Abstract
Six 12- to 14-month-old New Zealand White rabbits were diagnosed with copper toxicosis. These rabbits were part of a group of 110 purchased and shipped overnight for research purposes. On arrival, the group experienced an abrupt diet change. Eight died over 3 weeks and 6 were submitted for postmortem examination. Microscopic findings included severe centrilobular to midzonal hepatocellular necrosis with rhodanine stain–positive copper granules in the remaining hepatocytes. Mild periportal fibrosis and biliary hyperplasia, hemoglobinuric nephrosis, and splenic erythrophagocytosis were also observed. Hepatic copper concentrations were elevated, ranging from 319 to 997 ppm. Clinical disease was not previously observed in younger rabbits gradually transitioned from the supplier’s copper-supplemented diet. Copper toxicosis likely occurred in these rabbits from a combination of (1) increased duration of copper supplementation leading to increased hepatocellular stores and (2) stress leading to anorexia and release of hepatocellular copper stores similar to chronic copper toxicosis as described in sheep.
Chronic copper toxicosis in susceptible ruminant species refers to the acute presentation of clinical disease resulting from sudden release of massive hepatocellular copper stores. Excess copper is stored in hepatocytes, and significant accumulation can occur over time with defective hepatic metabolism, altered biliary and urinary excretion, and nutritional imbalances between copper and other trace elements. 4,5,7 Highly susceptible species, such as sheep and Boer goats, accumulate copper in the liver when fed copper at levels recommended for less susceptible species. 1,4,5,7 Hepatocellular damage, generally secondary to stress, releases hepatocellular copper stores into the bloodstream, leading to acute hemolysis. Clinical signs may include acute onset of depression, anorexia, and weakness with subsequent icterus and hematuria. Hereditary forms of copper toxicosis are reported in several breeds of dog, but unlike ruminants, these animals develop chronic-active hepatitis, and death results from hepatic failure rather than acute hemolytic crisis. 4,7
A group of 110 New Zealand White rabbits aged 12 to 14 months was purchased from an out-of-state supplier and shipped overnight to a research facility in Missouri. Neither the supplier nor the researchers reported clinical disease in other rabbits currently at or previously acquired from the supplier’s facility. The supplier fed a custom diet supplemented with copper, and although some of the supplier’s feed was usually included with shipments, none was sent with this shipment of rabbits. Upon arriving at the research facility, the rabbits were abruptly switched to Teklad Global High Fiber Rabbit Diet 2031C (Harlan Laboratories, Indianapolis, IN) and became anorexic. Some rabbits developed mucoid diarrhea. Over the course of 3 weeks, 8 rabbits died or were euthanized. Two rabbits found dead in their cages (rabbit Nos. 1–2), 1 euthanized rabbit (rabbit No. 3), and the livers of 3 additional euthanized rabbits (rabbit Nos. 4–6) were submitted to the University of Missouri Veterinary Medical Diagnostic Laboratory (MU-VMDL, Columbia, MO) for examination. The remaining 2 rabbits were found dead in their cages and not submitted for postmortem examination.
Tissue samples were fixed in 10% neutral buffered formalin for 24 hours and processed routinely. Paraffin sections were cut at 5 μm and stained with hematoxylin and eosin. Sections of liver were stained with rhodanine stain by routine methods. Copper concentrations in the liver and kidney were measured on a wet-weight basis at the MU-VMDL using flame atomic absorption spectrometry. Samples of the supplier’s feed and the researcher’s laboratory-grade diet were submitted to Michigan State University (Lansing, MI) for a Toxic Elements panel, which includes copper and molybdenum concentrations on a dry-weight basis.
