Abstract
An adult walleye (Sander vitreus) was submitted to Cornell University for evaluation of a hard pale-tan pharyngeal mass attached to the gill arches. Dozens of hard white conical structures radiated from the surface. Microscopically, conical structures were identified as denticles and rested on plates of dysplastic orthodentine, cementum, and acellular bone. A diagnosis of compound odontoma was made based upon the presence of proliferative epithelial and mesenchymal odontogenic tissues that recapitulated tooth structures normally present on gill rakers. Odontomas are classified as hamartomas and typically develop in immature diphyodont mammals. The pharyngeal location and lifelong regeneration of teeth in fish, however, both qualify the present diagnosis in the pharyngeal region of an adult teleost. Ontogenic and morphologic differences between mammalian and piscine dentition and differentials for tooth-bearing tumors in fish are presented within the context of a developmental anomaly.
An adult walleye (Sander vitreus), a piscivorous, freshwater game fish, was captured for sport in Lake Erie near Dunkirk, New York (42.5°N, 79.3°W) in late summer. The angler noted a large pharyngeal mass extending through the gill slit and elevating the right operculum. The partially filleted carcass was submitted to the Aquatic Animal Health Program at Cornell University in Ithaca, New York.
The fish was 58 cm long and in fair nutritional condition. Based on microscopic evaluation of scale annuli, the age of the fish was estimated to be 4.5 years. A firm to hard, pale-tan sessile mass (5 × 5 × 4 cm) was located on the floor of the oropharynx between the first and second right gill arches. The lateral free margin extended beyond the gill lamellae to displace the right opercula (Fig. 1), and the medial aspect obstructed over 80% of the oropharyngeal luminal area. The surface of the mass was covered with dozens of mineralized conical structures that projected perpendicular to the mass surface (Fig. 2). Cut surfaces revealed deep mucosal invaginations and a central hard, porous core. Occasionally, inverted toothlike structures (denticles) projected from the deep epithelial sulci toward the center of the mass. Both the mass surface and the gill lamellae directly apposed to the mass were enlarged by proliferative and ulcerated tissue.
