Abstract
Thymic depletion, presence of viral antigen, and changes in distribution and cytokine production of thymic macrophages were investigated in calves experimentally infected with a noncytopathogenic bovine viral diarrhea virus type (BVDV) 1 strain. Ten clinically healthy colostrum-deprived calves were used. Eight calves were inoculated with the virus and two were used as uninfected controls. Calves were sedated and euthanized in batches between 3 and 14 days postinoculation. At necropsy, thymus samples were collected for structural, immunohistochemical, and ultrastructural study and TUNEL (terminal deoxynucleotidyl transferase dUTP nick end labeling). From 6 days postinoculation, the thymic cortex was multifocally depleted with increased frequency of pyknosis and karyorrhexis, suggestive of apoptosis and confirmed by the TUNEL technique. Although the onset of lymphoid depletion was coincident with the detection of viral antigen by immunohistochemistry, the number of infected lymphocytes was very low through the experiment. There was an increase in number of macrophages in cortex and medulla, accompanied by ultrastructural changes indicative of phagocyte activation, and a decrease in cells expressing tumor necrosis factor-alpha (TNF-α) and IL-1α. These results suggest that the increase in number of these cells could be related to phagocytosis of cell debris and apoptotic lymphocytes. Furthermore, the results imply that, in contrast to the situation with classical swine fever virus, the lymphocyte apoptosis resulting from bovine viral diarrhea virus infection is not mediated by TNF-α or interleukin-1 alpha (IL-1α) production by virus-infected macrophages. This is the first study that describes this decrease in the number of thymic cells expressing TNF-α and IL-1α in cattle experimentally infected with bovine viral diarrhea virus type 1.
Bovine viral diarrhea virus (BVDV) is a Pestivirus in the family Flaviviridae, 25 which infects a large proportion of cattle worldwide and causes a number of clinical forms of the disease, including subclinical infection, reproductive failure, severe acute disease, and mucosal disease. 2 Two genotypes and two biotypes of BVDV have been recognized. 7,19,23 The two genotypes are called BVDV1 and BVDV2 and are now recognized as distinct species within the Pestivirus genus. 25 The two biotypes, cytopathic and noncytopathic, are based on the activity of the BVDV strain in cultured epithelial cell. 7 Recently, a third lymphocytopathic biotype, which correlates with high virulence strains in acute infections, has been proposed. 22
Thymus is a primary lymphoid organ essential for proper maturation and selection of T lymphocytes. 20 Atrophy of the thymic cortex due to lymphocyte apoptosis has been related to cytokine expression by monocyte–macrophages during experimental classical swine fever virus infection, another member of the genus Pestivirus closely related to BVDV. 24 Thymic depletion has also been described during BVDV1 and BVDV2 infection. 4,11,14,15,26 However, the role of macrophages in the pathogenesis of this thymic depletion remain unclear, although monocyte–macrophages have been identified as one of the target cells for this virus. 3,11,17
The purpose of this study was to investigate, in experimental animals, the relationship among thymic depletion, presence of viral antigen, and changes in distribution and cytokine production by thymic macrophages from colostrum-deprived calves experimentally infected with the low-virulence noncytopathogenic BVDV1 strain 7443.
Materials and Methods
Ten clinically healthy male Friesian calves, 8 to 12 weeks old, were obtained immediately after birth from a herd free of tuberculosis, brucellosis, bovine leucosis virus, and infectious bovine rhinotracheitis. Calves did not receive colostrum and were confirmed to be free of BVDV by antigen and antibody ELISA (enzyme-linked immunosorbent assay).
Calves were housed individually in the Animal Experimental Center of Cordoba under strict sterile conditions. Control calves were housed individually in another barn with the same conditions, so there was no contact between infected and uninfected animals. No other calves were housed in the center during the experiment.
Eight calves were inoculated (day 0) by intranasal instillation with 10 ml (5 ml per nostril) containing 105 median tissue culture infective dose per milliliter of noncytopathic BVDV1 7443 (courtesy of the Institute für Virologie, TIHO, Hannover, Germany). Two control calves were inoculated by intranasal instillation of tissue culture fluid and used as uninfected controls.
Clinical signs and rectal temperature were monitored twice daily. Blood samples were taken from all calves before virus inoculation and at several days postinoculation (dpi). In these samples, the total leukocyte count and percentage of lymphocytes, neutrophils, monocytes, and eosinophils were determined. BVDV antigen was detected in theses samples by ELISA (HerdChek BVDV Ag/Serum Plus, IDEXX Laboratories, Österbybruk, Sweden). Samples with corrected optical density > 0.300 were considered positive.
Animals were sedated with xylazine (Rompun 2% solution, Bayer Healthcare, Kiel, Germany) and killed in batches of two at 3, 6, 9, and 14 dpi by overdosing with thiopental sodium (Thiovet, Vet Limited, Leyland, Lancashire, United Kingdom). Control calves were killed at the end of the experiment. The experimental procedure was carried out in accordance with the Code of Practice for Housing and Care of Animals Used in Scientific Procedures, approved by the European Economic Community in 1986 (86/609/EEC amended by directive 2003/65/EC).
Histology and Immunohistochemical Study
At necropsy, tissue samples were collected from thymus and fixed in 10% neutral buffered formalin. Formalin-fixed samples were embedded in paraffin, sectioned, and stained with hematoxylin and eosin for structural study. For immunohistochemistry by the avidin–biotin–peroxidase complex method, tissue sections were dewaxed and rehydrated. Endogenous peroxidase activity was exhausted by incubation of the sections with 0.3% hydrogen peroxide in methanol for 30 minutes at room temperature. Sections were subjected to one of the following methods for retrieving antigen or increasing permeability: protease type XIV (Sigma-Aldrich Chemie, Steinheim, Germany) 0.1% in 0.01M phosphate buffered saline (PBS), pH 7.2 (8 minutes at room temperature); microwave heating in 0.01M citrate buffer, pH 3.2 (5 minutes from the beginning of boiling); Tween 20 (Merck, München, Germany) 0.1% in 0.01M PBS, pH 7.2 (10 minutes at room temperature). After pretreatment, sections were rinsed 3 times in PBS, pH 7.2, for 10 minutes and then covered with 1% normal horse serum (Pierce-Endogen, Woburn, MA) in 0.05M Tris buffer, pH 7.6, for 30 minutes at room temperature. Sections were then incubated with primary monoclonal antibodies at 4°C overnight. Details of primary antibodies used in this study, including dilutions and pretreatments, are summarized in Table 1. After primary incubation, the slides were washed in PBS (3 times for 5 minutes each) and incubated with biotinylated horse anti-mouse IgG secondary antibody (Pierce-Endogen), diluted 1:200 in 0.05M Tris buffer, pH 7.6, containing 1% normal horse serum for 30 minutes at room temperature. After 3 further 5-minute washes in PBS, samples were incubated with the avidin–biotin–peroxidase complex method (Vectastain ABC Elite Kit, Vector Laboratories, Burlingame, CA) for 1 hour at room temperature. All tissue sections were finally rinsed in PBS, incubated for 1 minute with chromogen solution (NovaRed Substrate Kit, Vector Laboratories), and counterstained with hematoxylin.
