Abstract
Brunner’s glands are submucosal glands located in the proximal duodenum. Hyperplasia of the Brunner’s gland has been reported rarely in humans and animals. We examined sections of the Brunner’s gland from 63 sand rats submitted for necropsy over 2 years. Of the 63 animals necropsied, 45 (71%) had evidence of hyperplasia defined as nodular expansion, dilated ducts, or intraductal papillary proliferation. The hyperplasia was graded as mild in 22 (49%) of the cases, moderate in 15 (33%), and marked in 8 (18%). We found an association with both increased age and evidence of gastric ulceration and hyperplasia of the Brunner’s gland. In sand rats with marked hyperplasia, 8 of 8 (100%) had evidence of gastric ulceration, compared to 13 of 18 (72%) in animals with no hyperplasia. Animals with marked hyperplasia were, on average, 8.4 months older than animals with no hyperplasia. There was no association with gender. The lesion in sand rats is histologically similar to that in humans.
Psammomys obesus, commonly called the sand rat or fat sand rat, is a diurnal desert animal belonging to the family Muridae and subfamily Gerbillinae. They are terrestrial mammals that naturally inhabit the salty desert areas of North Africa and east across the Arabian Peninsula. 19,22,28 Sand rats have proven to be useful animal models for numerous human diseases. They have served as a model for the study of otic cholesteatoma, spondylosis, renal function, and numerous diabetes-related disorders. 1,5,15,18 They have been found to be naturally infected with Leishmania major and have been identified as the most important reservoir host of zoonotic cutaneous leishmaniasis 17 in the Middle East and North Africa. 19,26,27
Brunner’s glands are submucosal glands located in the duodenum. They are most prominent near the junction with the pyloric stomach and dissipate distally. The glands consist of tall columnar cells with abundant, clear to mucinous cytoplasm with basally located nuclei. In the proximal duodenum, they can extend through the muscularis mucosa and into the overlying crypts. 23 It is theorized that chyme in the duodenum stimulates the glands to secrete an alkaline mucus that helps to neutralize the acid from the stomach; 6,21,24,31 however, there is not complete agreement on their physiologic role related to acid control. 6,20 The glands also secrete lysozyme, epidermal growth factor, and possibly IgA. 31
Materials and Methods
In 2008, the Veterinary Services Program at the Walter Reed Institute of Research (WRAIR) established 1 of the only 2 sand rat breeding colonies in the United States. All sand rats used in this study were maintained according to accepted animal care and use standards. All animals were on protocols approved by the WRAIR Institutional Animal Care and Use Committee and were carried out in facilities accredited by the Association for the Assessment and Accreditation of Laboratory Animal Care International. From September 2008 to September 2011, there were 138 sand rats routinely submitted for necropsy to the Department of Pathology at WRAIR. Standard necropsy tissue sampling was used; often the Brunner’s gland was included in the tissue sample as part of the proximal duodenum. In September 2010, five cases were submitted within the same week and histopathologic evaluation of proximal duodenum revealed remarkable variation in the size and cellular features of the Brunner’s gland. All subsequent necropsy submissions of the sand rat involved serial sections of the Brunner’s gland beginning 2–3 mm cranial to the pyloric sphincter and sectioning every 3–5 mm for a total of 5 sections. All tissues collected at necropsy were fixed in 10% neutral buffered formalin, routinely processed, paraffin embedded, sectioned at 4-μm thickness, and stained with hematoxylin and eosin.
All cases were initially microscopically evaluated by a board-certified veterinary pathologist. Three cases were tentatively diagnosed as hyperplasia of the Brunner’s gland and submitted to the Department of Veterinary Pathology at the Armed Forces Institute of Pathology. In consultation with the Department of Gastrointestinal Pathology, the diagnosis of Brunner’s gland hyperplasia was confirmed. To help establish consistent diagnostic criteria for the remaining cases, 4 board-certified veterinary pathologists, including the original pathologist, evaluated representative samples of the Brunner’s glands concurrently at a multi-head microscope. Diagnostic criteria for hyperplasia included moderate to marked nodular expansion of the gland by proliferative epithelium; an increase in the presence of dilated ducts lined by columnar or pseudostratified epithelium; intraductal papillary proliferation; or any combination of the three. With the criteria for hyperplasia established, the original pathologist reviewed all cases and scored the degree of hyperplasia as follows: none, mild, moderate, or marked.
