Abstract
Dendritic cells (DCs) are “professional” antigen-presenting cells with a critical role in the regulation of innate and adaptive immune responses and thus have been considered of great interest in the study of a variety of infectious diseases. The objective of this investigation was to characterize the in vivo distribution of DCs in bovine tissues by using potential DC markers to establish a basis for the study of DCs in diseased tissues. Markers evaluated included MHCII, CD208, CD1b, CD205, CNA.42, and S100 protein, the latter 2 being expressed by follicular dendritic cells whose origin and role are different from the rest of hematopoietic DCs. Paraffin wax–embedded tissues from 6 healthy Friesian calves were subjected to the avidin-biotin-peroxidase method, and the most appropriate fixatives, dilutions, and antigen retrieval pretreatments were studied for each of the primary antibodies. The most significant results included the localization of CD208-positive cells not only in the T zone of lymphoid organs but also within lymphoid follicles; CD1b-positive cells were mainly found in thymus and interfollicular areas of some lymph nodes; cells stained with anti–CD205 antibody were scarce, and their location was mainly in nonlymphoid tissues; and CNA.42- and S100 protein–positive cells localized in primary lymphoid follicles and light zones of germinal centers, although showing differences in the staining pattern. Furthermore, MHCII was established as one of the most sensitive markers for any DC of hematopoietic origin. These results increase our understanding of DC immunolabeling and will help in future DC studies of both healthy and diseased tissues.
Dendritic cells (DCs) are “professional” antigen-presenting cells (APCs) and are well known for their unique ability to present processed antigens to naive T lymphocytes. 20 In addition to their critical role in the regulation of the adaptive immune response, DCs serve as sentinels, recognizing the presence of invading pathogens and secreting proinflammatory cytokines involved in host defense and thereby linking both the innate and adaptive immunity. 35 These cells have a hematopoietic origin and express the major histocompatibility complex class II (MHCII) molecule. This is contrasted with the so-called follicular dendritic cells (FDCs) present in lymphoid follicles, which have a stromal origin, do not express MHCII, and present intact antigens to B cells in the form of surface immune complexes, being related to B-cell homeostasis. 39,41
Different authors have studied the role of DCs in various cattle diseases by means of in vitro infections of different subtypes of DCs, including monocyte-derived DCs 9,21,37 and afferent lymph DCs (veiled cells or ALDCs). 14,15 To our knowledge, this report is the first that describes in vivo distribution of DCs in bovine tissues on the basis of their differential expression of potential dendritic cell markers such as MHCII, CD208, CD1b, CD205, CNA.42, and S100 protein.
The MHCII molecule is expressed by professional APCs, which includes all subtypes of DCs as well as macrophages and B lymphocytes. APCs are responsible for T helper cell activation by means of a synapse established between the antigen-loaded MHCII molecule and the T-cell receptor. CD208, also known as DC-LAMP (dendritic cell–lysosome-associated membrane protein) was found to be specifically expressed in mature DCs located in T zones of lymphoid tissues, known as interdigitant dendritic cells. 6 This molecule is not present in any other cell type, with the exception of type II pneumocytes, 38 demonstrating the potential usefulness of this marker in diagnostics of DC-related pathology. CD1 molecules are a family of cell surface–associated glycoproteins now recognized as having a role in the presentation of lipid antigens to certain subpopulations of T cells, as MHC molecules do with protein antigens. 32 This family comprises several members, but not all are present in cattle. CD1b has been described as the most important CD molecule in this species. 36,42 The majority of CD1 molecules are strongly expressed in cortical thymocytes as well as in certain other cell types, to include DCs. 16 CD205, also known as DEC-205, is a C-type lectin that belongs to the same family of the macrophage mannose receptor and is primarily expressed on certain DCs and epithelial cells, acting as an endocytic receptor, and is involved in the capture of antigens from extracellular spaces and transferal to a specialized antigen-processing compartment. 8,18 In addition, bovine CD205 has been previously described as the WC6 antigen, showing a strong expression on ALDCs. 10 In this study, we used a monoclonal antibody (clone CNA.42) for the labeling of FDCs 34 and a polyclonal antibody for the detection of S100 protein, which is expressed in a wide variety of cell types, including FDCs. 17,23
Because of the complicated interactions between the distinct cells associated with both normal and pathologic mechanisms of action of the immune system, it is of critical interest to examine DCs not only in cell culture but also in vivo. This study aims both to standardize the immunohistochemical method for the detection of different potential DC markers but also to elucidate the histological distribution of these markers, some of which are commonly used in many in vitro studies, thus helping to expand the understanding of the various roles of DCs in different cattle diseases.
Materials and Methods
Six Friesian healthy male calves, aged 8 to 9 months, were obtained from farms free of tuberculosis, brucellosis, bovine leukosis virus, bovine viral diarrhea, and infectious bovine rhinotracheitis and were housed in the Animal Experimental Center of Cordoba University (Spain). Only those animals clinically healthy and with blood parameters within the normal range were included in the study. Parasitic coprological analyses were negative in all animals. Animals were sedated with xylazine (Rompun 2% solution; Bayer Healthcare, Kiel, Germany) and euthanized by overdose with thiopental-sodium (Thiovet; Vet Limited, Leyland, Lancashire, UK). This work was carried out in accordance with the code of Practice for Housing and Care of Animals used in Scientific Procedures, approved by the European Economic Community Union in 1986 (86/609/EEC) amended by the directive 2003/65/EC.
