Abstract
A 1.5-year-old Quarter Horse gelding with a history of chronic nasal discharge and leukocytosis presented with signs of increased lethargy and muscular pain. The horse quickly became recumbent and unable to rise and was euthanized due to a poor prognosis. At necropsy, severe bilateral guttural pouch empyema was observed, as well as numerous well-demarcated areas of pallor within the skeletal muscles of all major muscle groups. Polymerase chain reaction testing of the guttural pouch exudate confirmed an infection with Streptococcus equi subsp. equi, and an S. equi–associated immune-mediated rhabdomyolysis was initially considered to be the most likely diagnosis. This report briefly discusses the various etiologies that should be considered in cases of equine myopathy, and it demonstrates the complexity of these poorly understood muscular disorders.
A 1.5-year-old Quarter Horse gelding was referred to the Equine Internal Medicine Service at the Veterinary Medical Teaching Hospital at Texas A&M University in August 2008 for bilateral nasal discharge and leukocytosis of approximately 1.5 weeks duration. Previous treatments with flunixin meglumine (Banamine), enrofloxacin (Baytril), and fenbendazole (Panacur PowerPac) had resulted in no clinical improvement. The horse became increasingly lethargic and was stiff and slow to rise with continued bilateral nasal discharge. The owners reported that another horse diagnosed with an upper respiratory infection had recently been on the property.
Clinical and Gross Findings
On presentation (day 1), the horse was dehydrated and depressed with a stiff stilted gait and marked ventral edema in the pectoral and preputial regions as well as the right proximal hind limb. Rectal temperature was 101.1°F (38.4°C); heart rate was 80 beats per minute; and respiratory rate was 34 breaths per minute. Complete blood count results revealed a moderate leukocytosis with mature neutrophilia (white blood cell count, 19.9 × 103/μl [reference range, 5.4–14.3 × 103/μl]; segmented neutrophils, 90% [reference range, 30%–65%]; absolute neutrophils, 17 910 × 103/μl [reference range, 2260–8580 × 103/μl]), and initial biochemical analyses demonstrated severe elevations in AST (5865 g/dl [reference range, 134–643 g/dl]) and CK (4128 g/dl [reference range, 73-450 g/dl]) as well as a marked hyperphosphatemia (12.2 g/dl [reference range, 1.7–3.9 g/dl]).
Endoscopy revealed severe bilateral guttural pouch empyema, consistent with strangles. Since bacterial isolation of Streptococcus equi subsp. equi (S. equi) from nasal discharge, guttural pouch contents, or lymph node aspirates are considered the gold standard diagnostic approach for confirmation of S. equi infection, purulent exudate collected from the right and left guttural pouches was submitted to IDEXX/EBI for an S. equi polymerase chain reaction (PCR) and culture identification test. The guttural pouch contents cultured positive for S. equi and were also PCR positive for S. equi DNA. Serologic results for S. equi–specific antibodies were negative (< 1:200). However, ELISA results for S. equi M protein are frequently negative in these cases and therefore considered to be of little diagnostic value.
The horse received a 10-liter intravenous bolus of lactated Ringer’s solution every 6 hours, rifampin (5 mg/kg) by mouth every 12 hours, intravenous potassium penicillin (12 500 IU/lb) every 6 hours, intravenous flunixin meglumine (1.1 mg/kg) every 12 hours, and an intravenous single dose of dexamethasone (0.2 mg/kg). On day 2, the horse was recumbent, unable to rise, painful, and tachycardic with a heart rate of 84 beats per minute. Repeated AST and CK levels revealed that AST was significantly increased (6120 g/dl) and CK remained markedly elevated (2901 g/dl). The owners elected euthanasia due to the poor prognosis.
On gross examination, the guttural pouches bilaterally contained copious amounts of thick yellow purulent material (guttural pouch empyema). The retropharyngeal lymph nodes and tonsils were markedly enlarged and exuded similar purulent material on cut section (suppurative lymphadenitis and tonsillitis). Dozens of multifocal, dark red, poorly demarcated areas of consolidation, ranging from 0.5 × 0.5 × 0.5 cm to 2.3 × 2 × 1 cm, were within all lung lobes (pneumonia). The pericardial fat was diffusely transparent, gelatinous, and scant (serous atrophy of fat), and the heart contained dozens of multifocal to coalescing areas of pallor throughout the myocardium and endocardium. Multifocal to locally extensive, large (up to 10 cm), well-demarcated areas of pallor were within the skeletal muscles of all major muscle groups, including but not limited to the semimembranosus, semitendinosus, biceps femoris, pectoral, abdominal oblique, and sublumbar muscles (Fig. 1). The subcutaneous tissues were diffusely thickened, wet, and gelatinous and exuded copious amounts of clear fluid on cut section (generalized subcutaneous edema).
