Abstract
The normal embryonic development of organs and other tissues in mice and all species is preprogrammed by genes. Inactivation of a gene involved in any stage of normal embryonic development can have severe consequences leading to embryonic or postnatal developmental defects and lethality. Pathology methods are reviewed for evaluating normal and abnormal placenta and embryo, especially after E12.5. These methods include pathology protocols for necropsy and histopathology in addition to references that will provide additional knowledge for embryo assessment including histology atlases and advanced embryo imaging techniques.
Mice are often used in research to study functions of genes that have been modified by various genetic engineering methods. Complete or partial inactivation of a gene may lead to loss or modification of gene function in cells and tissues during any stage of gestation. The effects of gene modification can be ubiquitous in the embryo or targeted to specific tissues or cells. Modification of gene function by either spontaneous mutations or genetic engineering can result in an embryonic or postnatal lethality phenotype. 46 The aim of this article is to provide basic pathology evaluation procedures that will assist investigators in determining possible causes of embryonic and postnatal mortality. It should be noted, however, that the use of pathology and laboratory procedures described by us and other investigators may still not allow one to determine the cause of embryonic or postnatal mortality.
Stages of Gestation
Mouse development occurs after fertilization of the egg. Development in embryonic days (E) may be identified as going through 4 basic periods: include pre- and peri-implantation (E0-4.5), differentiation of the germ layers (E6-9), development of the cardiovascular system and placenta (E9-12), and tissue and organ differentiation (E12-19) (http://www.emouseatlas.org/emap/ema/theiler_stages/house_mouse/book.html; http://www.informatics.jax.org/greenbook). 31,38,44,47 The morning that a vaginal plug is found in mated females is generally defined as E0.5 of gestation. 23 Embryonic growth is at a specified rate and in stages, as defined by Theiler stage, head to rump length, and somite pairs. 23
Gene Expression
Genes are expressed in the various cells and tissues during gestation, and expression patterns have been described (http://www.emouseatlas.org/emage/home.php). Many genes function in normal development, growth, and differentiation of the embryonic organs and tissues. To determine the cause of embryonic death or to determine developmental defects found in live or dead embryos or neonatal, perinatal, or postnatal mice, it is crucial to first determine the cells and tissues expressing the gene in question. Several methods are available to assess gene expression, including in situ hybridization, immunohistochemistry, Lac-Z/β-gal expression, 2 and real-time qualitative PCR (q-PCR). 30,31 The effect of gene inactivation may be evident in the tissues normally expressing the gene. 14 Genes normally expressed at high levels during in utero development may have profound effects on organogenesis and differentiation. Secondary effects of gene mutations may also be found. A good example is the Nkx2-1 gene, the inactivation of which causes lack of normal lung development (lung bud branching is absent, which is the cause of death), absence of the pituitary gland and thyroid, and an abnormal hypothalamus. Mice live through gestation but die at birth or postnatally. 25 Pituitary development is altered not because of gene inactivation in the tissue but rather is secondary to inactivation of the Nkx2-1 transcription factor gene, which controls regulation of other genes important for pituitary pouch formation and thus pituitary development. 45
Necropsy Methods
The dam is euthanatized by carbon dioxide, the abdominal cavity is opened, and the uterus and ovaries are removed intact. 5,40 For embryos over E15, euthanasia may be induced by the injection of chemical anesthetics, induction of hyperthermia, or other methods specified by institutional policy, procedures, or guidelines. The uterus is spread out, and the location and sizes of placental sites (Fig. 1 ) are recorded on a uterus/embryo necropsy sheet as previously described. 49 Extraembryonic membranes (amnion, yolk sac, chorion, allantois) from each embryo are examined for viability (blood present in the yolk sac vessels) and then removed and detached from its placenta (Fig. 2 ).
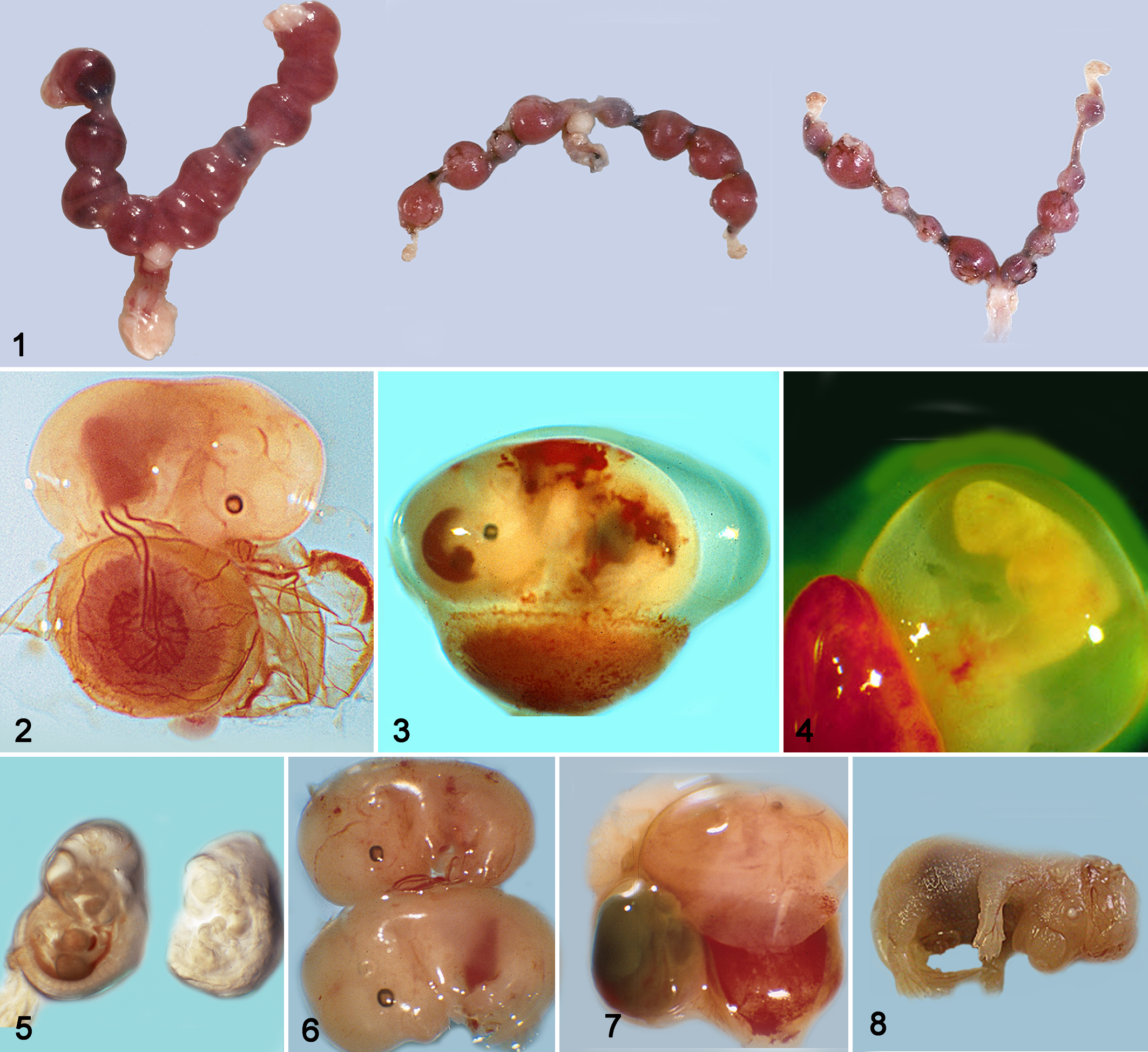
Mouse uteri at E12, E10, and E10 showing 1, 2, and 7 abnormal implantation (placental) sites.