Results
Signalment and Clinical Features
Of the 138 total sand rat cases submitted, 63 included sufficient sections of the Brunner’s gland to evaluate. There were 32 males, 30 females, and 1 animal whose sex could not be determined. The age ranged from 10 to 36 months, with an average of 20.4 months. In the 63 cases evaluated, 6 relatively healthy animals were euthanized for quality assurance or colony management purposes. The remaining 57 animals were found dead in the cage or were either moribund or in such poor clinical condition that humane euthanasia was required. At necropsy, 47 of these 57 animals (82%) had gross or histologic evidence of melena, gastric erosions, or gastric ulcers or a combination of the three.
Microscopic Pathology
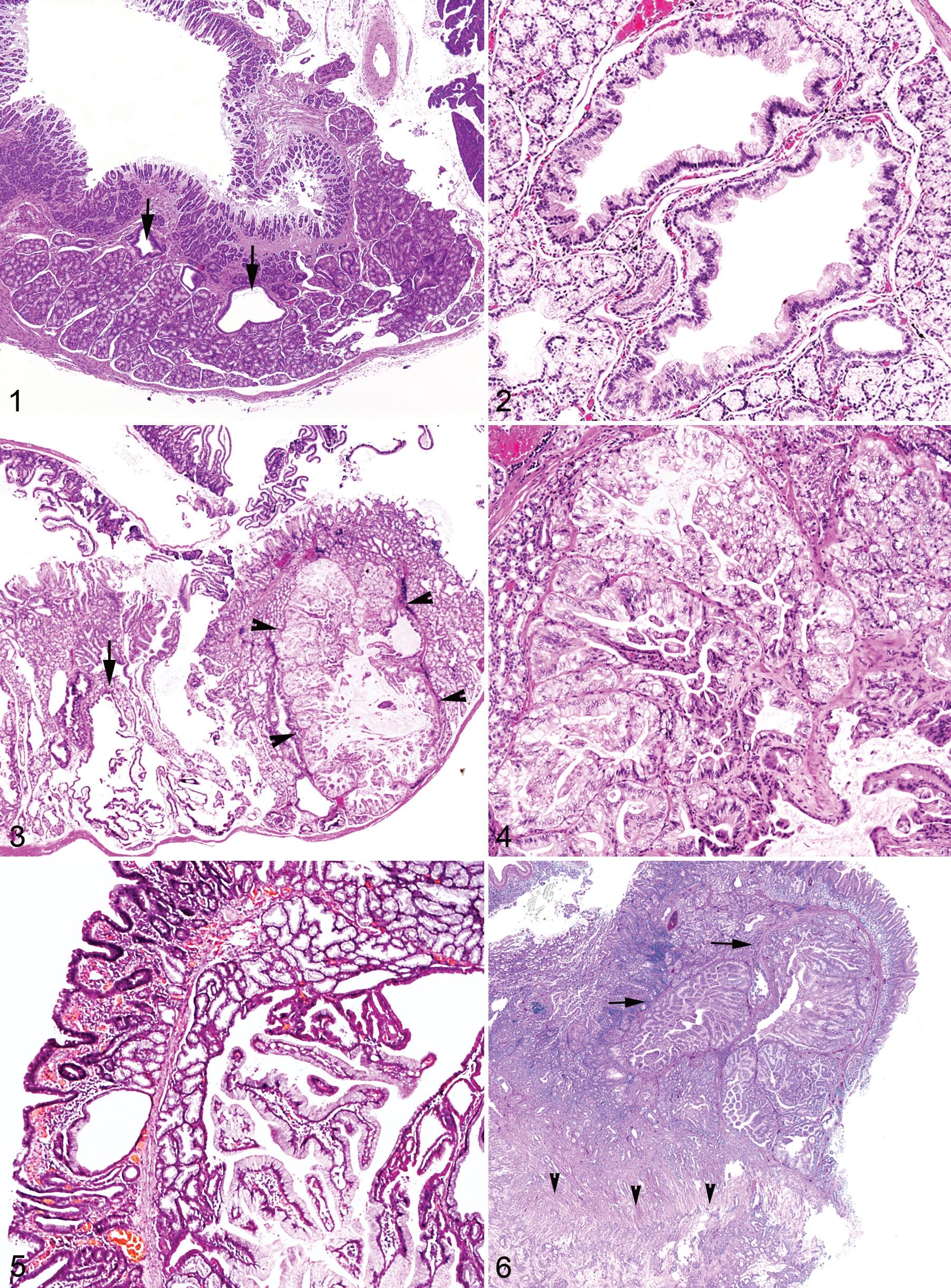
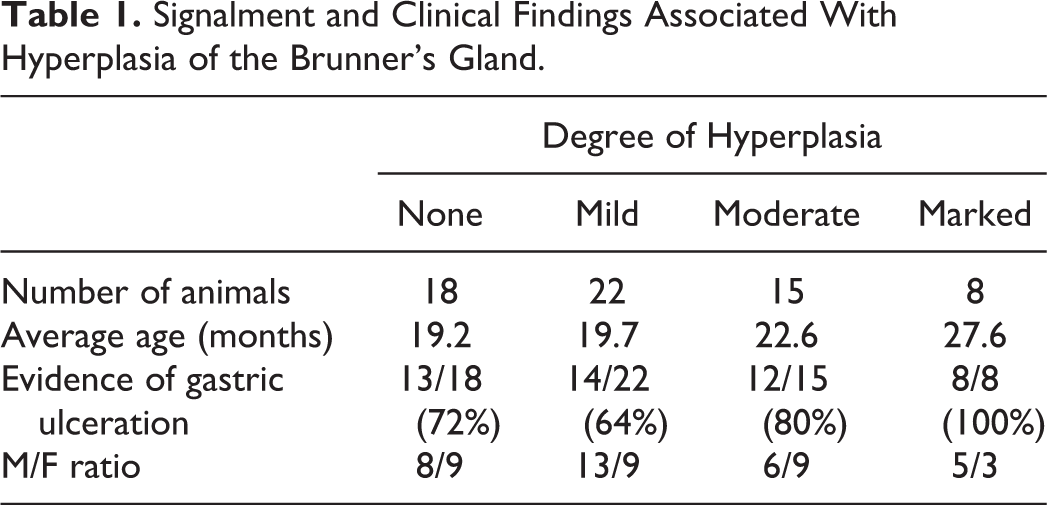
The proximal duodenum from an asymptomatic 3-month-old male sand rat that was euthanized as part of a separate study was obtained as a normal control (Fig. 1). Note that a normal sand rat can have what at first glance appear to be proliferative Brunner’s glands with few dilated ducts. Therefore, to be considered hyperplastic, the Brunner’s gland had to exhibit proliferation beyond this set point. Hyperplasia of the Brunner’s gland was observed in 45 of 63 cases (71%). Mild hyperplasia consisted of low numbers of dilated ducts that were lined by a tall columnar epithelium. Occasionally, there was mild to moderate displacement