Abstract
Systemic isosporosis, also known as atoxoplasmosis, is a common parasitic disease of passerines. Infection is thought to be endemic in wild birds with fulminant, fatal disease occurring under the influence of stress, concurrent infections, or immunosuppression. Here, we describe the histologic and immunohistochemical characteristics of the cellular infiltrate occurring in captive colonies of American goldfinches and house sparrows. Necropsies were performed on 9 birds, and histologic examination was performed on the intestines of 7 additional birds. Lesions were most severe in the proximal small intestines. Histologically, the changes ranged from variably intense infiltrates of lymphocytes that filled the lamina propria to sheets of large, atypical cells that expanded and obliterated normal mucosal epithelium and invaded through the wall of the intestine and into the ceolomic cavity. Both the smaller lymphocytes and large atypical cells were immunoreactive for CD3. Intracellular parasites consistent with Isospora were detected in the large atypical cells, but they were more easily detectable in the more differentiated lymphocytes. Polymerase chain reaction and virus isolation performed on tissues from 7 birds were negative for retroviruses and herpesvirus. The immunohistochemical results of this study and the destructive nature of the cellular infiltrate suggest that the lesion represents T-cell lymphoma. In birds, lymphomas are most often associated with herpes and retroviruses; the absence of these viruses suggests that the parasite initiated neoplastic transformation. Though much work needs to be done to prove the transformative nature of the lesions, these preliminary results suggest that passerine birds may be susceptible to parasite-associated lymphomas.
One of the more mysterious manifestations of coccidial infection in passerine birds is the development of systemic isosporosis, also known as atoxoplasmosis. This systemic disease has long been recognized in passerine birds; nonetheless, a great deal of uncertainty surrounds the pathogenesis of this disease and even the classification of the causative agent. Traditionally, the coccidial agent has been grouped in the genus Atoxoplasma; however molecular analysis of avian coccidian parasites has cast doubt on the traditional classification system of these avian parasites. 26 The disease syndrome atoxoplasmosis is associated with infection of mononuclear cells, necrosis in the liver and spleen, and infiltration of the intestines and multiple visceral organs with variable numbers of inflammatory cells, usually identified as macrophages, lymphocytes, and/or natural killer cells.3,10,15–18,20,22,28
In free-ranging birds, Atoxoplasma parasites are endemic, and population prevalence varyies between species and geographic location.5,6,27 Though infection in wild populations may not result in notable mortality, infection in captive birds can result in significant mortality.10,15 Stress from being in captivity and poor sanitary conditions resulting in increased shedding and accumulation of infective oocysts has been identified as contributing causes for the high morbidity in these facilities.17,18,28 These coccidia proliferate and undergo sexual maturation in the small intestine and have a systemic asexual phase. Merogony of Atoxoplasma parasites occurs in circulating mononuclear inflammatory cells and can result in proliferation of these cells.1,17,20,22,26
Clinical infection within passerine birds results in nonspecific signs of illness, including weight loss, reduction of pectoral musculature, and diarrhea. A subset of birds will remain subclinical.3,22 Histopathological descriptions of atoxoplasmosis typically describe an influx of variable numbers of mononuclear inflammatory cells, including lymphocytes, plasma cells, and macrophages. In some cases, there is effacement of the intestinal architecture by the infiltrating cells. Close examination of the infiltrating cells will often reveal merozoites within the cytoplasm of the infiltrating cells. 20 Aggregates of merozoites will occasionally indent the cell’s nucleus. 20
The identity of the infiltrating cells occurring during Atoxoplasma infection is often unclear. Many of the cells are obviously well-differentiated lymphocytes, but in some cases, the infected cells are not clearly lymphocytic. Ultrastructural analysis suggested they may be natural killer cells. 28 Furthermore, classification of some of the lesions has proven difficult. Various authors have classified this change as inflammatory, atypical lymphoproliferative, or sometimes neoplastic.3,10,15–18,20,22,28 Differentiating between severe lymphocytic or histiocytic inflammation and lymphosarcoma can be a diagnostic challenge. In domestic animals, the diagnosis may ultimately be made based on results of immunohistochemistry and destruction of intestinal architecture. 7 Nonetheless, PCR for clonality is important in distinguishing lymphoma from enteritis, as immunohistochemical results may be equivocal.
