Abstract
Matrix metalloproteinases (MMPs) are proteolytic enzymes involved with extracellular matrix degradation. They have been considered to be important for tumor growth and development of peritumoral edema. This retrospective study investigated the expression of MMP subtypes 9 and 2 in canine intracranial meningiomas and their association with peritumoral edema. Twenty-two cases of histologically confirmed grade I meningiomas based on human World Health Organization classification were enrolled. Tumor volume and peritumoral edema were measured by magnetic resonance imaging volumetry. The intratumoral MMP expression was semiquantitatively assessed by immunoreactivity scores and compared with the imaging data. MMP-9 was expressed in all the samples (22/22), whereas proMMP-2 was expressed in 21 of 22 meningiomas, and a/proMMP-2 was expressed in 9 of 22. The immunoreactivity scores were not statistically linked to the severity of peritumoral edema. None of the evaluated MMP expression parameters were statistically linked to the edema index. Although both edema index and MMP-9 expression were highest in meningiomas of the olfactory and frontal region, only the latter mounted up to statistical significance (P = .002) if compared with parafalx and convexity meningiomas of the parietal lobe. In summary, MMP-2 and MMP-9 expression by tumor cells, evaluated through immunohistochemistry, is not predictive of the formation of peritumoral edema in canine rostrotentorial meningiomas.
Matrix metalloproteinases (MMPs) comprise a family of zinc-dependent endopeptidases, capable of degrading components of the basement membrane and extracellular matrix (ECM). 5 Alteration to their normal expression can occur in a wide range of physiological adaptations and pathological conditions. 31
Tumor cells express MMPs, including MMP-2 and MMP-9, by direct contact with the stroma or as a response to soluble factors. Matrix metalloproteinases are also expressed within the surroundings of the tumor by fibroblasts, infiltrating immune cells and endothelial cells. Matrix metalloproteinases are involved at multiple stages of tumor progression, promoting establishment, tumor cell exfoliation, and invasive growth of a neoplasm, tumor angiogenesis, and metastasization. 17,31 Therefore, high MMP expression has been related to poor clinical outcome. 19 –21
Furthermore, MMPs have been implicated in the development of peritumoral edema (PTE) in brain tumors. 19 –21
Since both enzymes specifically degrade the basement membrane component collagen IV, their upregulation is thought to disrupt the blood-brain barrier (BBB). 7 This concept held true for MMP-9 expression that was positively correlated to the volume of PTE in a group of human meningioma patients. In contrast, no such correlation was seen with MMP-2 expression in the same tumors. 19
The peritumoral brain tissue exhibits significant edema in the majority of canine meningiomas. 12,28,30 The associated rise of intracranial pressure and mass effect markedly contribute to morbidity and mortality of the affected canine patients. 6,27 A vasogenic origin (in terms of BBB breakdown with extravasation of serum and leakage into the extracellular spaces) is implicated as the main cause of edema formation. 26,32 The BBB is composed of endothelial cells, pericytes, and ECM. Brain capillary endothelial cells are closely connected by tight junctions (TJs), which limit the paracellular pathway for solute diffusion. 26 The preservation of the TJs depends on 3 essential transmembrane proteins: claudin, occludin, and junction adhesion molecules. 26 A recent study provided direct evidence that MMP- 2 and MMP-9 open the BBB by degrading the TJ proteins occludin and claudin-5, 33 although there was no evidence that the neoplastic meningothelial cells resemble the direct source of these effector molecules. Endothelial cells and pericytes of the brain are surrounded by ECM proteins, such as collagen type IV, laminin, fibronectin, elastin, and various proteoglycans, which also are susceptible to enzymatic degradation. Hence, MMP-2 and MMP-9 have been the focus of several studies on PTE in brain tumors due to their substrate specificity for fibronectin, laminin, and collagen type IV. 15,19 Broad interpretation of the influence of MMPs on progression and morbidity in intracranial meningiomas has been somewhat skewed by the overall strong expression in higher grade meningiomas. Benign meningiomas appear to exhibit a larger variability of MMP expression, independent of the assessment method used, even though low-grade tumors are more prevalent and PTE resembles a considerable risk factor for patient survival. 3,8,19,20
Also in dogs, rostrotentorial meningiomas resemble the most common intracranial neoplasia, and benign meningiomas are the most representative. 27 Ninety-four percent of canine intracranial meningiomas (CIMs) are associated with PTE and have similar clinical relevance independent of the tumor subtype. 12,28,30 Despite this high prevalence, the mechanism(s) via which PTE develops in these cases remains unclear. Several investigations documented MMP-2 and MMP-9 expression in various canine tumors, 1,15,18 including a number of intracranial meningiomas. 15 However, this latter study focused on their relation to tumor cell proliferation defined by proliferating cell nuclear antigen (PCNA) expression and Ki-67.
The aims of this particular study were to investigate MMP-2 and MMP-9 expression in canine rostrotentorial grade I meningiomas and to clarify their involvement in the development of PTE to identify targets for new therapies. We also assessed whether there is an association between MMP expression and tumor location, tumor volume, or histologic subtype.