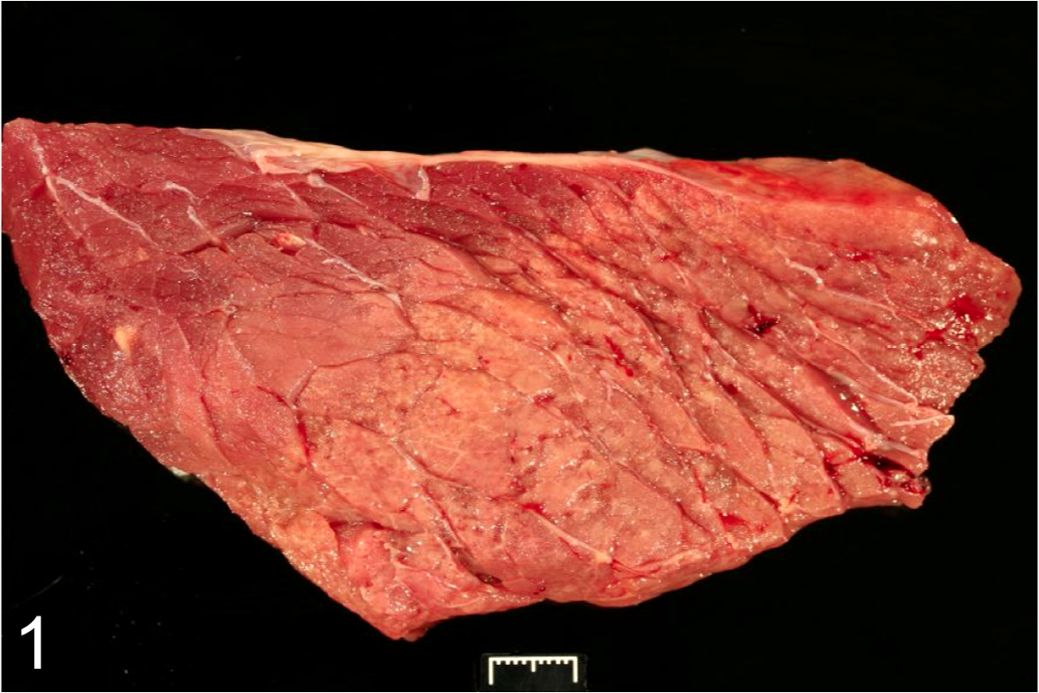
Semimembranosus muscle; horse. Note the clearly demarcated area of pallor.
The ureter of the left kidney was surrounded by a dark red, gelatinous mass, approximately 24 × 10 × 10 cm, encased within a firm, white, fibrous connective tissue capsule (organizing hematoma). The cortical surfaces of both kidneys contained dozens of pale, tan irregular, rough, and gritty foci measuring up to 1 × 1.5 cm and extending up to 0.5 cm into the cortex (mineralization). The bladder was markedly distended with approximately 2 liters of urine, and the abdominal cavity contained 1 to 2 liters of serosanguineous fluid (ascites).