A gross evaluation of the embryo and placenta is then performed. The rump to head length is recorded, as are any other gross findings. The placenta is examined to determine whether it appears grossly normal or abnormal. Depending on gestational age, the yolk sac, an appendage, the tail, or the entire embryo may be collected for genotyping. Any portion of the embryo not needed for histological examination may be used.
Mouse embryonic death may occur spontaneously 27 or may be induced by genetic engineering. Thus, any grossly abnormal embryo found may not only be an induced mutant. Examples of abnormalities and progressive gross changes leading to embryonic death are shown in Figs. 3–8. Growth retardation, growth arrest, or delayed development are reported, but we see embryonic lethality represented as early embryonic death (focal or diffuse degenerative and necrotizing lesions.
Histopathology Methods
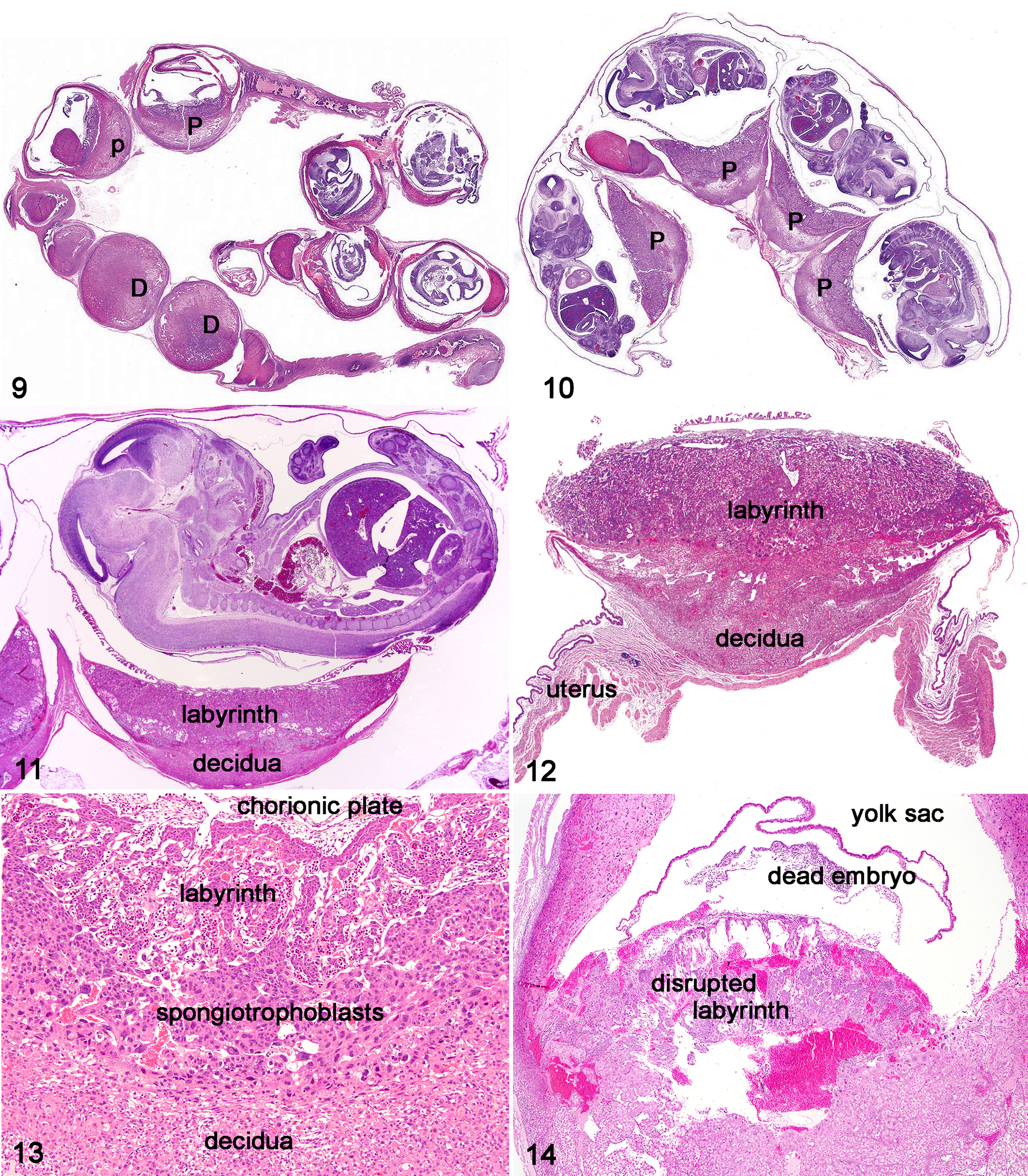
For evaluation of placenta and embryo histopathological characteristics, the placenta should be separated from the embryo. Embryos at most gestational stages are best fixed in Bouin’s fixative. 5,40 Embryo fixation is dependent upon gestational age: embryos less than E12 are fixed for 4 hours, E12.5–E16.5 for 24 hours, and greater than E16.5 for 24–48 hours. If in situ hybridization, immunohistochemistry, or molecular studies are to be done, 4% paraformaldehyde is the fixative of choice. Optimal embryo fixation and embedding protocols have been previously published. 5,23,24,40 The entire uterus can also be sectioned for overviews of uterus, placenta, and embryo (Figs. 9, 10). Dissection of all components (placenta, extraembryonic membranes, and embryo) usually results in the ability to perform more extensive examinations (Figs. 11–15). At embedding, embryos are oriented in the coronal (frontal), transverse, or sagittal plane. One tissue screening method for embryos older than E14 is as follows: at the time of necropsy, open the abdomen to allow better penetration of the Bouin’s fixative, and after optimal fixation, trim the embryo sagittally, making 1 cut down the middle (midsagittal) and trimming 1 remaining half sagittally into 2 pieces so that almost all tissues and organs can be found in the 3 long sections (except for small anatomical regions in the brain). Midsagittal sections alone make for good tissue scanning sections for most, but obviously not for all major organs (Fig. 15 ). The use of specific types of sections (frontal, sagittal, or transverse) is crucial for comparison, and the optimal type of sectioning can be organ specific. For example, a sagittal section of the embryo is excellent for an overall view of many organs, but the most useful section for the evaluation of the brain is coronal (frontal). Transverse sections are optimal for the evaluation of the spinal cord. With the use of a stereomicroscope, individual tissues of the larger later gestation embryos can be carefully dissected out of the body, trimmed, and embedded. The intact placenta is fixed in formalin (48 hours), not Bouin’s, with or without the uterus attached. Bouin’s is not used to fix the placenta because it alters the appearance of erythrocytes (Figs. 16–21). After fixation, the placenta is bisected adjacent to the umbilical vessels. Both halves are embedded face down within 1 paraffin block.