Materials and Methods
Case Selection and Data Acquisition
This retrospective study used imaging data and tissue samples from CIM cases presented to the Animal Health Trust from 2000 to 2005. Only rostrotentorial grade I meningiomas for which the amount and preservation of tissue were sufficient to conduct the whole panel of immunohistochemical procedures as described below were included. The clinical data set was analyzed for availability of interpretable magnetic resonance (MR) images. Dogs treated with corticosteroids prior to MR imaging were excluded from the study to avoid possible effects of this treatment on PTE. Tumor location was classified as olfactory bulb/frontal lobe, parietal lobe, occipital lobe, or temporal lobe.
Magnetic Resonance Volumetry
Volume measurements employed voxel-based MR morphometry using a 1.5 Tesla scanner (Sigma Echospeed System; General Electric Medical System, Milwaukee, WI).
Tumor volume (Vt) was measured, as previously described, 16 using a commercial software (Able software 3D-doctor; Able Software Corp 5, Lexington, MA), which allows the estimation of volumes outlining the tumor from stacks of consecutive transverse gadolinium-enhanced (0.05 mmol/kg, gadobenate dimeglumine) T1-weighted images (Fig. 1).
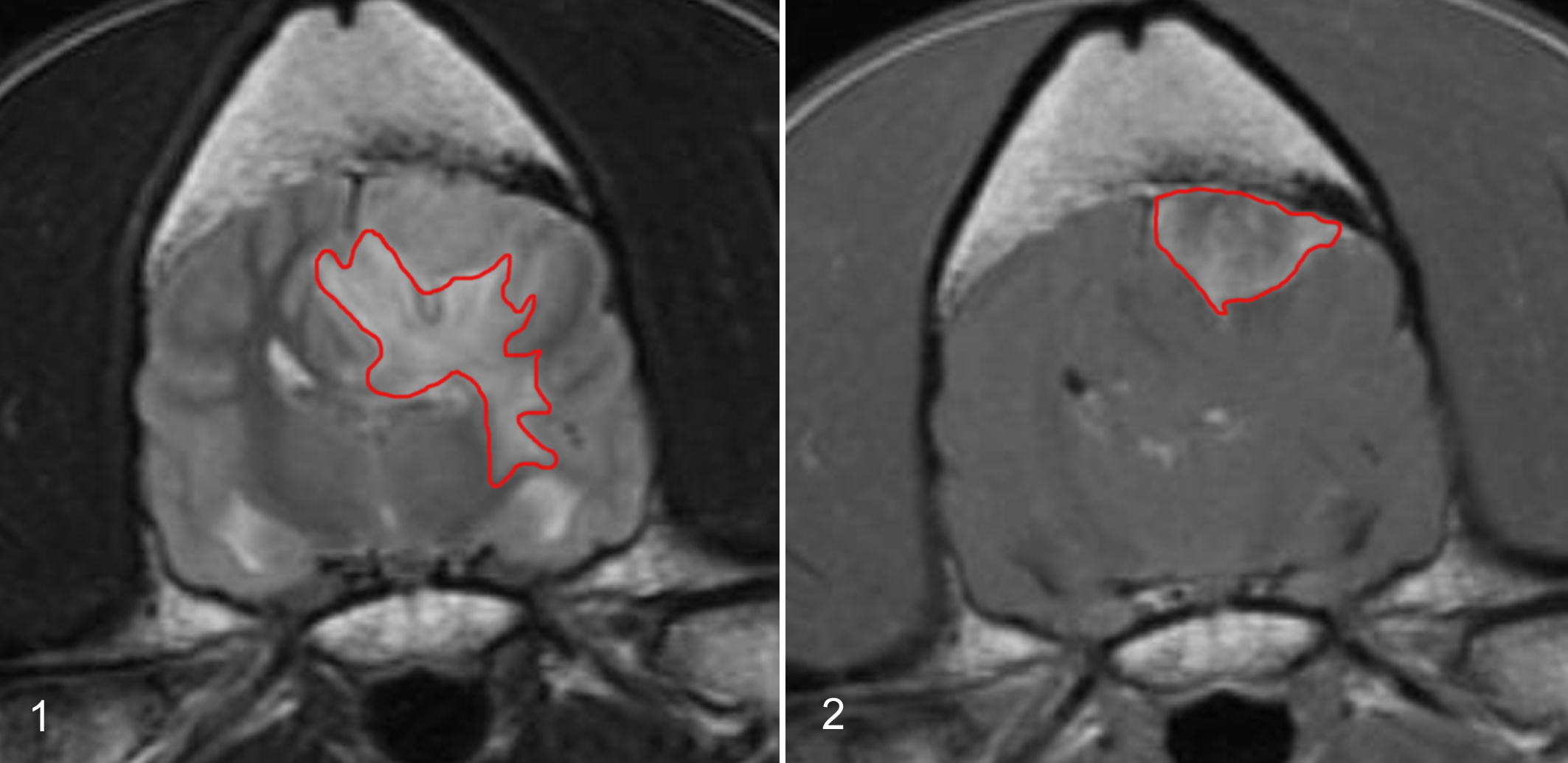
The edema volume (Ve) was measured based on the high signal intensity area around the tumor in T2-weighted images (Fig. 2) as previously described. 16 For all volume calculations, the field of view, matrix, slice thickness, and interslice thickness were taken into account. The normalized PTE was defined by the edema index (EI), which was calculated as the ratio of Ve to Vt. 20
Histopathology and Immunohistochemistry
All tumors fulfilling the inclusion criteria were reviewed by 2 investigators. Applicability of grade I (benign) was evaluated in accordance to histologic guidelines recommended by World Health Organization (WHO) Classification of Tumors of the Nervous System in humans. 10 Features that characterized grade I meningiomas included the presence of either ≤4 mitotic figures/10 high-power fields with each field representing 0.16 mm 2 or exhibiting no more than 2 of the following features: spontaneous necrosis, small cells, increased cellularity, nuclear atypia, or sheet-like growth patterns. 10
On the basis of their histologic appearance, meningiomas were further subclassified as meningotheliomatous, fibroblastic, transitional, psammomatous, angioblastic, papillary, granular, or myxoid. 11 Papillary meningiomas were included in the study only if their histologic appearance complied with the WHO criteria for benign grade I meningiomas. 10 We are aware that in humans, papillary meningiomas inflict a greater likelihood of recurrence and/or aggressive behavior 10 and that classification as grade I tumor in dogs 11 may underestimate the degree of malignancy if not individually inspected via histology.
