Abstract
Soft tissue sarcomas (STSs) develop from mesenchymal cells of soft tissues, and they commonly occur in the skin and subcutis of the dog. Although phenotypically diverse with frequently controversial histogenesis, STSs are considered as a group because they have similar features microscopically and clinically. Following resection, local recurrence rates are low in general but vary according to histologic grade and completeness of surgical margins. Complete margins predict nonrecurrence. Even most grade I STSs with “close” margins will not recur, but propensity for recurrence increases with grade. The frequency of metastasis has not been accurately estimated, but it is believed to be rare for grade I STSs and most likely to occur with grade III STSs. However, metastasis does not necessarily equate with poor survival. High mitotic index is prognostic for reduced survival time. Further research is needed to determine more precise estimates for recurrence rates and survival as related to completeness of surgical margins and to delineate potential differences in metastatic rate and median survival time between grades. Other potential indicators of prognosis that presently require further investigation include histologic type, tumor dimension, location, invasiveness, stage, markers of cellular proliferation, and cytogenetic profiles. Common issues limiting prognostic factor evaluation include biases from retrospective studies, small sample sizes, poor verification of metastasis, inconsistent STS classification and use of nomenclature, difficulties in differentiating STS phenotype, and diversity of the study population (stage of disease and treatment status).
Soft tissue sarcomas (STSs) are mesenchymal neoplasms derived from soft connective tissues. They can occur in any anatomical site of the body, most commonly involving the cutaneous and subcutaneous tissues. STSs account for between 8 and 15% of all cutaneous and subcutaneous tumors in the dog and are especially prevalent among middle-age to old, medium- to large-breed dogs. 18,33 Most of these tumors are locally expansile and grow between fascial planes. 33 They are usually surrounded by a pseudocapsule, tissue that resembles a capsule but is formed by the compression of peritumoral connective tissue and may contain or be confluent with tumor cells. Cutaneous and subcutaneous STSs have a low to moderate postsurgical recurrence rate (reported ranges of 7 to 30%) 7,42 and have low metastatic rate (reportedly up to 17% of cases). 11,32,33
The treatment of choice for canine cutaneous and subcutaneous STSs is surgical excision. 17,19,22,33 Curative-intent surgery aims for surgical excision of the primary tumor, the pseudocapsule, and a wide cuff of normal tissue. Overall median survival time following excision is long (reported ranges of 1,013 to 1,416 days), 11,32 and because affected dogs are usually old, they often die from other causes before succumbing to STS-related death. 5,11,32,40,50
Whereas most canine cutaneous and subcutaneous STSs have a good prognosis, the range of biological behavior is broad. Accurate prognostic information is needed to address the concerns of a patient’s owner and to select patients that may benefit from more aggressive treatment approaches, such as radical surgery, adjunctive radiation, and/or chemotherapy. For these reasons, several potential prognostic and predictive factors have been studied, and some of these are now pivotal to the clinical management of STSs. The aim of this review is to summarize the discoveries made and limitations encountered by studies that have evaluated prognostic and predictive factors for cutaneous and subcutaneous STSs of the dog.
STS Nomenclature
STSs encompass a heterogenous group of neoplasms, which depending on predominant histologic features, can ideally be further categorized according to presumed phenotype or tissue of origin. Nonetheless, they are widely regarded as a collective in the dog because they have similar histologic features and are believed to have similar biological behavior. 7,11,32 This approach was designed to foster simplicity; however, inconsistent use of STS nomenclature and poor correlation between tumor classification and histogenesis confuses veterinary diagnosticians and clinicians.