Differential Diagnoses
There are many differential diagnoses for skeletal muscle necrosis in horses. Based on the presence of bilateral guttural pouch empyema caused by S. equi, an immune-mediated myopathy secondary to S. equi was initially considered the most likely diagnosis in this case. However, a variety of infectious agents, nutritional deficiencies, toxicities, and inherited metabolic disorders can cause similar macroscopic findings in horses. 3–5,7–14,19–26 Several of these causative agents are listed in Table 1 , and they include equine influenza virus, 8,20 Clostridium spp., 3,8,12,14,20,26 Salmonella enterica subsp. enterica serovar Infantum, 7,15 Sarcocystis fayeri, 3,8,9,12,20,26 vitamin E/selenium deficiency, 8,12,20,26 ionophore toxicity, 12,20,26 cantharidin toxicity, 12,20,26 white snakeroot toxicity, 7,12,20,26 pasture myopathy, 7 polysaccharide storage myopathy (PSSM), 5,12,13,20,25,26 and exertional myopathies (rhabdomyolysis). 4,5,8,12,20,26
Causes of Myonecrosis in Horses
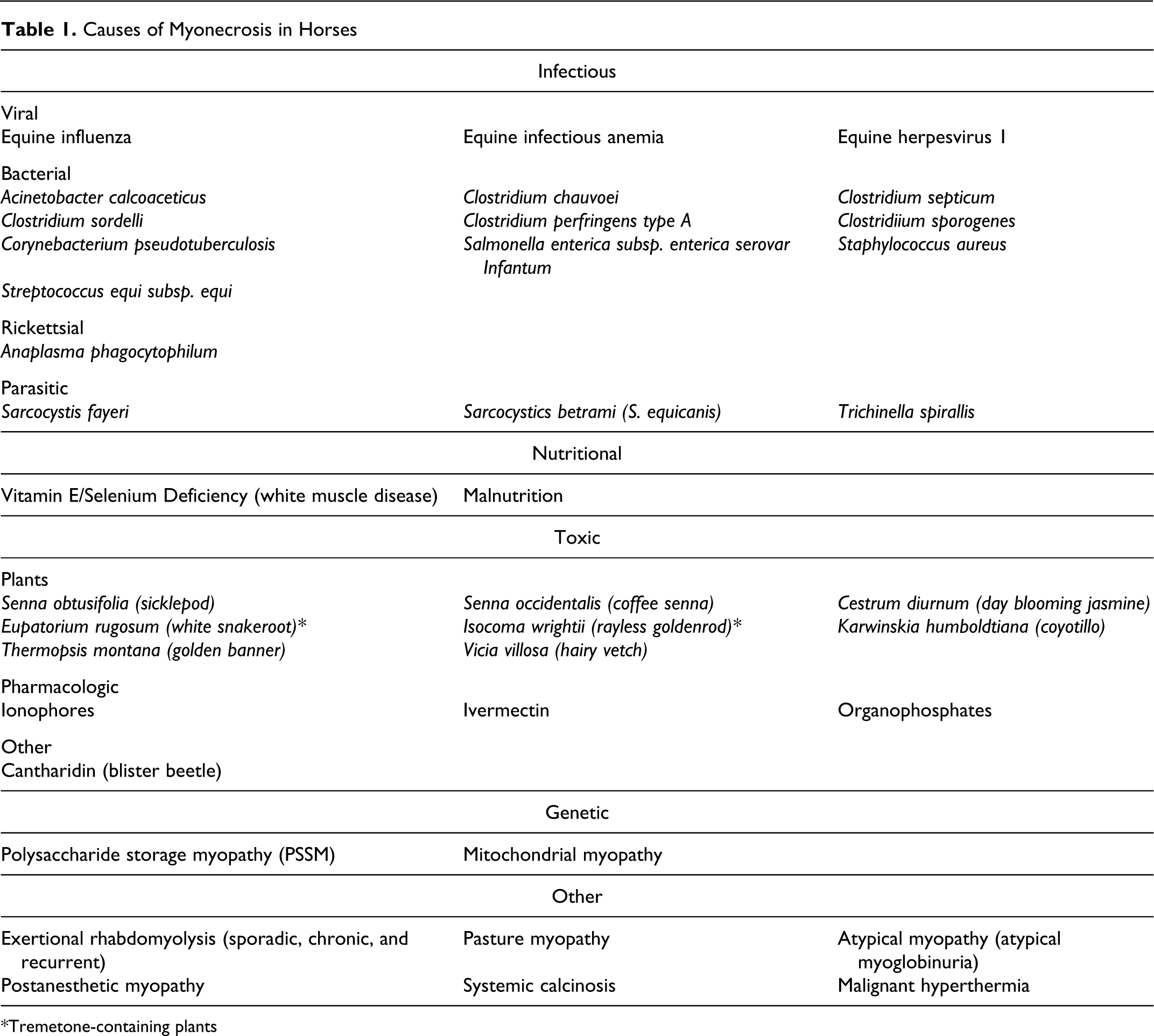
*Tremetone-containing plants
Microscopic Findings
Representative sections of selected tissues were fixed in 10% buffered formalin, processed routinely, sectioned at 5 μm, and stained with hematoxylin and eosin. Skeletal muscle sections were immunohistochemically stained for desmin (muscle marker), CD79a (B lymphocyte marker), CD3 (T lymphocyte marker), and CD18, which is expressed by a variety of leukocytes but most strongly by macrophages and granulocytes. On histologic examination, the skeletal muscle changes were multifocal and polyphasic. The majority of skeletal myofibers were lost and replaced by abundant fibrous connective tissue, proliferating satellite cells, and variable amounts of necrotic debris (Fig. 2). Multifocal areas of inflammation composed of numerous macrophages with fewer lymphocytes and occasional large multinucleated cells were