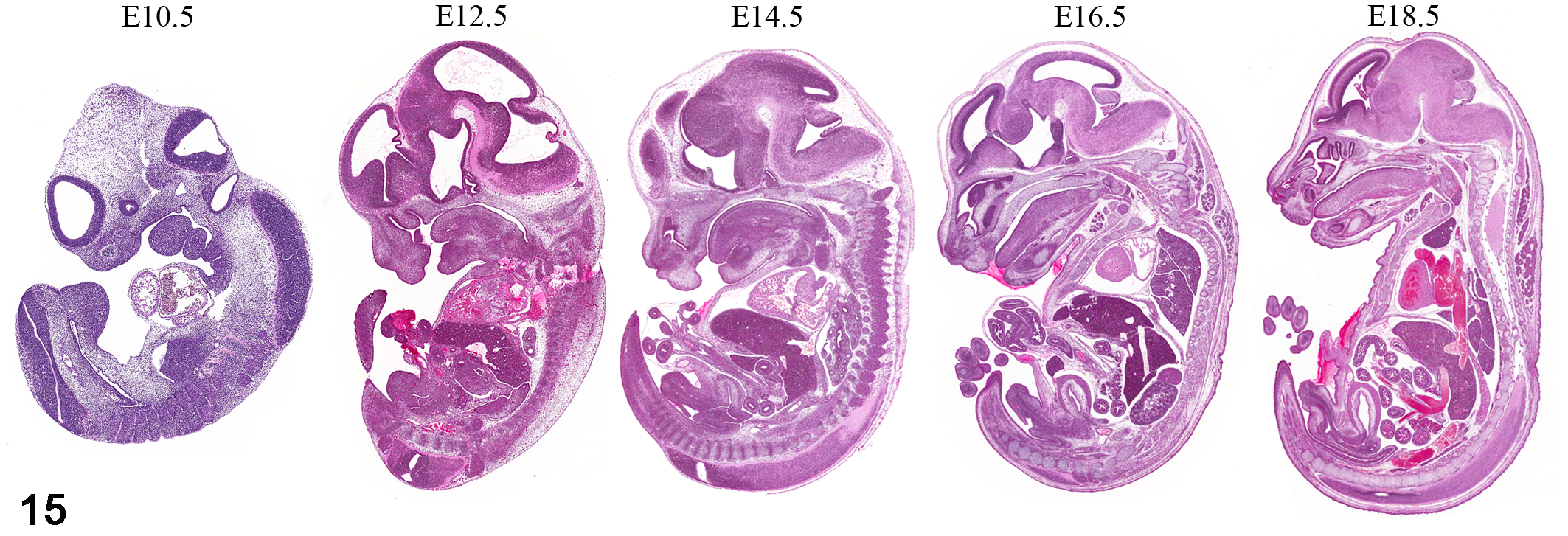
Mid-sagittal sections of the mouse embryo at different stages of gestation showing general organ development. Not to actual embryo size. Hematoxylin and eosin (HE) staining.
Histological Characteristics of the Normal Mouse Placenta
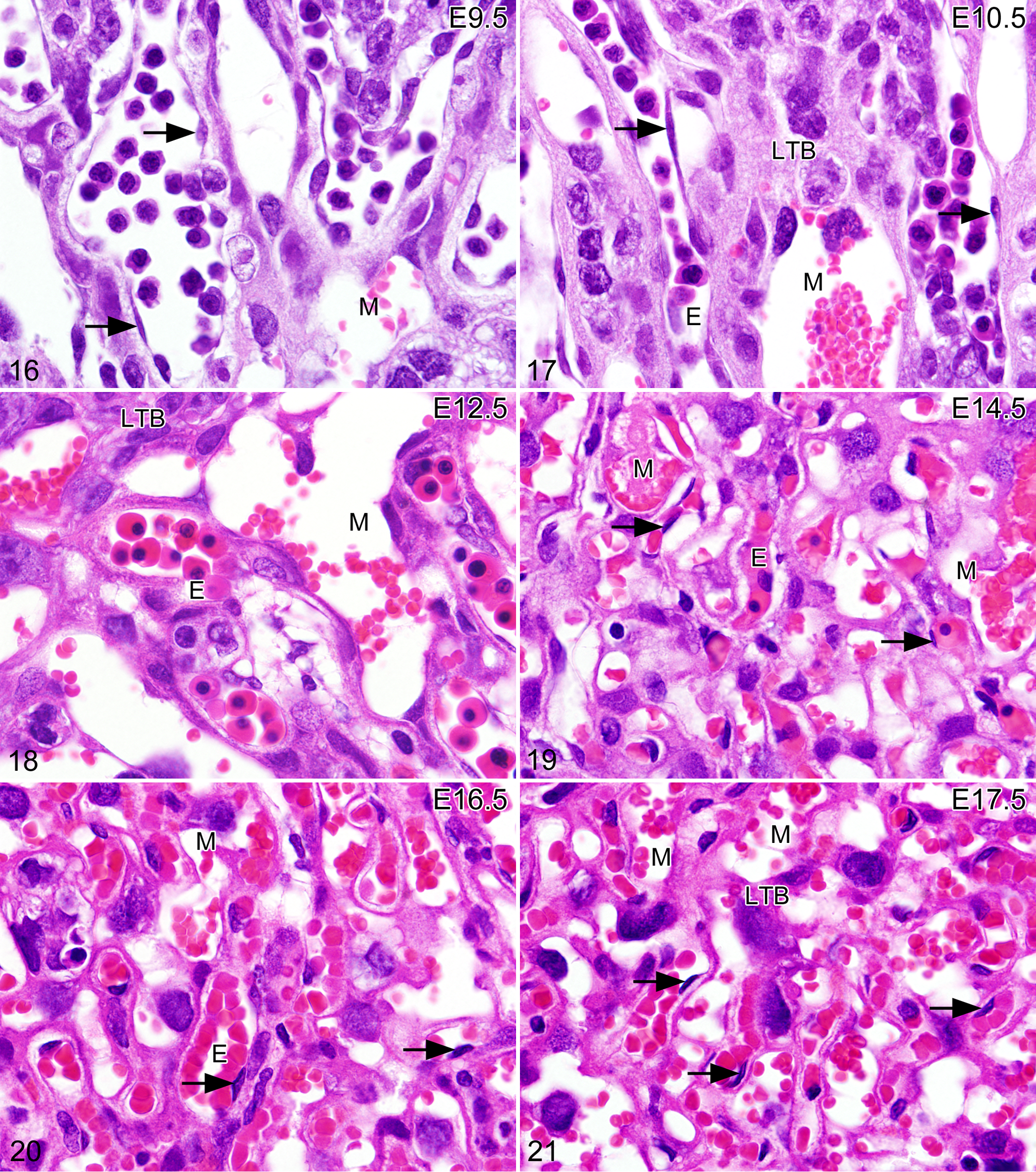
The mouse placenta can be described as discoid and hemotrichorial and has been compared to the discoid hemochorial placenta of humans and monkeys (http://placentation.ucsd.edu/). 16 The placenta, composed of both embryonic and maternal tissues, plays an important role in embryonic development and growth. The structure of the placenta allows nutrients to travel to the embryo via maternal blood and the trophoblast–embryonic blood interaction. The development of the mouse placenta has been well described. 6 –8,21,28,34,39,47,49,50 Pregnancy dating in the rat placenta is similar to that in mice. 9 By maturity of the placenta at E12, all structures are present (Figs. 11–13, 16–21) including the labyrinth with labyrinth trophoblasts and embryonic blood vessels lined by embryonic endothelium (Figs. 16–18). Vascular spaces are lined by embryonic labyrinth trophoblasts, contain mature maternal erythrocytes, and are also present in the spongiotrophoblast and giant cell trophoblast zones and decidua. The decidua, which is 100% maternal, is composed of blood vessels and macrophage-like cells expressing macrophage antigens. The metrial gland can be seen in the outer portion of the decidua. 47 If suboptimal sections are prepared of the placenta, one may see mostly decidua (Fig. 9 ). Normal age-related placental histological characteristics in wild-type embryos should be compared with those of the genetically engineered mouse when assessing mechanisms of embryonic death and possible abnormal placental development.
Many genes are expressed in the placenta, including many involved in adult carcinogenesis. 11,12,14,17,36,43 Inactivation of a number of genes that play a role in cancer development have also have been reported to result in embryonic mortality, especially involving the placenta. 48 Thus, how a gene functions in the embryo and placenta may differ greatly than those in adult mice. Immunohistochemistry studies and in situ hybridization can reveal cells and tissues expressing such genes (http://www.brain-map.org/; http://genex.hgu.mrc.ac.uk/). Inactivation of some genes can cause abnormal development and growth of the placenta and its membranes, leading to placental failure as an organ providing nutrition and oxygen to the embryo. 39,49 Without vital nutrients, embryonic tissues develop degenerative and necrotizing lesions, leading to embryonic death.