No significant macroscopic pathologic lesions were observed at necropsy. Samples from liver, intestine, and lung were subjected to microbiological routine cultures by using standard procedures, and no bacteria were isolated beyond the normal intestinal microflora. Tissue samples (0.5–1 cm thick) from a wide range of organs (Table 1) were immersed in 3 different fixatives: 10% neutral buffered formalin for 24 hours, Bouin’s solution for 8 hours, and zinc salts fixative for 24 hours. Samples were processed by routine methods and embedded in paraffin wax. Sections (4 μm) were stained with hematoxylin and eosin (HE) and examined microscopically.
Distribution of Cells Immunolabeled With Dendritic Cell Markers in Different Organs.
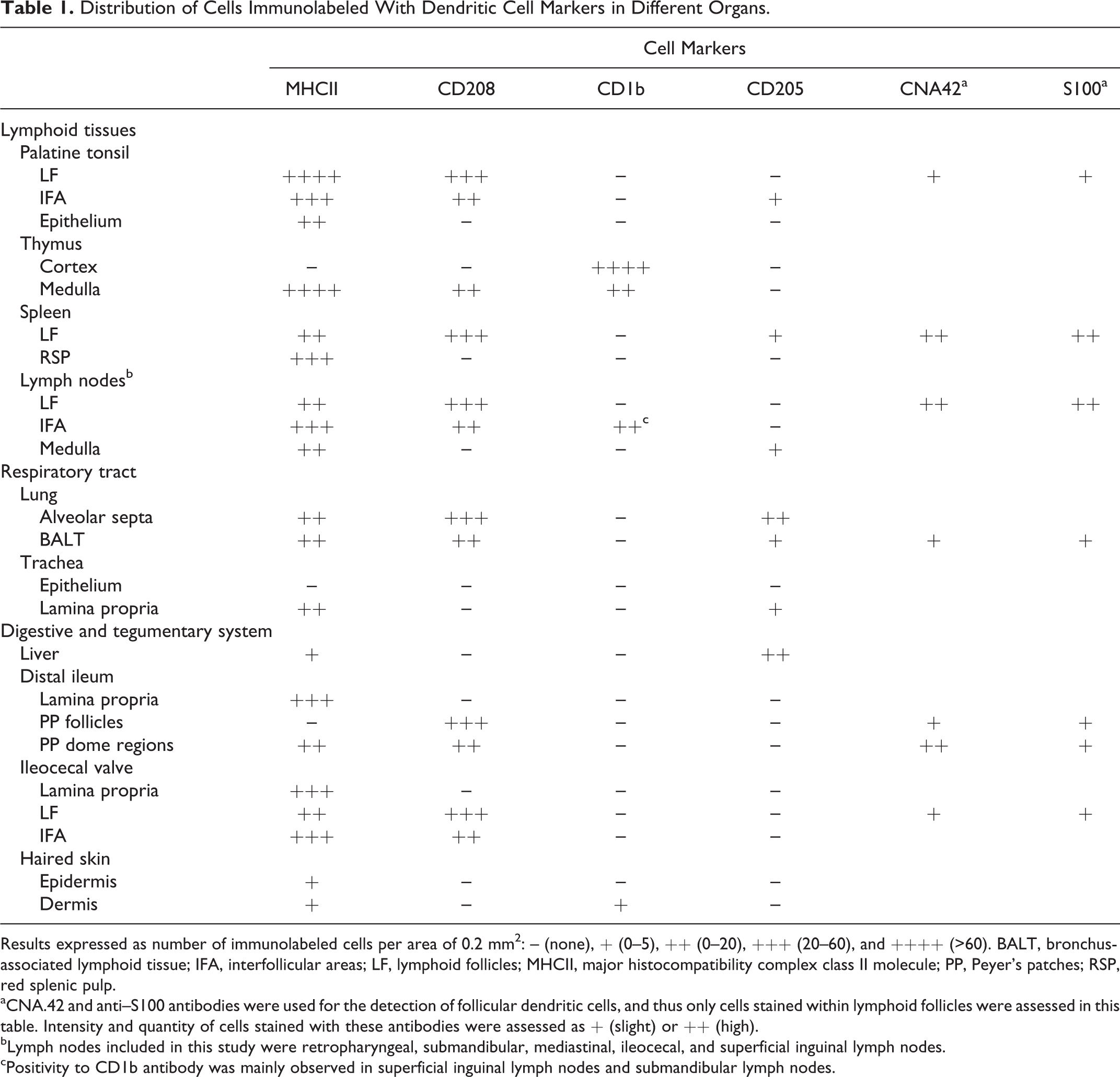
Results expressed as number of immunolabeled cells per area of 0.2 mm2: – (none), + (0–5), ++ (0–20), +++ (20–60), and ++++ (>60). BALT, bronchus-associated lymphoid tissue; IFA, interfollicular areas; LF, lymphoid follicles; MHCII, major histocompatibility complex class II molecule; PP, Peyer’s patches; RSP, red splenic pulp.
aCNA.42 and anti–S100 antibodies were used for the detection of follicular dendritic cells, and thus only cells stained within lymphoid follicles were assessed in this table. Intensity and quantity of cells stained with these antibodies were assessed as + (slight) or ++ (high).
bLymph nodes included in this study were retropharyngeal, submandibular, mediastinal, ileocecal, and superficial inguinal lymph nodes.
cPositivity to CD1b antibody was mainly observed in superficial inguinal lymph nodes and submandibular lymph nodes.