within areas of myodegeneration and necrosis, often dissecting through or surrounding affected myofibers. Remaining muscle fibers were often irregular, swollen, and vacuolated (degenerative) or shrunken (atrophy) and frequently contained multiple internal nuclei (regeneration). Necrotic fibers were often mineralized (Fig. 3). The majority of multinucleated cells stained strongly positive for desmin (Fig. 4), but a small number of multinucleated cells exhibited weak CD18 expression, suggesting that they were multinucleated giant cells. Many mononuclear inflammatory cells stained for CD79a and CD3, consistent with B lymphocytes and T lymphocytes, respectively. CD79a-positive cells were more common than the CD3-positive cells (Fig. 5).
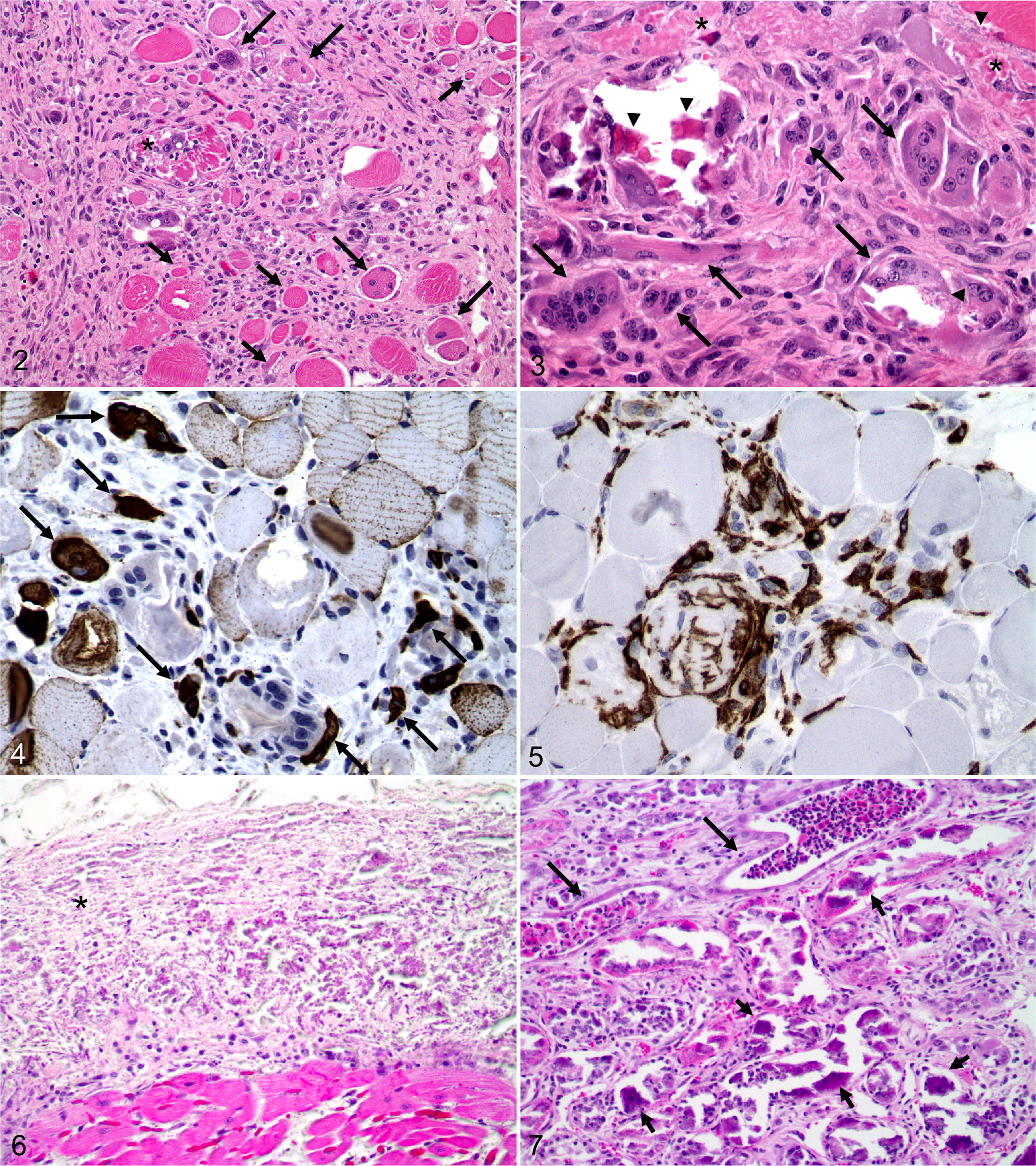
Skeletal muscle; horse. Skeletal muscle fibers are diffusely lost and replaced by abundant fibrous connective tissue infiltrated by a moderate number of mononuclear inflammatory cells. Note the area of necrosis (asterisk), as well as numerous degenerative (short arrows) and regenerative (long arrows) muscle fibers. HE.
Within the heart, a large amount of mineral was diffusely expanding the endocardium and proximal aorta (Fig. 6). The examined myocardial tissue was unremarkable. Bilaterally within the kidneys, the tubular epithelium was often necrotic, attenuated, or mineralized (Fig. 7). Tubules were frequently distended by a large number of viable and degenerative neutrophils admixed with sloughed epithelial cells and variable amounts of necrotic debris (cellular casts). Multifocal clusters of neutrophils and mixed mononuclear cells were also within the interstitium (tubulointerstitial nephritis). Areas of mineralization extended from the cortex into the medulla, and the lymphatics were diffusely dilated. A mild, focally extensive, fibrinous pneumonia with hemorrhage and edema was observed within the lung.