Five-micrometer sections were taken from each paraffin block. Immunohistochemical staining for MMP-9 immunoreactivity was performed by a polyclonal antiserum (1:100, rabbit anti–MMP-9, cat. Nr Rb-9234; Thermo Scientific, Fremont, CA). For assessment of MMP-2 expression, 2 monoclonal antibodies were used. The first (mouse anti–MMP-2 ab1, 1:250, clone CA-4001; Thermo Scientific) was directed against the latent proMMP-2, whereas the second antibody was directed against both latent and active a/proMMP-2 (mouse anti–MMP-2 ab4, 1:100, clone A-Gel VC2; Thermo Scientific).
Antigen retrieval was performed by microwave treatment (600 W) for 20 minutes in preheated pH 6.0 citrate buffer. After blocking the activity of endogenous peroxidases, sections were incubated for 30 minutes with normal goat serum (1:20; Vector Laboratories, Burlingame, CA). The slides were incubated with a biotinylated goat anti–rabbit Ig-antibody (1:200, BA-1000; Vector Laboratories) for MMP-9 and goat anti–mouse Ig-antibodies (1:200, BA-9200; Vector Laboratories) for both MMP-2 assays.
Then, an avidin-biotin kit was applied according to manufacturer guidelines (Vectastain ABC-Kit, VC-PK-4000; Vector Laboratories). Enzyme labeling was visualized by incubation with the chromogen 3,3′-diaminobenzidine (Kem-En-Tec Diagnostics, Copenhagen, Denmark). Nuclear counterstaining was achieved by Mayer’s hematoxylin. Negative control preparations involved omitting primary antibodies and their replacement with antibody diluent. Tissue sections of a canine osteosarcoma were used as positive controls for both enzymes. 14
Expression of the MMPs within the tumor was semiquantitatively assessed by 2 blinded observers.