There are several mesenchymal neoplasms derived from soft tissue that are conventionally excluded from the canine cutaneous and subcutaneous STS grouping, both in a diagnostic setting and in prognostic studies. These neoplasms—including histiocytic sarcoma, lymphangiosarcoma, hemangiosarcoma, synovial cell sarcoma, leiomyosarcoma, rhabdomyosarcoma, fibrosarcomas involving the oral cavity, and brachial plexus peripheral nerve sheath tumors (PNSTs; also termed schwannomas, neurofibromas, 12 or nerve sheath tumors 16 )—are specifically excluded from STS grouping because they can be consistently distinguished by microscopic features and/or anatomical location and because as a group, they exhibit more malignant biological behavior. 8,11,17,19,32,33,35,40,50 Thus, when applied to canine tumors of the skin and subcutis, the term soft tissue sarcoma is a grouping of tumors that is becoming a diagnosis of exclusion. This is in contrast to the grouping scheme for human STSs, which is not exclusionary. 52
Mesenchymal neoplasms, which veterinary diagnosticians and prognostic studies generally include within STSs, include PNSTs (nonbrachial plexus), fibrosarcoma, myxosarcoma, liposarcoma, perivascular wall tumors, pleomorphic sarcoma (also termed malignant fibrous histiocytoma [MFH]), malignant mesenchymoma, and undifferentiated sarcoma. PNSTs may be the most common type of cutaneous and subcutaneous STSs in the dog. 32 Most canine tumors previously called hemangiopericytomas are now considered to be either PNSTs 12 or perivascular wall tumors, although histogenesis remains controversial 36,44 and the use of these terms is not consistent within the international veterinary pathology community. Hemangiopericytomas are now believed to be a small subset of perivascular wall tumors, 2 but these may be difficult to discern from PNSTs without immunohistochemistry. 2
Canine cutaneous and subcutaneous STSs are difficult to tell apart at the light microscopic level because spindle or fusiform mesenchymal cells that form bundles, streams, and whorls, as well as intercellular collagenous matrix, are common components. 27,29 Pathologists look for areas within a tumor that resemble a mature tissue or for expression of certain cellular markers to determine phenotype (Table 1 ). However, these STSs frequently display more than one histologic pattern, and histologic patterns and immunohistochemical features may not be exclusive to any one cell type of origin. 2,12,56 There is currently no consistent immunohistochemical stain or group of stains that accurately separates different types. The complexity of phenotypic differentiation has led some authors to conclude that these neoplasms should simply be considered collectively as canine spindle cell tumors of soft tissue. 11,26,44,56 Alternate names for STS such as this have gained popularity because there is controversy about the use of the term sarcoma for neoplasms where the majority are not life-threatening and do not metastasize and because of the uncertainty in differentiating some forms of benign mesenchymal tumors of soft tissue from their low-grade malignant counterpart. For example, benign and malignant PNSTs have been described as distinct entities, 12,26,27,29 but there are no studies that have validated the association between their histomorphologic features of malignancy and biological behavior. Some pathologists use alternate terminology to refer to only a subset of STSs which cannot be further classified. 11,25,28,49