PSSM Testing
Fresh sections of haired skin and semimembranosus muscle were sent to the Equine Neuromuscular and Genetics Diagnostic Laboratory at the University of Minnesota. The muscle sections were routinely processed and stained with hematoxylin and eosin, periodic acid–Schiff, Gomori trichrome, and amylase periodic acid–Schiff stains. Due to a reportedly mild accumulation of subsarcolemmal glycogen within examined muscle sections, further testing was conducted for PSSM type 1 via DNA isolation and GYS1 genotyping. A DNeasy Tissue Extraction Kit (Qiagen, Valencia, CA) was used to extract genomic DNA from the hair roots according to the manufacturer’s protocol. The GYS1 genotype of the horse was obtained by a PCR–restriction fragment length polymorphism method that examined a 230–base pair segment of DNA containing GYS1 exon 6 and the flanking intronic sequence as previously reported. 13 Genotypes are generally classified with this method as homozygous normal R/R, heterozygous affected R/H, or homozygous affected H/H. This horse was reportedly homozygous normal.
Discussion
The clinical, gross, and histologic findings in this case were most consistent with severe, chronic–active rhabdomyolysis suggestive of S. equi immune-mediated myopathy, vitamin E/selenium deficiency, or systemic calcinosis. Among equine myopathies, “exertional” or exercise-induced rhabdomyolysis disorders are the most common, synonymous with the lay terms “tying up” or “Monday morning disease.” 3,4,8,12,20 Exertional myopathies can often be diagnosed on the basis of the history and clinical signs, as such horses typically present with a stiff/stilted gait, excessive sweating, and increased respiratory rate during or after exercise. 12,20 Since there was no historical evidence of exercise before the development of clinical signs in this case, exertional rhabdomyolysis was excluded from the list of differentials.