Details of Immunohistochemical Methodology

aCourtesy of Dr E. J. Dubovi, Cornell University, Ithaca, NY.
bSerotec, Oxford, United Kingdom.
cEndogen, Woburn, MA.
For negative controls, mouse nonimmune serum was used in place of specific primary antibodies. Additionally, positive control tissues from calves persistently infected with BVDV and negative control tissues from specific pathogen–free calves not exposed to BVDV were used for gp48 detection.
Apoptosis Detection
For the detection of DNA fragmentation, formalin-fixed samples were stained by the TUNEL method (terminal deoxynucleotidyl transferase dUTP nick end labeling; In Situ Cell Death Detection, POD Kit, Boehringer Mannheim, Indianapolis, IN), according to the manufacturer’s instructions.
Statistical Analysis
Positively labeled cells were counted in 20 randomly chosen areas of 0.20 mm2 of cortex and medulla. Cellular identification was based on morphologic features and size of cells. The numbers of positive cells were expressed as a mean ± SD. These values were evaluated for approximate normality of distribution by the Kolmogorov–Smirnov test. Differences between the means of control and inoculated animals were assessed by the Kruskal–Wallis test, followed by the Mann–Whitney U nonparametric test (GraphPad Instat 3.05, San Diego, CA). P < .05 represented a statistically significant difference between inoculated and control animals.
Ultrastructural Study
For ultrastructural study, samples were postfixed in 2% osmium tetroxide, dehydrated in acetone, and embedded in Epon 812 (Fluka Chemie AG, Buchs, Switzerland). Sections (50 nm) for ultrastructural examination were counterstained with uranyl acetate and lead citrate and examined with a Philip CM-10 transmission electron microscope.
Results
The two animals that served as controls and were not inoculated remained healthy during the entire observation period. In inoculated calves, there was a temperature increase at 3 and 7 dpi, compared to the controls (Fig. 1). Body temperature was normal from 9 dpi onward. Behavior and food intake were normal in all calves.
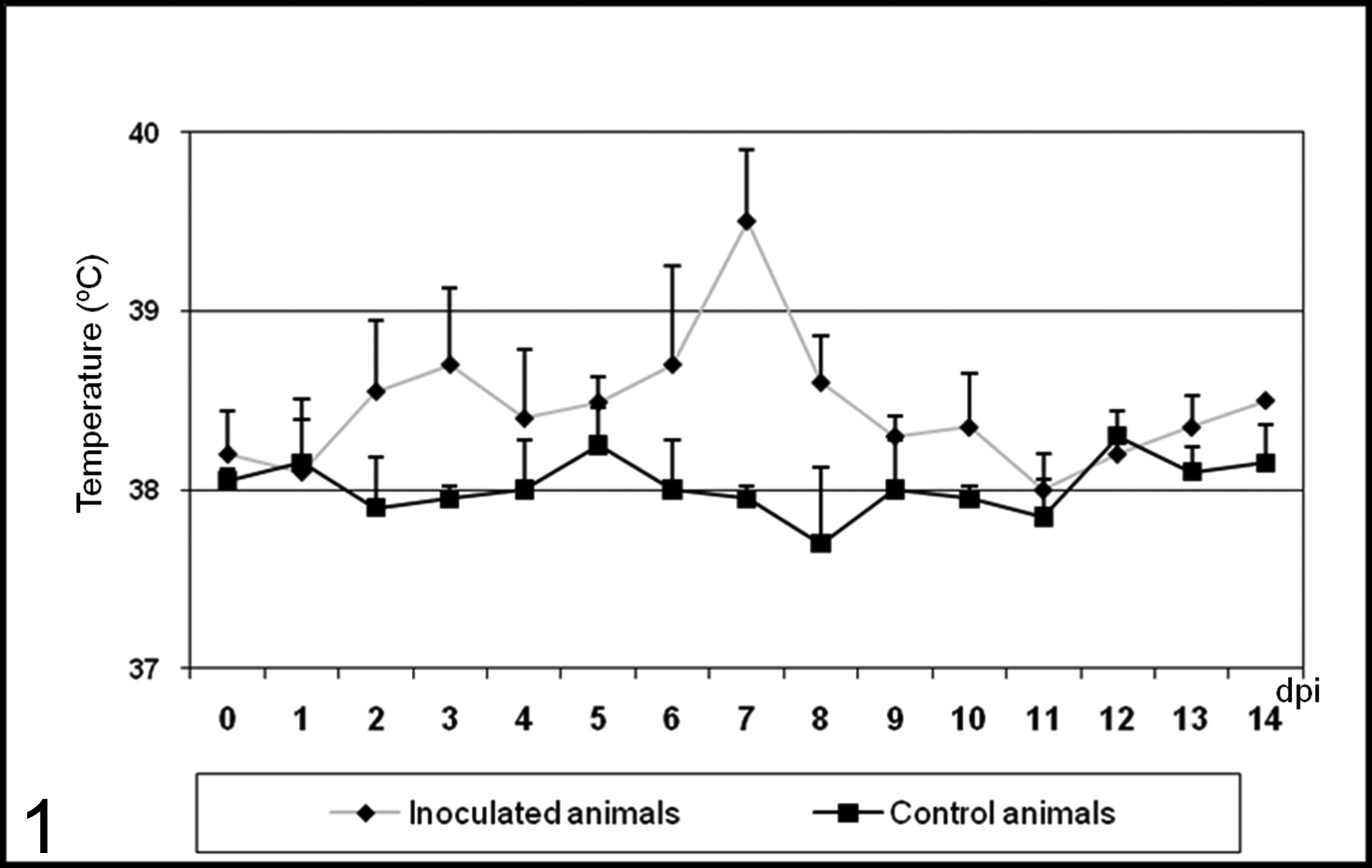
Mean ± SD rectal temperature (°C) from 0 to 14 days postinoculation (dpi) of controls and calves inoculated with noncytopathic bovine viral diarrhea virus type 1 strain 7443.
