine levels 32,43 have been described. Flow cytometry studies have also been performed on other mammals of the Mustelidae family, such as minks. These studies used reagents that were originally generated to recognize human, ovine, and bovine cells. 1,34
However, few studies have focused attention on the immunohistochemical characterization of immune system cells in formalin-fixed paraffin-embedded tissues in mustelids. In badgers (Meles meles), inflammatory responses in animals with tuberculosis have been characterized using some reagents against human and canine immune cells. 7 In ferrets, there are 2 reports that successfully used human antibodies in the differentiation of T- and B-cell lymphomas by immunohistochemistry (IHC). 2,17 Moreover, in recent years, several new infectious diseases have been described in pet ferrets, such as ferret systemic coronavirosis and pulmonary pseudomoniasis. 28,29 Interestingly, these diseases produce a similar pyogranulomatous inflammatory reaction. In this way, IHC would be a very useful tool for investigating the pathogenesis of these new diseases, through the characterization of the inflammatory response. But before this, standardization should be assessed of reliable immunohistochemical methods to detect different immune system cells in ferret tissues. Hence, the characterization and distribution of different types of immune cells in healthy ferret tissues will settle a basis for forward pathologic studies.
The aim of this study was to characterize the distribution of different immune system cells in different lymphoid and nonlymphoid organs of conventional, healthy ferrets. Therefore, IHC techniques were standardized in formalin-fixed paraffin-embedded and frozen ferret tissues using 8 antibodies: anti-CD3, anti-CD79α, anti-HLA-DR, anti-CD20, anti-lysozyme, CD163, SWC3, and Mac387.
Materials and Methods
Animals and Sampling
Five 3-year-old, male, neutered, healthy ferrets were used in this study. Animals were randomly selected from a stable, purposely bred colony. They were housed in standard ferret cages (Biosis Biologic Systems, SL, Spain), with stainless steel grid roofs, in an environmentally controlled room (19°–20°C, 40%–70% humidity, 12-hour light/dark cycle). They received a commercial pellet diet (Totally Ferret Active, Totally Ferret) and tap water ad libitum, until their use in this study. The colony was vaccinated for rabies (Im rab-3, Rhone Merieux, Inc) and dewormed with selamectine upon arrival at the laboratory, 3 years ago.
Blood samples were taken the day before euthanasia to test for influenza A virus infection and Aleutian disease. Animals were seronegative to influenza A virus (influenza A antibody competition multispecies ELISA, ID ScreenR, France). They were also negative for Aleutian disease virus, tested for by polymerase chain reaction in plasma (Applus Laboratories, Spain). Ferrets were euthanized by intravenous injection of sodium pentobarbital (100 mg/kg) under sedation with ketamine (5–10 mg/kg; Imalgene 1000, Merial, SA, Spain) and medetomidine (0.05 mg/kg; Domtor Pfizer, SA, Spain).
A complete necropsy was performed on all animals between 1 and 2 hours posteuthanasia, and no gross lesions were observed in any animal. The following tissues were collected during necropsy: mesenteric and mediastinal lymph nodes, spleen, thymus, bone marrow, small and large intestine, stomach, pancreas, liver, kidney, heart, lung, trachea, nasal turbinate, brain, skin, skeletal muscle, and eye. Tissue samples were embedded in OCT (Tissue-Tek, Torrance, CA), fast frozen using isopentane (M32631, Sigma-Aldrich Quimica, SA, Spain), precooled in liquid nitrogen, and stored at –80°C. Other portions of the same tissues were fixed for 48 hours in neutral-buffered 10% formalin. They were then embedded in paraffin wax, sectioned at 3 μm, and stained with hematoxylin and eosin (HE). Stomach sections were stained with Warthin-Starry silver impregnation for detection of Helicobacter spp. Kidney sections were stained with Prussian blue staining for the detection of hemosiderin deposits in tissues.
IHC in Formalin-Fixed Paraffin-Embedded Tissues
Serial 3-μm-thick sections of all tissues were cut and placed on silane-coated slides (3-trietoxysilyl-propylamine). Eight primary antibodies were used. The polyclonal anti-human antibodies tested were as follows: the anti-CD3 (Polyclonal Rabbit Anti-Human CD3, No. IS503, Dako), the anti-CD20 (CD20 Rabbit Polyclonal Antibody, No. RB-9013-P, Thermo Scientific), and the anti-lysozyme (Polyclonal Rabbit Anti-Human Lysozyme EC 3.2.1.17, No. A 0099, Dako). The monoclonal anti-human antibodies tested were as follows: the anti-CD79α (Monoclonal Mouse Anti-Human CD79αcy Clone HM57, No. M7051, Dako), the anti-HLA-DR (Monoclonal Mouse Anti-Human HLA-DR Antigen, Alpha-Chain Clone TAL.1B5, No. M0746, Dako) and the anti-myeloid/histiocyte antigen (Monoclonal Mouse Anti-Human Myeloid/Histiocyte Antigen Clone MAC 387, No. M0747, Dako). The mouse anti-swine monoclonal antibodies tested were as follows: the anti-CD163 (INIA research institute on-loan antibody against swine antigen 2A10/11, CD163 IgG1) and the anti-SWC3 (INIA research institute on-loan antibody against swine antigen, BA1C11, SWC3/CD172).
Different immunohistochemical protocols with different antigen retrieval methods and antibody temperature incubations were carried out. Successful protocols are represented in Table 1. Briefly, tissue sections were deparaffinized with xylene and rehydrated throughout with graded alcohols. Endogenous peroxidase activity was blocked by incubation with H2O2 3% in methanol for 30 minutes. Tissue sections were rinsed in 0.1M phosphate buffered saline (PBS; pH = 7.5). For the antigen retrieval, citrate, retrieval solution (Target Retrieval Solution, 10× concentrate, No. S1699, Dako), and EDTA buffers were tested at 98°C for 20 minutes. For the anti-Mac387, anti-CD163, anti-SWC3, and anti-lysozyme antibodies, an enzymatic digestion with proteinase K was also tested as antigen retrieval treatment. Afterward, Triton 0.2% for the anti-CD79α antibody and Tween 0.1% for the rest of antibodies, diluted in PBS for 10 minutes, were used to permeabilize the cell membranes. For the anti-Mac387, anti-CD163, and anti-SWC3 antibodies, both detergents were used in different attempts. Later, slides were blocked with 2% bovine serum albumin (85040C, Sigma-Aldrich Quimica) for 1 hour at room temperature (RT). Then primary antibodies were applied overnight (∼18 hours) at 4°C, except CD79α, which was incubated at RT. A biotinylated goat anti-mouse antibody (E-0433, polyclonal goat anti-mouse immunoglobuline biotinylated antibody, Dako) diluted 1:200 was used as secondary antibody for the HLA-DR, CD79α, and Mac387 antibodies, while a biotinylated goat anti-rabbit antibody (1:200) was used for the anti-CD3 antibody.