In this study, we describe the range of lesions seen in a group of American goldfinches (Spinus tristis; AMGO) and house sparrows (Passer domesticus; HOSP) that died during an outbreak of atoxoplasmosis in a captive colony. Both morphology and immunohistochemistry support a diagnosis of lymphosarcoma in the most severely affected portions of small intestine. This finding, in conjunction with lack of transforming viruses in the tissue, suggests the possibility that parasites may induce neoplastic transformation of infected cells.
Materials and Methods
Case Histories
During the summer of 2007, a research colony of 40 wild-caught American goldfinch (AMGO) was established as part of an NSF-funded project on Mycoplasma gallisepticum (MG) infection in passerine birds. The AMGO were caught in the Ithaca, NY, USA area between April 20 and April 24, 2007 under a Federal Bird Banding Permit (permit #22669) and housed in wire cages over a cement slab. The birds were divided evenly among 4 cages and had no contact with individuals from other cages. The AMGO cages were adjacent to similarly housed HOSP that were part of another research study. All birds received feed and water ad libitum, following the requirements of the Cornell University Institutional Animal Care and Use Committee. Prior to inoculation with MG on June 6, 2007, AMGO were treated daily with Tylosin at 1 mg/mL in 250 mL of drinking water for 3 weeks as part of the MG study protocol.
Mortality first occurred on July 13, 2007, and birds developed nonspecific clinical signs including diarrhea, lethargy, wasting, and poor flight capacity. Between July 13 and the termination of the experiment on August 23, 2007, 19 birds had died. Upon termination of the experiment, all birds were euthanized. A subset of these birds, including individuals that had recently exhibited clinical signs along with randomly chosen individuals, was submitted to the Cornell University Section of Anatomic Pathology for postmortem examination. Additional tissues were collected for molecular evaluation for viral infection. One week after the AMGO experiment was terminated, similar clinical signs developed in the adjacent colony of HOSP. Mortality rates were similar to that observed in the goldfinches.
Gross Necropsy and Histopathology
Whole birds (5 AMGO, 4 HOSP) or intestinal tracts (7 AMGO) were examined. After gross evaluation, tissues for histopathology were fixed in 10% neutral buffered formalin (NBF), routinely processed, and stained with either hematoxylin and eosin (H&E) or processed for immunohistochemistry. A section of colon from one AMGO was submitted for routine parasitology and lung from another AMGO submitted for bacteriology at the Cornell University Animals Health and Diagnostic Center.
Virus Isolation
Kidney and livers samples were obtained aseptically from 7 humanely euthanized AMGO and placed in phosphate buffered saline (PBS), pH 7.4. Single-cell lymphocyte suspensions were prepared by passing tissues through 70 μM nylon cell strainers (Falcon BD, Franklin Lakes, NJ) and centrifuging the filtrate over Ficoll Hypaque Plus (Amersham Biosciences, Piscataway, NJ). The cells in the interface were recovered, resuspended in PBS, counted, and seeded 2 × 106/mL into 24-well plates in LM Hahn medium (50% Leibovitz’s L-15 + 50% McCoy’s 5a modified supplemented with 10% tryptose phosphate broth, 0.2% of 10% NaHCO3, 0.01 mM 2-mercaptoethanol, 1% sodium pyruvate 100×, 2 mM glutamine solution, and antibiotics) with 10% fetal bovine serum (FBS) and 8% chicken serum. 24 Samples from 3 additional AMGO were prepared in a similar way, except that these cells were not centrifuged over Ficoll Hypaque Plus. At 5 days post-plating, aliquots of the lymphoid cell cultures were passed onto chicken embryo fibroblast (CEF) and chick kidney cell (CKC) cultures and at weekly intervals, 3 blind passages were made. CEF and CKC cells were prepared and maintained following standard techniques. 24
PCR for Retroviruses and Herpesviruses
RNA and DNA extraction
Cell pellets containing approximately 20 × 106 cells were resuspended in RNA STAT 60 (Tel-Test, Inc., Friendswood, TX) and stored at -80°C until RNA and DNA extraction. DNA was extracted using RNA STAT60 and DNASTAT60 following the manufacturer’s recommendations.