of the overlying duodenal epithelium. Moderate hyperplasia involved increased numbers of dilated ducts, some of which were lined by hyperplastic columnar epithelium that piled up 2 to 3 cell layers deep (Fig. 2). Epithelial cells that formed acini were moderately hyperplastic and filled with abundant amounts of clear to lightly eosinophilic cytoplasm. They often formed distinct lobules that raised the overlying duodenal epithelium. Marked hyperplasia was characterized by duodenal epithelium that was raised significantly by the underlying hyperplastic glands (Fig. 3), often with the formation of distinct nodules composed of proliferating papillary projections (Fig. 4). The incidence of hyperplasia was as follows: none, 18; mild, 22; moderate, 15; and marked, 8 (Table 1). The average age ranged from 19.2 months for animals with no hyperplasia to 27.6 months for animals with marked hyperplasia. Of the sand rats with no evidence of hyperplasia, 13 of 18 (72%) were diagnosed with evidence of gastric ulceration. In animals with hyperplasia, gastric ulceration occurred in 14 of 22 (64%) animals with mild hyperplasia, 12 of 15 (80%) with moderate hyperplasia, and 8 of 8 (100%) with marked hyperplasia. All animals with gastric ulceration were found either moribund or dead. The male/female ratio in animals with no hyperplasia was 8/9 (1 animal’s sex was not available), 13/9 in mild hyperplasia, 6/9 in moderate hyperplasia, and 5/3 in marked hyperplasia.