Antibodies Used in Immunohistochemistry.
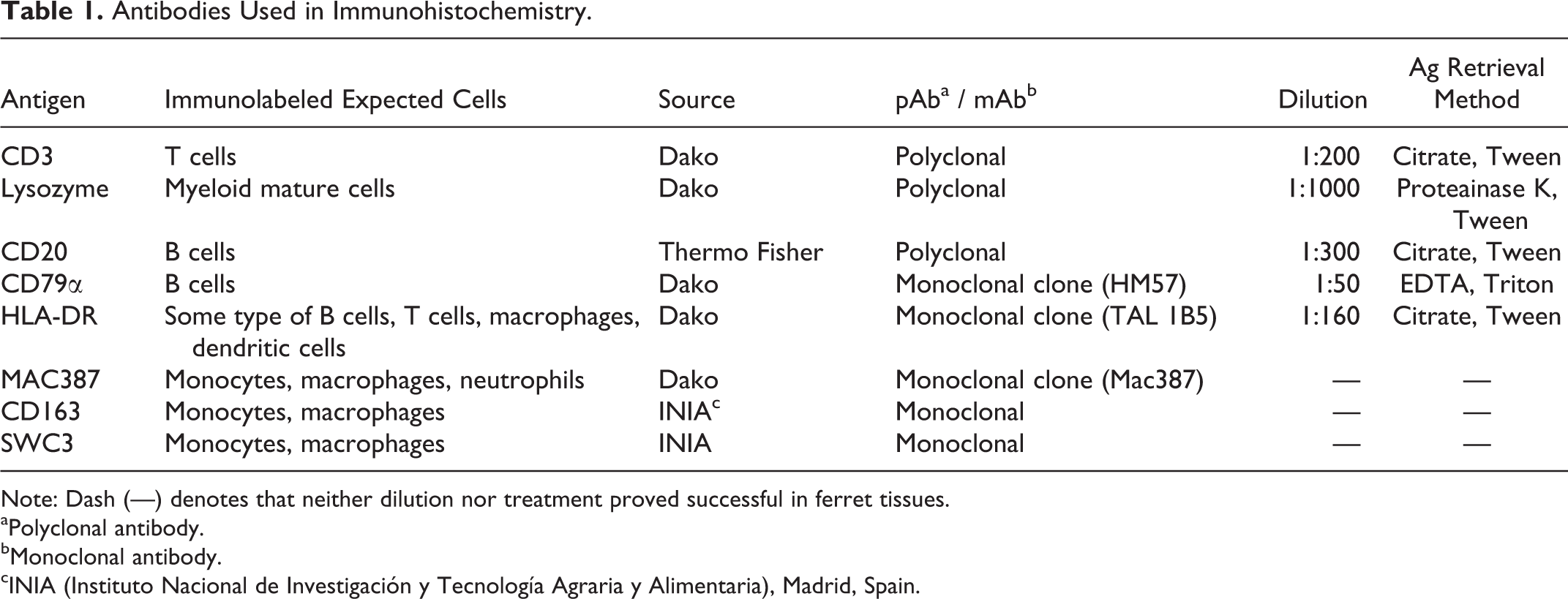
Note: Dash (—) denotes that neither dilution nor treatment has proved successful in ferret tissues.
aPolyclonal antibody.
bMonoclonal antibody.
cINIA (Instituto Nacional de Investigación y Tecnología Agraria y Alimentaria), Madrid, Spain.
An avidin–biotin–peroxidase complex (Pierce Biotin Quantitation Kit, Thermo Fisher Scientific) diluted 1:100 was applied at RT for 1 hour in the CD3 and HLA-DR detection. For CD79α, anti-lysozyme, anti-SWC3, anti-CD163, and anti-CD20 a polymer-based non–avidin–biotin–peroxidase system (Dako EnVision + System, Peroxidase-HRP) was used and applied directly to the slides and incubated for 30 minutes at RT. For Mac387, both methods were tested. Afterward, sections were incubated in diaminobenzidine (DAB) hydrogen peroxide solution, counterstained with Harris hematoxylin, dehydrated, covered with a coverslip, and examined under microscope. Swine lymph node and lung tissue sections, in which the specific primary antibodies were substituted with PBS or an irrelevant antibody, were used as negative controls.
Swine lymph node and lung tissue sections were used as positive control for all the primary antibodies, 9,12,14 except for CD20, in which canine lymph node sections were used as positive controls. 20
Immunofluorescence and IHC in Frozen Tissues
The anti-myeloid/histiocyte (Mac387), anti-CD163, and anti-SWC3 antibodies were also tested for immunofluorescence and IHC in frozen samples from spleen, lymph node, and lung. Briefly, frozen samples were stored at –80°C until they were cut into 7-μm-thick serial tissue sections using a cryostat (Leica Microsystems, Germany) at –25°C. They mounted on SuperFrost Plus glass slides (631-9483, VWR International Eurolab, SL, Spain).
Tissues were thawed for 30 minutes at RT and fixed using precooled acetone at –20°C for 10 minutes, then dried at RT for a further 10 minutes. Later, samples were rinsed 3 times with PBS (pH = 7.5) and blocked with 2% bovine serum albumin (85040C, Sigma-Aldrich Quimica) in PBS, containing 0.1% Triton X-100 (T8787, Sigma-Aldrich Quimica), 1 hour at RT. After that, sections were incubated overnight with the nondiluted primary antibody at 4°C. Then, the samples were rinsed with PBS and incubated with a goat anti-mouse (Alexa Fluor 488, Invitrogen, Spain) as a secondary antibody, diluted 1:300 in PBS for 1 hour at RT.
Nuclear counterstaining was performed using Hoechst 33258 (Sigma-Aldrich Quimica), diluted 1:500 in PBS for 10 minutes at RT. Samples were rinsed with PBS and sealed with antifade Vectashield mounting medium. Negative controls consisted of the same tissue but incubated with blocking buffer instead of the primary antibody. Swine lung and spleen tissues were used as positive controls. 9,13 For the IHC in frozen tissues, the same procedure was carried out using a different secondary antibody, a biotinylated goat anti-mouse antibody (1:200) incubated for 1 hour. Afterward, an avidin–biotin–peroxidase complex (Pierce Biotin Quantitation Kit, Thermo Fisher Scientific) diluted 1:100 was applied at RT for 1 hour. Finally, sections were incubated in DAB hydrogen peroxide solution, counterstained with Harris hematoxylin, dehydrated and covered with a coverslip, and examined under microscope. Tissue sections, in which the specific primary antibodies were substituted with PBS, were used as negative controls. Swine lymph node and lung tissue sections were used as positive controls for all the primary antibodies. 12,14
Double Immunohistochemical Staining for Codetection of Antibodies
Double immunohistochemical staining in formalin-fixed paraffin-embedded tissues was performed for the codetection of the antigens HLA-DR + CD79α, HLA-DR + CD20, and CD79α + lysozyme in lymphoid tissues (lymph nodes, spleen, thymus, small intestine, including Peyer patches and bone marrow), to elucidate the nature of several cells with myeloid morphology and positive staining for anti-CD79α antibody. The protocol used in this experiment was similar to that described in other studies 8,47 with minor modifications.