Polymerase chain reaction
Polymerase chain reaction (PCR) assays were used to determine the presence of retrovirus and herpesvirus sequences. Degenerate and specific primer sets previously shown to detect these agents in a wide range of species were used. Primers #1 (5′-CATACTGGAGCCAATGGTT-3′) and #2 (5′-AATGTTGTACCGAAGTACT-3′) for the detection of reticuloendotheliosis virus (REV) have been described.2,29 These primers amplify a 291-base pair (bp) fragment located in the long terminal repeat of REV. Primers #3 (5′-CTCGGATCCGTNYTNC CNCARGC-3′) and #4 (5′-ATRTACCTRCTRCAGCTGCTC-3′), designed to detect a 139-bp highly conserved fragment of the pol gene, were used to detect avian leukosis-like retroviruses. 8 Primers #5 (5′-AGCCTGCGGCTTAATTTGAC-3′) and #6 (5′-CAACTAAGAACGGCCATGCA-3′), designed for the detection of house finch (Carpodacus mexicanus) 18S RNA and amplify a 100-bp fragment of AMGO 18S RNA, were used as a positive DNA control.
A nested PCR was used to detect any herpesvirus sequences. This technique was originally developed for the detection of the DNA polymerase gene of Kaposi’s sarcoma-associated herpesvirus but can be used to detect most herpesviruses.19,21 Primers #7 (5′-GTGTTCGACTTYGCNAGYYTNTYCC-3′) and #8 (5′-GTGTTCGACTTYGCNAG YYTNTYCC-3′) are used for the first phase of the nested PCR. The 536-bp fragment is used in the second phase using primer #8 and primer #9 (5′-ACGTGCAACGCG GTGTAYGGNKT NCANGG-3′) to amplify a 236-bp product. Primers #7, #8, and #9 were a gift from Dr. James Casey, Department of Microbiology and Immunology, Cornell University. Integrated DNA Technologies, Coralville, IA synthesized all primers.
DNA from the REV-transformed cell line RECC-CU91, the avian leukosis virus (ALV)-transformed cell line LLCC-RP9 26 and Marek’s disease herpesvirus (MDV) strain RB-1B-infected chick kidney cells were used as positive controls for REV, ALV-like viruses, and herpesviruses, respectively.23–25
All PCR reactions used 100 ng of template DNA in a final volume of 50 µL consisting of 1 × PCR Amplitaq Gold Buffer, 0.64 µM dNTPs, 1.85 mM MgCl2, 2 µM of each primer, and 2.5 U AmpliTaq Gold with GeneAmp (Applied Biosystems, Foster City, CA).
PCR products were separated on 1.5% or 2% TAE agarose gels using Invitrogen “Trackit” as molecular marker. Gel images were analyzed with Syngene GeneTools (Synoptics Limited, Frederick, MD). The following PCR conditions were used. REV: denaturation at 95°C for 5 minutes followed by 30 cycles at 94°C for 30 seconds, 57°C for 30 seconds, 72°C for 2 minutes followed by a final extension at 72°C for 10 minutes. ALV-like viruses: denaturation at 94°C for 5 minutes followed by 10 cycles at 94°C for 1 minute, 37°C for 2 minutes, 72°C for 3 minutes, 30 cycles at 94°C for 30 seconds, 55°C for 1 minute, 72°C for 1 minute, followed by a final extension at 72°C for 6 minutes. 18 S RNA: denaturation at 94°C for 5 minutes followed by 35 cycles at 94°C for 30 seconds, 55°C for 40 seconds, 72°C for 1 minute, followed by a final extension at 72°C for 10 minutes. Herpesvirus first PCR: denaturation at 94°C for 5 minutes followed by 35 cycles at 94°C for 1 minute, 60°C for 1 minute and 72°C for 1 minute followed by the second PCR denaturation at 94°C for 5 minutes followed by 35 cycles at 94°C for 30 seconds, 55°C for 40 seconds, 72°C for 1 minute, and a final extension at 72°C for 10 minutes. At the end of all PCR reactions, products were kept at 4°C.