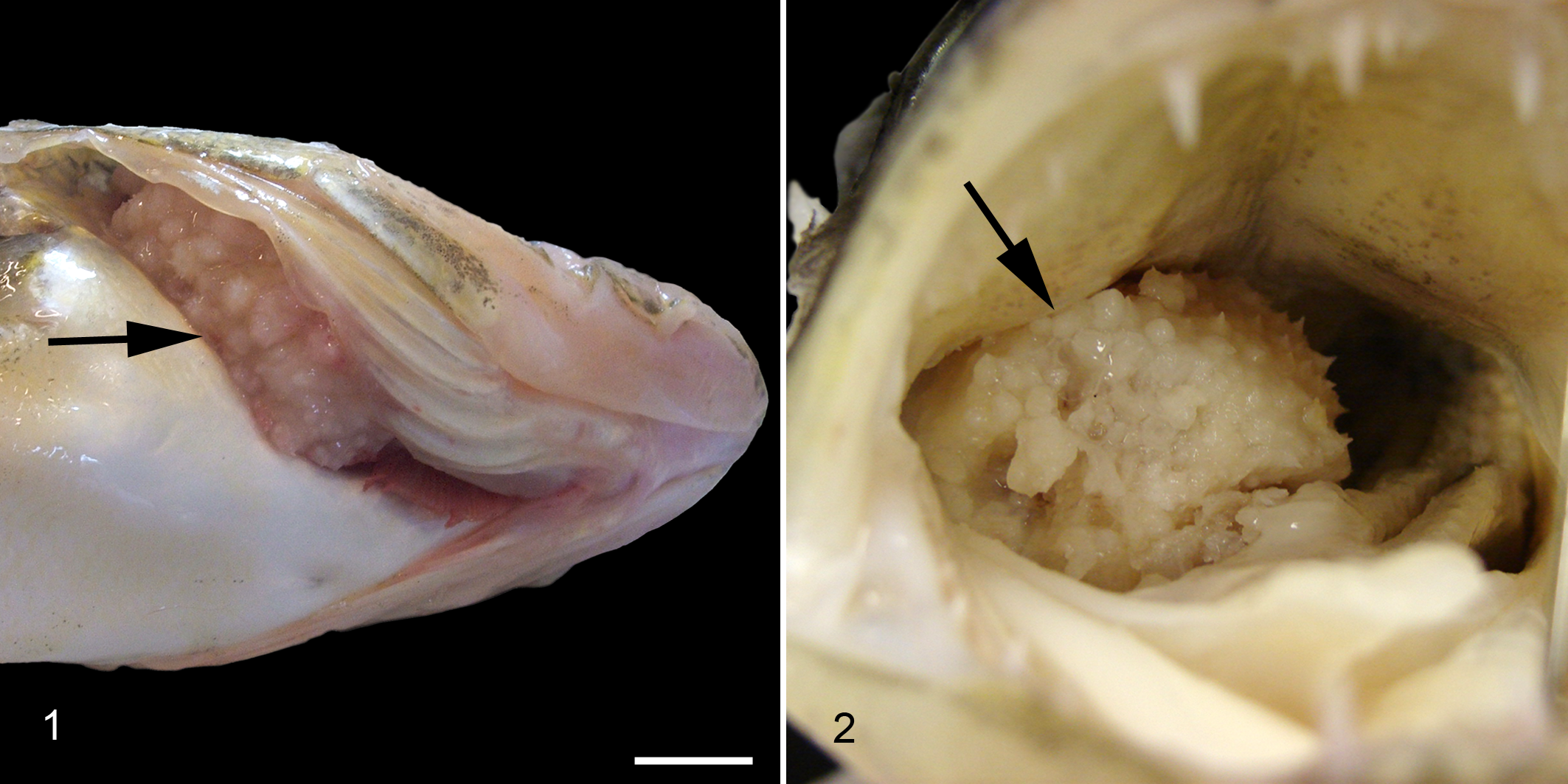
The mass and gill arches were removed en block, fixed in 10% neutral buffered formalin, and decalcified in 14% EDTA (ethylenediaminetetraacetic acid disodium salt, buffered to pH 7.1 with NaOH). Tissues were routinely processed for histology, sectioned at 4 μm, and stained with hematoxylin and eosin.
Microscopically, tooth-like structures, including enameloid organs and denticles, radiated from islands of acellular, nonpolarized, mineralized tissue identified as dysplastic orthodentine, cementum, or acellular bone based on the morphology of peripheral blastic cells (Fig. 3). Denticles were attached to mineralized tissue via ankylosis, represented at the broad base of dentin cones by a monolayer of amphophilic, parallel collagen stria (Sharpe’s fibers) oriented diagonal to the long axis of the tooth (Fig. 4). In the core of the mass, irregular islands of acellular bone were separated by poorly vascularized, loose, collagenous stroma that resembled stellate reticulum.
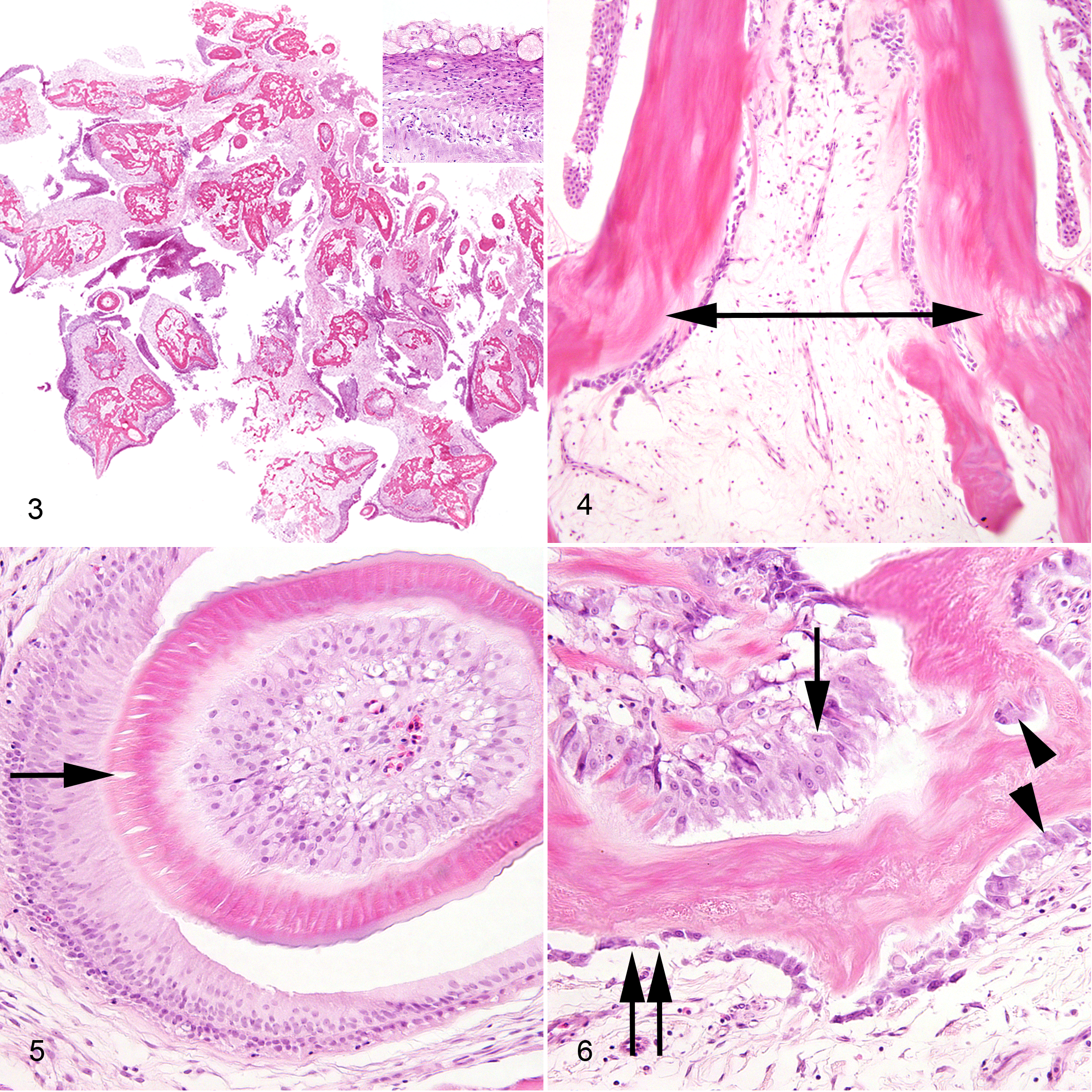
Denticles were composed of 2 layers of ameloblastic cells that palisaded along vacant crescents (enameloid was removed during decalcification) and orthodentine cones. Bilaminar ameloblastic cells consisted of outer and inner odontogenic layers and had distinct borders and moderate amounts of pale eosinophilic fibrillar cytoplasm. Nuclei were antibasilar and basilar, respectively, pale and oval to elongate, with finely stippled chromatin and 1 to 2 nucleoli (Fig. 5). Orthodentine cones were supported by dental papilla composed of central stellate reticulum bordered by packets of polygonal odontoblasts (called scleroblasts by some fish anatomists) that were more crowded in immature denticles. 6 Odontoblasts had distinct borders and moderate amounts of amphophilic, granular cytoplasm (Fig. 6). Nuclei were randomly dispersed, large and round and contained finely stippled, peripheralized chromatin with a prominent central nucleolus. Mitotic figures were rare in odontoblasts (2 mitoses counted in 10 400× fields) and were not detected in other cell types. Prominent Tomes fibers were represented by parallel clefts that extended perpendicularly from the apical border of odontoblasts into the overlying orthodentine (Fig. 5).