Nonexertional equine myopathies can be attributed to a number of etiologies that cause significant myonecrosis, including various infectious agents, nutritional deficiencies, toxicities, or metabolic diseases. 3–5,7–15,19–26 Viral infections associated with myonecrosis in the horse include equine influenza, equine infectious anemia, and equine herpesvirus 1. 8,20 In the United States, horses are routinely vaccinated for equine influenza and equine herpesvirus 1 and checked annually for equine infectious anemia. A viral etiology was considered unlikely in this horse given its current vaccination status and the lack of supporting lesions in other organs.
Several bacteria have been associated with equine rhabdomyolysis. 3,5,8,12,15,20–24,26 Clostridium perfringens type A, C. sordelli, and C. septicum are the clostridial organisms most commonly isolated from horses with clostridial myonecrosis. 8,14 However, infections with C. chauvoei and C. novyi type B have also been documented. 8,12,14,20,26 Clostridial myonecrosis is rapidly progressive and fatal, with coma or death resulting within 12 to 24 hours. 12,20 Muscular lesions are characterized by marked edema with regionally extensive to multifocal, dark red to black areas of necrosis and hemorrhage that are often emphysematous and maloderous. 26 Any muscle group can be affected, but lesions typically present in a limb or in the trunk and are often associated with an open laceration or recent injection site. 14,20,26 Although samples of muscle from this case were not submitted for bacterial culture, the lack of an associated wound, the prolonged clinical progression, and the gross and histologic appearance of the muscle lesions collectively eliminated clostridial infection as a potential diagnosis.
Myonecrosis due to Salmonella sp. infection—specifically, Salmonella enterica subsp. enterica serovar Infantum—has also been documented in the horse. 15 In this previous report, muscle lesions were the result of a disseminated Salmonella infection characterized by a multifocal “pyomyositis” closely associated with overlying cutaneous infarcts and areas of cellulitis. As there was no gross or histologic evidence of a suppurative response within examined skeletal muscles in this case, Salmonella infection was considered unlikely. Protozoal infections in horses caused by Sarcocystis fayeri or Sarcocystis bertrami are also reported to rarely induce a chronic myositis, with some evidence of myonecrosis in horses, but these infections are typically mild with no apparent clinical disease. 3,8,9,12,20,26 Parasites were not evident in the muscle lesions of this horse.
Nutritional myopathy due to vitamin E and selenium deficiency, also known as white muscle disease, is typically a disease of young farm animals, less than 1 year of age. 7,12,20,26 In horses, the usual age range is 1 day to 12 weeks; adult cases are rarely reported. 7,20,26 Nutritional myopathy is classically defined as a multifocal polyphasic myopathy, of which 2 distinct forms have been reported in the horse: a cardiac form and a skeletal form. 20 The cardiac form usually presents as a sudden onset of severe debilitation or acute death. The lesions include myodegeneration and necrosis that primarily affect the heart, diaphragm, and intercostal muscles. The skeletal form has a much slower clinical onset, characterized by stiffness and muscular weakness. 5,12,20,26 These horses may be recumbent and unable to stand and often have hard, swollen muscles that are painful on palpation. The semitendinosus, semimembranosus, gastrocnemius, and biceps femoris muscles are among the muscle groups most commonly affected. 20,26 Grossly, muscular lesions are often bilaterally symmetrical with well-demarcated dry areas of pallor, as well as intramuscular edema and fibrosis. 7,8,20,26 Histologically, these areas of myodegeneration and necrosis may exhibit varying degrees of dystrophic mineralization that can resemble systemic calcinosis. 7,8,20,22,26 Vitamin E and selenium levels in serum can be used to determine the vitamin E and selenium status of an animal, as well as selenium-dependent glutathione peroxidase levels in whole blood or selenium concentrations in the liver. 12,20 Nutritional myopathy is considered extremely rare in the region of Texas that this horse originated; therefore, selenium and vitamin E levels were not evaluated antemortem or postmortem. Because of the lack of testing, nutritional myopathy cannot be excluded as the cause for the lesions.
Systemic calcinosis is a well-recognized disease in humans and has recently been described in horses. 22 In horses with systemic calcinosis, calcium deposits are observed within the connective tissues of the lungs, kidneys, stomach, heart, skin, and skeletal muscle. Affected horses have a similar clinical presentation as the previously mentioned myopathies, with signs including progressive weakness and inability to stand. As with other rhabdomyolysis syndromes, affected horses have elevated muscle enzymes, specifically CK and AST, but also a pronounced hyperphosphatemia. Although systemic dystrophic calcification can be a consequence of uremia, most horses in this recent report were not azotemic. The mineralization of the endocardium, aorta, renal tubules, and multiple skeletal muscles in this case, in addition to the marked hyperphosphatemia observed clinically, suggest systemic calcinosis as a possible differential diagnosis. Hypervitaminosis D from ingestion of Solanum glaucophyllum has also been reported to cause dystrophic calcification of multiple tissues. This plant is not endemic in Texas.