Immunohistochemistry
Validation of reagents
In order to demonstrate the specificity and validity of the immunohistochemical technique in AMGO and HOSP tissue, sections of Harderian gland and spleen were stained using anti-CD3, IgA, IgG, IgM, Mac 387, BLA-36, CD79α, Pax-5, and Bu-1 primary antibodies. Sections were dewaxed in xylene, rehydrated through a series of solutions of decreasing ethanol concentrations (100%, 95%, and 70%), and placed in water. Immunoperoxidase staining for IgA was carried out on sections containing spleen and intestine, as described by Grodio et al. (2009). The sections were also stained for IgY and IgM following a similar protocol, with the following substitutions. When staining for IgY, a blocking step included incubation in 10% nonfat dry milk with 10% donkey serum. Sections were incubated with goat anti-bird IgG (Bethyl Laboratories, Montgomery, TX) diluted 1:600 in PBS with casein (Vector Laboratories, Burlingame, CA), followed by an incubation with biotinylated donkey anti-goat IgG secondary antibody (Jackson ImmunoResearch, West Grove, PA) diluted 1:200 in PBS. When staining for IgM, a blocking step included incubation in 10% nonfat dry milk with 10% goat serum. Sections were incubated with anti-bird IgM (Immunology Consultants Laboratory, Newberg, OR) diluted 1:300 in PBS with casein, followed by incubation with a biotinylated goat anti-rabbit IgG (Vector Laboratories) diluted 1:200 in PBS. 12
Staining of chicken and house finch spleen sections for B-lymphocyte marker Bu-1 was attempted by incubating with the mouse monoclonal antibody specific for chicken Bu-1 (Southern Biotech, Birmingham, AL), followed by incubation with biotinylated goat anti-mouse secondary antibody diluted 1:200 in a protocol similar to that used by Grodio et al. (2009), except with the antigen retrieval step omitted. 12
Specimens were also submitted to the histology laboratory at the College of Veterinary Medicine, Cornell University, for CD3, Mac 387, Pax5, and CD79α staining. Sections were subjected to antigen retrieval by incubation with pepsin. Primary antibodies (Zymed) were used according to techniques routinely used for diagnostic immunohistochemistry in the laboratory. Once reagents were validated, small intestinal sections from 9 AMGO were processed using similar procedures.
Results
Gross Findings
A total of 9 birds (5 AMGO and 4 HOSP) were presented for complete postmortem examination, and gross findings were similar in all birds. The carcasses were all in a moderate to marked state of autolysis. Seven of the birds were in good body condition, whereas the remaining 2 were in poor body condition, evidenced by a prominent keel and severe pectoral muscle atrophy. In 3 birds, there was mild diffuse hyperemia of the intestinal serosa accompanied by splenomegaly. Other findings included variably sized 3.0 ×1.0 mm to 10.0 × 2.0 mm flat, white tan foci on the liver of 2 birds and dozens of widely scattered pinpoint hemorrhages of the serosal surfaces of 1 bird.
Zinc sulfate and sugar flotation of the colon contents from 1 bird identified parasites consistent with Isospora spp. Bacterial culture of the lungs from another animal revealed mild growth of Klebsiella, which was likely a contaminant, as there were no associated lesions in the lungs.
Intestinal lesions were similar in carcasses and from the 7 additional AMGO for which intestine only was submitted. There was mild to moderate thickening of the proximal small intestine with the walls ranging from 2 to 4 mm thick. The affected areas were homogenously tan to white, slightly turgid on palpation, and bulged on section (Fig. 1). Though the severity of this lesion varied from bird to bird, the distribution of the lesion in each bird was similar. The thickening of the intestine was most prominent in the duodenum and proximal jejunum and lessened toward the ileum.
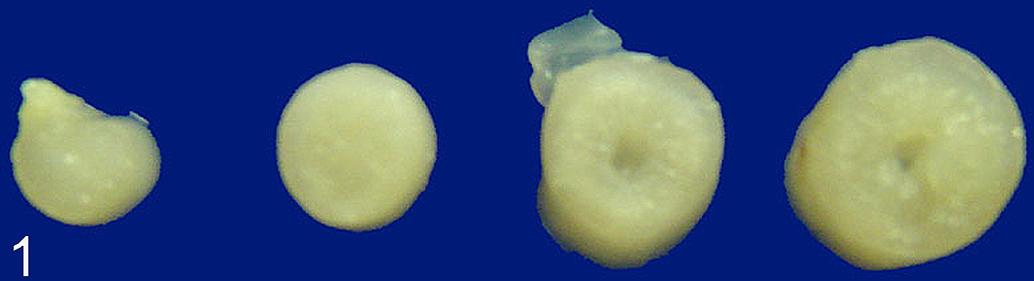
Intestine, American goldfinch. Gross appearance of intestines from affected bird showing increased thickening of the intestinal wall. The tissue from the most affected segments of intestine (far right) is pale and bulges slightly on cut section. Formalin fixed.