Total white blood cell count (mean preinoculation value, 8.3 × 103/μl) was reduced in all inoculated animals between 2 and 6 dpi, with a decrease in total number of lymphocytes and neutrophils. At 3 dpi, a nadir was reached (mean value, 4.1 × 103/μl). Lymphocyte and neutrophil numbers were on average 74% and 36% below the control values, respectively. From 8 dpi, the number of neutrophils and lymphocytes remained within the range of variation seen in the controls.
Virus antigen was detected in blood between 5 and 12 dpi, with a peak at 6 dpi (Fig. 2). Virus antigen was not detected in controls.
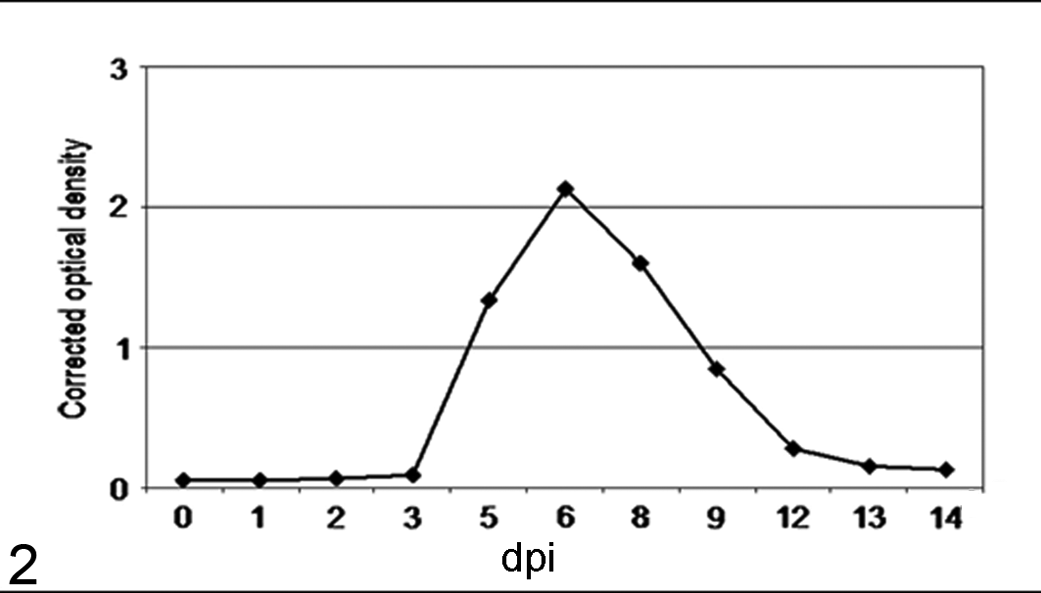
Quantification by ELISA of bovine viral diarrhea virus antigen in blood samples from inoculated animals.
Histologic Findings
No lesions were observed in thymus of control calves and at 3 dpi. From 6 dpi, the thymic cortex was multifocally depleted with increased frequency of pyknosis and karyorrhexis, suggestive of apoptosis. The onset of apoptosis coincided with an increase in size and number of macrophages containing phagocytized cell debris (tingible bodies). This lymphoid depletion was also observed in medulla. Lesions were more severe from 9 dpi (Figs. 3, 4), giving rise to the “starry sky” appearance.
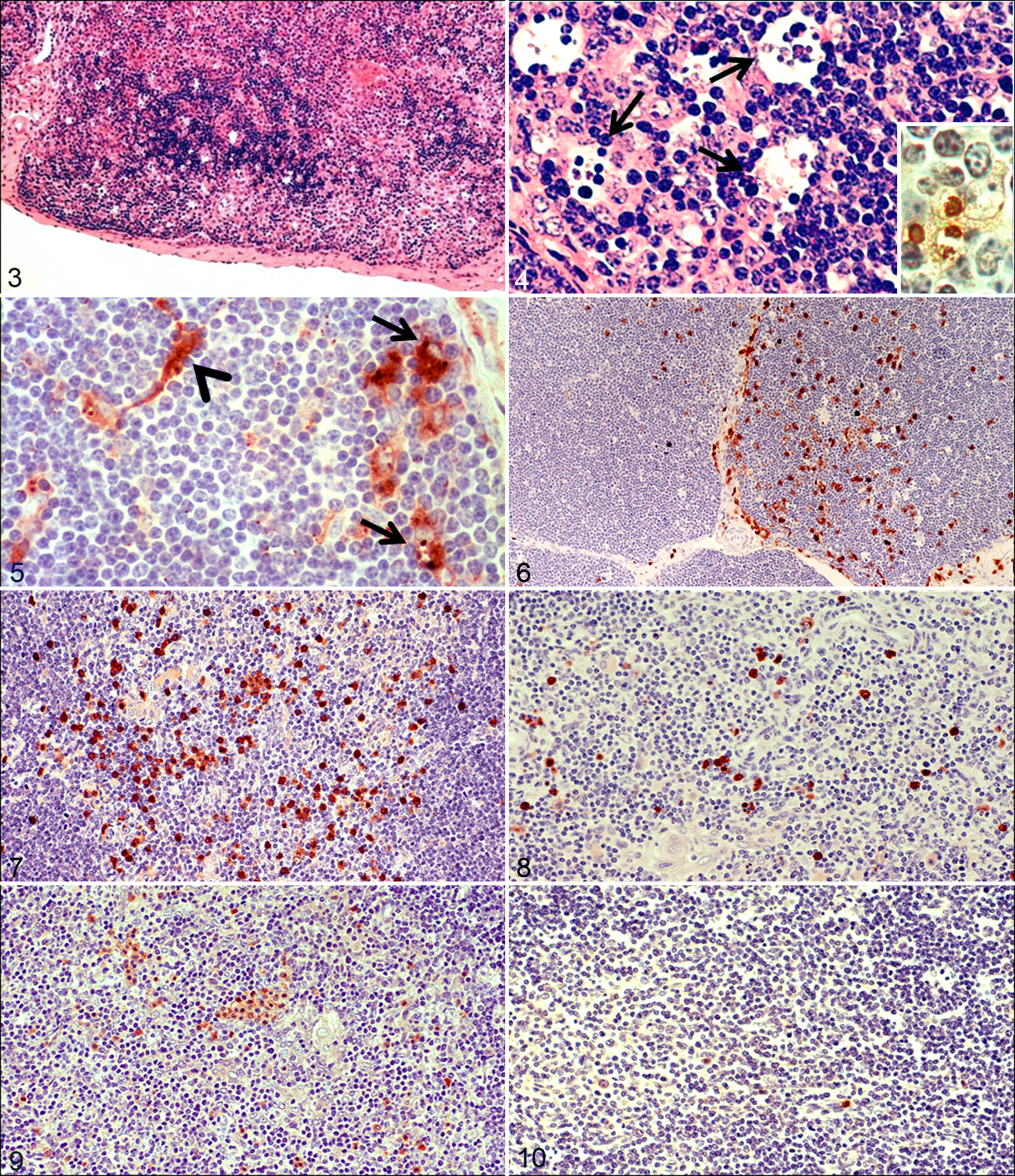
Thymus; calf infected with bovine viral diarrhea virus (BVDV), 9 days postinoculation (dpi). Multifocal depletion of lymphocytes in the thymic cortex. HE.