Briefly, paraffin-embedded tissue samples were dewaxed, cut to 3 μm in thickness, and blocked to eliminate the endogenous peroxidase activity using 3% H2O2 in methanol. Antigen retrieval was carried out with citrate buffer at 98°C for 20 minutes and later permeabilized using Triton 2% for the anti-CD79α primary antibody and Tween 1% for the anti-CD20 primary antibody in PBS for 15 minutes. Slides were blocked with 20% normal goat serum diluted in PBS for 1 hour at RT. The primary staining corresponded to the incubation with the antibody against CD79α diluted 1:25 and against CD20 diluted 1:300 in blocking buffer. Antibodies were incubated overnight at RT and 4°C, respectively. After incubation of the primary antibody, tissues were rinsed for 15 minutes with PBS. Later, a secondary goat anti-mouse antibody, conjugated with a polymer-based non–avidin–biotin–peroxidase system (Dako EnVision + System, Peroxidase-HRP) was applied directly on the slides and incubated for 30 minutes at RT. Finally, sections were incubated in DAB hydrogen peroxide solution for 10 minutes. The presence of positive staining was visualized in brown.
For the second staining, samples were rinsed and permeabilized 3 times with Tween 1% diluted in PBS and blocked again with normal goat serum 20% diluted in PBS. After that, tissue sections were incubated with the second primary antibody anti-HLA-DR (diluted 1:80 when combining with CD79α) and (diluted 1:50 when combining with CD20) in blocking buffer overnight at 4°C. Later, the slides were rinsed in PBS for 15 minutes and incubated in an alkaline phosphatase conjugate goat anti-mouse Ig (H+L) secondary antibody (1010-04, Southern Biotechnology), diluted 1:100 in PBS.
For the second staining using the polyclonal antibody anti-lysozyme, slides were incubated with anti-lysozyme diluted 1:1000 in blocking buffer overnight at 4°C. Later, the slides were rinsed in PBS for 15 minutes and incubated in the alkaline phosphatase conjugate goat anti-rabbit secondary antibody (EXPOSE Rabbit Specific AP [red] Detection IHC Kit, ab94738, Abcam, UK) without dilution. The presence of positive staining was visualized in blue using nitro blue tetrazolium chloride 5-bromo-4-chloro-3-indoxyl (NBT-BCIP; 11 681 451 001, Roche). Immunolabeling of HLA-DR and lysozyme positive cells were visualized in blue.
Codetection was visualized as a deep violet color. Samples were not counterstained. Negative controls consisted of the incubation of a sequential sample with PBS instead of the primary antibodies. Positive controls, consisting of ferret lymph node sections, were included for each antibody separately. To check the unspecific cross-reactions between antibodies, the whole technique was carried out in each control.
Cell Quantification
A semiquantitative assesment of positive cells in IHC was assessed for every tissue and antibody. Positivity was considered and represented with different numbers depending on the percentage of positive cells per high-power field. This was measured in 4 areas of each organ of every animal. The mean and standard deviation results for each organ for all 5 animals were recorded. The proportion of positive cells was scored as follows: (0) nil (fewer than 5 positive cells), (1) scarce (less than 25% of positive cells), (2) slight (25%–50% positive cells), (3) moderate (50%–75% of positive cells), and (4) intense (>75% of positive cells).
Results
Histopathology
Minor histopathologic lesions were observed in some animals. Four of the 5 ferrets showed mild to moderate lymphocytic infiltration with few eosinophils in the lamina propria of the stomach, consistent with mild gastritis. These cells were mainly T lymphocytes and HLA-DR-positive cells, with low numbers of B cells. A small number of bacillary bacteria were detected with Warthin-Starry special stain in the lumen of the stomach. However, no spiral-shaped bacteria consistent with Helicobacter spp. were observed in any animal.
In the kidneys of all animals, a mild accumulation of a brown, finely granular pigment in the cytoplasm of tubular epithelial cells was observed. This pigment was negative following Prussian blue staining. No relevant microscopic lesions were observed in the remaining tissues.
Immunohistochemistry
Relevant results for lymphoid and nonlymphoid tissues are shown in Tables 2 and 3, respectively.
Comparative Immunohistochemical Staining Distribution in Different Lymphoid Areas.
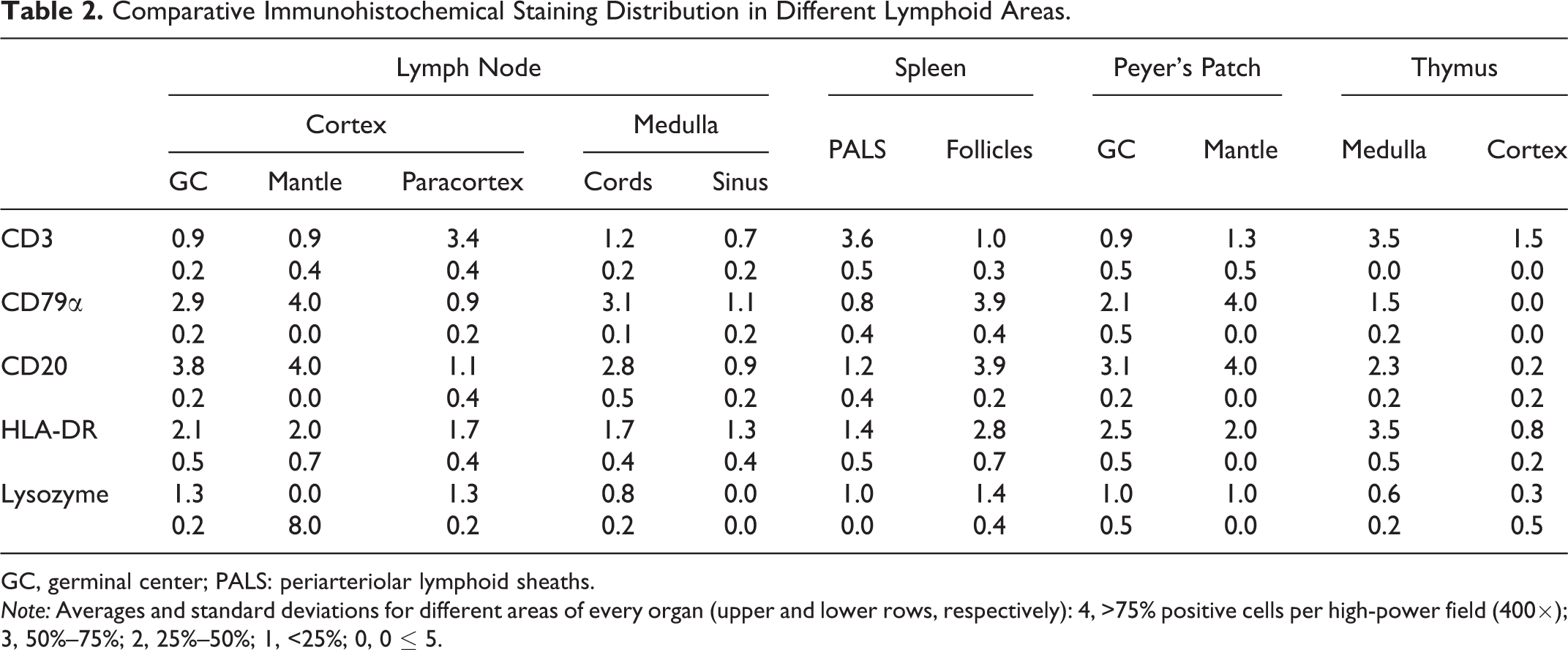
GC, germinal center; PALS: periarteriolar lymphoid sheaths.