Two monoclonal antibodies (CD1b and CD205) directed against bovine antigens were used in this study. Anti–S100 antibody has been raised against S100 protein isolated from cow brain, and CNA.42 monoclonal antibody cross-reacts with FDCs in different species, including cattle. 34 Studies by Ababou et al 1 and Grüneberg et al 13 have demonstrated that the clone TAL.1B5 (anti–human HLA-DR α chain) also binds to an intracellular epitope of the BoLA-DR α chain. Given the close phylogenetic relationship between cattle and sheep, an anti–mouse CD208 antibody that cross-reacts with sheep 38 was used in this study (Table 2).
Primary Antibodies Used in the Present Study.
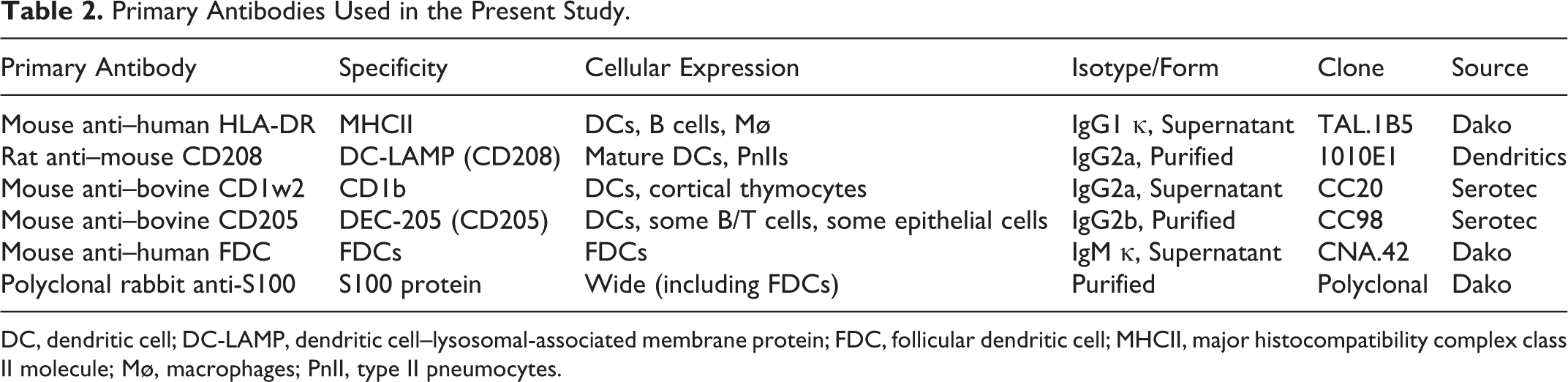
DC, dendritic cell; DC-LAMP, dendritic cell–lysosomal-associated membrane protein; FDC, follicular dendritic cell; MHCII, major histocompatibility complex class II molecule; Mø, macrophages; PnII, type II pneumocytes.
The avidin-biotin-peroxidase complex method was used. 30,31 Tissue sections (3 μm) were dewaxed and rehydrated using graded ethanol, and endogenous peroxidase activity was exhausted by incubation of the sections with H2O2 3% in methanol for 45 minutes at room temperature. Tissue sections were subjected to one of the following antigen retrieval pretreatments for the stated antibodies (Table 3): no pretreatment, when no antigen retrieval methods were performed; Tween 20 (Merck, Munich, Germany) 0.1% in 0.01M phosphate-buffered saline (PBS), pH 7.2 (10 minutes at room temperature); protease type XIV (Sigma-Aldrich Chemie, Steinheim, Germany) 0.1% in PBS (7 minutes at room temperature); and microwave heat–induced epitope retrieval (HIER) in 0.01M citrate buffer at different pH levels (3.2, 6, and 9), in addition to different time points from the onset of boiling. After pretreatment, sections were given three 5-minute rinses in PBS. For the primary antibodies MHCII, CNA.42, CD1b, and CD205, tissue sections were covered with 1% normal horse serum (Pierce-Endogen, Woburn, MA) in 0.05M Tris-buffered saline (TBS), pH 7.6, for 30 minutes at room temperature and incubated with the primary monoclonal antibodies at 4°C overnight. For the primary antibodies against CD208 and S100 protein, 1% normal rabbit serum and 20% normal goat serum, respectively, replaced normal horse serum. Details of the commercial primary antibodies tested in this study are summarized in Table 2. After primary incubation, slides were washed in PBS (3 times for 5 minutes each) and then incubated with the secondary antibodies for 30 minutes at room temperature. Biotinylated horse anti–mouse IgG secondary antibody (Pierce-Endogen) diluted 1:200 in TBS containing normal horse serum 1% was used for the primary antibodies MHCII, CNA.42, CD1b, and CD205. Biotinylated rabbit anti–rat Igs secondary antibody (Dako, Glostrup, Denmark) diluted 1:100 in TBS containing normal rabbit serum 1% was used for the primary antibody against CD208. Biotinylated goat anti–rabbit IgG secondary antibody (Vector Laboratories, Burlingame, CA) diluted 1:200 in PBS containing normal goat serum 1.5% was used for the polyclonal primary antibody anti-S100. After 3 further 5-minute washes in PBS, samples were incubated with the avidin-biotin-peroxidase complex (Vectastain ABC Kit Elites; Vector Laboratories) for 1 hour at room temperature in the dark. All tissue sections were finally rinsed in TBS, and labeling was “visualized” by application of a chromogen solution (NovaRED Substrate Kit; Vector Laboratories). Slides were counterstained with Mayer’s hematoxylin. Mouse or rat nonimmune sera were used in place of specific monoclonal primary antibodies as negative controls.