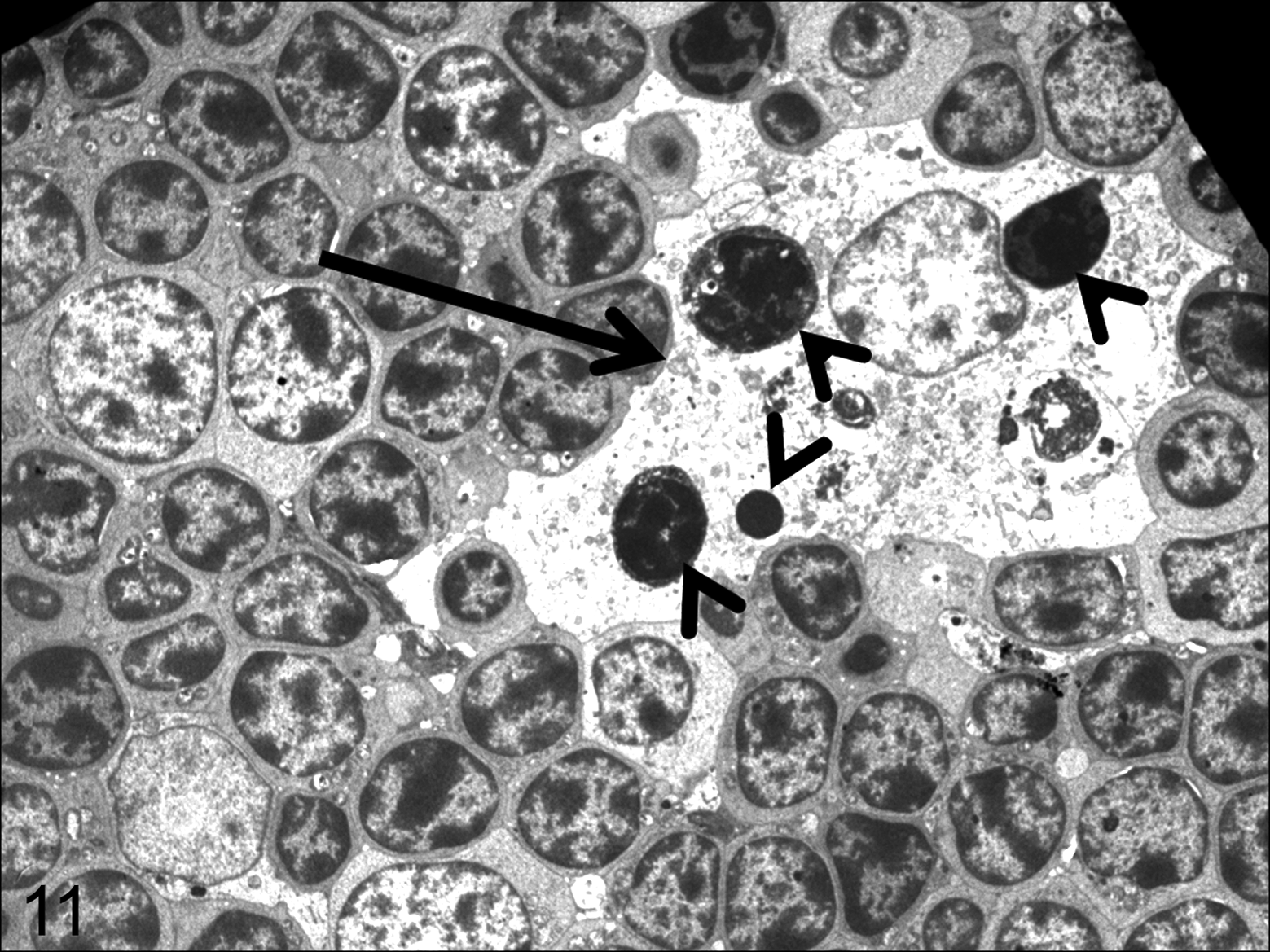
Thymus; calf infected with bovine viral diarrhea virus, 6 days postinoculation. Macrophage (arrow) with morphologic features indicating phagocyte activation: increase in cell size and presence of phagocytized cell debris, mainly apoptotic bodies (arrowheads), characterized by condensation of chromatin with fragmentation of nuclei and cytoplasm. Transmission electron microscopy.
Study of Apoptosis by the TUNEL Technique
The TUNEL technique confirmed the apoptosis observed in the histologic study; TUNEL-positive cells were detected both in control and inoculated animals. These cells included pyknotic lymphocytes and macrophages displaying phagocytosis of strongly labeled apoptotic bodies (Fig. 4, inset). The number of TUNEL-positive cells increased in number from 6 dpi in cortex and medulla, compared to the number in control animals euthanized on day 14 and infected animals euthanized on 3 dpi.
Immunohistochemical Study
BVDV antigen was not detected by immunohistochemistry in tissues of control animals or the infected calves necropsied at 3 dpi. From 6 dpi, viral antigen was distributed multifocally in thymus. Positive cells were identified as macrophages, most of them containing phagocytosed apoptotic bodies, star-shaped thymic epithelial cells, and small numbers of lymphocytes. The number of cells containing viral antigen was different among individual lobules. At 9 dpi, the widest distribution and highest numbers of cells containing BVDV antigen were seen (Fig. 5).