Note: Averages and standard deviations for different areas of every organ (upper and lower rows, respectively): 4, >75% positive cells per high-power field (400×); 3, 50%–75%; 2, 25%–50%; 1, <25%; 0, 0 ≤ 5.
Comparative Immunohistochemical Staining Distribution in Different Nonlymphoid Organs.
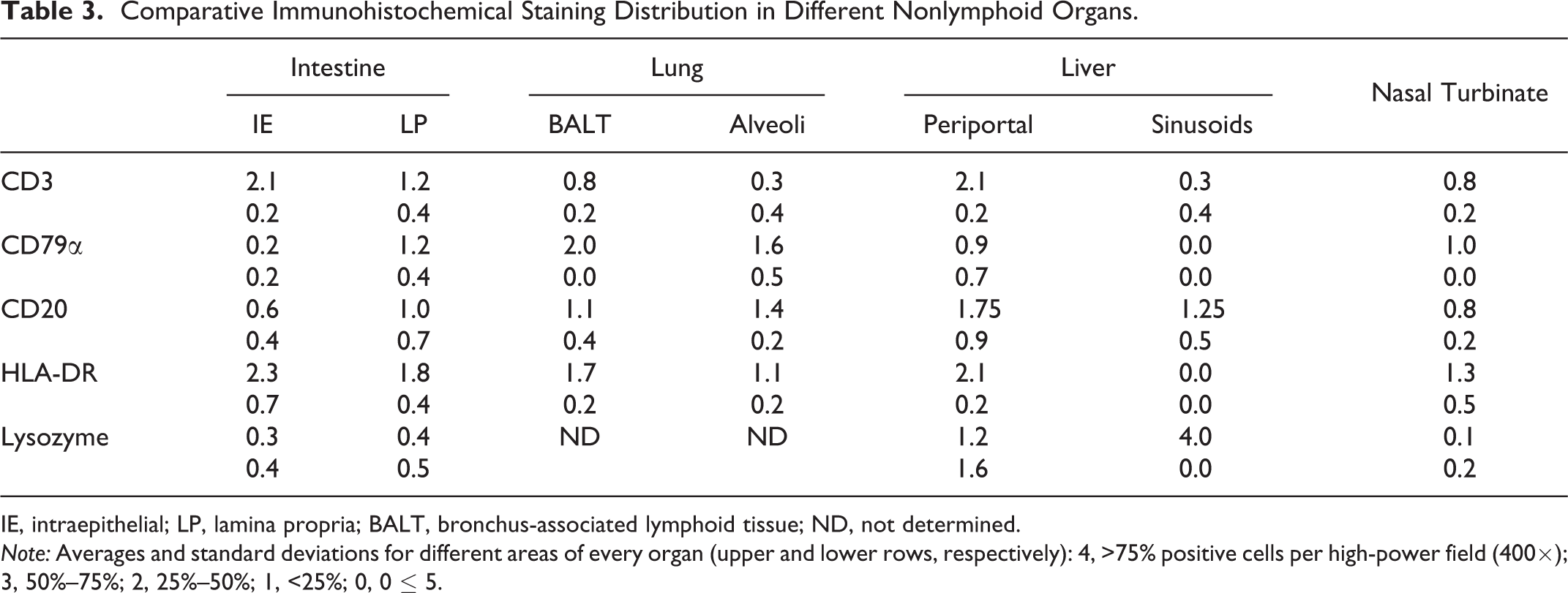
IE, intraepithelial; LP, lamina propria; BALT, bronchus-associated lymphoid tissue; ND, not determined.
Note: Averages and standard deviations for different areas of every organ (upper and lower rows, respectively): 4, >75% positive cells per high-power field (400×); 3, 50%–75%; 2, 25%–50%; 1, <25%; 0, 0 ≤ 5.