Distinctions Among Types of Soft Tissue Sarcoma in the Dog
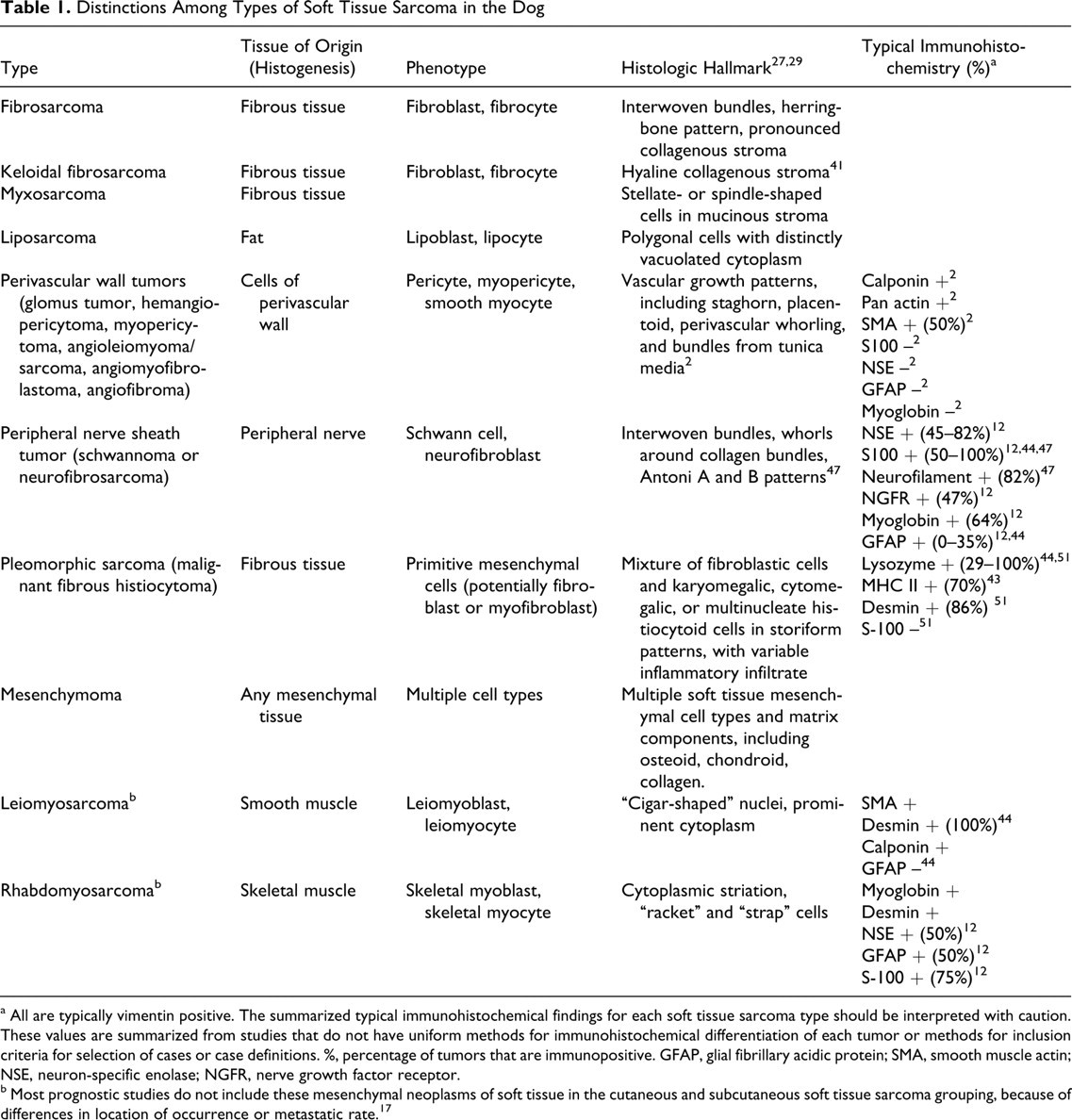
a All are typically vimentin positive. The summarized typical immunohistochemical findings for each soft tissue sarcoma type should be interpreted with caution. These values are summarized from studies that do not have uniform methods for immunohistochemical differentiation of each tumor or methods for inclusion criteria for selection of cases or case definitions. %, percentage of tumors that are immunopositive. GFAP, glial fibrillary acidic protein; SMA, smooth muscle actin; NSE, neuron-specific enolase; NGFR, nerve growth factor receptor.
b Most prognostic studies do not include these mesenchymal neoplasms of soft tissue in the cutaneous and subcutaneous soft tissue sarcoma grouping, because of differences in location of occurrence or metastatic rate. 17
Limitations of Canine STS Prognostic Studies
Numerous studies describe prognostic and predictive factors for canine STSs, but few have involved properly implemented survival analysis purposely aimed at improving STS prognostication. Indeed, much of the information regarding prognosis of STS has been gathered from studies designed for other purposes, such as comparing efficacy of two or more treatment modalities. 11,23,28,38,46,48 In these studies, the objective is to assess a treatment strategy rather than just assess prognostic variables. These studies are subjected to a variety of selection and treatment biases that confound evaluation of prognostic variables. Furthermore, the majority of studies that examine prognostic factors for canine STS involve limited sample sizes (ie, < 100 dogs) with questionable statistical power. 3 –6,8, 23 –25,28,32,34, 36 –39, 45, 46, 49 –51 These studies may have failed to detect statistically significant differences in disease outcome that could have been attributed to a prognostic factor, especially because many evaluated an inappropriately high number of prognostic variables relative to the number of events that occurred in the study population. 55 Estimates for rates of metastasis or recurrence are also not precise in these studies. In addition, most studies evaluating prognostic factors for canine STS were conducted retrospectively. 3,5,7,11,23,32,46,48 –50 These studies can be troubled by a variety of biases and limitations, 55 including loss to follow-up, inability to review original slides, 11 lack of standard treatment and/or follow-up protocols, inability to confirm metastasis, and inability to provide accurate measurements of tumor size. 48 Another common drawback of STS prognostication studies is a lack of multivariable analysis. 4,5,7,11,20,25,37,40,46,49,50 In these studies, numerous prognostic variables were explored independently without adjusting for the effects of other prognostic variables. There is some disparity in the way in which canine STSs are grouped among studies. For example, some studies include visceral STSs, 4,34,48 and some studies include histiocytic sarcomas, 25,48 leiomyosarcomas, 48 haemangiosarcomas, 18 and synovial cell sarcomas. 45,48 This confounds evaluation of prognostic factors because most of these neoplasms probably behave more aggressively than conventional cutaneous and subcutaneous STSs. Finally, several studies have dissimilarity between patients in terms of natural disease history (eg, length of time that a tumor was present, inclusion of patients with recurrent tumors or detectable metastasis); 5,23,46 these may have failed to detect true differences in prognosis.