Gross and Microscopic Findings
The most severe gross lesions were observed in rabbit No. 3. The liver was diffusely yellow-brown with an accentuated lobular pattern, and the gallbladder was markedly distended (Figs. 1, 2). The kidneys were dark red and the urinary bladder contained dark red urine (Figs. 1, 3). In addition, the spleen was congested. The liver from rabbit No. 2 was moderately yellowed with an accentuated lobular pattern and the gallbladder was distended. The urinary bladder was empty. Postmortem autolysis impaired meaningful interpretation of gross necropsy findings in rabbit No. 1. Rabbit No. 3 had the most representative microscopic lesions. Severe hepatocellular necrosis was observed in midzonal areas, and hepatocytes, especially in periportal areas, were distended with cytoplasmic accumulations of numerous blue-green granules that stained positive for copper with rhodanine stain (Figs. 4 –6). Mild periportal fibrosis and biliary hyperplasia were also observed. Sinusoids in the spleen were congested and erythrophagocytic macrophages were often observed (Fig. 7). Renal tubules contained eosinophilic granular material (hemoglobin casts) and proteinaceous fluid (Fig. 8). The liver from rabbit No. 2 also had severe centrilobular and midzonal hepatocellular necrosis with rhodanine-positive granules. Moderate hemosiderosis was observed in the spleen. Periportal hepatocytes in rabbit No. 1 were often distended with cytoplasmic, rhodanine-positive granules. The microscopic features of the livers from rabbit Nos. 4 to 6 were similar to rabbit No. 3, with the most severe lesions observed in rabbit No. 5 and the least severe in rabbit No. 6.
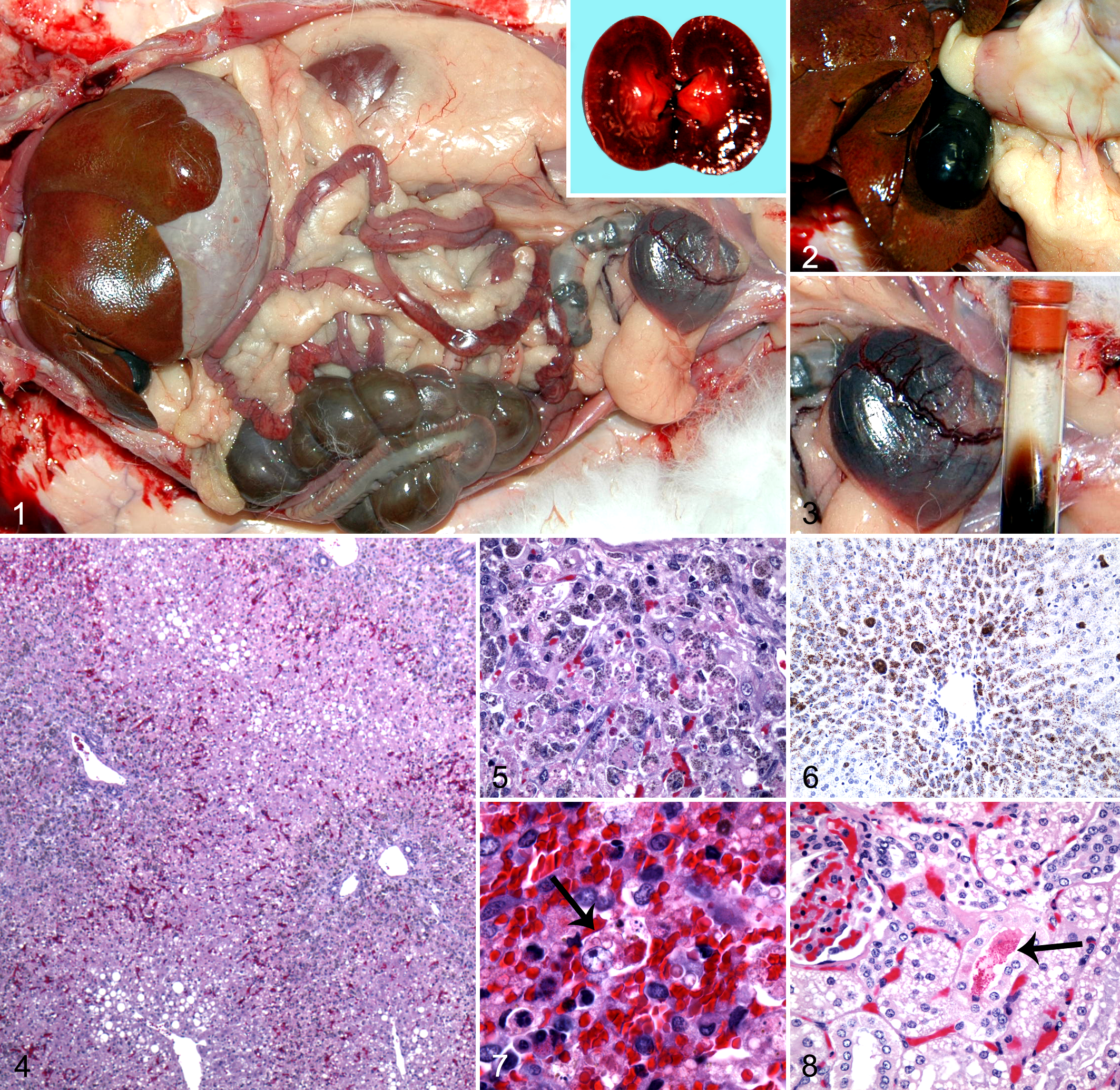