Signalment and Clinical Findings Associated With Hyperplasia of the Brunner’s Gland
Discussion
In humans, the nomenclature for growth-related lesions of the Brunner’s gland is not well established. There is considerable interchangeability between the terms hyperplasia, hamartoma, and adenoma. 20,23 Hyperplasia of the Brunner’s gland seems to be well established. 6,20,21,25 Hamartoma of the Brunner’s gland is histologically distinct from hyperplasia consisting of lobulated Brunner’s glands interspersed with fibrous, lymphoid, and adipose tissue. 14 However, some authors use size to distinguish between a diagnosis of hyperplasia and hamartoma, with lesions greater than 5 mm diagnosed as hamartoma. 20,23 There have been some reports of adenomas of the Brunner’s glands, 24,29 but the histologic distinction between hyperplasia and adenoma is not clear. There have been 3 authors to report adenocarcinoma of the Brunner’s glands. 3,12,25
In animals, hyperplasia or cystic dilation of the Brunner’s gland is even more rarely reported than in humans, but there is still some disagreement on whether to classify the lesion as cystic, hyperplastic, or neoplastic. 2,4,10,16 In addition, we could find only one report of adenocarcinoma of the Brunner’s gland in a species other than human. In reviewing this one reported case, it is not clear that the lesion is similar to those we observed in sand rats; furthermore, it is not clear how the authors differentiated it from adenocarcinoma originating from duodenal epithelium. 4 In transgenic mice, the incidence of hyperplasia of the Brunner’s gland was reported as high as 48% in females. 30 Certainly, to our knowledge, spontaneous hyperplasia of the Brunner’s gland has not been reported in any species at an incidence rate that approximates what we observed in our sand rat colony.
In the sand rat cases we examined, we did not use the term adenoma to describe any of the lesions. The presence of ducts and lack of dysplasia are more consistent with a diagnosis of hyperplasia. In addition, we concur with previous authors 20 that the difference between hyperplasia and adenoma is arbitrary. These proliferative lesions of the Brunner’s gland in the sand rat seem to differ mostly on gradation and we could find no histologic criteria to consistently diagnose adenoma. Finally, although some of the hyperplastic lesions were expansive, they all maintained normal architecture and cellular polarity and were not considered dysplastic or neoplastic. Interestingly, Figure 5 is a case of Brunner’s gland hyperplasia in a human (published with permission from the American Registry of Pathology). Note the similarities to the marked hyperplasia in the sand rat (Fig. 4). In both cases there are numerous dilated ducts and hyperplasia of papillary projections. We did examine 2 cases from our sand rat population of adenocarcinoma at the site of the Brunner’s gland. Both were older males, 2.8 and 3.1 years of age. In each case, the duodenum was markedly expanded by neoplastic glandular cells that formed acini and tubules and invaded the underlying lamina propria and tunica muscularis producing a marked scirrous response (Fig 6). Upon close examination it appears in both cases that the neoplastic cells arose from duodenal epithelium; however, a Brunner’s gland origin could not be completely ruled out. One of these 2 cases of adenocarcinoma at the site of the Brunner’s gland was referred to the Armed Forces Institute of Pathology and reviewed by the Department of Gastrointestinal Pathology. They concurred that the adenocarcinoma arose from the duodenum and stated that there was no definitive way to distinguish the neoplastic duodenal epithelium from Brunner’s gland epithelium. In reviewing the human cases reported as Brunner’s gland adenocarcinoma, there is only one report in which mucosubstance histochemical staining methods were used to distinguish Brunner’s gland epithelium. 12 Unfortunately, we did not have access to similar techniques and could not apply them to our 2 cases. Regardless, based on our cases and a search of the literature, Brunner’s gland adenocarcinoma in any species, if it does occur, is exceedingly rare.
The pathogenesis of Brunner’s gland hyperplasia is unknown. The prevailing hypothesis in humans is that the glands undergo compensatory hyperplasia in reaction to increased gastric acid production. 8 This is supported by reports of patients that undergo renal transplant and therefore experience increased gastric acid production, having an increased incidence of Brunner’s gland hyperplasia. 7 However, in one study, increased gastric acid was found in only 45% of patients with Brunner’s gland hyperplasia while 20% had decreased acid. 9 In addition, reports of Brunner’s gland hyperplasia in patients with conditions associated with increased gastric acid production, such as Zollinger-Ellison syndrome are rare. 11,21 We were unable to measure the levels of gastric acid at the time of death of these sand rats. However, there is a strong association with clinical evidence of gastric ulceration and Brunner’s gland hyperplasia, at least in the case of moderate to marked hyperplasia. All animals (8/8) with marked hyperplasia and 12 of 15 (80%) with moderate hyperplasia had evidence of gastric ulceration. Complicating this association, however, is the fact that 13 of 18 animals with no hyperplasia had evidence of gastric ulceration. However, of the total 136 sand rats submitted to necropsy, 98 had evidence of gastric ulceration. Thus in our sand rat colony, over the course of 2 years, the incidence of gastric ulceration was 72% (98/136), which is the same rate found in the sand rats diagnosed with no hyperplasia of the Brunner’s gland (13/18). Other sand rat colonies report a similar high incidence of gastric ulcers (personal communication, Michelle V. Chenault). Therefore, given the markedly high incidence of gastric ulceration in our sand rat colony, the increased incidence in sand rats with moderate to marked hyperplasia (80% and 100%, respectively) supports the association between gastric ulceration and Brunner’s gland hyperplasia.