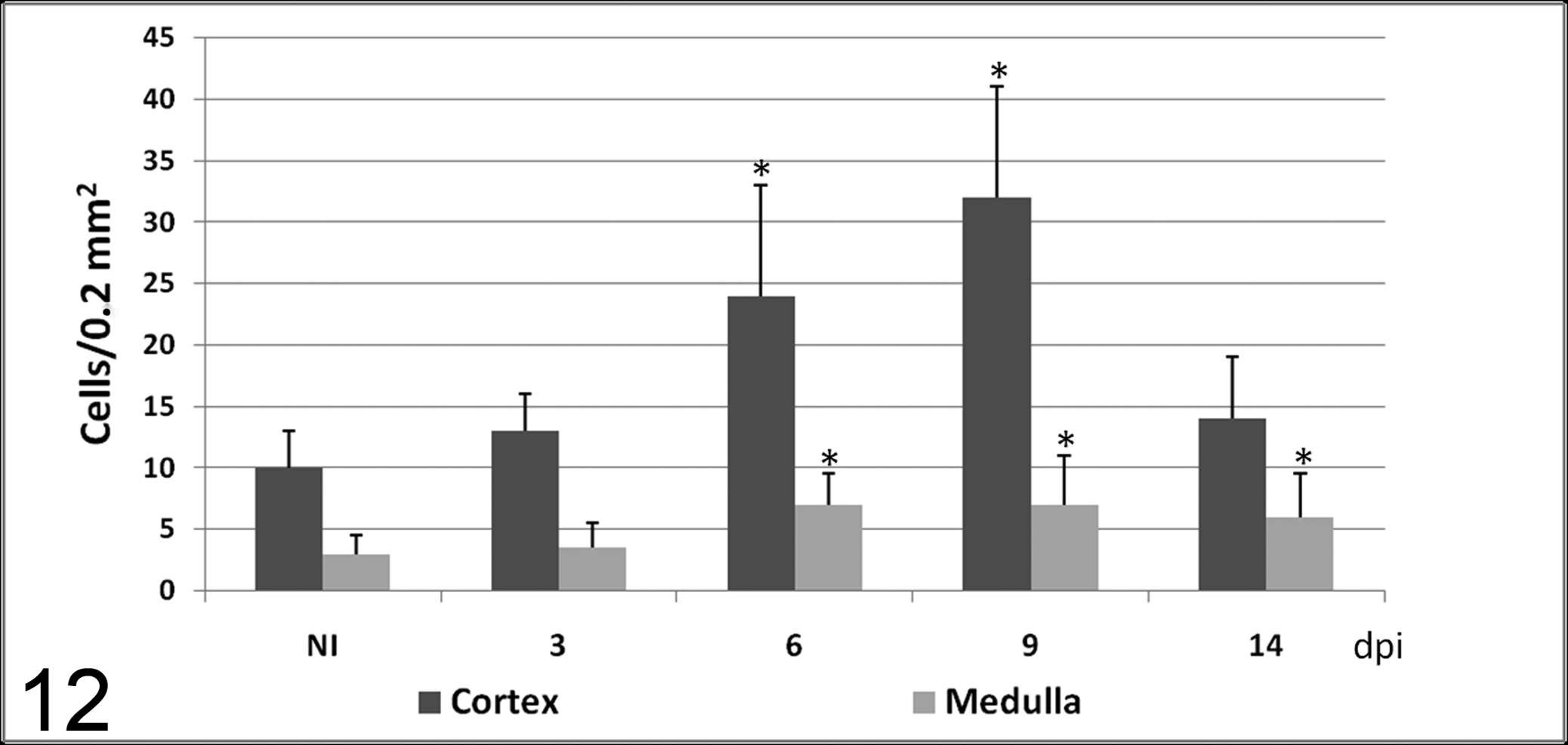
Number of cells (mean ± SD) detected by the TUNEL technique in cortex and medulla in calves noninoculated (NI) and inoculated with the noncytopathogenic bovine viral diarrhea virus type 1 strain 7443. dpi, days postinoculation. * P < .05 (between the inoculated group and controls).
Thymic macrophages were identified with MAC387 antibody. In control animals, positive macrophages were detected in cortex and medulla, although there were more immunolabeled macrophages in the medulla than in the cortex throughout the experiment. From day 6, a significant increase was noted in the number of macrophages immunolabeled with this marker, many of them containing phagocytosed apoptotic bodies (Fig. 6). The number of immunolabeled macrophages in the cortex was different among individual lobules, giving rise to a higher standard deviation at 6 and 9 dpi.
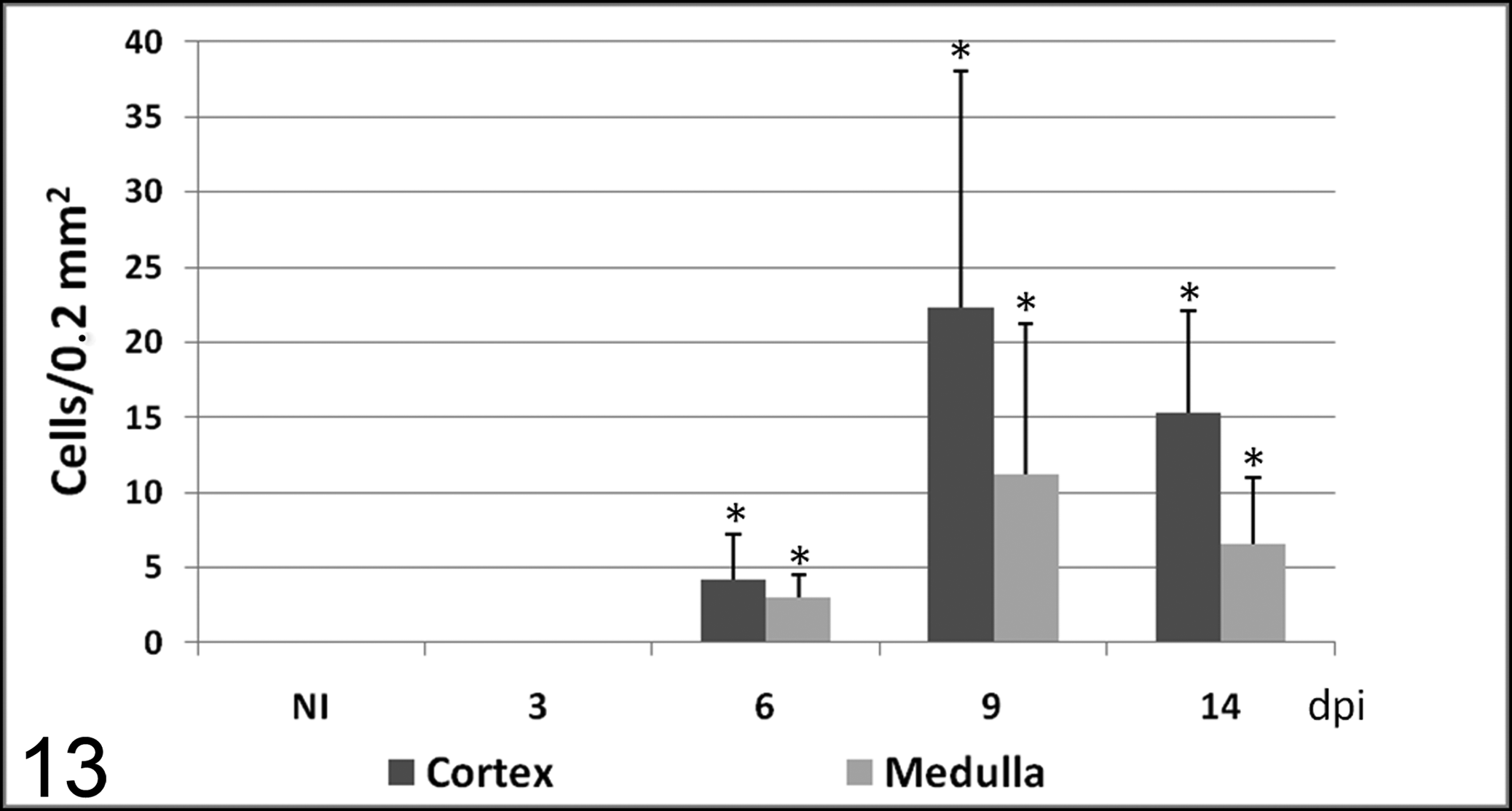
Number of cells (mean ± SD) immunolabeled with 15C5 antibody (specific for bovine viral diarrhea virus protein gp48) in cortex and medulla in calves noninoculated (NI) and inoculated with the noncytopathogenic bovine viral diarrhea virus type 1 strain 7443. dpi, days postinoculation. * P < .05 (between the inoculated group and controls).