Histopathology
Histopathological changes in the intestine were similar in 15 of 16 birds. The tissues of the last bird were too autolyzed for interpretation. The severity of the lesion varied between birds and within different sections of intestine. The mildest lesions consisted of moderate expansion of the lamina propria by large numbers of small, morphologically normal lymphocytes. In the most severe lesions, there was transmural effacement of the intestinal architecture by small lymphocytes along with larger, 10- to 15-μm diameter round cells with indistinct cell margins, a moderate amount of eosinophilic cytoplasm, round to indented nuclei with loose, finely stippled chromatin, and a prominent central nucleolus (Figs 2 and 3). The larger cells were most dense in the deepest layer of the lamina propria, and the cellular infiltrate would occasionally dissect through the intestinal wall and extend onto the serosal surface (Fig. 2). A small proportion of the larger cells contained a moderate number of eosinophilic intracytoplasmic granules. The mitotic rate within the large-cell population was variable and ranged from 0 to 1 mitotic figure per high powered field. No mitoses were noted in the well-differentiated lymphocytes. Organisms consistent with coccidial merozoites were present in the cytoplasm of infiltrating cells. The organisms were most easily detected in moderately enlarged cells, and pockets of organisms often caused indentation of the nuclei (Fig. 2). Organisms were present in the larger atypical cells, but were less easily detected than in cells showing less atypia.
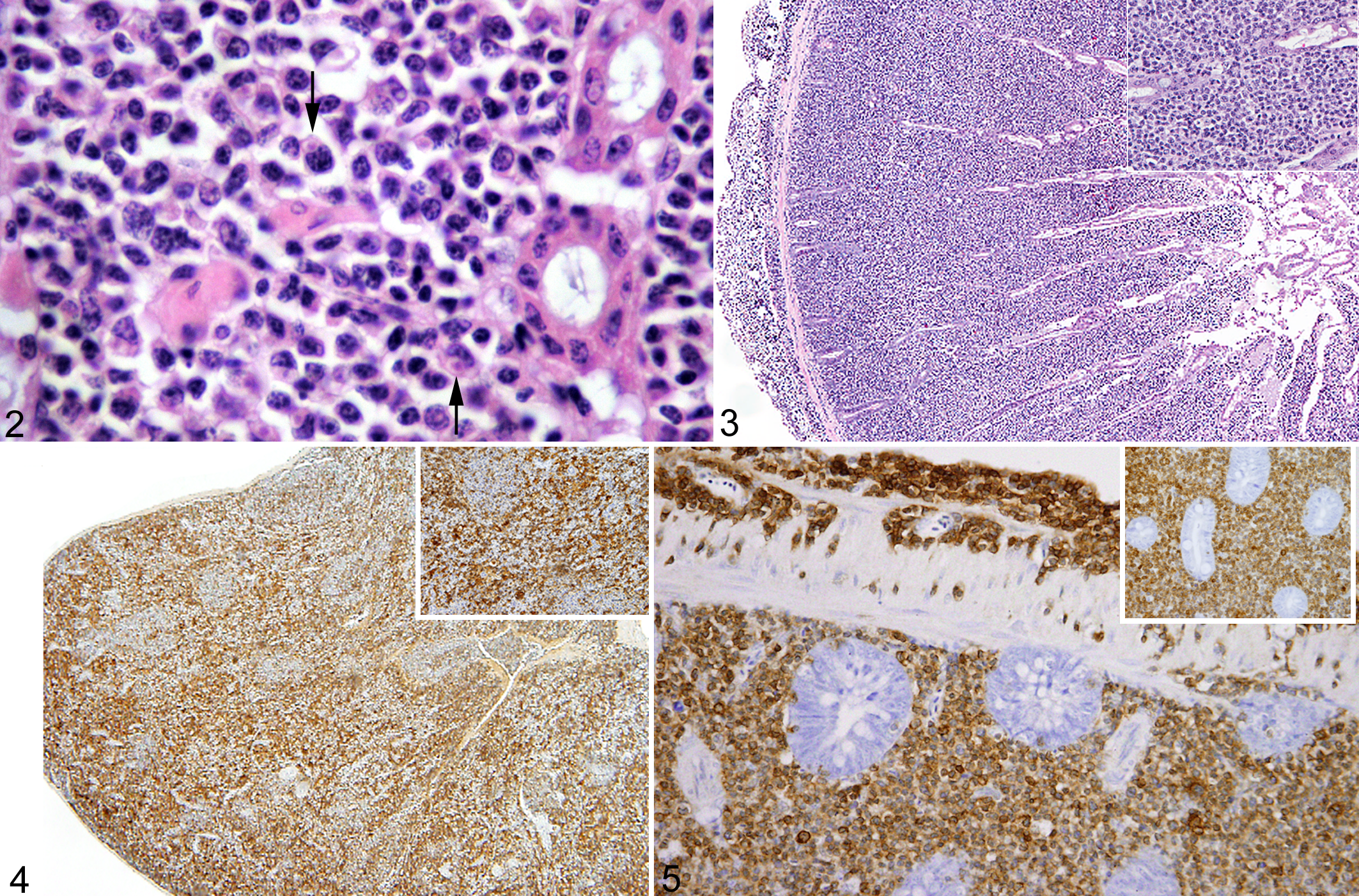
Lesions in the more distal small intestine and ileum were less severe. The lamina propria in these sections of intestine was infiltrated by small numbers of lymphocytes, plasma cells, Mott cells, and histiocytes. The larger cells described in the proximal intestine were rare.
Within the cytoplasm of variable numbers of enterocytes in each bird, there are various life stages of coccidia, which are consistent with Isospora spp. The cells contained 10- to 30-µm vacuoles containing numerous 2-µm diameter, round basophilic organisms (merozoites); 2-μm, slightly elongated basophilic organisms (microgametes); or larger 3- to 5-µm diameter eosinophilic organisms (macrogametes). Macrogamonts were the most prevalent sexual stage noted in the small intestines of these birds; no oocysts or sporozoites were noted.