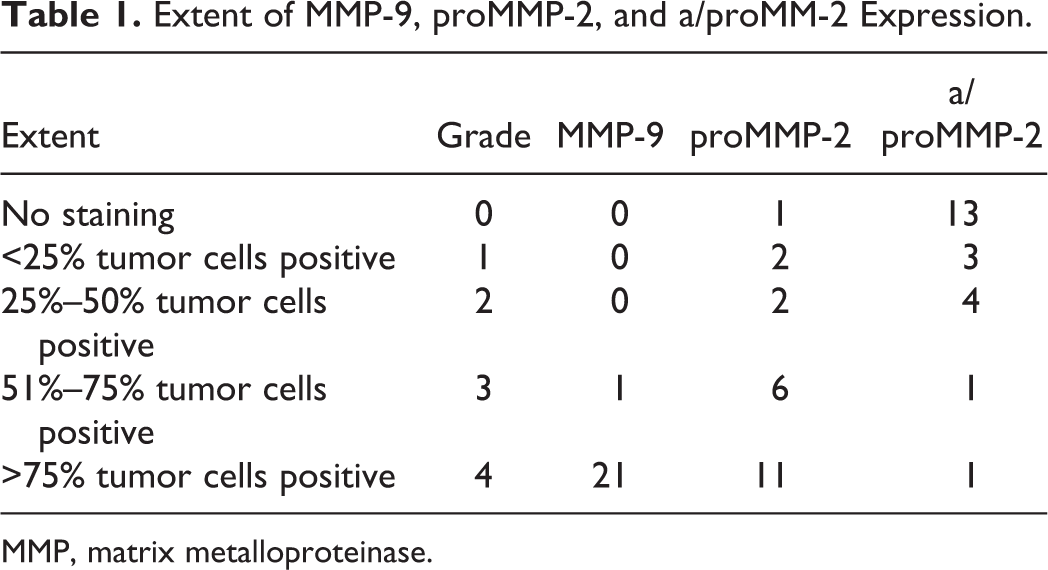
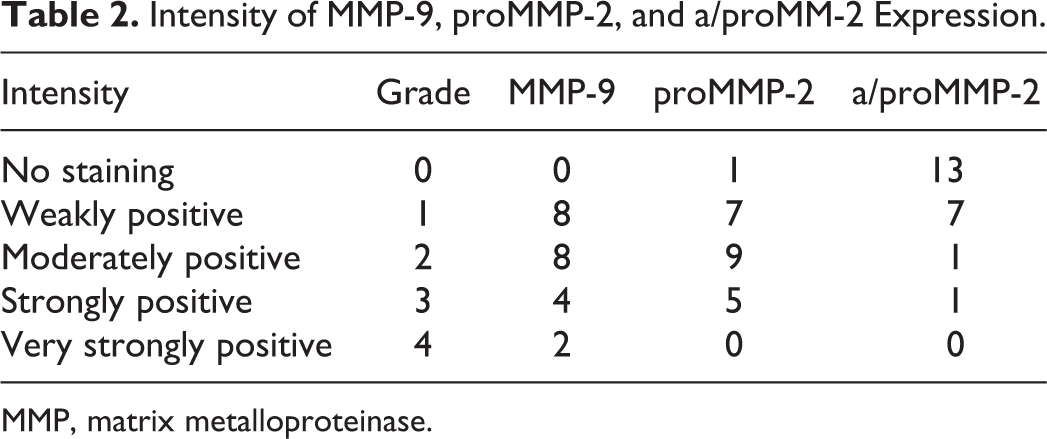
The tumor staining was recorded as being either not present or positive in <25%, 25% to 50%, 51% to 75%, or >75% of tumor cells (Table 1). Integer values were assigned as scores according to percentage of positive cells (0–4). The signal intensity was averaged for the entire tumor mass and graded as mild, moderate, strong, or very strong and accordingly scored as integer values (1–4) (Table 2). Integer values were both added 22 and multiplied 23 to provide 2 dedicated immunoreactivity scores (IRS). In cases of interobserver discrepancy, the mean values were assigned.
Extent of MMP-9, proMMP-2, and a/proMM-2 Expression.
MMP, matrix metalloproteinase.
Intensity of MMP-9, proMMP-2, and a/proMM-2 Expression.
MMP, matrix metalloproteinase.
Statistical Analysis
Subgroup comparisons of Vt and Ve were carried out using Student’s t-test and 1-way analysis of variance (ANOVA) after confirmation of normality. The interdependency between individual MMP scores, EI, and Vt was assessed by the Spearman rank correlation coefficient. The variation of the different MMP scores and EI between histologic subtypes and tumor locations was assessed. Direct comparison of individual parameters between 2 groups was made using the 2-tailed Mann-Whitney test. The Kruskal-Wallis test was used for nonparametric multiple comparison of the values between the different tumor subtypes. For all tests, P < .05 was considered indicative of significance. For multiple tests, a Bonferroni post hoc correction was implemented.
Results
Magnetic resonance images and tissue samples from 22 dogs were included in this study. Twenty samples had been obtained surgically and 2 samples were harvested at necropsy.