To establish that the hepatocellular copper accumulations leading to toxicosis occurred at the supplier’s facility prior to shipment, livers from a 3.5-month-old rabbit (rabbit No. 7) and a 9-month-old rabbit (rabbit No. 8) were harvested at the supplier’s facility and sent directly to the MU-VMDL. These rabbits were clinically healthy prior to euthanasia and fed the same diet as rabbit Nos. 1 to 6. Representative sections were collected and placed in formalin overnight prior to processing. As these liver samples were shipped fresh without ice, both specimens had diffuse, moderate to severe, postmortem autolysis. Rabbit No. 7 had a relatively normal liver, whereas the liver from rabbit No. 8 exhibited diffuse hepatocellular degeneration and necrosis with multiple enlarged macrophages and Kupffer cells containing green-brown granules in the cytoplasm. Diffuse, moderate copper accumulation was observed with rhodanine staining in rabbit No. 7, and severe accumulations were observed in rabbit No. 8.
Copper Concentrations
The hepatic and renal copper concentrations are listed in Table 1. Hepatic and renal copper concentrations from the 12- to 14-month-old group (rabbit Nos. 1–6) ranged from 319 to 997 ppm (reference range, 8–50 ppm) and 8.2 to 110 ppm (reference range, 4–6 ppm), respectively. The hepatic copper concentrations in the samples submitted directly from the suppler were similarly elevated. The copper and molybdenum concentrations in the custom feed from the supplier were 368.7 ppm and 2.1 ppm, respectively. Concentrations were 16.2 ppm and 1.3 ppm, respectively, in the laboratory-grade diet fed at the research facility.
Copper Concentrations in Liver and Kidney (ppm).a
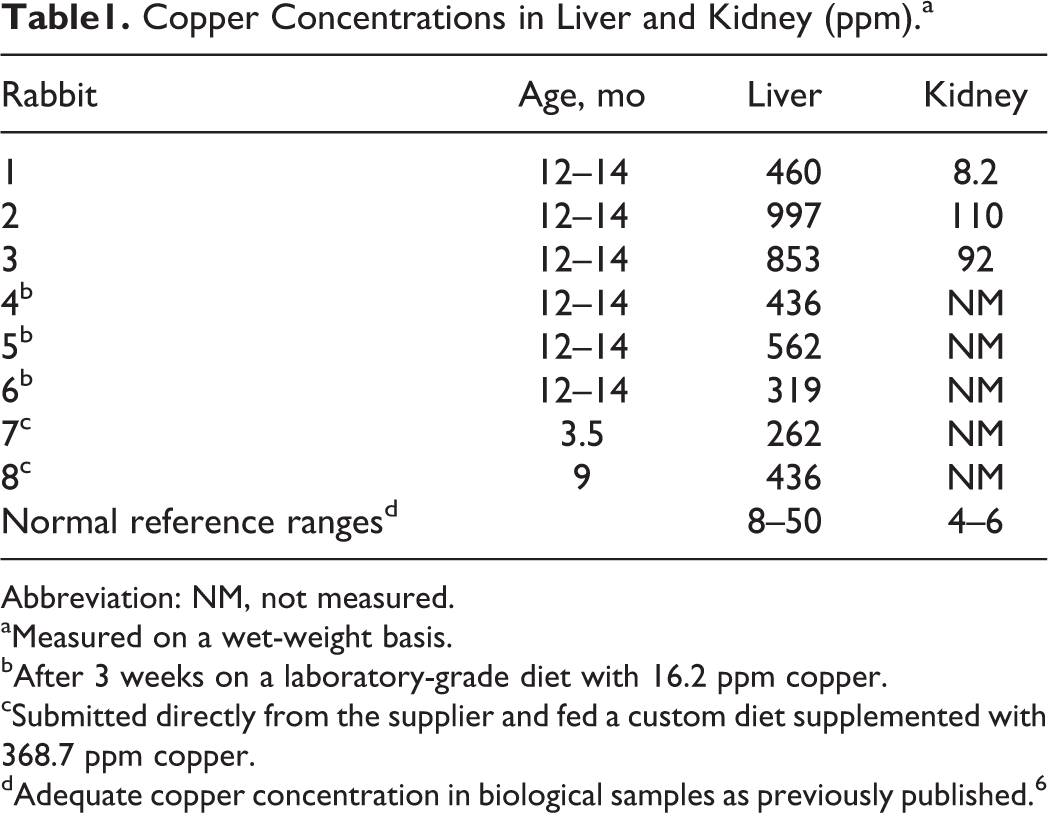
Abbreviation: NM, not measured.