Mononuclear infiltrates with intracellular parasites similar to those seen in the intestine were seen in other organs of the 9 birds for which additional tissues were available for examination. The most commonly affected extra-intestinal organs were the liver (9/9 birds) and kidney (6/9 birds). In fewer cases (3/9 birds), there was infiltration of the lung, spleen, proventriculus, and pancreas, and in 1 bird, cellular infiltrates and organisms were present in the bone marrow. In all cases, the lesions consisted of perivascular aggregates of uniform lymphoblastoid cell infiltrates. Pancreas was present in 6 of the 7 samples of birds for which only intestine was submitted. In 3 animals, cellular infiltrates and parasites were present in the pancreas.
Immunohistochemistry
The specificity of the primary antibodies for immunoglobulins and CD3 was validated by staining lymphoid organs of AMGO and HOSP. Anti-IgG,-IgA, and -IgM antibodies stained plasma cells in the Harderian gland, but not the smaller lymphocytes. The smaller lymphocytes were positive for CD3. Additionally, CD3 reacted specifically with cells in the T-cell areas of the spleen (Fig. 4).
The anti-B cell and macrophage antibodies (Pax 5, Mac 387, and CD 79α) and the avian B-cell marker Bu-1 did not produce consistent results. Bu-1 did not react with the house finch control tissues, thus it was not used on the AMGO or HOSP intestinal tissues. There was faint immunoreactivity for BLA 36 in the B-cell areas of the AMGO spleen, and scattered cells were immunoreactive for Pax 5 or Mac 387. There was no immunoreactivity against CD79a in the AMGO spleen.
The infiltrating cells in the intestines were negative for immunoglobulins, Pax 5, Mac 387, and CD79a but the cell membrane of both the small lymphocytes and larger atypical cells were positive for CD3 (Fig. 5). The proportion of CD3-positive cells decreased in the distal intestine. Approximately 80 to 98% of the cells in the proximal intestine and 15 to 70% of the cells in the distal intestine, respectively, were positive for CD3. The smaller lymphocytes had the strongest membrane staining, whereas the membrane staining of the larger atypical cells varied with section, weak in some and strong in others.