Location and Histologic Subtypes
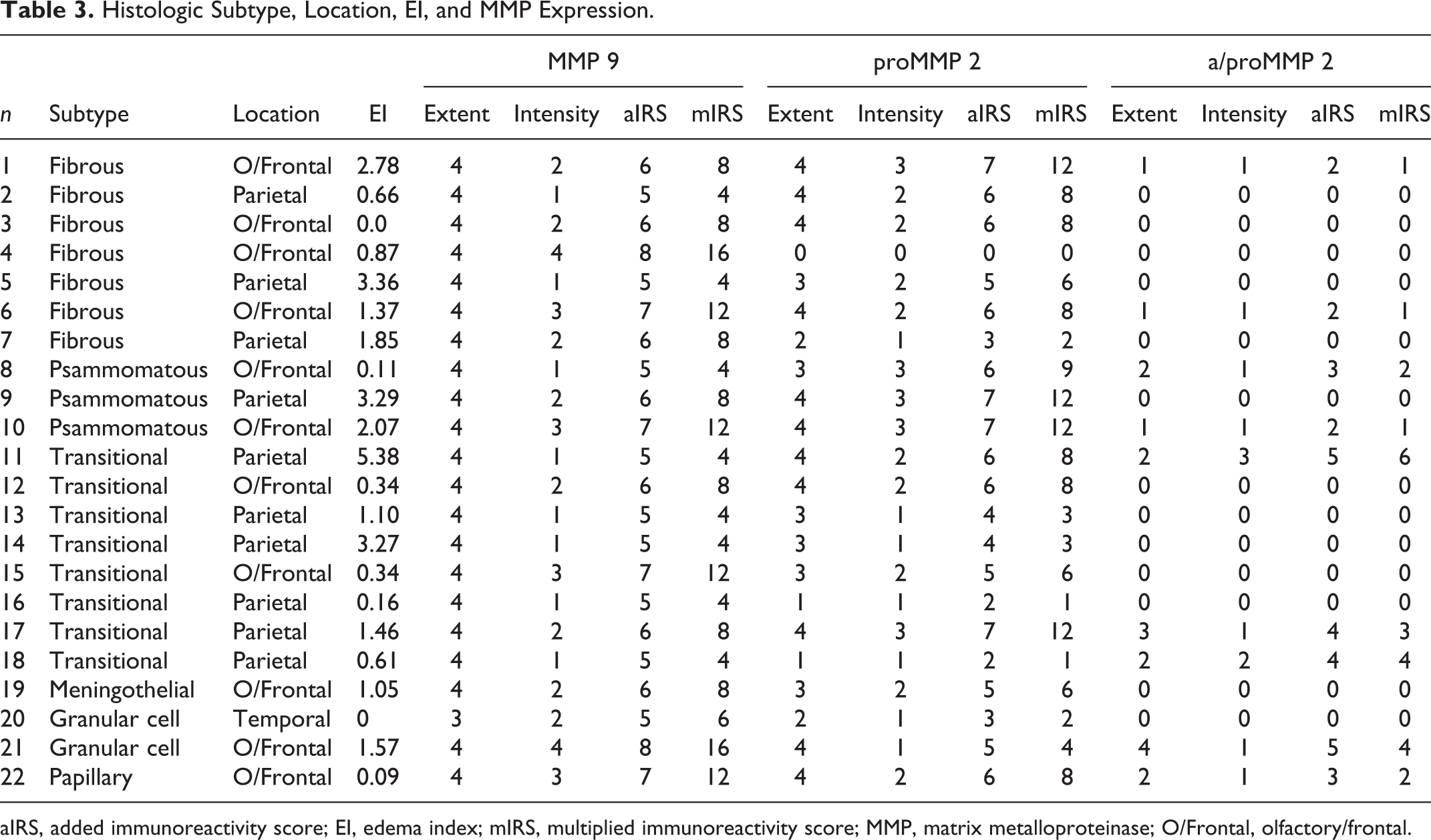
Each dog had a single intracranial tumor. Tumors were confined to the rostral and middle cranial fossae. Eleven of the 22 meningiomas (50%) were located in the region of the olfactory bulb/frontal lobe; in 10 cases (45.4%), the meningioma was confined to the parietal lobe and in 1 case (4.6%) to the temporal lobe. Histomorphologically, the tumors were classified as transitional meningioma in 8 cases (36.3%), fibrous in 7 cases (31.8%), psammomatous in 3 cases (13.6%), granular cell in 2 cases (9%), and meningothelial and papillary meningioma in 1 case each (4.5%). To provide an adequate group size, we pooled the histologic subtypes in 3 groups only: transitional (8/22), fibrous (7/22), and other histologic subtypes (7/22) (Table 3). Furthermore, the single case of a temporal lobe tumor was omitted from the statistical comparison between tumor locations. Tumor subtype and location were not statistically linked (P = .09).
Histologic Subtype, Location, EI, and MMP Expression.
aIRS, added immunoreactivity score; EI, edema index; mIRS, multiplied immunoreactivity score; MMP, matrix metalloproteinase; O/Frontal, olfactory/frontal.
Tumor Volume (Vt) and Edema Volume (Ve)
The Vt ranged from 0.03 to 8.84 cm3 (mean ± SD, 3.5 ± 2.6 cm3; median, 2.63 cm3). Peritumoral edema was present in 20 of 22 cases. The Ve ranged from 0.0048 to 11.7 cm3 (mean ± SD, 3.37 ± 3.27 cm3; median, 2.36 cm3). There were no significant differences in Vt and Ve between meningiomas of different subtypes (P > .15 for Vt; P > .94 for Ve) or different locations (P = .23 for Vt; P > .19 for Ve) (Fig. 3).