Standards for conducting a veterinary oncologic prognostic study have recently been described in detail. 55 The themes described above exemplify the difficulty in adhering to these standards and, at the same time, indicate the need to more closely evaluate the prognostic variables conventionally used for canine STS diagnosis. Whereas many studies lack methodological rigor, it is nonetheless informative to describe consistently observed trends. Therefore, these types of studies are included in this review, and limitations are designated where essential. It is emphasized that summarized trends cannot replace investigation using proper study design and analysis. Many uncertainties stemming from canine STS prognostication research remain to be resolved, and it is imperative that future studies conform to the methods necessary to do so.
Prognostic and Predictive Factors
Several prognostic and predictive factors for canine STS have been historically considered in a clinical setting, including histologic type, histologic grade, tumor dimensions, tumor location, previous treatment, clinically evident invasiveness, and completeness of surgical margins. 17,19,22,33 However, as reviewed below, there is limited scientific support for many factors conventionally suspected to have prognostic significance, whereas there is compelling evidence for others. No single factor consistently predicts the outcome of an STS; therefore, no single factor can be used to dictate STS management.
Histologic Type
In humans, all STSs were historically treated as a group for the purposes of prognostication; however, accumulating evidence suggests that STS types exhibit unique patterns of spread, recurrence, and prognosis. 9,15 Several studies have considered histologic type as a potential prognostic factor for canine STS, but none have adopted methods necessary for meticulous measurement of detailed differences in disease progression among STS types. Also, none of these studies have provided detailed methods for differentiation of STS type. Several studies have found no significant differences in survival time or local recurrence among different STS types, but the statistical power to detect these differences is questionable given the small numbers of study animals and restricted sample sizes for certain STS types (particularly, pleomorphic sarcoma [MFH], liposarcoma, and myxosarcoma). 37,39,48,49 In one study, STS type (malignant PNST versus other types) was not prognostic for tumor recurrence or metastasis when adjusted for mitotic index (MI) or grade. 32 Associations between STS type and MI and/or grade suggest that disparate behavior between STS types, if existent, could be attributed to differential propensity for high grade and/or MI. 7,23,32 Several studies describe trends that suggest fibrosarcoma may carry a worse prognosis or that PNST may carry a better prognosis than other STS types; 3,5,7,25,37 however, robust analytic methods are needed to confirm and measure differences in prognosis.
The degree to which STS types similarly behave is presently unknown, and it is possible that true differences in prognosis exist. However, differences are not clinically apparent; thus, STS type currently has little bearing on the clinical management of these tumors. 17,33 An important step in detecting potential differences is to define methods for accurate classification. Recent publications have examined or attempted to better define different types of STSs, such as pleomorphic sarcoma (MFH), 43,51 keloidal fibroma and fibrosarcoma, 41 liposarcoma, 4 perivascular wall tumors, 2 and PNSTs. 26,47 These studies improve precision of terminology and diagnosis, even though there is considerable overlap of histologic patterns among STS types. Studies are needed that compare prognosis among sufficient numbers of accurately classified tumors from each STS type category. Such studies would seem to require the prospective use of a large panel of immunohistochemical markers, including those applied to frozen sections. 2
Among pathologists, there is widespread inconsistent use of diagnostic terms for various STS types. For example, the diagnoses PNST, perivascular wall tumors, and hemangiopericytoma are not consistently assigned, with some pathologists preferring the use of one term over another. If veterinary oncologists and pathologists are to produce prognostic research that has clear translational relevance and results that can be readily integrated into a body of knowledge, STS types need to be classified consistently (preferably as described under STS Nomenclature) so that studies can be accurately compared.
