Abstract
B6.Cg-
The skin contains nerves that innervate the erector pili (smooth muscle), which raise the hair in involuntary response to environmental conditions such as low temperature, fear, or other stimuli. Below the hypodermal fat layer of the skin is a layer of striated skeletal muscle, the panniculus carnosus, innervated by motor neurons allowing the animal to move or twitch its skin. 3
The B6.Cg-
Variations in this transgenic construct, for example, using the “brainbow mice,” provides tools to follow individual nerves. 9 This investigation determined that these mice could be used successfully to investigate the innervations of the skin and appendages either by direct visualization of the excited YFP or indirectly by immunohistochemical detection of the YFP protein.
Materials and Methods
Mice
B6.Cg-
Labeling Nerves and Neuromuscular Junctions
The YFP16 skin samples were also incubated with tetramethylrhodamine-conjugated α-BTX (1:1000; Alexa 594 α-Bungarotoxin, catalog #B13423, Invitrogen [Molecular Probes], Carlsbad, California) diluted in PBS/0.3% Triton 100/1% bovine serum albumin (BSA) for 90 minutes at room temperature in the dark, followed with 3 washes for 15 minutes each in PBS/0.3% Triton 100. Tissues were placed on a slide and cover slipped with 80% glycerol. The slides were visualized with a confocal microscope (E600, Nikon, Tokyo, Japan) and imaged (RTColor camera, Diagnostic Instruments, Sterling Heights, Michigan). For frozen sections, fresh skin was lightly fixed in 4% PFA for 1 hour at 4°C and then transferred to 30% sucrose at 4°C overnight. The skin was embedded in Optimal Cutting Temperature compound (Tissue Tek, Torrance, California) at –30°C, and serial 10-μm sections were placed on Fisher Plus slides (Fisher Scientific, Pittsburgh, Pennsylvania). Slides were washed 3 times for 5 minutes in PBS and then incubated with α-BTX (1:1000) diluted in PBS/0.3% Triton 100/1% BSA for 90 minutes at room temperature in the dark. Slides were washed 3 times for 10 minutes in PBS/0.3% Triton 100 and were subsequently treated with 4′,6-diamidino-2-phenylindole (DAPI, 1:10000) for 5 minutes in the dark. Slides were again washed 3 times for 5 minutes in PBS and cover slipped with Fluoromount (Sigma, St. Louis, Missouri). Slides were examined and imaged (SP5 AOBS confocal microscope, Leica).
For immunohistochemistry, YFP16 skin was fixed overnight in Fekete’s acid-alcohol-formalin solution, transferred to 70% ethanol, embedded routinely, and sectioned at 6 µm. Using an autostainer (Discovery XT, Ventana Medical Systems, Tucson, Arizona), slides were double labeled using rabbit anti-green fluorescent protein (GFP, 1:6000, ab290, Abcam, Cambridge, Massachusetts) and rabbit or mouse anti-smooth muscle actin (SMA, 1:200, NB600-531, Novus, Littleton, Colorado, or 1:400, A 2547, Sigma). Detection kits used for labeling were UltraMap anti-Rabbit Alkaline Phosphatase with Naphthol-Fast red chromogen (red) and either OmniMap anti-Rabbit Horse Radish Peroxidase or DABMap anti-Mouse Horse Radish Peroxidase with diaminobenzidine chromogen (brown) (Ventana Medical Systems, Tucson, Arizona). When the mouse anti-SMA was used, the MoMap kit (Ventana) was also used to block nonspecific binding of anti-mouse antibodies. Additional sections were stained with hematoxylin and eosin (HE), phosphotungstic acid hematoxylin (PTAH), and Bodian’s stains, the latter 2 to emphasize striated muscle with cross-striations.
Results