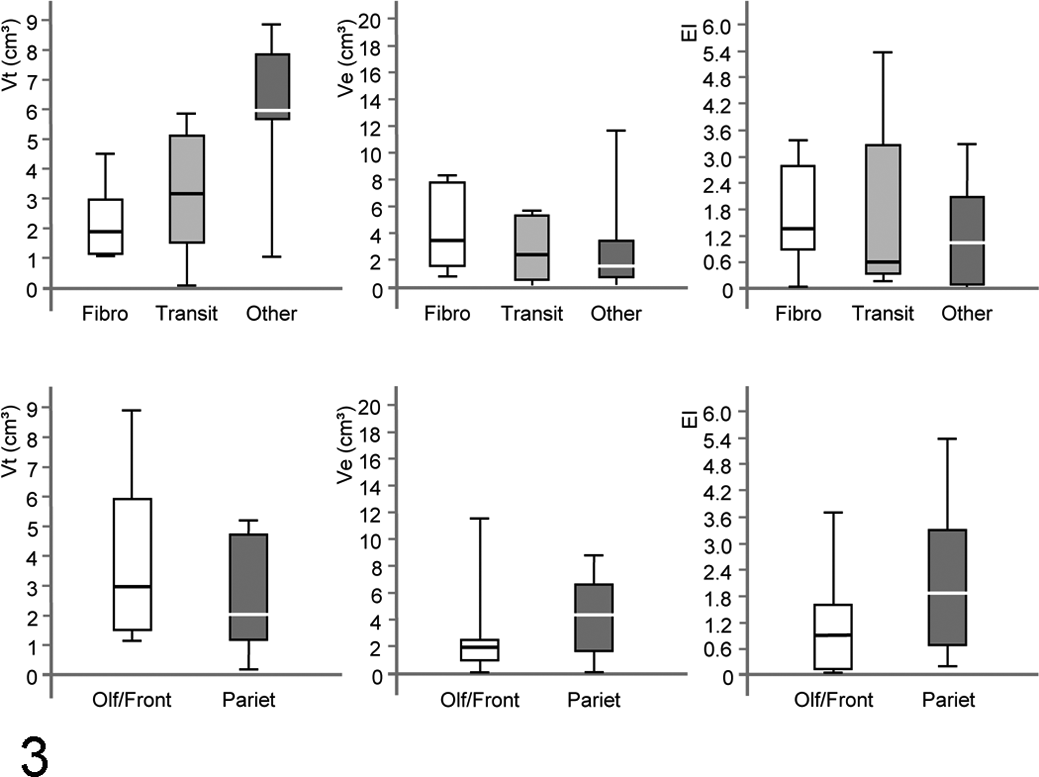
Box plots displaying the tumor volume (Vt), edema volume (Ve), and edema index (EI) with respect to different histologic subtypes (fibro, fibroblastic; transit, transitional) and locations (olf/front, olfactory bulb/frontal lobe; pariet, parietal lobe).
The EI ranged from 0 to 5.38 (mean ± SD, 1.44 ± 1.42; median, 1.08). There were no significant differences in EI between meningiomas of different subtypes (P > .85) or different locations (P > .06) (Fig. 3). However, the EI in olfactory/frontal meningiomas was subsignificantly (P = .0055) higher if compared with parietal tumors.
Matrix Metalloproteinase Expression
Matrix metalloproteinase-9 was expressed in all the samples (22/22), whereas proMMP-2 was expressed in 21 of 22 meningiomas and a/proMMP-2 was expressed in 9 of 22 (Table 3).
Matrix metalloproteinase-9 was expressed in more than 75% of tumor cells, independent of tumor type and location, with a mean ± SD additive immunoreactivity score (aIRS) of 5.95 ± 1 and a mean ± SD multiplied immunoreactivity score (mIRS) of 7.91 ± 3.93 (Table 3). There was no significant association between MMP-9 expression and tumor subtype (P ≥ .10) (Fig. 4). Frontal meningiomas, independent of their type, exhibited a much higher intensity of MMP-9 expression when compared with the parietal meningiomas (P = .003). This site-specific expression pattern had an impact on both IRS (P = .003), so that all 3 values differed significantly between the 2 locations (Fig. 4).
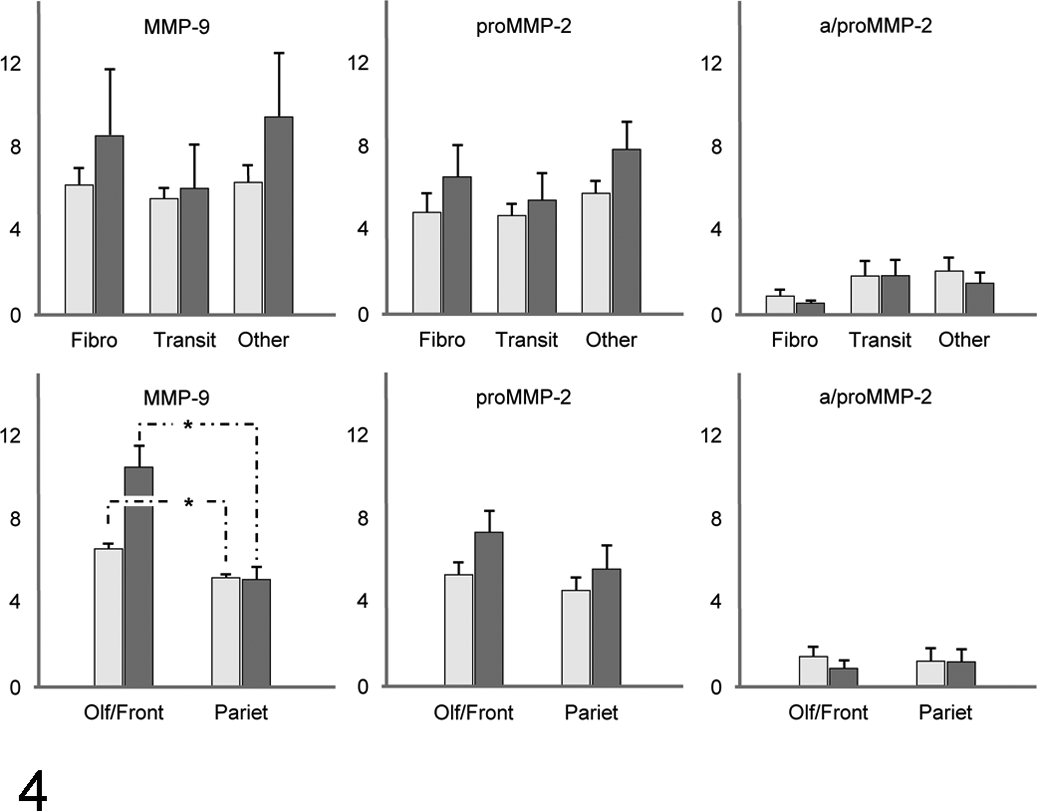
Bar charts of the immunoreactivity scores (IRS); additive IRS (aIRS; bright gray) and multiplied IRS (mIRS; dark gray) of matrix metalloproteinase-9 (MMP-9), proMMP-2, and a/proMMP-2 sorted for histologic subtypes (upper graphs) (fibro, fibroblastic; transit, transitional) and location (lower graphs) (olf/front, olfactory bulb/frontal lobe; pariet, parietal lobe).