The embryo can be aged, somewhat, by the appearance of erythrocytes and their precursors in the embryonic vessels of the placenta (Figs. 16–21) and yolk sac. These findings may be related to the mouse strain. Immature erythroblast-like cells (Figs. 16, 17) are seen in early placental development, whereas erythrocyte maturation occurs over the next 3 days (E11–E14) and a population of mostly metarubricytes is observed (Figs. 18, 19). Histologically, by day 15, most embryonic erythrocytes have extruded their nuclear material (Figs. 19, 20). Degenerative changes commonly develop in the maternal decidua and placental trophoblasts (Fig. 21 ) as the placenta ages, including atrophy, vacuolation, hydropic change, and necrosis. Degenerative changes in wild-type and mutant placentas must be compared to determine whether there is an increase of these changes or even appearance of degenerative changes not normally seen, including hyperplasia, dysplasia, and multinucleated giant cells. 49
Causes and Evaluation of Embryonic Mortality After E12.5
Genetic engineering has produced many mouse developmental defects resulting in embryonic lethality and postnatal mortality. Information for many of the embryonic lethal mutants can be found at the Jackson Laboratory Mouse Genome Informatics Web site (http://www.informatics.jax.org) under Phenotypes & Disease Models. In an accompanying article, the authors discuss evaluation of embryonic lethality prior to E12.5. 30 We will review embryonic lethality as it applies to placental lesions prior to E12.5, embryonic lesions after E12.5, and after birth (due to abnormal in utero development).
Necropsy Strategy for Determining the Causes of Embryonic Lethality
It has been estimated that perhaps 5%–15% of wild-type fertilized embryos may be embryonic lethal. 6,27,32 Thus, not all dying or dead embryos are a result of induced genetic changes. If null mice are absent after birth, it is most likely that the null mutation is embryonic lethal. To determine whether embryonic lethality begins post implantation and to assess the histopathogenesis of lethality, a strategy would be to begin collecting litters at E16. If null embryos are still absent and abnormal placentation sites can be seen, work backward and collect litters at 2-day gestation intervals (E14, E12, E10) to pinpoint the initial developmental abnormality leading to defects or lethality.
Gross Abnormalities of the Uterus, Placenta, and Embryo
Initial evidence of embryonic mortality is a decrease in size and change in color of the normal placental site in the uterus (Fig. 1). As the embryo dies, the placenta also undergoes degenerative and eventually necrotizing lesions. The number of abnormal placental (implantation) sites that develop during gestation depends on the cause and the nature of the defect during normal development of the placenta or embryo. If vascular abnormalities develop in the placenta, hemorrhagic lesions may be seen (Fig. 14 ). 18 Initial embryonic changes may include loss of a normal heartbeat or abnormal appearances or loss of blood vessels or erythrocytes within the visible blood vessels. Focal hemorrhages may appear, and large areas of hemorrhage are often indicative of megakaryocyte or platelet defects (Fig. 3 ), almost always after E13. As more severe gross changes develop, there is loss of normal embryo structure as it becomes more amorphous in nature (Figs. 4–6). Eventually, the embryo dies. Over time, the embryo is gradually reduced in size and becomes a discolored, amorphous structure, unrecognizable as an embryo (Fig. 7 ).