Degree of Resection and Completeness of Surgical Margins
To avoid local tumor recurrence, conventional surgical recommendations for STS follow the Enneking classification of musculoskeletal tumors, 21 with curative intent surgeries typically requiring a “wide” or “radical” excision. 17,19,33 Wide excision of STS attempts to achieve 30 mm laterally and one fascial plane or 30 mm of tissue depth from the palpable edge of the tumor but still work within the same anatomic compartment as the mass. Radical excision involves removal of the entire compartment containing the tumor, typically including muscle, bone, and/or limb. Other terminology used to refer to the degree of resection include narrow excision (variably defined but often referring to < 3 mm of normal tissue marginating the mass) and marginal excision (plane of dissection passes through the pseudocapsule). Trends suggest that STSs removed with wide or radical excision have the lowest recurrence rate, 6 whereas recurrence is more frequent with narrow excision and most frequent with marginal excision; 11 however, statistically significant differences have yet to be demonstrated. Local recurrence of marginally excised STS is nonetheless fairly common, reportedly 13 of 45 (29%) 11 and 10 of 27 (37%) 10 for tumors marginally excised in primary practice and referral centers, respectively. A relationship between degree of resection and survival has yet to be clearly demonstrated. Although it has been explored by several studies 10,11,39,50 and is a significant prognostic factor for survival on univariable analysis of dogs with liposarcoma, 4 robust studies are needed that include an adequate number of events for the level of prognostic variables examined in multivariable analysis. The lack of a sizable influence of degree of resection on patient survival would call to question the need for radical treatment, such as amputation, unless the tumor is causing physical impairment of the patient.
There has been little investigation of the extent to which degree of resection (assessed macroscopically at surgery) corresponds to completeness of surgical margins (assessed microscopically following processing of a biopsy), but incomplete surgical margins frequently correspond to masses excised with intralesional, marginal, or narrow approaches.
3,5,10
To our knowledge, no study has evaluated completeness of surgical margins as a continuous variable (ie, minimum distance between neoplastic cells and surgically created section edge). Instead, completeness of surgical margins is considered as a categorical variable, and there is wide variation in the methods used to define categories. Incomplete (“dirty”) margins are usually defined as having neoplastic cells at surgical margins; however, some studies include tumors having neoplastic cells within 1 mm of surgical margins.
48
Close (also referred to as “marginal” or “narrow”) margins have been variably defined as neoplastic cells within ≤ 1 mm of surgical margins or as absence of tissue outside the pseudocapsule;
40
neoplastic cells 1 to 3 mm from the surgical margin;
50
neoplastic cells within < 5 mm of the surgical margin;
6
and neoplastic cells within < 10 mm of the surgical margin.
48
Complete margins have been variably defined as tumor pseudocapsule ≥ 1 mm from the surgical margin;
40
neoplastic cells > 3 mm from the surgical margin;
3,50
neoplastic cells > 5 mm from the surgical margin;
6
and neoplastic cells > 10 mm from the surgical margin (wide).
48
Some studies provide little detail as to how completeness of surgical margins was categorized.
11,46
It is very difficult to translate research findings to diagnostic use when there is such wide variation among studies and when methods cannot be imitated. To improve consistency among studies, it is recommended that researchers examining completeness of margins consider them as a continuous variable. If a categorical variable must be used, it is recommended that the following terminology be adopted, based on the methods used by studies from which prognostic significance of margin evaluation was found:
Incomplete margins: Neoplastic cells are continuous with at least one surgical margin in any plane.
Close margins: Distance between surgically created tissue edge and neoplastic cells is less than 3 mm thickness,
50
or surgical margins do not contain normal tissue outside the pseudocapsule.
40
Complete margins: Distance between surgically created tissue edge and neoplastic cells is at least 3 to 5 mm.
3,6,50
Research that considers completeness of surgical margins as a continuous variable, using multivariable analysis that accounts for histologic grade, would facilitate validation of these categories.