CD3
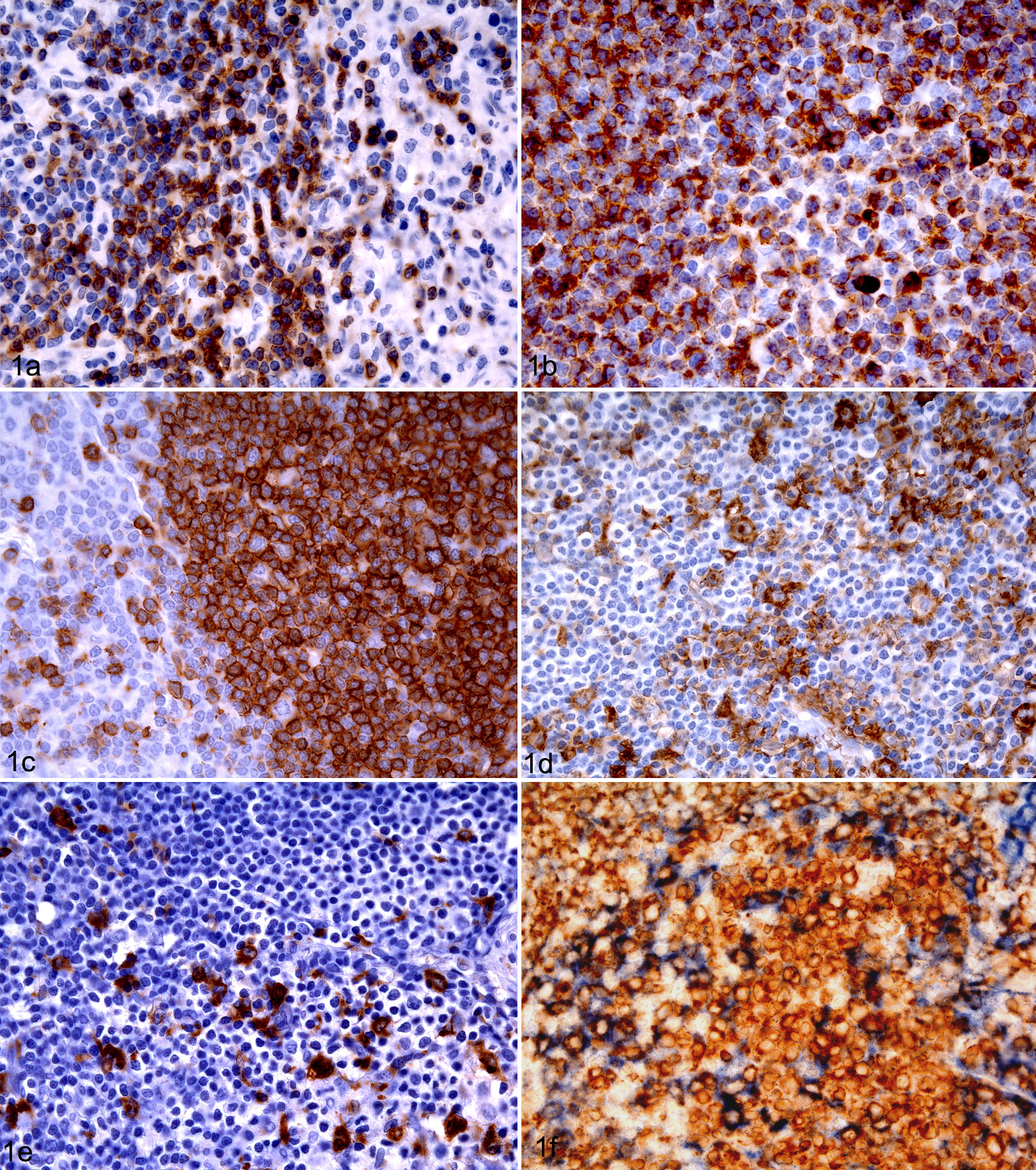
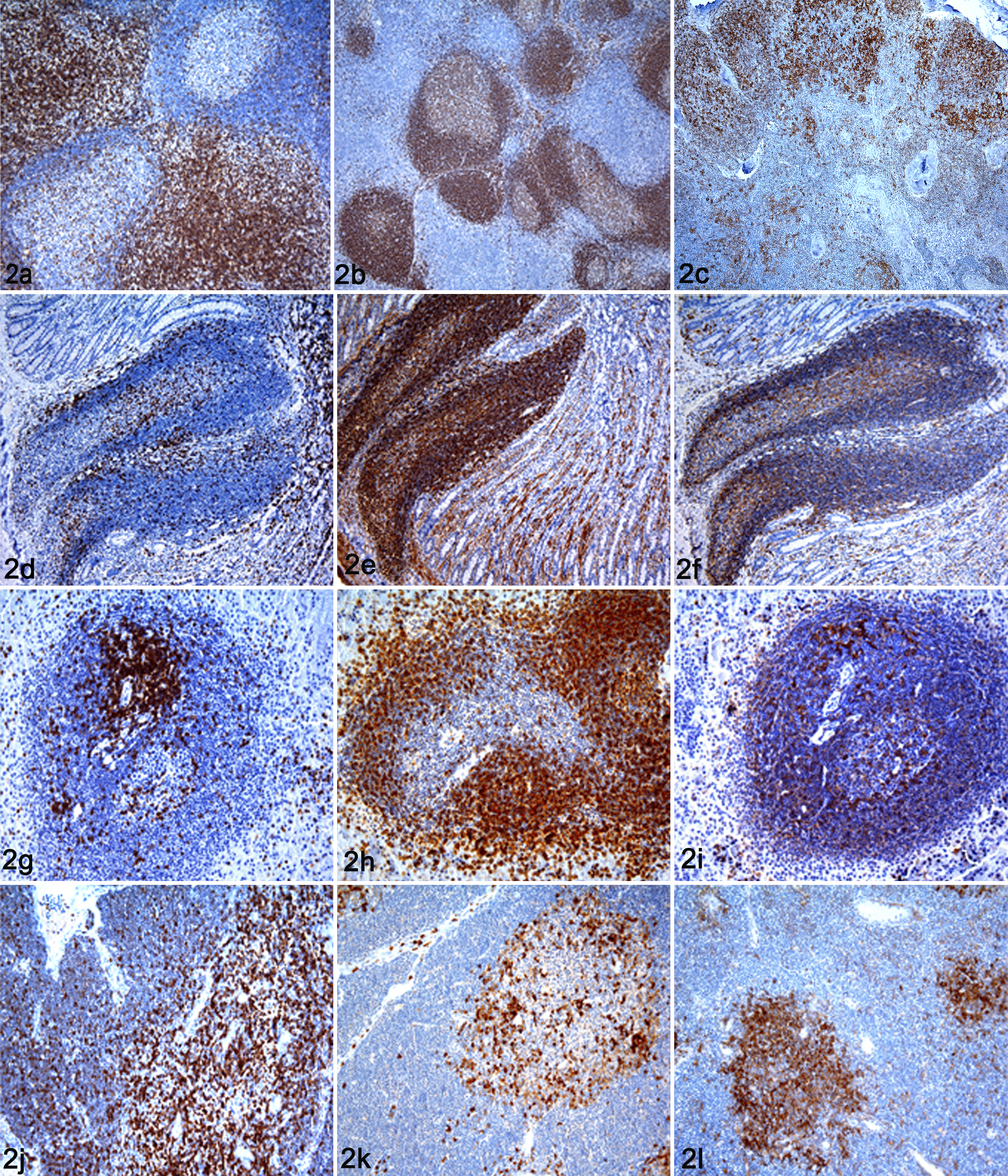
The anti-CD3 antibody labeled the surface and cytoplasm of round cells consistent with lymphocytes in formalin-fixed paraffin-embedded tissues (Fig. 1a). These cells were located in T-cell areas of the lymph nodes (Fig. 2a), Peyer patches (Fig. 2d), spleen (Fig. 2g), and thymus (Fig. 2j). In the lymph node, these areas corresponded to interfollicular and paracortical areas and to periarteriolar lymphoid sheaths (PALS) in the spleen. Moderate to abundant numbers of positive cells were found in cortical and medullary areas of the thymus, with higher concentration of CD3-positive-labeled cells in the medulla. A small number of positive cells were observed forming a marginal layer of CD3-positive-labeled cells around Peyer patches. A very small number of CD3-positive-labeled cells were located, scattered in the follicular germinal centers of lymph nodes, spleen, Peyer patches, bone marrow, and medullary areas of lymph nodes.