Evaluation of the Placenta
The development of the placenta, which is complete by E12.5, may fail because of a variety of developmental membrane defects (eg, failure of allantois to fuse to the chorion, yolk sac failure, mesodermal/endodermal defects, lack of normal erythropoiesis in the yolk sac, placental defects in vasculogenesis, trophoblast formation, and decidua formation). 35,43,49 The dam is often heterozygous or homozygous null for a gene, and thus the expression of the gene in the decidua may be altered, possibly affecting decidua and placental development. 15
If the placenta fails to develop normally, the consequences are disruption of nutrition and oxygen to the developing embryo. Embryonic growth is restricted. Lack of normal growth and development leads to initiation of embryonic lethality often by E10 and commonly before E13. Histologically in the embryo, one can see focal, multifocal, or diffuse necrosis of various cells and tissues. After embryonic death, the remaining tissue is resorbed but may leave residual lesions at the placental site (Fig. 1). The structure of the labyrinth and other anatomical regions may be compromised, causing disruption of normal placental anatomy and, therefore, causing hemorrhage within the labyrinth.
Phenotypes From E12 to Birth
Gene inactivation causing initial developmental changes at E9–E10 may also cause initial histological changes in the placenta and embryo at that time. These progressive changes could result ultimately in embryo death within a few days or over a period time (several days or longer). Thus, at E12, embryos dead or dying from earlier developmental alterations can be observed as smaller placental sites in the uterus (Fig. 1). Embryos with initial changes after E12 may survive the entire gestation period or die after E12 but prior to birth. Common causes of embryonic death after E12 include heart defects involving failure of the cardiac conduction system and septation 40 and hematopoietic defects involving erythroid and megakaryocyte differentiation. 20 The majority of dying or dead embryos found after E12 have defects that have started prior to E12. However, developmental defects initiated in any stage of gestation for brain, kidney, liver, and lung may allow the embryo to survive in utero but could result in neonatal or postnatal mortality.
Phenotypes in the Perinatal Period and Beyond
Mice may survive the gestation period with an organ or system developmental defect, especially defects of the skin, skeleton, lungs, kidneys, liver, and central nervous, immune, endocrine, and reproductive systems. Defects involving the placenta, erythroid, or platelet development and the heart often result in intrauterine mortality. Defects initiated in utero may be lethal at some age postnatally, such as cystic kidneys, 1,19 or mice may be viable with nonlethal lesions such as loss of 1 kidney or reproductive failure. Death, secondary to these defects, can occur quickly at the time of birth, such as when lack of normal lungs or brain dramatically impairs normal postnatal viability. When postnatal mortality occurs, investigators can return to the embryo and neonate to study the histopathogenesis of the postnatal lethal lesions and determine the developmental defect that led to death. Developmental defects, however, may also be compatible with life, with or without clinical consequences. In humans, these include cleft lip and palate, neural tube defects, ciliopathies, polycystic kidney disease, laminopathies, deafness, and the muscular dystrophies. Some mouse models exist for these human conditions.