Immunoreactivity Produced by Various Primary Antibodies on Calf Tissues Fixed in Formalin Solution and Subjected to Various Antigen-Retrieval Pretreatments.
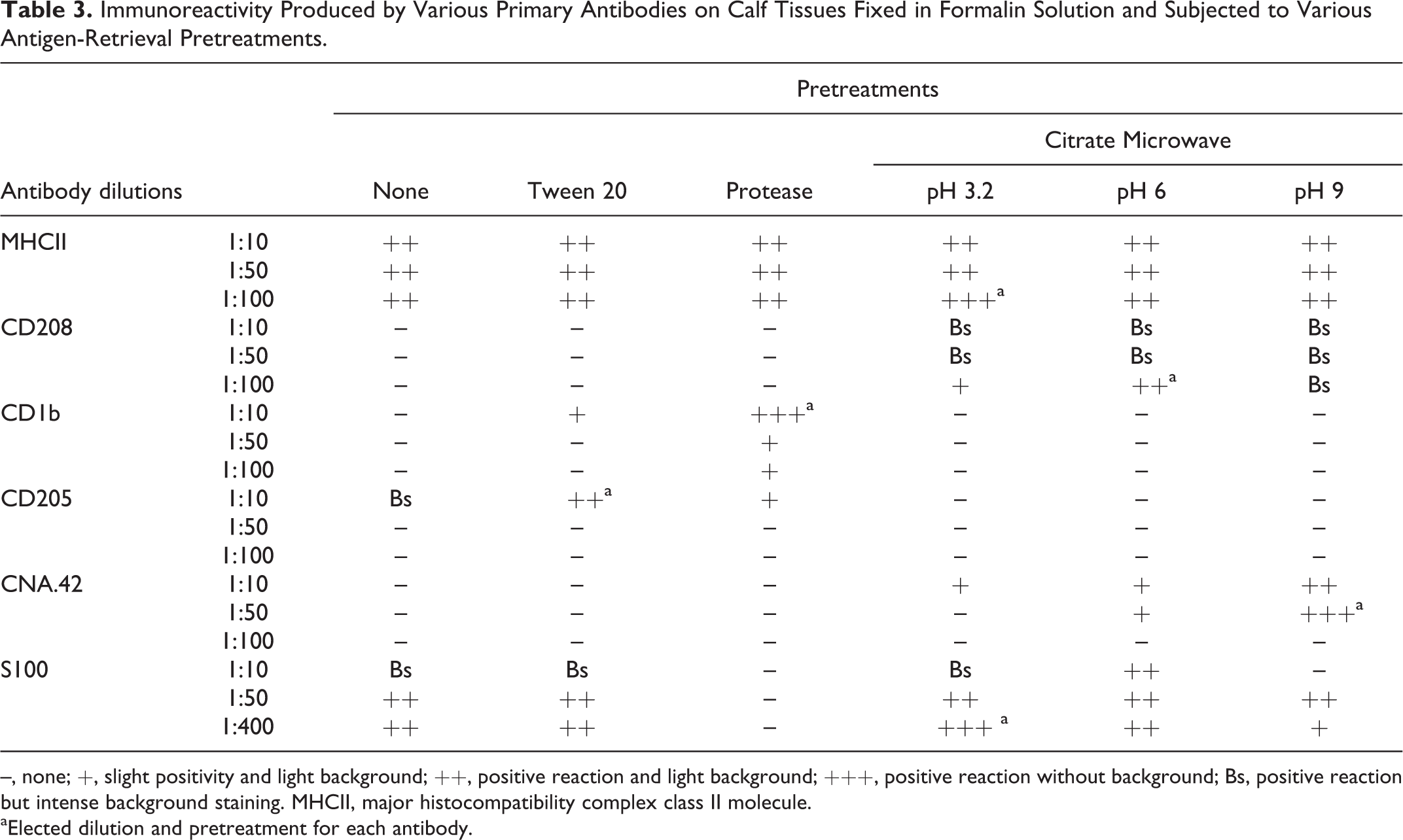
–, none; +, slight positivity and light background; ++, positive reaction and light background; +++, positive reaction without background; Bs, positive reaction but intense background staining. MHCII, major histocompatibility complex class II molecule.
aElected dilution and pretreatment for each antibody.
A semiquantitative assessment of the immunolabeled cells was performed by 2 experienced observers in 25 fields of 0.2 mm2 randomly chosen. Results were expressed as number of immunolabeled cells per area of 0.2 mm2: – (none), + (0–5), ++ (0–20), +++ (20–60), and ++++ (>60). Intensity and quantity of cells stained with CNA.42 and anti–S100 antibodies were assessed only within lymphoid follicles as + (slight) or ++ (high) (Table 1).
Results
Tissues used in this study were assessed by a board veterinary pathologist who certified the absence of histopathological lesions. Signs of inflammation indicative of infectious or toxic agents were not observed in any of the animals included in the study.