In nonlymphoid tissues, moderate numbers of CD3-positive cells were observed in periportal areas of the liver and lamina propria and intraepithelial compartment of the intestine. A small number of CD3-positive-labeled cells were located in the bronchus associated lymphoid tissue (BALT) of the lung and multifocally distributed in the interstitium of pancreas. A very small number of CD3-positive-labeled cells were observed in the lamina propria of trachea and nasal turbinates. None or a very limited number of CD3-positive-labeled cells were observed in the heart, skin, muscle, eye, subarachnoid space, and the choroid plexus of the brain. Variable numbers of CD3-positive-labeled circulating cells were detected in the blood vessels of all tissues studied.
CD79α
The anti-CD79α antibody positive labeled the cell membrane and cytoplasm of small round cells in formalin-fixed paraffin-embedded tissues. These cells presented a centrally located nucleus and a narrow rim of cytoplasm, consistent with lymphocytes (Fig. 1b). These cells were localized in B-cell areas, mainly in the lymphoid follicles of the lymph nodes (Fig. 2b), Peyer patches (Fig. 2e), and spleen (Fig. 2 h). There was a denser concentration of positive labeling in the mantle of lymph nodes and Peyer patches. Smaller numbers of CD79α-positive-labeled small cells were located in the thymic medulla (Fig. 2k) and the dome region of Peyer patches. High numbers of CD79α-positive small cells were also located in the bone marrow and medullar cords of the lymph nodes.
In nonlymphoid tissues, moderate numbers of immunolabeled small cells were observed in the lungs and intestinal lamina propria. Cells were located within the bronchiolar epithelium, mucous glands, BALT, peribronchiolar connective tissue, and alveolar septa. Low numbers of positive small cells were observed in the periportal areas of liver, tracheal lamina propria, meninges, and choroid plexus of the brain. In the pancreas, kidney, skeletal muscle, and eye, very few positive cells were detected in the interstitial connective tissue. Variable numbers of CD79α-positive-labeled circulating cells were detected in the blood vessels of all tissues studied.
Interestingly, a subpopulation of CD79α positive cells characterized as showing irregular cell borders, abundant cytoplasm, and eccentric and round-to-oval nucleus were detected in the follicular germinal centers (Fig. 1b) of the spleen, lymph nodes, and Peyer patches. In the thymus, a moderate amount of these cells were also observed in the thymic medulla.
HLA-DR
The HLA-DR antibody labeled different cell types in formalin-fixed paraffin-embedded tissues. Some HLA-DR-positive cells were small and round with a centrally located nuclei and a narrow rim of cytoplasm consistent with lymphocytes. Besides these, medium-sized cells with a moderate amount of cytoplasm and oval-to-round nuclei consistent with cells of the macrophage/dendritic lineage were also detected by using this cell marker (Fig. 1d).
Moderate numbers of HLA-DR-positive lymphocyte-like cells were observed in the mantle of lymphoid follicles in lymph nodes, Peyer patches, and spleen. Moderate numbers of HLA-DR-positive macrophage/dendrite-like cells were also observed in the follicular germinal centres in the lymph nodes (Fig. 2c), Peyer patches (Fig. 2f). and spleen (Fig. 2i). These cells were considered as follicular dendritic cells and tingible body macrophages. Low numbers of positive macrophage/dendrite-like cells were detected in medullar and paracortical areas of the lymph nodes. They were considered interfollicular dendritic cells and tingible body macrophages. In the thymus, a small number of positive cells were located in the cortex and were also considered to be tingible body macrophages. A moderate to high number of positive cells were observed in the thymic medulla (Fig. 2l). These cells were considered to be epithelial reticular cells or interdigitating cells of the thymus.
In nonlymphoid tissues, moderate numbers of HLA-DR lymphocyte–positive cells were present in the periportal areas of the liver and lamina propria of the intestine. In the lung, positive labeling was present in lymphocyte-like cells located in the bronchiolar lamina propria. In the liver, the anti-HLA-DR antibody labeled Kupffer cells located in the sinusoids; in the lung, macrophage/dendrite-like cells were located in the alveolar lumen and wall, and were considered to be pulmonary alveolar macrophages. A moderate number of positive cells consistent with macrophage/dendrite-like cells were also observed in the intraepithelial compartment and lamina propria of the intestine, nasal turbinate, and trachea. In the interstitium of the kidney, bone marrow, pancreas, heart, meningeal and choroid plexus of the brain, muscle, skin, and eye, HLA-DR labeled a very low number of macrophage/dendrite-like cells. Variable numbers of HLA-DR-positive-labeled circulating cells were detected in the blood vessels of all tissues studied.
CD20
The anti-CD20 antibody immunolabeled the membrane of small round cells, with scarce cytoplasm and a central round nucleus, consistent with lymphocytes in formalin-fixed paraffin-embedded tissues (Fig. 1c). These cells were located forming primary and secondary folices in lymph nodes immunolabeled mantle and germinal center with equal intensity (Fig. 3a), Peyer patches (Fig. 3c), and white pulp of the spleen (Fig. 3e). In the follicular germinal centers, the anti-CD20 antibody also immunolabeled slightly larger round cells with large amounts of cytoplasm and a bigger central round nucleus, consistent with immunoblasts. Moderate numbers of CD20-immunolabeled small cells were located in the thymic medulla (Fig. 3g).