TNF-α and IL-1α had a similar distribution pattern. In thymic cortex, there were low numbers of both cytokines, and no significant differences were observed between infected and control animals. However, numerous groups of immunolabeled cells, mainly macrophage-like cells and occasionally neutrophil-like cells, were observed in thymic medulla of control animals and infected animals necropsied at 3 dpi. From 6 dpi until the end of the experiment, the number of cells immunolabeled for both mediators was significantly lower than in control animals necropsied at day 14 and in infected animals necropsied at 3 dpi (Figs. 7–10).
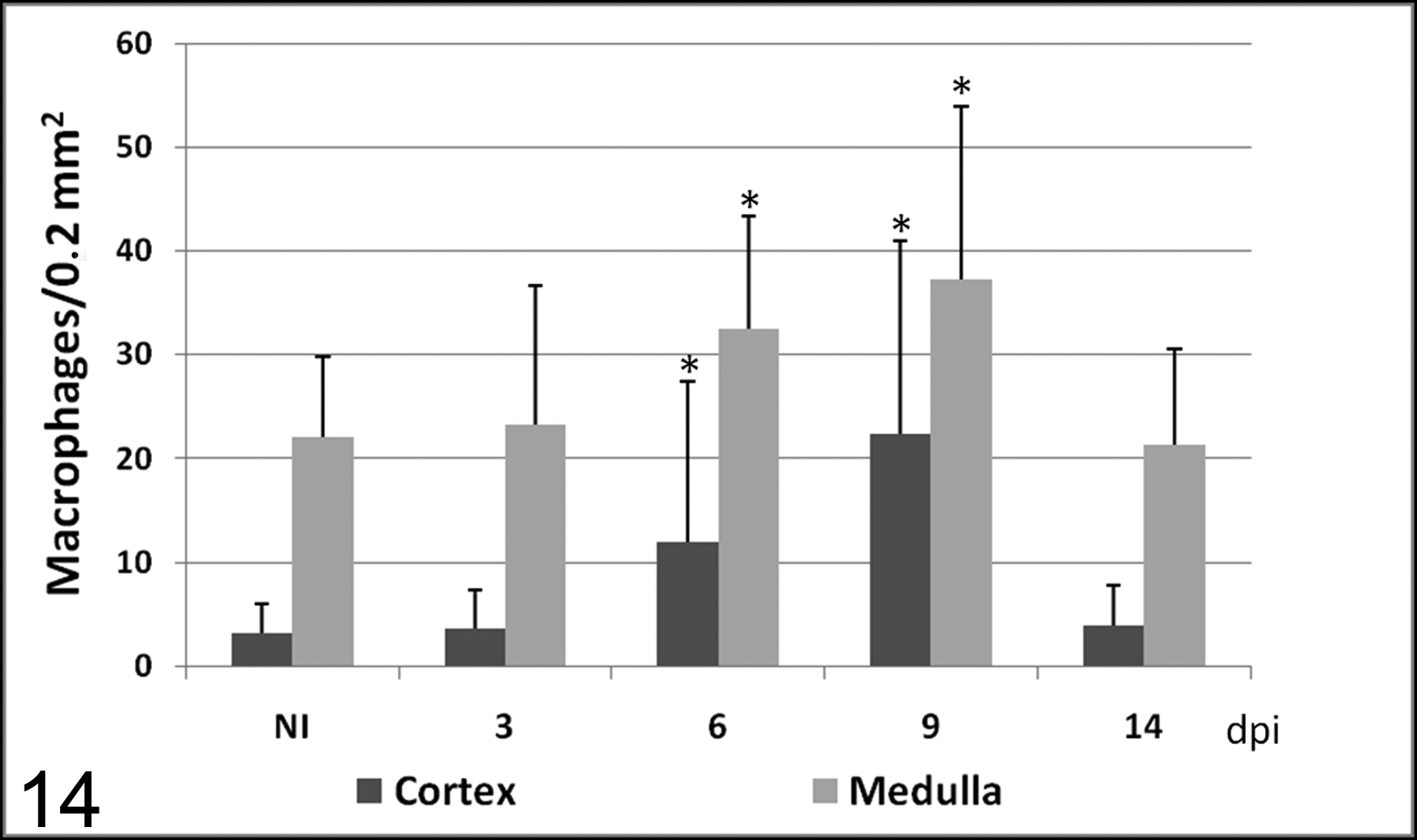
Number of macrophages (mean ± SD) immunolabeled with MAC387 antibody in cortex and medulla in calves noninoculated (NI) and inoculated with the noncytopathogenic bovine viral diarrhea virus type 1 strain 7443. dpi, days postinoculation. * P < .05 (between the inoculated group and controls).