Optimization of the Immunohistochemical Method
Despite using other fixatives such as Bouin’s solution or zinc salts fixative, the best results were obtained with buffered formalin. Tissue expression of the MHCII molecule was observed with each of the different antibody concentrations as well as with the different unmasking pretreatments, although the higher dilution (1:100) and the unmasking method with citrate at pH 3.2 during 6 minutes at subboiling temperature were considered the most effective (Table 3). Immunolabeling against CD208 was possible with the higher dilution (1:100), although only when using the HIER methods at pH 6 during 20 minutes at subboiling temperature. This technique appeared to be the most appropriate, since staining using pH 3.2 was too weak, and pH 9 gave rise to intense background staining. The most suitable antigen retrieval method for the CD1b molecule was enzymatic digestion with protease but required the highest concentration of anti–CD1b antibody (1:10) to obtain the best results. In the case of anti–CD205 antibody, the only dilution that yielded positive results was the most concentrated (1:10), with the HIER methods being ineffective and the detergent permeabilization with Tween 20 being considered the most appropriate. For the detection of FDCs with CNA.42 monoclonal antibody, use of the HIER methodology was necessary, with the best results obtained at pH 9 during 30 minutes at subboiling temperature and with a 1:50 antibody dilution. S100 protein expression was observed with all of the unmasking methods except for enzymatic digestion, although the best results were obtained with citrate at pH 3.2 during 6 minutes at subboiling temperature and with a 1:400 antibody dilution.
Quantification and Location of Immunolabeled Cells
Immunolabeled cells were counted for each animal, with minor differences among animals being observed and included within the stated intervals (Table 1).
MHCII followed by CD208 appeared as the primary molecules expressed in the different tissues, both in intensity of labeling and quantity of labeled cells. All lymphoid organs displayed a high amount of MHCII-positive cells. These variably sized cells were typically stellate or polygonal and had homogeneous cytoplasmic staining. Immunopositive cells in palatine tonsil and lymph nodes could be clearly differentiated from one another in the interfollicular areas (Fig. 1). A general faint staining was observed in the majority of lymphoid follicles, and in some of these lymphoid follicles, more strongly immunostained cells were noted and mainly located in the lymphoid follicle light zones (Figs. 1, 12a). Immunolabeled cells were also observed in the medulla of lymph nodes, although to a lesser degree in comparison with the cortex. The cells identified within the medullary regions had a very characteristic dendritic morphology (Fig. 2). Immunostaining observed in the spleen was evident in red splenic pulp and periarteriolar lymphoid sheaths, displaying many immunopositive dendritic-shaped cells (Fig. 3), in contrast to lymphoid follicle staining being weaker if more diffuse. In the thymus, immunolabeling against MHCII was restricted to the medulla, where strong immunoreactivity was observed, including many cells with a dendritic morphology (Fig. 4). Rare immunopositive cells were observed in the pulmonary alveolar septa and periportal areas of the liver. Positive cells were also noted within the tonsillar epithelium and, to a lesser degree, in the epidermis and dermis of the skin, frequently showing dendritic morphology (mainly in tonsillar crypts epithelium) (Fig. 5). MHCII-positive cells in the distal ileum were located in the lamina propria as well as within the dome region of the Peyer’s patches but not within Peyer’s patches follicles (Fig. 6a), in contrast to the immunopositivity observed in the ileocecal valve lymphoid follicles.