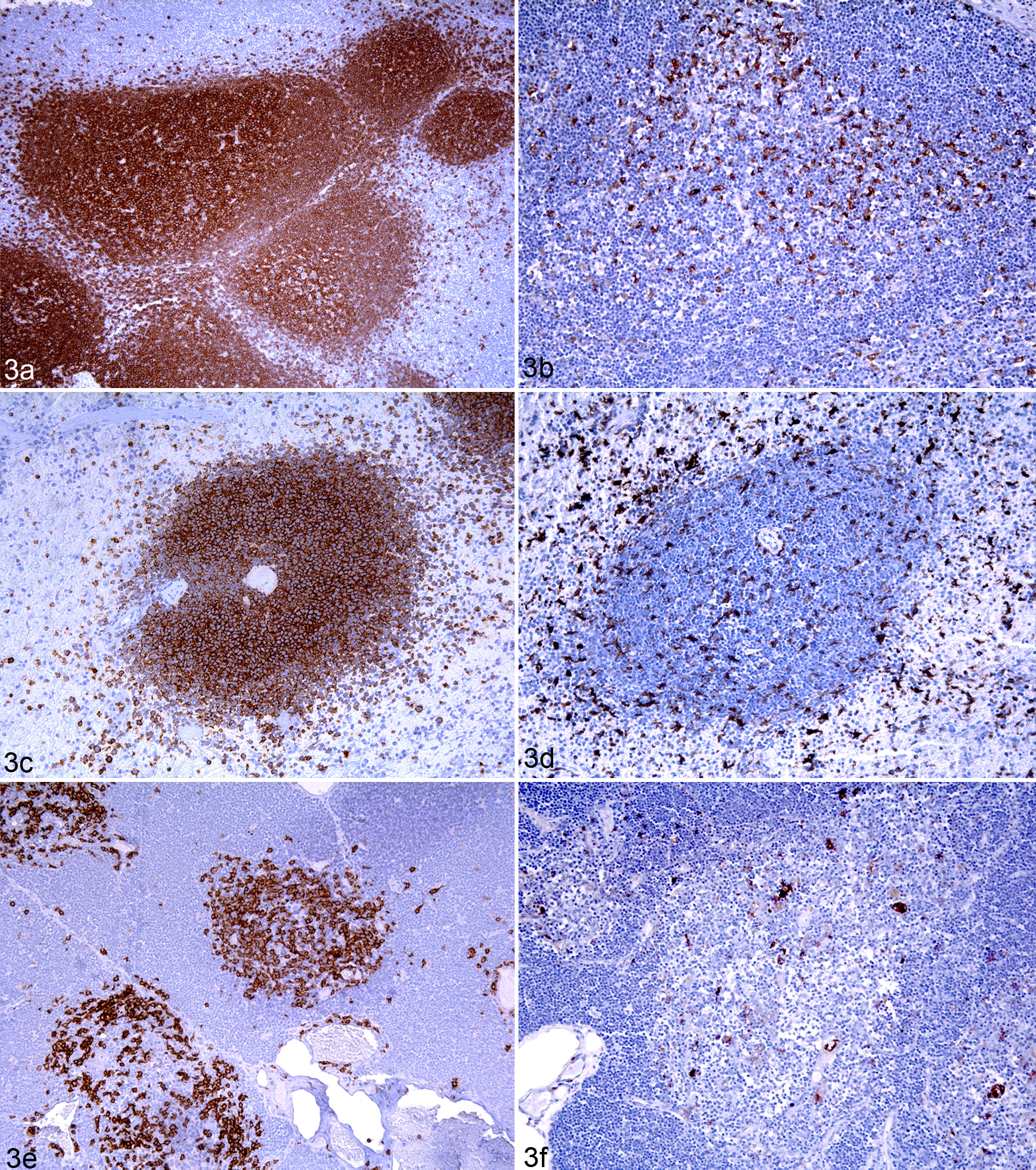
Interestingly, the CD20 antibody also immunolabeled a subpopulation of large macrophage/dendritic positive cells characterized as showing irregular cell borders and abundant cytoplasm with an eccentric and a round-to-oval nucleus. These cells were detected in the follicular germinal centers of the spleen, lymph nodes, and Peyer patches. In the thymus, a moderate amount of these cells were also observed in the thymic medulla. High numbers of CD20 positive-labeled small cells were also located in the bone marrow and in the medullary cords of the lymph nodes.
In nonlymphoid tissues, the anti-CD20 antibody immunostained the membrane of small round cells consistent with lymphocytes. Low numbers of positive cells were observed in the lung (alveolar septa and BALT) and in the liver (periportal areas and sinusoids). The anti-CD20 antibody also labeled low numbers of cells in the lamina propria of the intestine, trachea, meninges, and choroid plexus of brain. In the pancreas, kidney, heart, skeletal muscle, and eye, very few positive cells were detected in the interstitial connective tissue. Variable numbers of CD20-positive-labeled circulating cells were detected in the blood vessels of all studied tissues.
Lysozyme
In formalin-fixed paraffin-embedded tissues, the anti-lysozyme antibody immunolabeled the cytoplasm and membrane of round-to-polyhedral, medium-size cells with abundant and irregular cytoplasm and round to “bean-shape” exocentric nucleus consistent with macrophages (Fig. 1e). High numbers of positive polyhedral cells were located in the red pulp of the spleen. Low numbers of positive cells were located in the germinal centers of lymphoid follicles in the lymph nodes (Fig. 3b), Peyer patches (Fig. 3d), spleen (Fig. 3f), and paracortical areas and cords of lymph node and thymus (Fig. 3h). In the bone marrow, moderate to large numbers of positive cells were also observed and were interpreted as myeloid leukocytes.
In nonlymphoid tissues, large numbers of positive cells were observed between hepatic sinusoids of the liver. Low numbers of positive cells were located in the tracheal lamina propria, meninges, and choroid plexus of the brain. In the pancreas, skeletal and heart muscle, kidney, stomach, and eye, very few positive cells were detected in the interstitial connective tissue.
The anti-lysozyme antibody also immunolabeled the cytoplasm of different nonlymphoid cell types—namely, ciliated epithelial cells of the trachea; glandular cells in bronchus, trachea, and stomach; and high endothelial venules of lymph nodes and renal proximal tubules. Variable numbers of lysozyme-positive circulating cells were detected in the blood vessels of the different tissues studied.
Mac387, CD163, and SWC3
No positive labeling was detected in any ferret tissue using the anti-Mac387, anti-CD163, and anti-SWC3 antibodies in frozen or formalin-fixed paraffin-embedded tissues.
Double Immunohistochemical Staining
To clarify the origin of the CD79α- and CD20-positive cells with a nonlymphocytic morphology, a double IHC was carried out using different combinations of antibodies. The intention was to find out if these cells could belong to the macrophage/dendritic lineage. Low to moderate numbers of cells were labeled with both anti-CD79α and anti-HLA-DR in germinal centers of lymphoid follicles, spleen, and Peyer patches (Fig. 1f). A smaller number of cells were also labeled with both anti-CD20 and anti-HLA-DR antibodies in the germinal centers of lymphoid follicles. No coexpression of anti-CD79 + anti-lysozyme antibodies was observed in follicular germinal centers of lymphoid follicles in lymph nodes, spleen, and Peyer patches and in medullar areas of the thymus.