As with human STS, 14 completeness of surgical margins is an important prognostic factor for recurrence. STSs with complete margins are less likely to recur, 32 and they have significantly longer tumor-free intervals than those without. 11,40 Trends described for recurrence rates of STS with incomplete and close margins are similar (reports ranging from 2 of 12 [17%] to 10 of 36 [28%]); 32,40,50 however, differences in recurrence rates have not been statistically measured. Recurrence rates for STS with tumor pseudocapsule > 1 mm from the surgical margin are as low as 0 of 30 (0%), regardless of grade. 40 However, the median time to recurrence in this study was 12 months and minimal follow-up time was 6 months, so it is possible that recurrence rates were underestimated. 40 Further study is needed to measure and statistically compare differences in recurrence rates and survival for complete, close, and incomplete margins and to determine if extent of margin completeness (in terms of minimal distance between neoplastic cells and surgically created section edge) imparts any prognostic benefit. Because recurrence rates appear to be different between general practice and referral centers (reported from 20 of 116 [17%] 40 to 29 of 104 [28%] 11 as compared to 4 of 53 [8%] 5 to 11 of 75 [15%] 32 ), studies evaluating prognostic factors for recurrence (including completeness of margins) will need to target the study population of the clientele of interest. Furthermore, completeness of surgical margins has not yet been evaluated as a prognostic factor for survival by using appropriately focused study design and analytic methods.
Many prognostic studies concerned with completeness of surgical margins account for tissue selection methods to some degree, 3,32,40,50 but there is some variation. Several factors affect the ability to determine the thickness between and relationship of neoplastic cells and surgical margins, including tissue handling by surgeons; incision of larger tumors; inking of margins; contraction of muscle, fat, and fascial tissue; suturing of tissue to prevent slippage and artifactual exposure of a pseudocapsule; number of sections prepared; and areas targeted during tissue selection (trimming). These factors should be specified and addressed by a study's methods so that they are adopted by those wishing to apply the findings of a study in a diagnostic or clinical setting. Recommended guidelines for submission, trimming, and margin evaluation of tumor biopsies have recently been described. 31 Future STS research aiming to reexamine completeness of surgical margins as a prognostic factor should adhere to these standards to improve consistency among studies and reproducibility of results. Specifically, for cutaneous and subcutaneous tumors that may be STSs, it is necessary to ink surgical margins. Before large tumors are fixed, transverse cuts can be made through the overlying skin and into the tumor in such a way that the tumor remains in one piece and retains its shape without artifactual slippage of its margins. There are no studies that indicate superiority of any single tissue-trimming methodology, and there are limitations for each: Radial sectioning evaluates the least percentage of margin circumference; tangential sectioning cannot provide a numeric measurement for the distance between neoplastic cells and the surgically created perimeter of a biopsy; and parallel sectioning may overlook small tumor projections that occur between the sections evaluated. For these reasons, sections derived from more than one tissue-trimming method should ideally be collectively evaluated for each biopsy. The accuracy of even rigorous methods for margin evaluation is largely unknown, and given the infiltrative and tentacular propensity of canine STS, there is considerable potential for error. Future studies are necessary to determine the accuracy of and most suitable methods for STS surgical margin evaluation. Such research would seem to require a prospective approach, meticulous sectioning techniques, and long-term follow-up for tumor recurrence.
Histologic Grade
Grade is considered to be the most important prognostic factor for STS of humans. 14 The grading system adopted for assessing STS in dogs was first developed for use in people and is one of two systems used internationally for assessing STS in humans. 53 It was first applied to canine cutaneous and subcutaneous STSs in the late 1980s. 32,37,38 It takes into account tissue differentiation, MI, and necrosis (Table 2 ).
Grading System for Cutaneous and Subcutaneous Soft Tissue Sarcoma in the Dog a

a From Trojani et al. 53
b Combined differentiation, mitotic, and tumor necrosis scores.