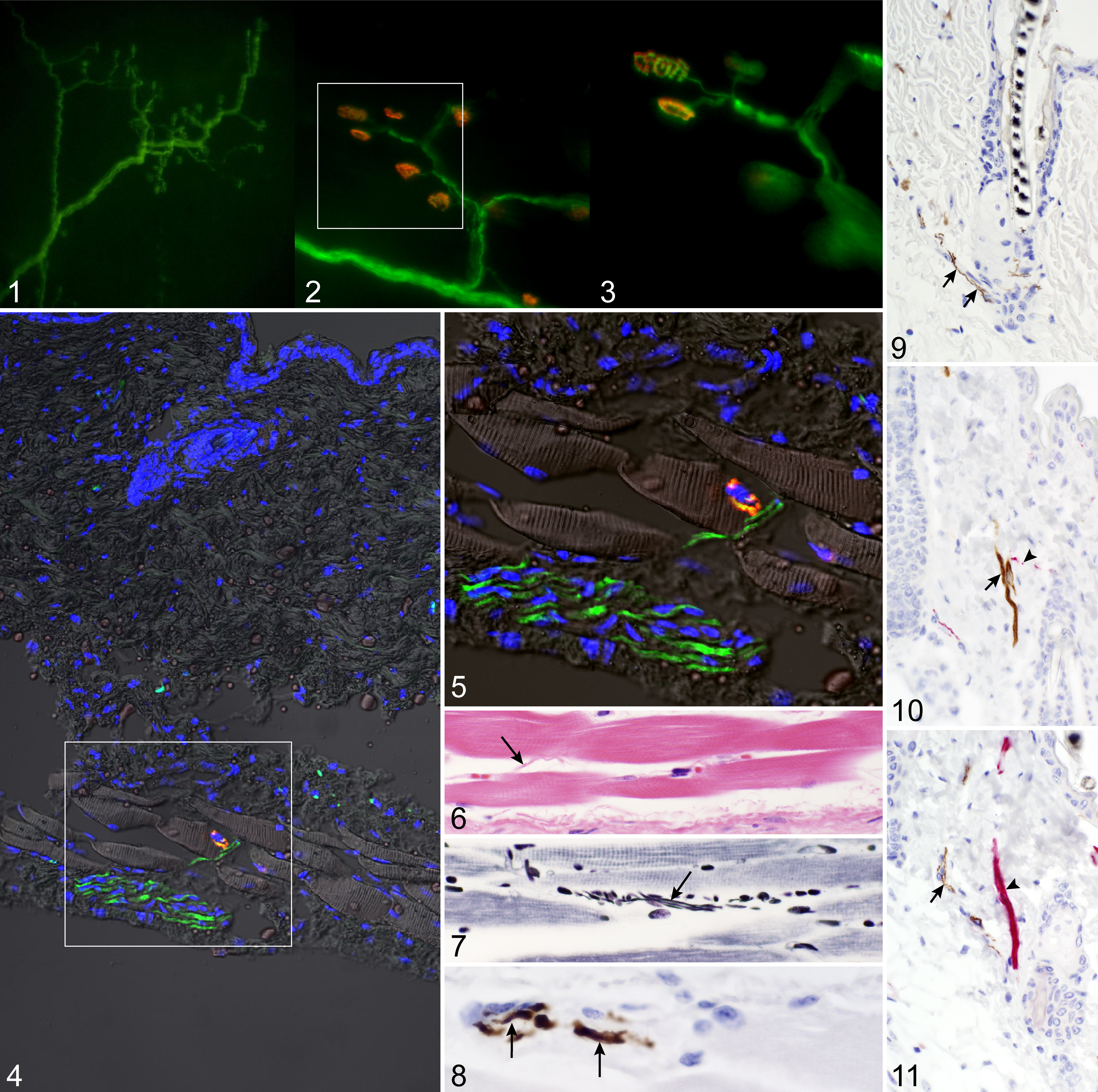
Direct examination of lightly fixed dorsal and ventral skin confirmed YFP expression in both the motor axon and the neuromuscular junction (Fig. 1). Frozen sections verified YFP expression of the motor axon and the neuromuscular junction that was labeled with α-BTX (Figs. 2, 3). The panniculus carnosus muscle was identified by its striated structure and subcutaneous location to the dermis associated with a nerve bundle, an axon, and a neuromuscular junction clearly innervating the muscle (Figs. 4, 5). The HE barely identified an axon, whereas the PTAH stain highlighted cross-striations in the panniculus carnosus muscle and axons (Figs. 6, 7). Immunohistochemistry, using paraffin-embedded sections of YFP16 dorsal skin, identified nerves in the panniculus carnosus (Fig. 8) and innervating the erector pili muscle and surrounding the bulge region (Fig. 9). Double labeling with anti-GFP and anti-SMA using both horseradish peroxidase–diaminobenzidine and alkaline phosphatase in either combination confirmed innervation of the erector pili muscle (Figs. 10, 11).
Discussion
Whole mount YFP expression in skin is similar to motor axon and neuromuscular junction expression in large muscle bellies. 4,6,14 The tetramethylrhodamine-conjugated α-bungarotoxin effectively labeled the neuromuscular junction in the panniculus carnosus muscle. Vertical frozen skin sections, using light acetone fixation coupled with short processing time, caused the fluorescent protein to precipitate, which in turn resulted in no fluorescence when excited (data not shown). Tissue lightly fixed in 4% PFA and then incubated overnight in 30% sucrose 13 effectively preserved the fluorescence of the reporter protein and the integrity of the skin structure while allowing visualization of YFP expression in the motor axon in conjunction with tetramethylrhodamine-conjugated α-bungarotoxin labeling of the neuromuscular junction. Nerves of various sizes could be detected by this method, as they could by using classical histological stains. When YFP was detected using immunohistochemistry, very small nerves were easily detected innervating the erector pili muscles and traversing the dermis. Double labeling for SMA and YFP confirmed innervation of the erector pili muscle. Although these labels are very specific in the skin, one must be careful to not misinterpret mast cell reactivity when using avidin–biotin–peroxidase methods, because these present as false positives. 1,2
Expression of the YFP16 is driven by the neuronal enhancer of the Thy1 (CD90.2) promoter. Thy1 has expression outside the nervous system, but with this transgene most expression outside the nervous system is suppressed. There may be nonspecific labeling of inflammatory cells in response to injury based on studies investigating nerve regeneration in vivo following injury. 10,11
The value of these mice and the methods used here to visualize interactions between nerves and muscle in and under the skin can be numerous, ranging from analysis of wound healing to defining innervations of the hair follicle and adjacent structures and how this changes in various diseases. 5,7,12
Footnotes
Acknowledgements
We thank Dr. R. Burgess for providing the mice and advice on their use, C. Rosales and N. Gott for technical assistance, and J. Hammer for assistance with figures.
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the National Institutes of Health (AR49288, AR047204, and AR054407 to J.P.S.).