In contrast to MMP-9 expression, proMMP-2 and a/proMMP-2 expression did not correlate with tumor location (P ≥ .30). ProMMP-2 and a/proMMP-2 expression did not correlate with histologic subtypes (P ≥ .22) (Fig. 4).
Correlation Between MMP-9 and MMP-2
Matrix metalloproteinase-9 and MMP-2 (proMMP-2 and a/proMMP-2) were expressed independently without any detectable correlation (P ≥ .07) (Fig. 5). However, sorted by histologic subtypes, there was a significant correlation between the intensity of MMP-9 expression and proMMP-2 expression in transitional meningiomas (P = .04, r = 0.7).
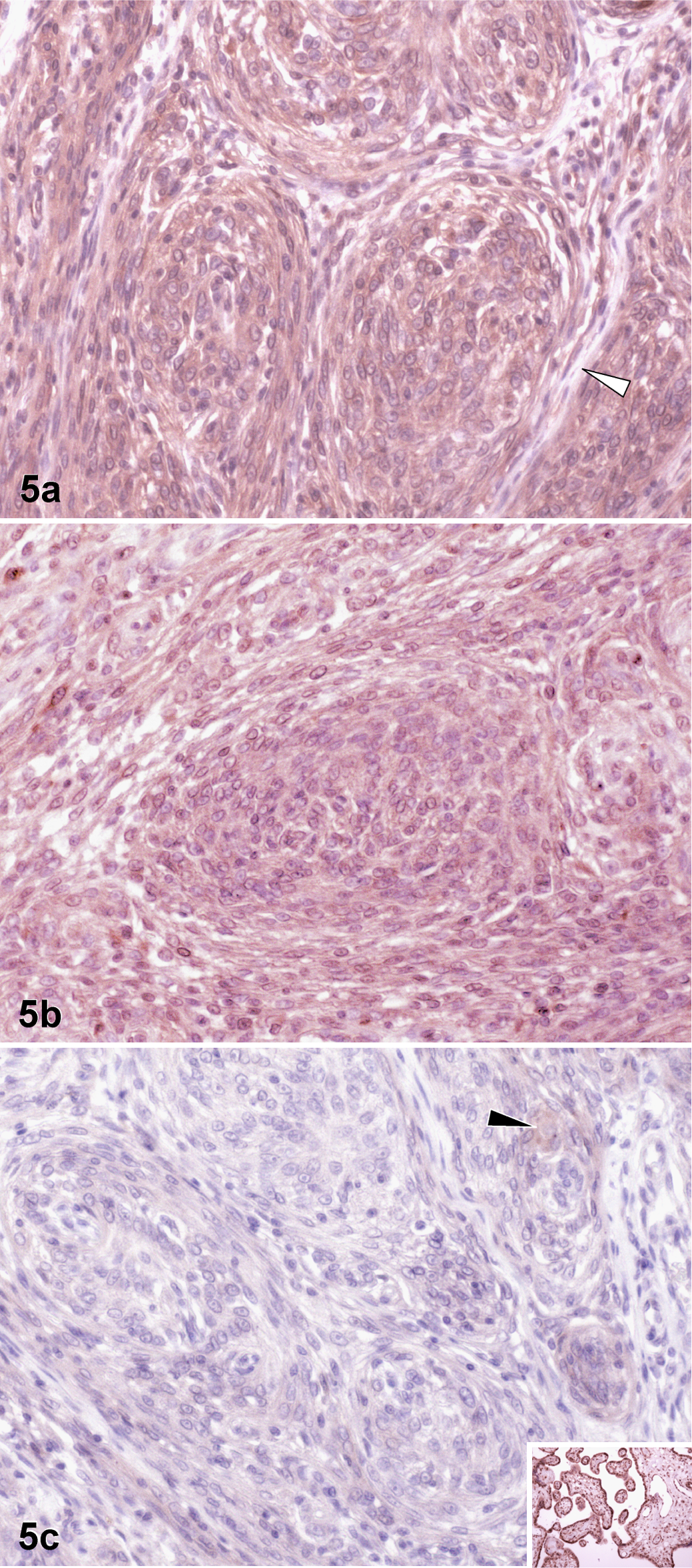
Brain; dog No. 6. Example of the immunohistochemical pattern in 1 single meningioma: note the strong staining for matrix metalloproteinase-9 (MMP-9) (a) and proMMP-2 (b) and much weaker staining for a/proMMP-2 (c). The white arrowhead: immunonegative areas in between tumor cell whorls (a). The black arrowhead: weakly stained tumor cells (c). Inset: positive control for a/proMMP-2.