Scattered throughout the mass stroma were pools of pyknotic nuclear debris with minimal leukocytic infiltrates. Alternatively, the superficial mucosal epithelium adjacent to the compressed gill lamella was variably ulcerated and extensively infiltrated by lymphocytes, histiocytes, and fewer neutrophils. Deep mucosal invaginations penetrating the mass were lined by abundant goblet cells and therefore resembled odontogenic crypts (Fig. 3, inset). Based on the presence of multiple ectodermal and mesodermal odontogenic tissues (some of which formed enameloid organs and denticles) and the absence of an inflammatory nidus, this mass was diagnosed as a compound odontoma.
Tumors of odontogenic epithelium with odontogenic mesenchyme in domestic animals include ameloblastic neoplasms, inductive odontogenic tumors, and odontomas. 9 Several subtypes have been reported in fish. Epizootic ameloblastomas occur in wild populations of salmon and cunners. 5,8,17 Isolated reports of spontaneous odontogenic tumors in fish include fibroameloblastomas (referred to as “soft odontomas”) in salmon, an odontogenic epithelioma in a cod, and odontogenic fibroma in gilthead sea bream. 2,17,21
In mammals, odontomas do not generally present a diagnostic challenge. In fish, however, the wide distribution of dental lamina and continuous production of teeth can complicate the diagnosis. Microscopic evaluation of any proliferative lesion involving the lips, mouth, or pharynx can entrap developing teeth, giving the false impression of a proliferative odontogenic mass. Such masses include neoplasms and reactive or inflammatory lesions. To illustrate this point, Francis-Floyd et al report several tooth-bearing lip fibromas associated with a retrovirus in angel fish. 3 A comment subsequently submitted to the journal by an expert in human oral pathology in response to the angel fish study suggested the diagnosis be amended to “odontoma.” 4 In an attempt to clarify this issue, Harshbarger provided criteria for differentiating odontogenic tumors from hyperplastic tooth-bearing lesions induced by trauma. 7 This work characterized the latter lesion as pedunculated, symmetric, and typically inflamed with an edematous fibrous stroma. The mass in this walleye was broadly attached to the gill rakers, asymmetric, minimally inflamed, and contained a dense mineralized core with areas of cell-poor stroma resembling stellate reticulum.