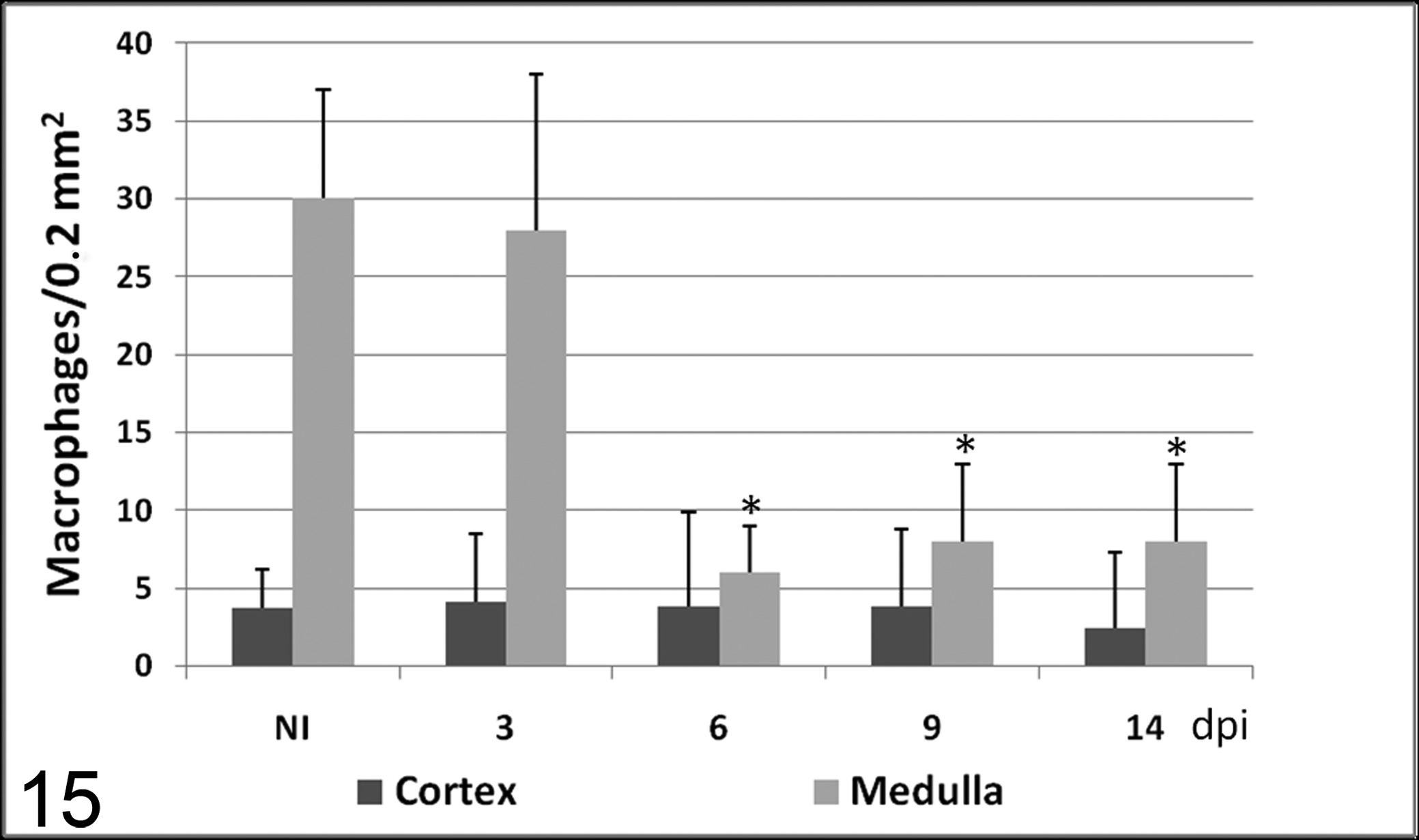
Number of macrophages (mean ± SD) immunolabeled with TNFα antibody in cortex and medulla in calves noninoculated (NI) and inoculated with the noncytopathogenic bovine viral diarrhea virus type 1 strain 7443. dpi, days postinoculation. * P < .05 (between the inoculated group and controls).
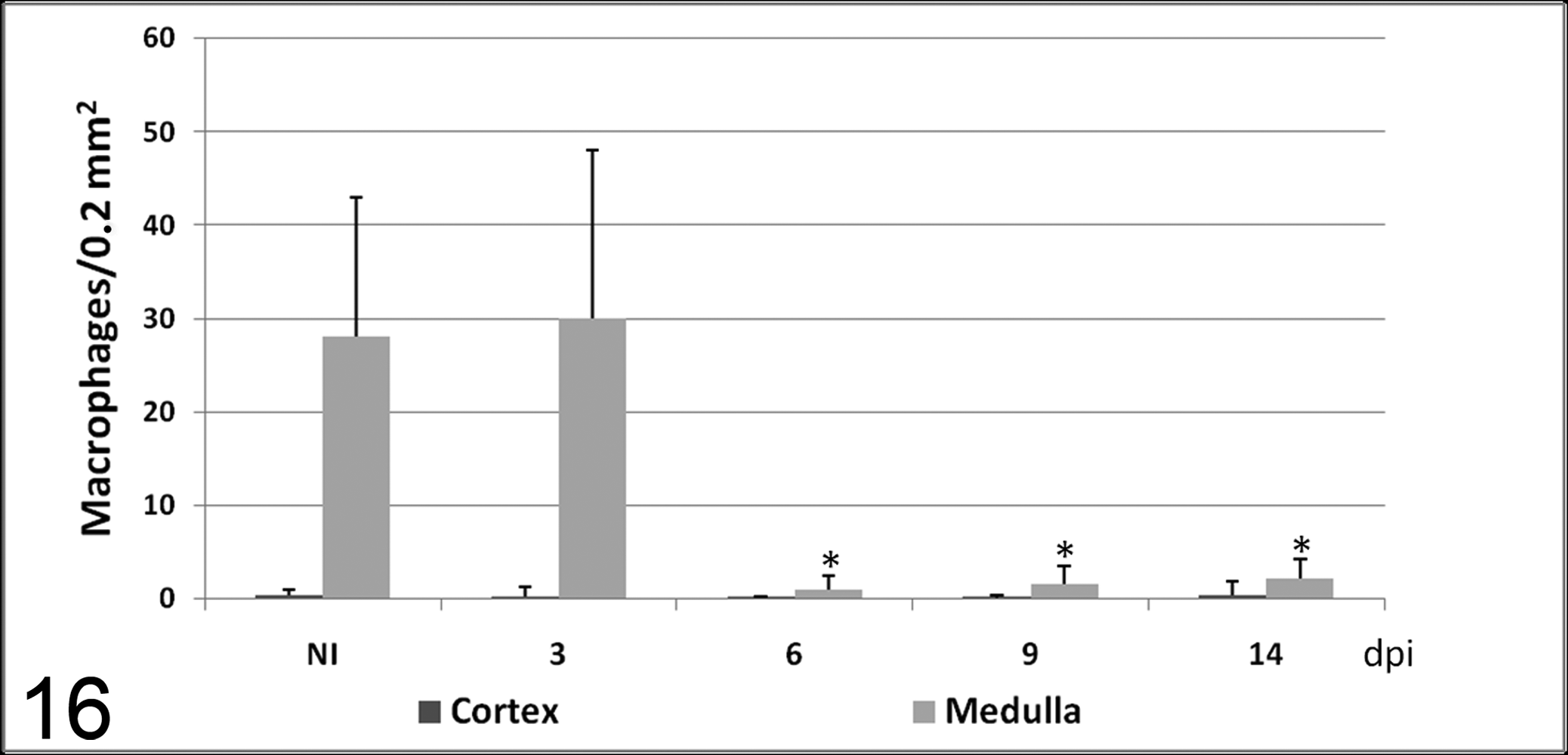
Number of macrophages (mean ± SD) immunolabeled with IL-1α antibody in cortex and medulla in calves noninoculated (NI) and inoculated with the noncytopathogenic bovine viral diarrhea virus type 1 strain 7443. dpi, days postinoculation. * P < .05 (between the inoculated group and controls).