Location of the tumors did not have a significant influence on the correlation between any of the MMPs’ expression parameters (P ≥ .19).
Correlation Between MMP Expression and EI
Regarding the calculated edema values, none of the different MMP expression parameters were statistically linked to EI (P ≥ .27).
Discussion
In this study, we investigated MMP-2 and MMP-9 expression in rostrotentorial grade I intracranial meningiomas and their association with PTE. Furthermore, we assessed whether there is an association between MMP expression and tumor location or histologic subtype.
Our study confirmed the previous observation that intracranial meningiomas in dogs express both MMP-2 and MMP-9. 15 In the cited study, MMP-2 and MMP-9 were expressed in 70% and 69.3% of benign meningiomas, and the expression was reported to be independent of the tumor cell proliferation by means of PCNA and Ki-67 immunoreactivity.
In human meningiomas, the extent of MMP-9 expression was shown to be associated with severe PTE in neuroinvasive anaplastic and aggressive tumors, whereas the immunoreactivity in benign meningiomas only subsignificantly was linked to a higher EI. 9,19,20 Similar to the human cases, we did not find an association between the severity of PTE, as defined by the EI, and the expression of MMP-9 in benign CIM. Further studies on high-grade meningiomas in dogs may produce different results.
Concerning tumor location, it is noteworthy that the intensity of MMP-9 expression and subsequently both IRS were significantly higher in the frontal lobe when compared with the parietal lobe. Human benign meningiomas did not respectively differ in MMP-9 immunoscores in between the lobes. 9 Even though not much is known about the spatial expression pattern and local activity of MMPs within the meninges, species-specific gene expression may account for this difference. In animals, site-related variations in the expression of particular MMPs have been observed in the healthy mammalian brain. 2,24,29 In humans, however, there were no significant differences in the expression of MMP-9 among different regions of the central nervous system. 4,20 Further investigations are required before drawing further conclusions as to whether this observation reflects a characteristic of the region or the state of the tumor cells.
The specific analysis of MMP-2 subtypes in our dogs produced less clear results. All investigated meningiomas exhibited a much stronger immunopositivity for proMMP-2, which recognizes the latent enzyme only, than for a/proMMP-2, which tackles both active and latent states. There was generally an excellent interobserver agreement regarding the scored parameters’ extent and intensity. Since data on the comparative avidity and affinity of both antibodies are not available, the immunohistochemical results may not match completely with the functional state of MMP-2. The investigation therefore remains to be validated by enzymological assays. Our study did not show a clear association between EI and expression of proMMP-2 or pro/aMMP-2, and this lack of interdependency is in accordance with previous findings in human meningiomas. 19
There was no detectable correlation between MMP-2 and MMP-9. However, further comparison of the expression profiles identified a positive correlation between the intensity of MMP-9 and proMMP-2 in transitional meningiomas. A human study also attempted to establish the relationship between MMP-9 and MMP-2 in meningiomas of different subtypes. The investigators observed that whereas meningothelial meningiomas apparently have the weakest expression and the transitional subtype showed an intermediate intensity, fibrous meningiomas presented with the strongest expression of both MMP-2 and MMP-9. 25 The MMP-9 results could not reproduced in a selective study on benign supratentorial meningiomas in humans that screened for differences among meningothelial, transitional, fibrous, psammomatous, angiomatous, microcystic, and secretory meningioma subtypes. 9 Our analogous study also failed to exhibit significant differences between fibrous and transitional meningiomas in the forebrain of the dog.
Only experimental studies have indicated that the expression of MMP-9 and its activity may be downregulated after treatment with dexamethasone. 13 In our study, dogs that received corticosteroids prior to MR imaging were excluded, but the dosage and duration of corticosteroid administered after MR imaging until the surgery varied between individual dogs, which might have influenced the MMP-9 expression variably. A study based on MMP expression and PTE in humans also used dexamethasone prior to sample collection, but this was not considered to affect the potential effect on the expression and therefore the results of the study. 20
Conclusion
Matrix metalloproteinase-2 and MMP-9 do not seem to play a major role in the formation of PTE in grade I CIM.
Footnotes
Acknowledgements
We are very grateful to Karin Stingl, Ludwig-Maximilians University of Munich, for optimizing the immunostaining. We also thank Prof Dr Juergen Schlegel, Technical University of Munich, for his helpful suggestions regarding the immunoreactivity scores.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This study was supported by the Animal Health Trust-Centre for Small Animal Studies Research Fund.