There are numerous reports of mouse mutants that breathe briefly at birth and subsequently die. For some of these mutants, the cause of respiratory insufficiency is unknown. In our experience with these cases, lung histological status is usually normal or only slightly deviant from normal. Death may be related to the breathing mechanism, rather than a defect in lung anatomical, histological, or physiological characteristics. Research of the breathing mechanism is a specialty involving knowledge of respiratory physiological characteristics and nervous system gene expression patterns. An excellent example of neonatal respiratory insufficiency was reported in necdin (Ndn) null mice. 37 Absence of the gene normally expressed during neuronal development resulted in disruption of pre-Bötzinger complex, which is essential for the generation of respiratory rhythm. In this case, the investigators knew the anatomical brain location of Ndn gene expression before they created their mutant mice, which aided significantly in their ability to identify the cause of death.
Resources for the Evaluation of Mouse Embryo Organ Development
With the rapidly expanding use of genetically engineered mice in biomedical research and the widespread development of mouse models to study the pathogenesis of human diseases, additional and improved histology atlases are required to evaluate embryonic malformations and lethality. Pathologists and biomedical researchers have historically relied on atlases for illustrations and descriptions of normal structures relevant to the most important developmental events in the embryonic mouse. 5,40,23,24,47 Other useful resources include an online tutorial of normal mammalian development using scanning electron micrographs (http://www.med.unc.edu/embryo_images/), a high-resolution magnetic resonance histology atlas of the embryonic and neonatal mouse, 33 and the Edinburgh 3D mouse embryo anatomy atlas (http://www.emouseatlas.org/emap/home.html).
The complexity of organ development underlies the need for such resources. For example, the developing heart undergoes a complicated 3-dimensional structural change when the heart tube loops to the right during embryonic days 8.5–10.5 and the venous pole subsequently moves cranially and dorsally. 40 Concomitant with the rightward looping, lengthening heart tube and further ballooning of the future chambers take place. Additional structural changes occur until the final cardiac architecture is achieved. Although the liver doesn’t undergo such complicated architectural changes, there are changes at the cellular level, which can best be illustrated with high-magnification images. 5 Embryonic liver venous drainage and embryonic hematopoiesis are also important factors to consider.
Knowledge of how the organ was sectioned (frontal, sagittal, or transverse) is crucial for comparison and the optimal type of sectioning can be organ specific. For the heart and hepatobiliary system, frontal, transverse, and sagittal views at each stage of development may be useful (Figs. 22–25). Providing all 3 views can be helpful when there are structures that can be seen in one view but not in another view. When appropriate, high-magnification images can illustrate important cellular detail such as apoptosis in the outflow tract cushions of the heart (Figs. 26,27) and hematopoietic cells, hepatoblasts, and erythrocyte morphological changes in the liver. 5,40 Special stains can be used to illustrate specific cellular and tissue morphology such as macrophages within erythroblastic islands and the presence of granulocyte progenitors in the developing liver. 5
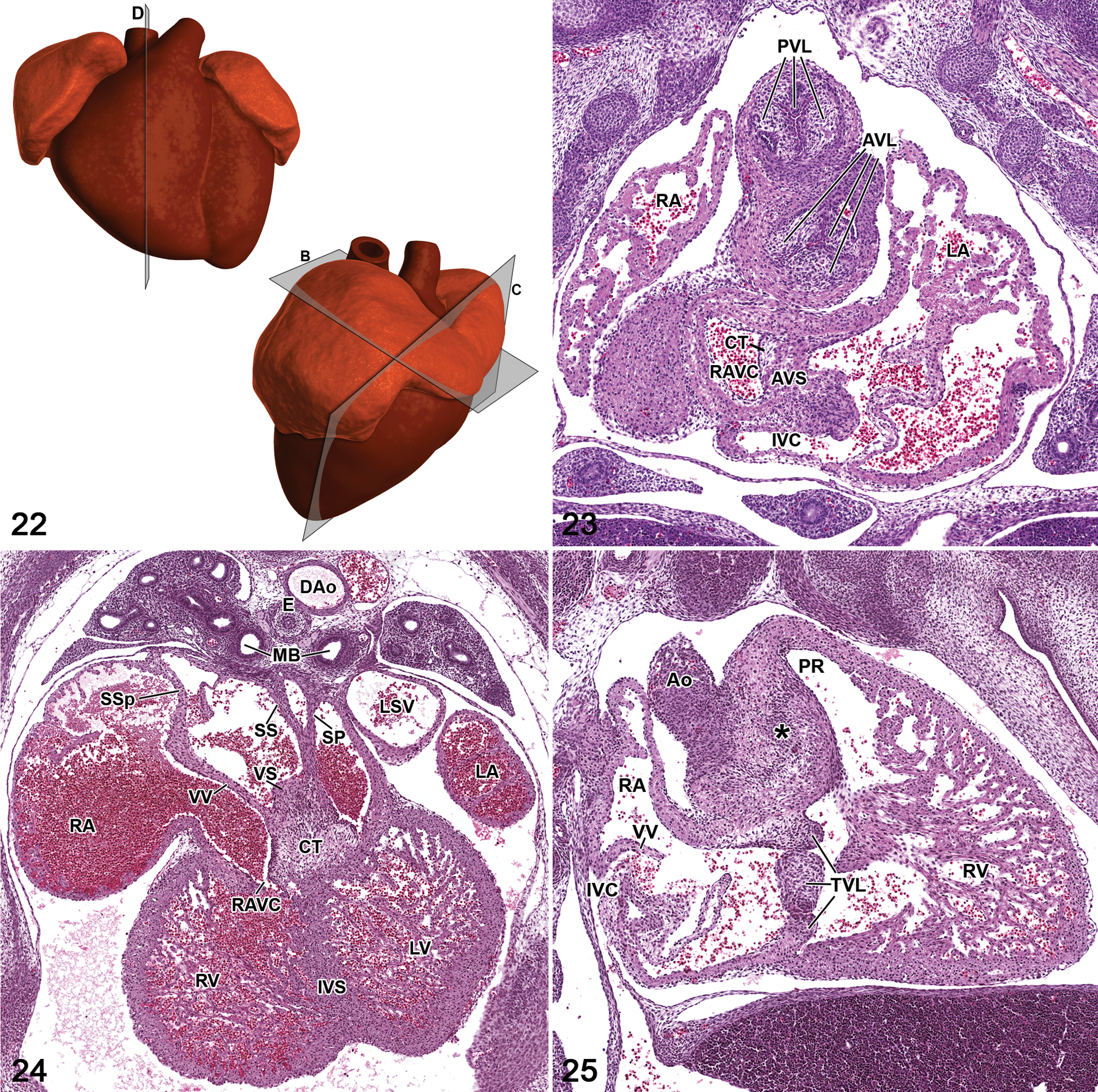
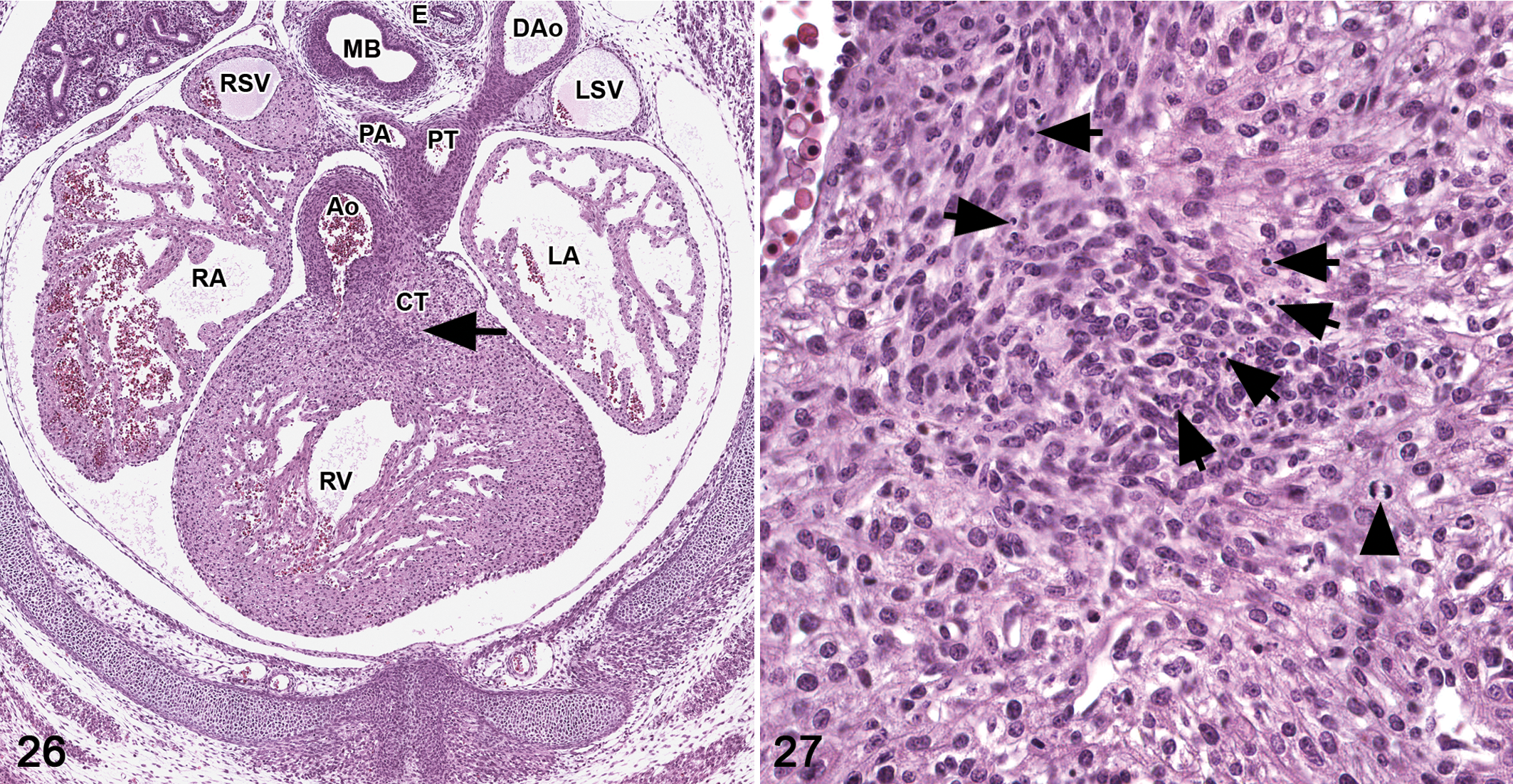
The overall goal for histology atlases and online resources is to provide tools that pathologists and biomedical scientists can use for proper evaluation of developing mouse organs. Critical to the phenotypical evaluation of any embryo organ system is a thorough knowledge of early mouse development, main developmental events, normal histological characteristics, all features unique to that organ system, major abnormalities seen at each embryonic stage, and important early postnatal developmental events.