Ultrastructural Findings
Ultrastructural examination revealed changes consistent with lymphocyte apoptosis in the thymus of infected animals and control calves. Apoptotic cells exhibited condensation and margination of chromatin as well as fragmentation of the nucleus and cytoplasm. These changes became more marked from 6 dpi, with the appearance of condensed nuclear chromatin and highly electron-dense membrane-bound cell fragments (apoptotic bodies) either free or phagocytized by macrophages (Fig. 11). From 6 dpi, macrophages displayed other ultrastructural changes indicative of phagocyte activation, such as an increase in cell size and an increase in cytoplasmic lysosomes.
Discussion
This is the first sequential study of the histopathologic changes produced in thymus during an experimental infection with noncytopathogenic BVDV1 and their relationship with virus distribution and cytokine production by macrophages (see Figs. 12–16). Our results suggest that BVDV1-induced thymic lymphoid depletion has a different mechanism than that in classical swine fever 24 and is not the consequence of an increase of TNF-α and IL-1α production by thymic macrophages. By contrast, this study showed reduced frequency of cells expressing these cytokines coincident with the onset of lymphoid depletion.
Colostrum-deprived calves inoculated with the noncytopathogenic BVDV1 strain 7443 developed lymphoid depletion of thymus from 6 dpi, when viral antigen was detected by immunohistochemistry, similar to that reported for other BVDV1 and low-virulence BVDV2 strains. 4,13,15,16,26 This lymphoid depletion has been associated with lymphocyte apoptosis, 13 which was evidenced in this experiment by the TUNEL technique.
The onset of thymic depletion was coincident with the detection of viral antigen by immunohistochemistry, but viral antigen was present mainly in macrophages and thymic epithelial cells. The number of infected lymphocytes was very low through the experiment. These results agree with previous reports about high-virulence BVDV2 that detected thymic depletion but very low infection of lymphocytes by immunohistochemistry. 6,17 These results suggest that viral infection of lymphocytes plays no more than a minor role in the lymphoid depletion. More studies, focusing on viral detection in early phases in the disease, are necessary to confirm that infected lymphocytes are not dying soon after they begin to express detectable viral antigen.
BVDV antigen has been found in monocyte–macrophages during BVDV infection by immunohistochemistry, immunocytochemistry, and electron microscopy, 3,11,17,18 and some authors have suggested that virus was replicating in these cells and not merely being processed. 16 In our work, BVDV antigen was detected by immunohistochemistry in thymic macrophages, which could represent infection of these cells and/or phagocytosis of other infected cells. The increase in number of macrophages in cortex and medulla was coincident with the onset of lymphoid depletion, which was accompanied by an increase of tingible body macrophages in histologic study. Ultrastructural study showed changes in the macrophage population indicative of phagocyte activation. These results suggest that, although active viral replication could be present in macrophages, the increase in number of these cells could be related with phagocytosis of cell debris and apoptotic lymphocytes.
The increase in number of macrophages was not accompanied by ultrastructural changes in macrophages indicative of secretory activity or by an increase in the number of cells expressing TNF-α or IL-1α. Expression of both cytokines was decreased from 6 dpi until the end of the experiment. These results agree with previous studies regarding cytokine production during BVDV infection. Jensen and Schultz in 1991 suggested that BVDV infection of bovine monocytes could result in the production of an inhibitor of IL-1 activity that inhibits thymocyte proliferation. 10 In 1996, Adler et al showed that TNF-α production in bovine bone marrow macrophages is decreased at 24 hours after BVDV infection. 1 Lee et al in 2008 demonstrated that in vitro infection with cytopathic and noncytopathic BVDV strains decreases TNF-α and IL-1β gene and protein expression in bovine peripheral blood monocytes. 12 TNF-α production has also been investigated in primary bovine fetal muscle cells, showing a transcription of TNF-α mRNA 82% lower in cells infected with noncytopathogenic BVDV strains as compared to the mock-infected cells. 27 According to these studies, the decrease of cytokine expression by thymic macrophages could be a direct effect of the virus.
All these data regarding the effects of BVDV infection on cytokine production have been obtained from in vitro studies. This is the first study that describes this decrease in the number of thymic cells expressing TNF-α and IL-1α in cattle experimentally infected with BVDV1. This decrease was coincident with the presence of viral antigen in thymic monocytes from 6 dpi and with the onset of the lymphoid depletion. Both cytokines are secreted constitutively in thymus, and in this organ, they are known to be involved in the differentiation and proliferation of thymocytes, playing a crucial role in T-cell production. 5,8,9,21,28 So, a downregulation of these cytokines could contribute to the BVDV-induced immunosuppression by interference with the mechanisms of thymocyte differentiation and proliferation. Our methods do not quantify total cytokine production and cannot exclude that few cytokine-producing cells are producing large amounts of cytokines in these animals, so more studies are necessary to confirm this hypothesis. This decrease on cytokine production would not explain lymphocyte apoptosis, which could be related to the presence of viral antigen observed in other thymic populations, such as thymic epithelial cells, which play a crucial role in thymocyte selection mechanisms in the thymic cortex.
In summary, thymic lymphoid depletion following inoculation of colostrum-deprived calves with BVDV1 has a different mechanism than that observed in classical swine fever and is not likely a consequence of an increase of TNF-α and IL-1α production by thymic macrophages. We hypothesize that the decreased expression of these cytokines could be one of the mechanisms contributing to BVDV-induced immunosuppression, due to the important function of these cytokines in T-lymphocyte maturation and activation of the immune response. However, the downregulation of cytokines would not alone explain the increase in apoptosis observed during the experiment. The apoptosis may elicit an increase in number of macrophages with a phagocytic function. More studies are necessary to clarify the pathogenesis of thymic lymphoid depletion, the cause of lymphocyte apoptosis, and the role of other thymic populations infected with the virus, such as thymic epithelial cells.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by the Ministry of Education and Science (grant numbers AGL 2003-00252, AGL 2006-01536).