Virology
The lymphoblastoid cell cultures did not yield any permanent cell lines, and cytopathic effects were not noted. Cytopathic effects were not detected in any of the passages of CEF and CKC cultures inoculated with the lymphoblastoid cells. All PCR assays for the oncogenic viruses were negative, whereas the positive controls for REV, ALV-like viruses, and herpesviruses yielded amplicons with the expected sizes. The PCR for 18S RNA produced the expected size amplicons for all AMGO samples, indicating that DNA was of good quality.
Discussion
Atoxoplasmosis is a poorly understood disease. This lack of understanding has led to a great deal of confusion regarding the pathogenesis of infection and the classification of the causative organism.4,13,26 It is thought that the sexual stages of the coccidian reproduction occur in the intestines and that the asexual merozoite stages are able to infect cells of the immune system and cause systemic infection. Recent work by Schrenzel et al. (2005) has revealed that the avian coccidia group is closer to Eimeria than Isospora, but further classification requires the establishment of criteria for subdividing species within suborder Eimeriina. 26
Similar confusion has surrounded the classification of the lesions of atoxoplasmosis. The lesions in passerine species have been variably described as inflammatory, atypical lymphoproliferative, and neoplastic.3,10,15–20,28 Middleton (1983) noted lesions in 3 goldfinches similar to those seen our birds and described the syndrome as either lymphoproliferative or neoplastic. 18 Though parasites were present in the infiltrating cells, Middleton suggested that an underlying lymphoproliferative disorder caused the death of the birds, and that the coccidia were secondary to immunosuppression by the neoplasm. Swayne et al. (1991) described the entity in Nashville warblers (Vermivora ruficapilla) and commented that the lesions resembled lymphosarcoma but that they were more likely the result of a dysregulated immune response. 28
The lesions described in this report are similar to lesions previously described in HOSP and goldfinches. However, based on the morphology of the infiltrating cells, immunohistochemistry findings, and the invasiveness of the lesions, we propose that the lesions represent lymphosarcoma rather than a lymphoproliferative disorder.
Distinguishing between lymphosarcoma and heavy lymphocytic enteritis can be challenging. In mammals, alteration or effacement of the villous architecture, infiltration into the submucosa, and cellular atypia are criteria used to denote neoplasia. 7 Even with these criteria, final diagnosis often relies on immunohistochemistry or clonality assays to demonstrate that the infiltrating cells all exhibit a similar immunophenotype and a clonal population. 7 The birds described in this study exhibited a spectrum of changes that led the authors to believe that the associated parasitic lesions began as lymphoplasmacytic enteritis and progressed to lymphosarcoma. In the mildest lesions, the intestinal lamina propria was infiltrated by a mixture of small lymphocytes and plasma cells. In more severe lesions, the cellular infiltrates were composed of a more monomorphic population of larger, atypical cells that expanded and effaced the normal architecture of the lamina propria and thickened the intestinal wall. In the most severe lesions, the cellular infiltrate invaded the muscularis mucosa and extended into the ceolomic cavity.
Immunohistochemistry supports the possibility that the infiltrating cells underwent neoplastic transformation. These results highlight the progression of a mixed inflammatory response to a more monomorphic infiltrate. The large atypical cells were uniformly immunoreactive with CD3. CD3 is generally a marker for T-lymphocytes, but in birds, it can also mark natural killer cells, as there is evidence of a common progenitor for these 2 cell types. 11 The staining of these tissues with CD3 suggests that the infiltrating cells are either poorly differentiated T cells or natural killer cells. Ultrastructural analysis by Swayne (1991) supports a natural killer cell origin, as electron microscopy of the infiltrating cells was consistent with NK cells. 28 Immunohistochemical reagents were not available to look for AMGO-specific NK cells, and NK cells can be difficult to identify, even in species in which they have been better characterized. NK lymphomas have been described in humans, but they are often diagnoses of exclusion. Granzyme activity has been used to differentiate NK cells from other lymphoid cells, however, to the author’s knowledge, such reagents have not been previously validated for birds and generation of a proper positive control was not possible for this study. Thus, we are unable to determine if the cells are of T- or NK-cell origin.