Numerous toxic agents are reported to cause myonecrosis in horses. 7,12,20,26 Ingestion of tremetone-containing plants such as Eupatorium rugosum (white snakeroot) and Isocoma wrightii (rayless goldenrod) often cause myonecrosis in horses and may even cause death when 0.5% to 2% of the horse’s body weight is ingested. Other toxic plants associated with myonecrosis include Senna obtusifolia (sicklepod), Senna occidentalis (coffee senna), Cestrum diurnum (day blooming jasmine), and Vicia villosa (hairy vetch). Feed additives and pharmacologic agents such as ionophores, organophosphates, and ivermectin reportedly cause myonecrosis in horses. 12,20,26 Based on the clinical history in this case, a toxicity was considered an unlikely cause for the rhabdomyolysis.
Seasonal pasture myopathy and atypical myopathy or atypical myoglobinuria have also been reported in horses. 5,7 In these instances, pastured horses present with an acute, severe, nonexertional rhabdomyolysis characterized by muscle stiffness and weakness progressing to recumbency. Horses are often lethargic and tachycardic and have dark red-brown urine consistent with myoglobinuria. Outbreaks of this syndrome affect multiple horses simultaneously and are frequently associated with inclement cold weather. Gross and histologic lesions of pasture or atypical myopathy include myodegeneration and myonecrosis of cardiac and skeletal muscles. This diagnosis is largely a diagnosis of exclusion. Pasture or atypical myopathy was not considered in this case, based on the clinical history and positive identification of S. equi as a potential causative agent.
Inherited metabolic disorders such as PSSM also cause myodegeneration and necrosis in horses. 5,12,20,25,26 PSSM is characterized by an abnormal accumulation of glycogen and glycogen-related polysaccharide within skeletal muscle fibers. Quarter Horses, Paint Horses, Appaloosas, Draft Horses, Draft Crossbreeds, and Warmbloods are the breeds most frequently affected, but rare cases in Thoroughbreds have been reported. Clinical signs range from a recurrent, exertional rhabdomyolysis to progressive weakness, muscle fasciculations, and, less commonly, muscle atrophy, renal failure, and respiratory distress. 19,25,26 Gross pathologic findings may be absent or include pale streaks within affected muscles consistent with myodegeneration and necrosis. 25 A diagnosis of PSSM is dependant on histologic examination and demonstration of periodic acid–Schiff-positive and amylase-resistant inclusions within skeletal muscle fibers. 19,25,26 However, recent reports have demonstrated that a mutation in the skeletal muscle glycogen synthase gene (GYS1) is highly associated with PSSM in horses, particularly the Quarter Horse and Belgian Draft breeds, so genetic testing may also be diagnostic. 13 Since the horse in this case was a young Quarter Horse with clinical signs of progressive muscular weakness, samples of haired skin and semimembranosus muscle were collected and submitted to the Equine Neuromuscular and Genetics Diagnostic Laboratory at the University of Minnesota for PSSM testing. Based on the results, PSSM was definitively ruled out as a possible cause in this case.
S. equi and its sequelae are commonly associated with equine rhabdomyolysis and myonecrosis.3,5,8,12,20,22–24,26 Bacterial isolation of S. equi from nasal discharge, guttural pouch exudate, or lymph node aspirates is considered the gold standard for the diagnosis of strangles. 20,23,24 S. equi PCR is an additional diagnostic tool often used in conjunction with bacterial culture to confirm S. equi infection. 23,24 However, PCR cannot differentiate between viable and nonviable bacterial organisms, so a positive result does not always indicate an active infection. 23,24 Serum levels of S. equi M protein can also be tested using an ELISA, but false negatives are common, particularly if exposure has occurred within the last 7 days. 18,23,24 Immunofluorescent staining of skeletal muscle for Lancefield group C carbohydrate, S. equi M protein, and a recently identified S. equi myosin-binding protein, Se18.9, is also reportedly effective in the identification of S. equi antigens in skeletal muscle. 19 An S. equi immune-mediated myopathy was considered the most likely diagnosis in this case, based on the positive bacterial culture and PCR results, as well as the history, clinical signs, and gross and histologic pathology. Repeat ELISA testing for S. equi M protein and immunofluorescence of skeletal muscle tissues were not attempted due to the unavailability of serum and frozen tissue, respectively.