Modalities for Imaging the Mouse Embryo in 3D: High-Resolution Anatomical and Anatomical Pathological Methods, Low-Resolution Histological and Histopathological Methods
Besides the availability of enhanced histology atlases for evaluating mice, one can also use advanced imaging techniques to assist with the traditional histological methods of the embryo assessment. Using conventional histological methods to identify and characterize possible developmental defects is labor intensive and time consuming. High-throughput 3D imaging methods improve screening embryos for possible developmental phenotypes. Imaging is basically a high-resolution anatomical and anatomical pathological method but a low-resolution histological method. Technological advances in the 2 primary methods of nondestructive, noninvasive imaging—micro–magnetic resonance imaging (μMRI) and micro–computed tomography (μCT)—allow the imaging of small specimens such as mouse embryos (Fig. 28). Other optical modalities of high-throughput imaging, such as episcopic fluorescence imaging capturing (EFIC) and optical projection tomography (OPT), have been developed to examine mouse embryos in 3D. These imaging methods are discussed below as they apply to mouse embryo phenotyping.
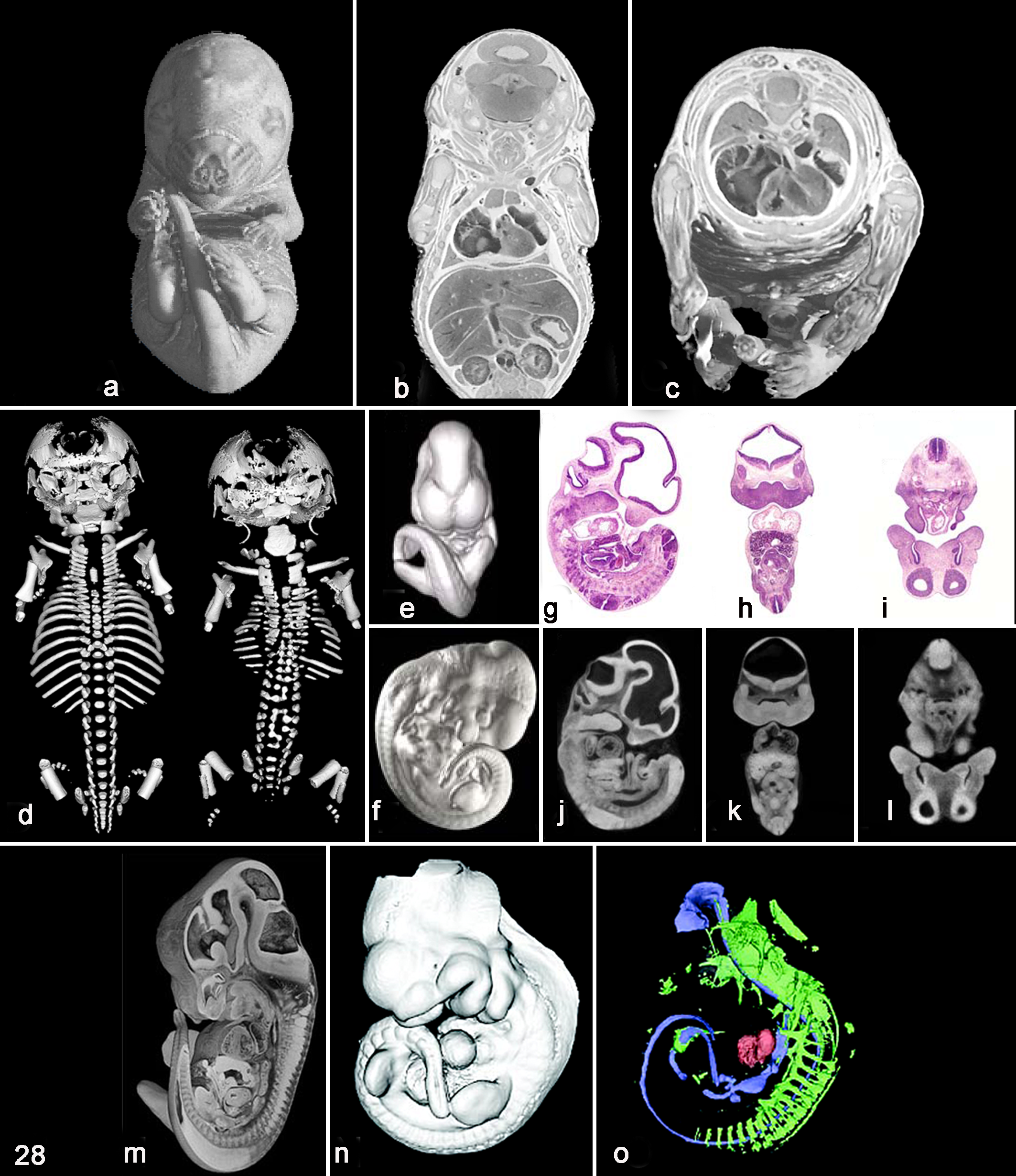
Representative images for methods of imaging mouse embryos in 3 dimensions. (a-c) μMRI of an E17.5 mouse embryo at 19.5-μm isotropic resolution. Isosurface, coronal, and transverse rendered images. (d) 3D rendering of a μCT scan (10-μm resolution) of the skeletal system from an E18.5 mouse embryo. Dorsal view of a wild-type mouse versus a transgenic littermate overexpressing human COX-2. (e-l) 3D rendering from a μCT scan of an E11.5 mouse embryo. (E, F) Isosurface renderings of a μCT-scanned embryo. (g-l) Comparison of hematoxylin and eosin (HE) and computed tomography sections of an E11.5 mouse embryo scanned at 8 μm. (m) High-resolution episcopic microscopy (HREM) used to compile a series of 2-μm, 2D block surface images into a 3D image from an E15.5 mouse embryo. (n-o) Fluorescence OPT imaging of multiple signals within an E10.5 mouse embryo with HNF3β antibody. (n) 3D rendering of an isosurface generated from the voxel reconstruction of the autofluorescent signal. (o) The isosurface has been rendered transparent, enabling the 3D shapes of the antibody staining patterns to be observed within the embryo (blue, HNF3β expression; green, neurofilament). The high autofluorescence of blood in the heart has been rendered in red. Figures 28a–c courtesy of the Duke Center for In Vivo Microscopy, supported by NIH/NCRR P41RR005959 and NCI U24 CA092656. Figures 28e–l reprinted according to the Creative Commons Attribution License; 2006. Johnson et al. Virtual histology of transgenic mouse embryos for high-throughput phenotyping. PLoS Genet. 2006;2:e61. Figure 28m courtesy of Dr. T. J. Mohun, supported by the Medical Research Council through the MRC National Institute for Medical Research. Figures 28n–o reprinted from Sharpe J, Ahlgren U, Perry P, et al. Optical projection tomography as a tool for 3D microscopy and gene expression studies. Science. 2002;296:541-545. Reprinted with permission from AAAS.