Discussion
A detailed knowledge of normal immune system cell distribution is required to better understand the pathologic changes of many infectious and noninfectious diseases. In recent years, the ferret has become a reliable animal model for infectious and degenerative diseases. 5,24,42 In addition, this species has become a popular companion animal and new diseases have been described, such as influenza, systemic coronavirosis and pulmonary pseudomoniasis. 28,29,31 However, there are still no detailed descriptions of the main ferret immune system cells. In this study, both standard and double IHC techniques were performed toward that end. Furthermore, a histologic description was made of the previously uncharacterized antibodies CD20, lysozyme, and HLA-DR.
Animals from this study were clinically healthy, although 4 presented a mild to moderate lymphocytic infiltration in the stomach, mainly formed by CD3- and HLA-DR-positive cells. The cause of this infiltration is currently unknown because animals tested negative to Aleutian disease by polymerase chain reaction in plasma and no spiral-shaped bacteria compatible with Helicobacter spp. were detected in Warthin-Starry stain.
The anti-CD3 and anti-CD79α antibodies were formerly used to characterize ferret lymphomas. 2,17 In our study, the distribution of T and B cells in lymphoid tissues of healthy ferrets correlated with that described in other mammal species. 7,9,22
The anti-CD20 antibody has been described as a B-cell marker in other species, such as dogs and mice. 20,44 In humans, dogs, and mice, the anti-CD20 antibody is expressed in pre–B cells and in mature B cells. 13,20,44 In ferret tissues, CD20 immunolabeled the membrane of B cells located in B-cell areas, and it intensely immunostained the germinal center of lymphoid follicles, impeding differentiation between the germinal center and the mantle zone. The strong immunostaining found in follicular germinal centers makes it difficult to differentiate between the different shapes and structures of the cells within the germinal center, as described in other species. 20 The intense expression of CD20 in germinal centers clearly stands out when compared with the CD79α immunolabeling, what could be the result of difference in expression between the CD79α and CD20 molecules, which is dependent on the stage of B-cell maturation. Resting naïve B lymphocytes in the mantle express a higher number of CD79α molecules, and the activation of germinal center B cells into mature B cells downregulates CD79α expression. 37 In contrast, the CD20 antigen expression increases on B cells located in lymphoid germinal centers, as they mature. 13,20
The primary purpose of MHC-II molecules is to allow antigen-presenting cells and B cells to display the epitopes of the invading pathogen to the T cells. In humans, histocompatibility class II antigens are expressed in a limited number of cell types: macrophages, monocytes, B cells, a variable number of T cells, 3 dendritic cells, 30 and in some types of epithelial cells, such as thymic epithelial cells and pneumocytes type II. 10 In this study, it was possible to identify these cells for the first time in ferret tissues and to study their distribution using the anti-HLA-DR antibody. This antibody strongly labeled cells that presented different characteristics. Some had round central nuclei and a small rim of cytoplasm; these were considered B or T lymphocytes, 18 depending on their expression of CD20, CD79α, or CD3 molecules, as well as their location and morphology. It is worth mentioning that in our study, not all CD79α- and CD20-positive B cells were immunolabeled with the anti-HLA-DR antibody, even though the expression of MHC-II molecules in all B cells has been reported in different mammal species and humans. 7,18 The other subpopulation of HLA-DR-positive cells were large, irregular cells that were recognized as macrophages or dendritic cells, depending on their shape and distribution in the lymphoid and nonlymphoid organs. 41,45
The anti-human lysozyme antibody has been described as a useful marker to immunostain cells of the monocyte/macrophage series in different mammal species and humans. 9 , 35 , 36 In ferret tissues, the anti-lysozyme antibody immunolabeled mature peripheric myeloid cells and different types of resident macrophages. However, the anti-lysozyme antibody failed to immunolabel resident macrophages in the lungs as described in other species, 9,37 which could be a consequence of incomplete formalin penetration during tissue fixation.
In addition to B cells, the CD79α and the CD20 antibodies immunolabeled atypical B cells with a macrophage/dendritic morphology, located in nontypical B cell areas. The expression of B-cell markers in large cells with an atypical morphology has been described in humans at follicular germinal centers and scattered in the thymic medulla. 15,16,19 Furthermore, a subpopulation of large interfollicular B cells showing a dendritic morphology have been described in humans. 25 These cells were positive to the B-cell markers CD79α and CD20 yet negative to dendritic and macrophagic markers. In the present work, double IHC techniques combining different B-cell markers with the HLA-DR antibody (expressed in follicular dendritic cells, 11 interdigitating cells, and macrophages 3 ) and the macrophagic marker lysozyme have been used to elucidate the origin of these atypical B cells.
Results showed that these cells did not coexpress the lysozyme and the CD79α antigens but coexpressed both B-cell markers (CD20 and CD79α) with the HLA-DR antibody, suggesting that these cells did not have a macrophagic origin and that they have a B-cell origin, despite their morphology.
The results obtained in this work led us to hypothesize that these atypical large B cells found in lymphoid germinal centers and thymic medulla might be a subpopulation of B cells similar to those described before in humans and mice 15,19,25 or, less likely, follicular dendritic cells. The lack of reliable dendritic markers that cross-react with ferret tissues impeded us from further testing this hypothesis. Hence, further investigation into the immunophenotyping of ferret cells should be undertaken to further elucidate the origin of these cells.
The monoclonal swine antibodies CD163 and anti-SWC3 were described as useful markers for immunostaining cells of the monocyte/macrophage lineage in swine tissues. 12,14 The monoclonal human anti-L1 (Mac387) antibody has been described as a usefull marker for immunostaining macrophages or polymorphonuclear cells in different mammal species. 6,7,9,22 Different protocols for the standardization of the IHC technique with these antibodies (in frozen and paraffin-embedded ferret tissues) were implemented in the present study. However, despite the positive results obtained in swine tissue controls, the Mac387, CD163, and SWC3 antibodies did not cross-react with ferret tissues in this study.
The current study investigated the normal distribution of immune cells in lymphoid and nonlymphoid paraffin-embedded tissues of conventional healthy ferrets, using immunohistochemical techniques with different antibodies. This work represents a preliminary histologic investigation of the distribution of certain immune system cells in conventional healthy ferrets. This will help to establish a basis for future research, which could lead to a better understanding of immune responses in ferrets. Moreover, it contributes to the standardization of several immunohistochemical markers in this species, which may help in the diagnosis and research into different infectious and noninfectious ferret diseases.
Footnotes
Acknowledgements
We are grateful to Lola Pérez and Rosa Valle for their technical assistance. We are also grateful to Aaron Casley for the English correction and editing and to Dr Neus Prats from Almirall Laboratories for providing the animals.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was partly funded by Instituto de Salud Carlos III (“Programa especial de investigación sobre la gripe pandémica,” GR09/0023, GR09/0040, GR09/0039) and the Servei de Diagnòstic en Patologia Veterinària of the Universitat Autònoma de Barcelona.