Three immune-mediated myopathies have been associated with S. equi infection: (1) an acute rhabdomyolysis form characterized by severe myonecrosis and histiocytic inflammation, (2) an infarctive form of purpura hemorrhagica with coagulative necrosis and necrotizing vasculitis affecting multiple organs, and (3) an immune-mediated polymyositis form involving rapid muscular atrophy and a large number of lymphocytes within affected muscle tissue. 12,20,24 Based on the degree of bacterial infection and muscular necrosis in this case, the acute rhabdomyolysis form of S. equi immune-mediated myopathy was initially considered the most likely diagnosis. However, the degree of muscular fibrosis and mineralization was more typical of a chronic, rather than acute, process. Previous reports of immune-mediated myopathies suggest that the acute rhabdomyolysis form primarily affects young Quarter Horses less than 2 years of age, which matches the signalment of the horse in this case. 19,24 In S. equi immune-mediated myopathy, affected horses typically present with submandibular swelling and/or guttural pouch empyema and later develop signs of rhabdomyolysis that may include a stiff gait, difficulty rising, dark brown urine, and eventually firm, swollen, and painful epaxial and gluteal muscles. The muscle pain is nonresponsive to aggressive antimicrobial and anti-inflammatory therapies, and most horses become recumbent and unable to rise within 24 to 48 hours of hospitalization, often developing relentless pain that necessitates euthanasia.
The mechanism for S. equi immune-mediated myopathies is unknown. 24 Proposed mechanisms include the presence of cross-reacting streptococcal antibodies that target skeletal muscle myosin, direct muscle invasion by the bacteria, or toxin production. 6,19,24,26 Four S. equi superantigens (SePE-H, SePE-I, SePE-L, SePE-M) have been identified and described on the basis of their high immunomodulating capacity in the pathogenesis of equine strangles. 2,16,17,19,26 The strong mitogenic responses elicited by these S. equi superantigens are very similar to those induced by Streptococcus pyogenes, the group A Streptococcus implicated in necrotizing myositis and toxic shock syndrome of humans. 1,2,16,17 Streptococcal superantigens have a high affinity for binding major histocompatibility class II molecules. 16,17 The simultaneous binding of MHC II and T-cell receptor molecules with a specific V-β region results in the activation of numerous antigen presenting cells and T cells, causing a sudden and massive systemic cytokine release and subsequent toxic shock. The major cytokines released in response to superantigens include tumor necrosis factor α, the prime mediator of shock, tumor necrosis factor β, and interleukins 1, 2, and 12, which can further potentiate the inflammatory response and induce fever. A different Streptococcus species, S. pneumoniae, is also reportedly associated with an acute infectious form of rhabdomyolysis in humans, similar to the acute rhabdomyolysis form of S. equi immune-mediated myopathy. 6,19
The multifocal polyphasic changes within skeletal muscle in this case, as well as the marked fibrosis and mineralization of necrotic myofibers, indicate a more chronic lesion than what is typically associated with the rhabdomyolysis form of S. equi immune-mediated myopathy. Ultimately, this case was diagnosed as an S. equi immune-mediated myopathy with features of chronicity, although other etiologies cannot be completely excluded. This report demonstrates the complexity of determining the etiology in equine myopathy cases and emphasizes the importance of having a complete clinical history, performing a detailed postmortem examination, and requesting appropriate ancillary testing.
Footnotes
Acknowledgements
We thank Dr Stephanie Valberg for her comments regarding polysaccharide storage myopathy testing, Dr Andy Ambrus for assistance with immunohistochemistry, and John Roths for his photographic support.
This case was presented at the 18th annual C. L. Davis Foundation South Central Division Meeting, October 2008, College Station, Texas.
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The authors received no financial support for the research, authorship, and/or publication of this article.