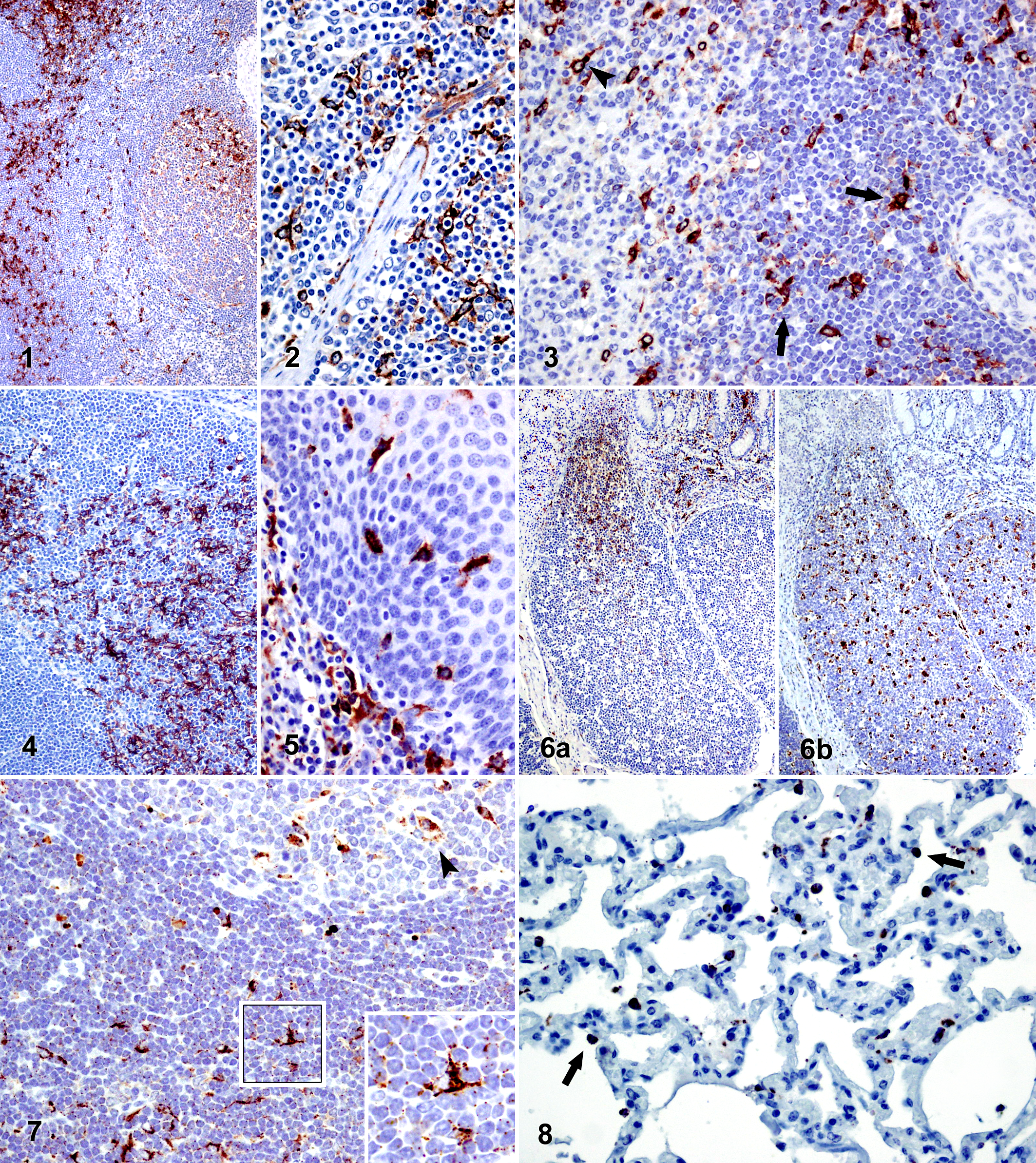
Staining against CD208 (DC-LAMP) was cytoplasmic and had a dark granular appearance. Positive cells were mainly stellate or polygonal and were widely distributed in lymphoid tissues. A slight generalized staining was observed as tiny dark dots that could be appreciated on the cell surfaces (Fig. 7, inset). This was more evident as the pH of the HIER method was increased. Immunopositive cells were located in the interfollicular areas and, to a greater extent, within the lymphoid follicles (Figs. 7, 12b), where the cells were homogeneously distributed, with no differences observed between the dark and light zones of the germinal center, unlike the differences noted with anti–MHCII and anti–FDC antibodies. In general, not all the lymphoid follicles of the same lymph node presented the same quantity of immunopositive cells, and the medulla of lymph nodes was observed to be free of CD208-positive cells. Immunostained cells against CD208 had a round morphology and were located on the alveolar surfaces in the lung (Fig. 8). A moderate amount of immunolabeled cells that were predominantly stellate was identified in the periarteriolar lymphoid sheaths of the spleen and within the thymic medulla. In addition, numerous large round immunopositive cells were detected within the ileum Peyer’s patches and the lymphoid follicles of the ileocecal valve (Fig. 6b).
Very few organs were noted to have CD1b-immunopositive cells. Cortical thymocytes were uniformly immunopositive along with some cells with a dendritic morphology located in the thymic medulla (Fig. 9). Most lymph nodes were immunonegative for anti–CD1b antibody, with the exception of the submandibular and superficial inguinal lymph nodes and, to a lesser extent, the mediastinal lymph nodes, where numerous dendritic-shaped immunolabeled cells were identified in the interfollicular areas. Some scattered immunopositive cells were also demonstrated in the dermis of the skin.
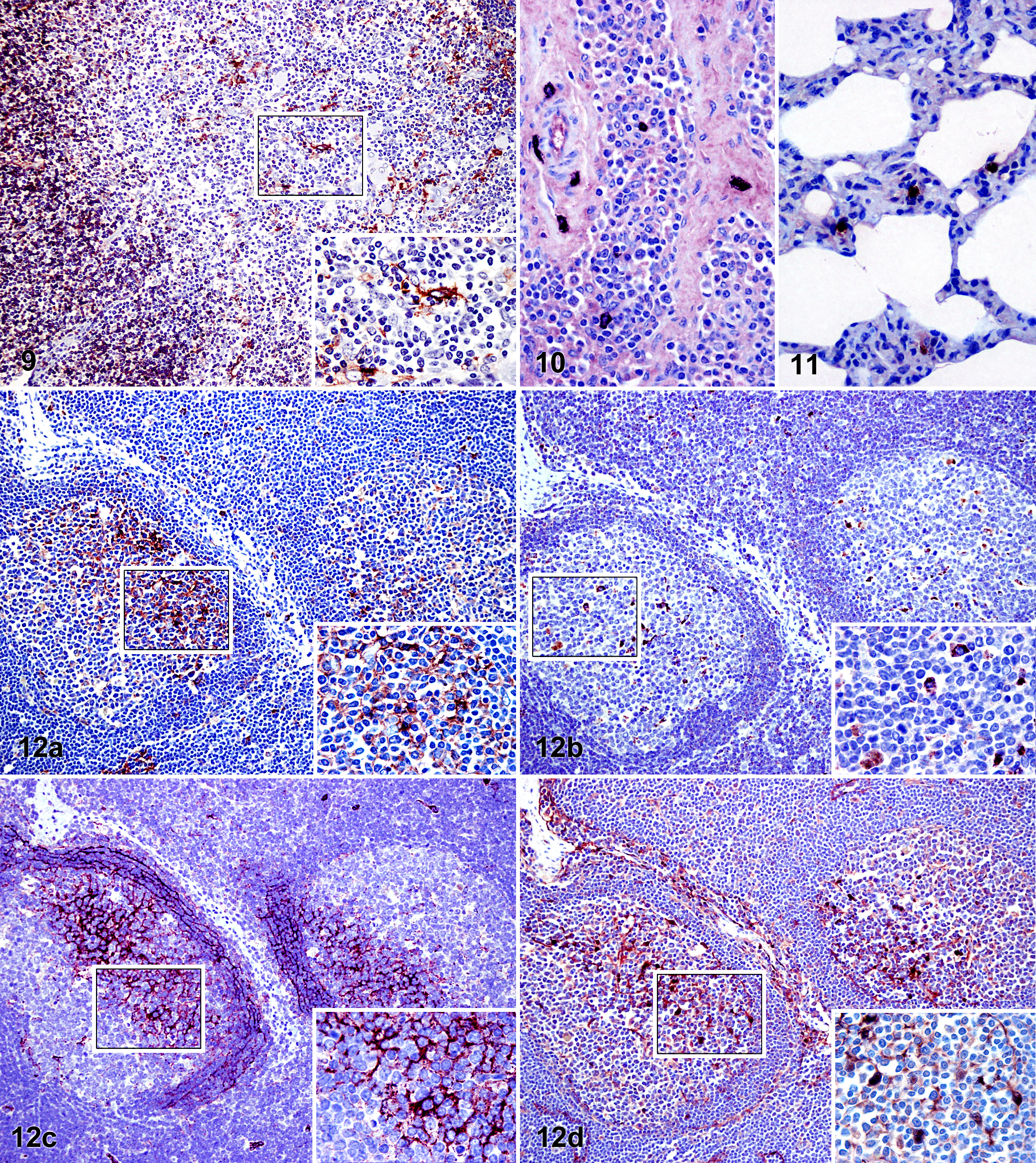
Immunolabeling against CD205 demonstrated a granular appearance, located in the cytoplasm of cells with either round or spindle morphology. In general, few immunolabeled cells were observed by using this antibody, and these were located in uncommon regions such as in tissue adjacent to tonsil mucous glands or to large blood vessels (data not shown), in trabeculae of lymphoid organs, or in the connective tissue of hepatic portal spaces, central veins, and Glisson’s capsule. It was difficult to find any immunopositive cells within the interfollicular areas or the lymphoid follicles (Fig. 10). A greater number of immunolabeled cells were identified in the lung, located in the alveolar septa, the pulmonary pleura, or surrounding bronchioles and arteries (Fig. 11).
Use of the CNA.42 antibody yielded a staining pattern in the form of a network distributed among lymphocytes of the primary lymphoid follicles, as well as the light zones and mantle zones of germinal centers (Fig. 12c). Curiously, the lymphoid follicles of palatine tonsil, Peyer’s patches, and ileocecal valve demonstrated only a scarce staining intensity, unlike the stronger labeling intensity noted in the lymphoid follicles of the examined lymph nodes and the spleen. Expression of S100, also observed in the primary lymphoid follicles and the light zones of the germinal centers, showed organic differences similar to those observed with CNA.42, although staining against S100 was located in both the cell cytoplasm and the nucleus (Fig. 12d). Both antibodies (CNA.42 and anti-S100) yielded immunostaining of cell types other than cells located within the lymphoid follicles, which are not included in the aims of this study.