In addition to gastric ulceration, Brunner’s gland hyperplasia in sand rats appears to be associated with age. Animals with marked hyperplasia were, on average, 8.4 months older than animals with no hyperplasia, and those with moderate hyperplasia were 3.4 months older. Also, of the 63 sand rats included in this study, 6 animals were sentinels and were submitted for necropsy as part of the colony quality assurance program. These animals did not have any evidence of gastric ulceration and theoretically would not have any physiologic abnormality that would produce hyperplasia of the Brunner’s gland. We found hyperplasia in 5 of 6 of these sentinel animals; 3 scored as mild and 2 moderate. The average age of these animals was 25.2 months, which was 6.0 months higher than the average age of those animals with no hyperplasia. We also analyzed the data for an association between gender and Brunner’s gland hyperplasia. As indicated in Table 1, hyperplasia of the Brunner’s gland in sand rats appears nearly equally distributed between males and females.
There have been some reports associating Brunner’s gland hyperplasia with Helicobacter pylori infection in humans. 13 However, given the relatively high incidence of H. pylori infection in humans and the relatively low incidence of Brunner’s gland hyperplasia, it is difficult to conclude a link between the two. In our sand rat population we did not find histologic evidence of Helicobacter sp. infection in the sand rats studied using routine HE. In addition, 3 cases involving gastric ulceration were stained with Warthin-Starry and found to be negative. However, as part of our colony management program, 10 sand rats were tested for evidence of Helicobacter sp. infection over the course of 2 years (April 2009–March 2011). Fecal samples were submitted for PCR. All 10 sand rats tested positive for Helicobacter sp. Four of the 10 samples were from animals in this study. Of these 4, two had mild hyperplasia of the Brunner’s gland, one moderate, and one marked. Because we did not study Helicobacter sp. infection prospectively and did not establish the infection status of each animal, there is not sufficient evidence to exclude Helicobacter sp. infection as a cause of Brunner’s gland hyperplasia in the sand rat. However, given the high rate of both gastric ulceration and Brunner’s gland hyperplasia in this unique species, defining the role of Helicobacter sp. in the sand rat as it relates to one or both of these conditions is a logical course for further study.
In conclusion, to our knowledge this is the first report of a relatively high incidence of Brunner’s gland hyperplasia in any species. The histologic features of Brunner’s gland hyperplasia in sand rats are similar to that reported in humans. From our data it appears in the sand rat that age and gastric ulceration are associated with Brunner’s gland hyperplasia. Further study in the sand rat to establish a link between gastric ulceration, levels of gastric acid, and Helicobacter sp. status could help establish the sand rat as a model for this condition in humans.
Footnotes
Acknowledgments
We thank Drs. Jeff Wolf, Kathleen Funk, and Debra Banas of Experimental Pathology Laboratories, Inc. for their expert insight on the histologic evaluation of Brunner’s glands; Dr. Michelle Chenault for establishing the sand rat colony at the WRAIR; the WRAIR veterinary technicians and animal husbandry staff, especially Robert Blesh and Tesafaye Mekonnen, and the WRAIR Pathology Department, especially Mr. Rex Hartzoge, and MAJ Gwynne Kinley for their support.
Declaration of Conflicting Interests
Material has been reviewed by the Walter Reed Army Institute of Research. There is no objection to its presentation and/or publication. The opinions or assertions contained herein are the private views of the authors, and are not to be construed as official, or as reflecting true views of the Department of the Army or the Department of Defense. Research was conducted in compliance with the Animal Welfare Act and other federal statutes and regulations relating to animals and experiments involving animals and adheres to principles stated in the Institute for Laboratory Animal Research’s Guide for the Care and Use of Laboratory Animals, 2011, 8th edition.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