Micro–Magnetic Resonance Imaging
μMRI is a nondestructive imaging modality that provides accurate geometric representation of mouse anatomy and can also be used when phenotyping the mouse embryo (http://www.birncommunity.org/data-catalog/duke-center-for-in-vivo-microscopy-civm-high-resolution-mri-images). Magnetic resonance imaging is based on the alignment of hydrogen atoms of the specimen within a strong magnetic field, subjecting them to pulses of radio waves and then detecting the 3D distribution of further radio waves that are emitted as a response to the excitation. 42 High-contrast images can be obtained of soft tissue structures at about 25 μm resolution in the mouse (Fig. 28a-c). Since the first studies using μMRI, it has emerged as a technique to facilitate high-throughput phenotyping of genetically engineered mice and ENU mutagenesis screens and to investigate development effects secondary to teratogens and the environment. 41 As a preferred instrument for studying soft tissue structures, μMRI has been most powerful in the rapid identification of cardiac malformations such as atrial and ventricular septal wall defects, outflow tract malformations, and aortic arch remodeling defects. 3,4,41 Advances in instrumentation, such as stronger magnets, and replacement of complicated methods of sample preparation by simple immersion staining techniques have allowed for enhanced visualization of soft tissues and, more recently, mineralized tissue. 13 Disadvantages of using μMRI include increased costs attributable to complex instrumentation typically housed at large medical/research centers and scan time. Low image resolution is another drawback. At 25-μm resolution, μMRI is not capable of differentiating cells and some tissues.
Micro–Computed Tomography
Traditional methods for examining the skeletal system of the mouse embryo involve a dual-stain procedure for the detection of cartilage (alcian blue) and bone (alizarin red). Limitations of this conventional embryo skeletal staining method consist of time-consuming and laborious specimen preparation, tissue integrity loss, and fragile samples, which are difficult to image since they easily fragment when handled. μCT is a nondestructive tomographic imaging method that circumvents many of these problematic issues. Mathematical models create a 3D reconstruction of internal organs, vessels, and bone from a large series of 2D X-ray data taken around a single axis of rotation from a series of digital images. 26
μCT is used extensively for imaging mineralized tissue, since the radio opacity of mineralized bone makes it easy to detect (Fig. 28d). Skeletal analysis can be accomplished at a lower resolution of 38 μm, thus decreasing scan time, computational power, and expense. 29 For bone metrics, μCT also allows for the generation of accurate data such as bone volume and average bone mineral density, which is not possible in conventional-stained embryos.
Widespread use of μCT for nonmineralized tissues has been restricted by the low X-ray contrast of soft tissues; however, recent advances in contrast agents and instrumentation with increased x-ray strength are changing this limitation. 26 In comparison with μMRI, μCT offers higher resolution of soft tissue scans, 6–10 μm versus 25 μm (Fig. 28e, f, j-l). Histology equivalents of Fig. 28j-l are also shown (Fig. 28g-i). Scanning time is rapid for μCT, 2 hours versus 9–14 hours. 22 Since scanning time is decreased, costs associated with μCT are also reduced. As for μMRI, instruments for μCT are expensive, typically are housed at large medical/research centers, and require a skilled technician for instrument operation and software rendering of 3D images.
Episcopic Fluorescence Image Capturing
EFIC is a histological method that provides accurately aligned, high-resolution (2 μm) digital images that can be examined through computer animations of stacked images, surface rendering, or volume rendering procedures. Embryos E10.5 or later are routinely processed according to gestational age and blocked in paraffin. Since the tissue has the intrinsic property of autofluorescence, episcopic fluorescence microscopy is used during sectioning to capture digital images of the freshly cut block surface. 52 Because the block surface rather than individual cut sections is imaged, precise alignment of the specimen is obtainable. In addition, tissue distortions produced by routine histological sectioning and staining are avoided, since digital images are acquired prior to sectioning and no special staining is required of the specimen. Embryos less than E10.5 are not optimal for EFIC given that there is little tissue to for autofluorescence. Stacked images can be looped with QuickTime or the public domain software Image J. Although it is not possible to discern specific cellular structure and only the largest intercellular spaces can be identified, recent developments in this field have allowed for the 3D visualization of molecular markers such as GFP and LacZ in the embryo. In high-resolution episcopic microscopy (HREM), embryos are embedded in a resin with added fluorescein dyes, which enhance the captured digital image (Fig. 28m). 51,52
Optical Projection Tomography
With regard to 3D imaging of the mouse embryo, Sharpe, 42 the inventor of OPT, which is based on the ability of light to be transmitted through the sample, describes the technique as filling the gap between confocal microscopy and μMRI. OPT is the optical equivalent of μCT, which scans and records a quantitative shadow of the object, whereas OPT uses image-forming optics to create a focused image on a CCD camera chip. Digital images can be acquired from specimens up to 15 μm thick (embryos <E10.5) with a resolution of 5–10 μm. Like EFIC, OPT offers the advantage of visualizing RNA and protein expression in an intact embryo; however, OPT allows for the visualization of molecular markers in a 3D rendered image (Fig. 28n-o). 42
Although attempts have been made to render conventional 2D histological mouse embryo specimens into 3D structures by aligning numerous digital images of serial cut, HE-stained sections, the results cannot compare with more sophisticated, 3D imaging modalities. Whereas 2D histological analysis is the only method to ascertain high-resolution images at the cellular level, this level of detail is not necessarily required for the identification of anatomical development defects. Tables summarizing the advantages and limitations of the 3D modalities for the detection of a morphological phenotype in a developing mouse embryo are available for review. 10,22
As described in this article, postimplantation evaluation of the mouse embryo can be challenging. Using the described basic pathology methods, along with provided histological atlas references and possible 3D imaging techniques, the pathologist can determine embryonic lethality with confidence and assist other biomedical researchers using genetically engineered mice.
Summary
We have provided information on pathology methods and procedures that may be used for evaluation of the causes of mouse developmental defects and embryonic lethality due to induced gene manipulation. Determination of gene expression in tissues and cells and optimal necropsy, histopathological, and imaging methods will make such evaluations much easier. Resources such as Web sites, books, and other publications can also contribute. Collaboration between pathologists, developmental biologists, and other scientists is important to determine the causes of embryonic developmental defects and lethality.
Footnotes
Acknowledgements
We are grateful to Dr Deborah Gillette and Beth Mahler for photoediting, Dr Piper Treuting for the whole pregnant uterus slides, and Deborah Devor-Henneman for previous assistance with mouse embryology. We would also like to thank the journal reviewers who contributed to the editing of this paper.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported, in part, by the Intramural Research Program of the National Institute of Environmental Health Sciences, National Institutes of Health.