aMeasured on a wet-weight basis.
bAfter 3 weeks on a laboratory-grade diet with 16.2 ppm copper.
cSubmitted directly from the supplier and fed a custom diet supplemented with 368.7 ppm copper.
dAdequate copper concentration in biological samples as previously published. 6
Discussion
Copper is an essential trace element that serves as a cofactor for enzymes involved in many biologic processes. Copper homeostasis is tightly regulated in the body because copper facilitates the generation of reactive oxygen species. Absorbed in the small intestines and transported in a protein-bound state in the blood to the liver, copper is stored within hepatocytes, secreted into plasma, or excreted in the bile. Excess copper will accumulate in hepatic lysosomes if the excess overwhelms biliary secretion. Over time, excessive accumulations cause hepatocellular cell membrane damage and death with release of copper stores into the bloodstream. If the liver is able to reabsorb the copper and compensate for the hepatic damage, the animal remains subclinical, but hepatic fibrosis and regeneration are evident microscopically.
A chronic form of copper toxicosis is described in ruminants. The “chronic” descriptor is based on the duration of copper exposure vs the clinical presentation. These animals remain subclinical until some stressor, such as pregnancy, disease, or transportation, triggers acute breakdown of copper-containing lysosomes in the liver. Hepatocellular cell membrane damage and death result in large amounts of copper being released into the bloodstream, causing methemoglobin formation, lipid peroxidation of erythrocyte cell membranes, and intravascular hemolysis. Severe acute anemia causes hypoxia, which further increases hepatocellular damage and release of copper into the bloodstream. Hemoglobinuric nephrosis can result from intravascular hemolysis, further exasperating the toxicosis. Affected animals usually exhibit sudden onset of depression, anorexia, and weakness with subsequent hemoglobinuria, icterus, and potential death within 1 to 2 days. 4,5,7 In this group of rabbits, the clinical progression and microscopic findings combined with hepatic and dietary copper concentrations are consistent with chronic copper toxicosis. Although hepatocellular copper accumulation is chronic in nature, the gross and microscopic lesions are consistent with acute hemolysis similar to copper toxicosis in sheep and goats.
Toxic levels for copper are not established in rabbits. In cattle, sheep, and goats, hepatic copper levels more than 250 ppm (wet-weight basis) are suggestive of toxicosis, and levels greater than 500 ppm are considered toxic in most mammalian species. 6 Rabbits are considered sensitive to copper, and toxicosis was previously reported at hepatic copper concentrations of 532 and 541 ppm (wet-weight basis). 2,6 According to our investigation, hepatic concentrations of 319 ppm with stress led to toxicosis.
Published recommended dietary copper levels for rabbits vary between 3 and 10 ppm (dry-weight basis), with higher levels, up to 25 ppm, recommended for breeding does and fur production. 3,6 In meat production, supplementation with levels ranging from 100 to 400 ppm (dry-weight basis) is used to increase growth performance. 3 In this report, the feed was supplemented with 368.7 ppm of copper (dry-weight basis), which is more than 30 times greater than recommended levels but within levels reported to enhance growth performance. Molybdenum enhances excretion of copper, and copper-to-molybdenum ratios (Cu:Mo) >20:1 result in severe hepatic copper accumulation in sheep. The Cu:Mo ratio in the supplier’s diet was 176:1. These rabbits likely had excessive hepatocellular accumulations of copper from high levels of copper supplemented in their feed. The copper stores were released during the stress of transport, handling, and abrupt diet change. Susceptible species, such as sheep, have decreased ability to excrete excess copper in the bile. 5 Further studies are necessary to determine if decreased biliary secretion of copper is associated with copper sensitivity in rabbits.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