Discussion
It is well known that DCs are a heterogeneous cell population categorized according to their location, functionality, and cell marker expression. The present study has focused on the comparison and characterization of the location of dendritic cells of Friesian calves based on their expression for different cellular markers. This was accomplished by using fixed and paraffin wax–embedded tissues, which preserve an optimal cell morphology, in comparison with frozen tissues, in which there can be a significant loss of morphology, making the identification of a precise location of the antigen-expressing cells within the tissue much more difficult. 2,5,33 Furthermore, frozen tissue handling may involve a higher degree of difficulty to produce cryostat sections of consistent quality.
The detection of the MHCII molecule is frequently conducted in tissue sections, although the vast majority of these studies are based on frozen tissue sections and typically for purposes other than detecting DCs. 24,25 The novel information presented by this study employed the MHCII molecule to describe the type of immunolabeled cells within a wide variety of organs, thereby providing a standardization of the immunohistochemical methodology, as well as taking advantage of the fact that DCs are strong expressors of this molecule and that fixed paraffin-embedded tissues preserve the characteristic morphology of these cells, allowing for a mapping of their localization within select organs. To our knowledge, this is the first report of the use of this list of antibodies in the detection of DCs in bovine paraffin-embedded tissues.
This study revealed that of the antibodies examined, the MHCII antibody yielded the greatest quantity of labeled cells in the different tissues analyzed. This is due to the expression of the molecule not only in DCs but also in macrophages and certain populations of B cells, as well as being inducible in endothelial cells. Despite this variety of positive cells, use of this antibody also permits the detection of all DC populations, unlike other markers that detect only certain subpopulations. A striking finding in the results obtained with the anti–MHCII antibody was the marked staining polarization in most of the germinal centers of secondary lymphoid organs, which coincided to a great extent with the immunolabeling observed with the use of CNA.42 and anti–S100 antibodies, which was restricted solely to the light zone (and adjacent mantle zone in the case of CNA.42) of the germinal centers. This observation was possible by using serial sections of the tissue immunostained with the antibodies of interest (Fig. 12a,c,d). It is known that FDCs do not internalize or present processed protein antigens in the context of MHCII molecules, 41 a fact that would establish as incompatible the coincidence in location of FDCs in light zones with the strong expression of MHCII. However, it has been shown that this cell type can acquire MHCII molecules not expressed by FDCs themselves, 7 which may explain why a strong immunostaining against MHCII in light zones can be observed, where FDCs are located. Human studies have demonstrated the CNA.42 antibody as an immunomarker for FDCs for each level of maturation, ranging from FDC precursors to activated FDCs. 19 Furthermore, the S100 protein is a molecule expressed by activated FDCs, which may explain why CNA.42 yielded a network staining pattern that was widely distributed and anti-S100 gave rise to the staining of individual cells.