While tissues from this fish were not evaluated for oncogenic retrovirus, the potential role of these viruses in the development of this lesion is considered unlikely for several reasons. Oncogenic tumors in walleye associated with walleye dermal sarcoma viruses and walleye discrete epidermal hyperplasia viruses are typically located within the dermis and epidermis, respectively, and do not arise from mucosal surfaces. 1 The size of the mass and the season in which this fish was sampled (late summer) are discordant with reports for oncogenic tumors in walleye, which develop over winter and present as large masses in the spring, and finally regress by late summer. 1
In contrast to diphyodontic mammals, toothed fish, reptiles, and amphibians are polyphyodonts and retain the capacity to replace teeth throughout life. 20 The location of dental tissues in teleosts is highly variable throughout the taxa, and extraoral dental lamina is associated with bone plates of the skull and mucosal surfaces of the lips, mouth, oropharynx, and esophagus. Even within specific genera of teleosts, there is enormous variation in the composition and distribution of odontogenic tissues. 11,12
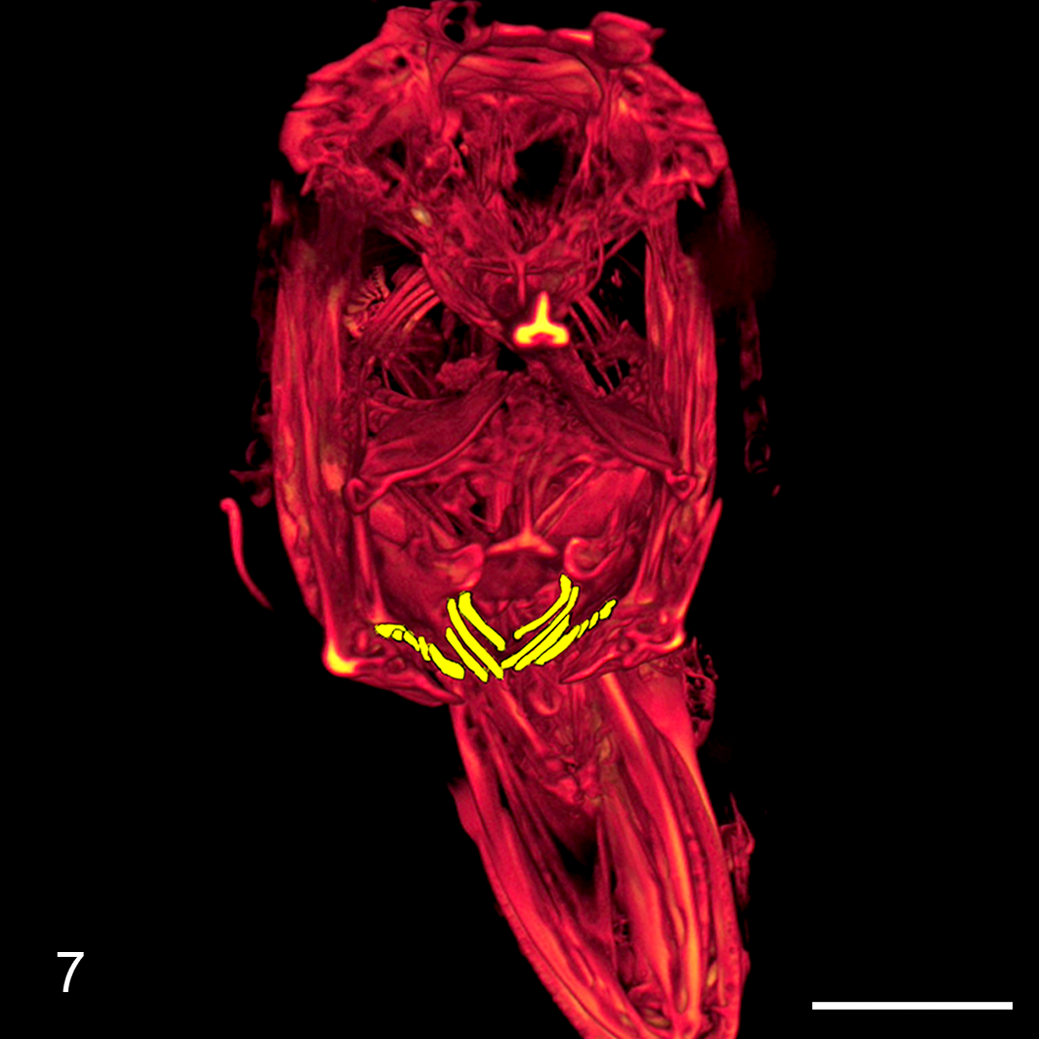
Like most piscivorous fish, walleye have prominent canines distributed along the premaxillary, maxillary, and mandibular arcades, the vomer bone, and the soft palate. 19 In addition, extraoral pharyngeal teeth, commonly called “gill rakers” and coincidentally termed “denticles” by fish anatomists, are distributed along the dorsal and ventral gill arches (Fig. 7). 6 Pharyngeal denticles have evolved to fulfill a variety of functions in teleosts, but in walleye they serve to trap prey and grind flesh. 6,19 The loss of pharyngeal denticles in some fish taxa is considered an indicator of more recent evolutionary divergence. 10
In addition to the unique distribution and development of dental tissues in fish, the histologic composition differs significantly compared to that of mammals, particularly with respect to the hard tissues of the teeth. 6 Enamel tissues in fish are referred to as “enameloid” because the matrix is nonprismatic and forms along collagenous fibers. 16 While the ectodermal origin of enamel is accepted in mammals, the origin of enameloid in fish is unknown. Neither amelogenins nor enamelins have been detected in fish. 12,13,16 Heterogeneous dentin-like substrates in fish include vasodentine, orthodentine, and osteodentine and are present not only in teeth but also throughout integument to function as protective armor plating. 6,15 Tomes fibers, described in the dentin-like tissues in the present case, are unique microscopic features of mammalian dentin and fish orthodentine; they are absent in vasodentine and osteodentine. 6 The teeth of most teleosts attach to plates of acellular bone, also called “bone-like tissue,” that lacks appositional growth, osteocyte lacunae, and Haversian canals. Bone is not typically a proliferative component of odontomas in mammals, but it may be present as reactive, metaplastic, or induced mesenchyme. 14