The majority of canine STSs classified by this system are grade I. 38,40 Grade I tumors have the lowest likelihood of recurrence following excision, but recurrence is also related to completeness of surgical margins, as previously discussed. 40 When surgical margins are complete, grade I tumors appear to rarely recur. 6,40,50 Even when surgical margins are close, grade I tumors recur infrequently (3 of 41, 7%). 40 Similarly, metastasis of grade I tumors is considered to be rare, and its occurrence is sometimes debated among clinicians and pathologists. Lymph node and pulmonary metastasis are reported for grade I STSs; 3,32,49 reportedly, 4 of 31 (13%) grade I STS tumors metastasized in one study, 32 1 of 15 (7%) in another, 49 and 4 of 48 (8%) combined grade I and II STS tumors in another. 39 However, histologic confirmation of metastasis was reported only by McKnight et al, 39 who did not specify if metastases were from grade I or II tumors. Studies of canine STS in general have not always employed thorough follow-up protocols and/or reported histologic verification of metastasis, relying on diagnostic imaging techniques and/or cytology to demonstrate metastasis and leaving uncertainty about estimates for metastatic rate. 3,7,11,23,32,34,40,48,49
Grade II tumors occur with intermediate frequency. 23,38,40 Grade II tumors with complete margins also appear to rarely, if ever, recur. 40 Grade II tumors with close margins recur more frequently than grade I tumors (14 of 41, 35%), and when they recur, they have a shorter tumor-free interval than grade I tumors. 40 The metastatic rate of grade II tumors is uncertain and reported to be 2 of 27 (7%) in one study, 32 3 of 9 (33%) in another, 49 6 of 22 (27%) in another, 23 and 4 of 48 (8%) in one that combined grade I and II tumors. 39 Comparisons of the difference in metastatic rate between grade I and II tumors have been hampered by the small number of metastatic events, varying stage of disease in the study population (eg, inclusion of primary reexcision cases), and methods used to detect metastasis or follow-up. 23,32,49 Further study is needed to establish whether or not there is a difference in metastatic rate of grade I and II tumors.
Grade III tumors are least common, constituting around 7 to 17% canine STSs of the skin and subcutis. 23,38,40 They are considered to have the greatest potential for recurrence and metastasis; however, information on these tumors in humans and dogs is necessarily based on studies with relatively small numbers of grade III tumors to evaluate. 48 Recurrence of grade III STS with complete surgical margins has been observed following wide surgical excision, 6,48 but the rate of recurrence when surgical margins are complete appears to be infrequent. 40 Grade III STSs with close margins recur more frequently than do lower-grade tumors (3 of 4, 75%), 40 and there is concern that grade III STSs may be more difficult to completely resect than lower-grade tumors. 23 Analysis of a larger number of grade III tumors is needed. Metastasis most frequently occurs with grade III STSs, but metastatic rate has not been accurately measured. Reports of metastatic rate for grade III tumors include 2 of 9 (22%), 7 of 17 (41%), and 17 of 39 (44%), but these values should be generalized with caution because they are not precise estimates and they have been derived from study populations receiving different treatments. 23,32,48
There is no clear consensus regarding the association between STS grade and patient survival. In some reports, grade is prognostic for survival, 32 whereas in others it is not. 23,39,40,49 However, the latter studies had smaller study populations and, therefore, a more limited number of events per grade to allow reliable detection of true survival differences. Tumor grade may not have a profound effect on survival because most STSs are relatively slow growing and can be managed locally without affecting the patient’s quality of life, metastases also appear to be slow growing and may not affect survival, 23,32,37,46,48 and most dogs diagnosed with STSs are elderly and likely to die from other causes before metastatic or local disease become fatal. 5,10,11,32,40,50
Histologic grade is an intrinsically subjective test and therefore subject to intraobserver and interobserver variation, which impedes translation of research findings to clinical use.
55
In human STS, there was a 75% agreement between 15 pathologists when 25 cases were evaluated with an STS grading scheme similar to the one described here.
13,49
Agreement for differentiation was lowest (61%) of the 3 categories analyzed.