The present study describes for the first time in bovine tissue samples the expression of CD208 (DC-LAMP), known as an exclusive marker for mature DCs in T regions (interdigitant DCs) and type II pneumocytes. 6,38 CD208 expression in alveolar surfaces coincides with the previously described expression of this molecule in type II pneumocytes. However, we unexpectedly found CD208-positive cells not only in interfollicular areas but also within lymphoid follicles; given the known exclusive expression of CD208 to DCs in lymphoid organs, these immunopositive cells found within lymphoid follicles might be considered the previously described germinal center DCs (GCDCs). 11,12 However, this consideration would be mistaken since GCDCs, whose origin is hematopoietic, should express MHCII, a fact that was ruled out by using serial sections immunostained with both antibodies (Figs. 6, 12a,b); CD208-positive cells were located both in MHCII-positive light zones and MHCII-negative dark zones. The typical follicular location, round morphology, and large size suggest that these CD208-positive cells are in fact tingible body macrophages.
Numerous MHCII-positive cells were detected in the thymic medulla, most of them having a stellate shape compatible with that of DCs, which have significant importance in both positive and negative selection during T-cell development. 3 In this same location, we also observed dendritic-shaped cells immunolabeled against CD208, albeit to a lesser extent. This observation may be explained by the fact that during maturation, DCs acquire a higher capacity to form and accumulate MHCII-peptide complexes, a process that requires a generalized activation of the lysosomal function; 40 the CD208 molecule is included in the lysosomal-associated membrane protein family, which is why these CD208-positive cells in thymic medulla may represent a population of mature DCs, which corresponds to similar descriptions in humans. 4
Langerhans cells (LCs) were originally described as immature DCs present in the epidermis, although currently this term has been generalized to include DCs present in all surface-stratified epithelium. 27 This may support the hypothesis that considers MHCII-positive cells observed in tonsillar epithelium as LCs (Fig. 5), as these demonstrated stronger immunopositivity and were more numerous in comparison to those of the epidermis, further highlighting the importance of DCs in the tonsil due to its strategic location in the entrance of numerous airborne pathogens.
It is well established that DCs are present within the dome regions of the intestinal lymphoid tissue, 28 coinciding with the presence of stellated MHCII-positive cells that we found in those intestinal sections examined. However, Peyer’s patches follicles barely stained against the MHCII molecule. In ruminants, ileal Peyer’s patches are considered primary lymphoid organs, 22 in which there is no germinal center reaction as observed in jejunal Peyer’s patches, 43 which may explain the lack of MHCII expression at this level, similar to that which occurs in the cortex of thymus, another primary lymphoid organ. However, these MHCII-negative Peyer’s patches follicles demonstrated a great quantity of widely distributed CD208-immunopositive cells, supporting the hypothesis that considers these cells to be tingible body macrophages.
Previous studies have determined the phenotypic characteristics of both intestinal and respiratory tract DCs in sheep 2,26 by using antibodies against CD205 and CD1b molecules. ALDCs are strong expressors of CD205, 10 and other tissues have been shown to express this molecule as well. 10,29 Contrary to results obtained by Akesson and collaborators 2 in sheep, we did not find cells expressing CD205 in the ileum in this study of Friesian cattle. However, a considerable amount of CD205-immunolabeled cells were detected in the parenchyma and pleura of the lung, which is consistent with other ovine studies. 26 In comparison with other DC markers, very few cells demonstrated immunolabeling against CD1b in the intestine and the lung of sheep. 2,26 Likewise, our results demonstrated that both organs were negative to the expression of CD205 and CD1b. Antibodies for detecting CD205 and CD1b used in this study and in ovine studies from Akesson et al 2 and McNeilly et al 26 are bovine specific. Despite this fact, the absence of expression in this study of CD1b in the intestine and the respiratory track and of CD205 in the intestine suggests that the observed species differences between sheep and cattle are due to differences in the technique sensitivity employed; ovine studies have been carried out in frozen tissues, whereas this study used tissue samples that had been fixed and embedded in paraffin wax. The latter technique permits an optimal preservation of the cellular morphology and tissue architecture, although it may mask or alter the 3-dimensional structure of antigens, which is why antigen retrieval methods are required. 33
In summary, despite the existence of other molecules displayed by DCs, the recognition of MHCII expression appears to be one of the most sensitive methods for the detection of any DC of hematopoietic origin in tissue sections. The staining pattern observed with the monoclonal antibody detecting CD208 suggests that the expression of this molecule in bovine lymphoid tissues is restricted not only to interdigitant DCs but also to tingible body macrophages, a finding that has not been previously described. Although further studies will be needed to confirm this novel finding, this study has demonstrated that CD208 detection allows for the differentiation of mature DCs from all other DCs, a fact of significant interest in the diagnostics and study of infectious diseases. This study further supports the tissue expression of CD1b in dendritic-shaped cells, even though these cells comprised a reduced population out of the total number of the existing DCs and were located in specific sites such as the thymus. Therefore, although global utility is limited, CD1b will provide valuable information on the role of thymic DCs, which is of significance due to the organ’s importance in the central immunotolerance. Both CNA.42 and anti–S100 antibodies stained FDCs, but only the latter allowed for the identification of FDCs as isolated cells. Taken together, these results provide a useful general view of the different staining patterns of potential DC markers and will help in future DC studies with pathologic tissues.
Footnotes
Acknowledgements
We thank the farm “Las Rozuelas del Valle” (Torrecampo, Spain) for providing the animals.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a grant from the Andalusian Regional Government-ERDF (P09-AGR-4671). F. Romero-Palomo was supported by a predoctoral grant associated with project P09-AGR-4671.