In the present case, acellular bone was a proliferative component of the mass and was differentiated from orthodentine by the presence of peripheral low-cuboidal osteoblasts with deeply basophilic cytoplasm and polarized nuclei. Cementoblasts were large and cuboidal, with the characteristic perinuclear halo and prominent central nucleolus. Additionally, cementoblasts, unlike odontoblasts and osteoblasts, were occasionally isolated within mineralized lacunae. Odontoblasts were easily identified as packeted, polygonal to fusiform cells with amphophilic, granular cytoplasm representing abundant rough endoplasmic reticulum and nuclei bearing a single basophilic, peripheralized nucleolus. Odontoblasts comingled with linear arrays of poorly mineralized orthodentine subdivided by Tomes fibers, while osteoblasts and cementoblasts remained external to their respective matrices (Fig. 6). 6
The odontoma in this fish is unique compared to those described in mammals. As such, we feel that this case provides an excellent illustration of comparative odontogenic pathology. In mammals, most odontomas occur in immature animals, while the presentation of this tumor in an adult fish can be attributed to the lifelong generation of teeth in polyphyodonts. In mammals, compound odontomas typically expand within the mandible or maxilla and contain denticles or simply organoid proliferations of enameloid organs and dental matrices. 18 The sessile tumor in this fish projected into the pharynx and was characterized by florid proliferations of well-formed denticles, tooth buds, and odontogenic rests with minimal inflammation. Finally, some of the mineralized tissues identified microscopically in this mass, including orthodentine and acellular bone, are unique to ectothermic animals.
Footnotes
Acknowledgments
We thank Donald Einhouse of the Lake Erie Unit, New York State Department of Environmental Conservation, for providing the specimen and Mark Riccio of Cornell’s Biotechnology Support Unit for providing the microcomputed tomography images.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This effort was supported in part through Contract 006010 between the New York State Department of Environmental Conservation and Cornell University funding the Fish Pathology Laboratory contract.