Regardless of whether the infiltrating cells are NK cells or T cells, the destructive and infiltrative nature of the lesion, the invasion of large atypical cells through the intestinal wall, and the consistent staining of the infiltrating cells with CD3 suggest neoplastic transformation of the cells. Conclusive demonstration of the neoplastic nature of the cells in the lesions will require studies into additional surface markers of the infiltrating cells, analysis of the clonality of different cell types, cell cycle disturbances, and demonstration of how the parasitic infection interacts with the dysregulated pathways. The findings of this study indicate that these further studies are warranted, as they could yield important information on the pathogenesis of systemic isosporosis.
The relationship between the development of the observed lesions and the infection with the parasite is also unclear. It is possible, as Middleton suggests, that the birds develop lymphosarcoma for other reasons and the parasites are able to replicate because of the subsequent immunosuppression. 18 It is also possible that the parasitic infection contributes to the development of lymphosarcoma. The consistency of the lesions, the level of association with coccidian parasites, and the horizontal spread of the disease throughout the AMGO and HOSP colonies suggests an infectious etiology for the lesions. In chickens, viruses such as avian leukosis and Marek’s disease virus are known causes of lymphoma and lymphoproliferative lesions. 23 To rule out these viruses as a contributing cause, PCR, using primers known to detect a wide array of retroviruses and herpesviruses, was performed.19, 24 No retroviruses or herpesviruses were detected in the lymphocytes from these birds. The lack of known oncogenic viruses in the tissues indicates the need to examine whether the coccidia are involved in the genesis of lymphosarcoma in the AMGO and HOSP.
Proof of parasitic causality would require demonstration that the intracellular organisms alter the cell regulatory cycle and that these changes persist in the absence of the parasite. Parasite-induced oncogenesis is rare but has been reported. For example, Spirocerca lupi is noted to cause sarcomas in infected dogs. 9 The bovine parasites Theileria parva and Theileria annulata, the etiologic agents of East Coast fever and tropical theileriosis, respectively, have been shown to induce a severe lymphoproliferative response that progresses to lymphosarcoma.14, 27 Theileria parasites have the ability to transform host leukocytes and induce clonal proliferation of T cells, and to a lesser extent, B cells.14,27 Though we cannot determine whether the cellular atypia observed in the lesions described in this study would persist if the parasites were removed, we did note that the coccidian parasites were most easily detected in more well-differentiated cells. The parasites were present in the large, most atypical cells, but they were subjectively much harder to detect in the cytoplasm of these cells. Though these findings are hardly conclusive, they do suggest further study is needed to determine the relative distribution of parasites in different infiltrating cell populations.
A complete understanding of the pathogenesis of atoxoplasmosis and the nature of the lesions will require additional research. However, this study demonstrates that further investigation into the oncogenetic potential of the parasite is warranted. First, more comparative studies describing the immunophenotype of the infiltrating cells are needed. These studies are needed to determine whether the CD3-positive cells are of T-cell or NK-cell origin. Furthermore, there is evidence that the observed immunophenotype of the cells in these birds is not universal. Maslin et al. (2009) describes the infiltrate in canary fledglings as being of B-cell origin, with little contribution by CD3-immunoreactive cells. With the exception of faint immunoreactivity of splenic B cells with Pax-5 antibody, we were unable to validate other B-cell markers for use in our passerine species. However, we were able to show that the T-cell markers did not react with B cells and that the infiltrating cells were not immunoreactive for immunoglobulins. Second, the relationship between parasitic infection and the morphologic changes in the infiltrating cells should be determined. If the parasite is responsible for a neoplastic transformation, it could do so by either directly or indirectly causing chronic inflammation. Chronic inflammation has been shown to predispose to intestinal lymphoma in humans. 7 Further understanding of these relationships would not only help elucidate the pathogenesis of this poorly understood disease, but also contribute to our overall knowledge of how chronic inflammation may progress to lymphosarcoma.
Footnotes
Acknowledgements
The authors would also like to thank the Cornell University College of Veterinary Medicine Histopathology Laboratory for processing of histologic preparations, Dr. James Casey for providing the primers for herpesvirus PCR, and Dr. Meena Haribal for the information on the house sparrows.
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
This study was funded through the NSF grant EF-06227054 (A. Dhondt) under the NSF-NIH Ecology of Infectious Diseases Program.