13,49
Similar studies have not been conducted for canine STS. Veterinary pathologists often debate the reproducibility of STS grading, particularly for the differentiation category. Some guidelines are proposed here to improve consistency of use among pathologists; these are based on our practices and the grading system used for human STS:
53
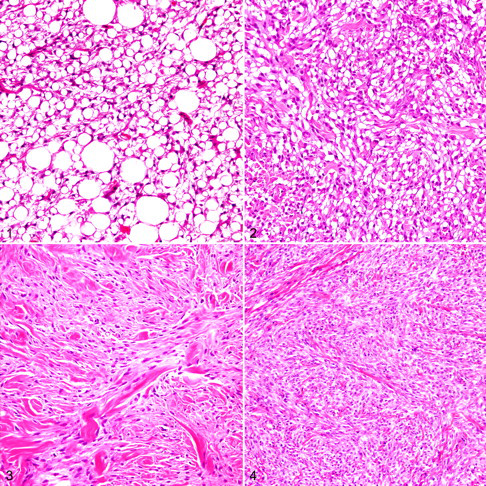
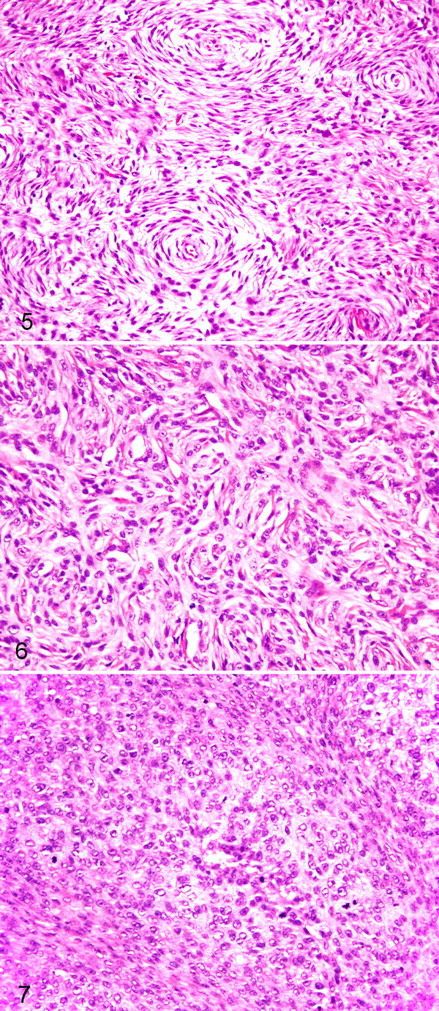
Evaluation of 1 section per 2 cm of tumor diameter is critical if accurate grade results are to be obtained. Areas used for grading should be well fixed and not overly complicated by inflammation or hemorrhage. MI is the number of mitotic figures in 10 contiguous (where possible) high-power fields (400×). It should be measured in the most cellular part of the tumor and the area with highest mitotic activity. If the count is close to the cutoff, recounting is recommended for confirmation. Foci of necrosis, hypocellular areas, and zones of ulceration should be avoided. Necrosis should be differentiated from mucinous or hyaline change, hemorrhage, and surgery- or biopsy-associated trauma. Differentiation represents a combination of histologic type and true differentiation. The histologic type of a tumor is generally determined from its architecture and is best assessed at low power. Type is indicated by classic histologic patterns (Table 1). If a type can be determined, it is rated either score 1 (well differentiated) or score 2 (poorly differentiated) (Figs. 1–6). Well-differentiated tumors are those that most closely resemble normal adult mesenchymal tissue (Figs. 1, 3, and 5). Conversely, poorly differentiated tumors have a modest resemblance to normal adult mesenchymal tissue while at the same time being clearly of a particular tissue type (Figs. 2, 4, and 6). If the type of a tumor cannot be determined or if indicative histologic patterns are difficult to discern, it is undifferentiated by definition, score 3 (Fig. 7). We emphasize that degree of differentiation does not account for uncertainty of histogenesis (ie, although PNST and PWT have controversial histogenesis, these have resemblance to a normal adult mesenchymal tissue and should not be given a score of 3 if characteristic patterns are evident).
Grade has now been used in canine cutaneous and subcutaneous STS diagnosis for more than 20 years. It has proven to be a useful prognostic indicator, especially in terms of predicting recurrence and for indicating concern for metastasis (grade III tumors). To better clarify the relationship between grade and metastasis, future studies should ideally include a thorough follow-up protocol to detect and confirm metastasis. This would seem to require histologic verification of metastases at necropsy because clinical examination, diagnostic imaging, and cytology may lead to overestimation of metastasis (eg, other diseases appear similar radiographically, or cytology may not accurately differentiate between neoplastic and reactive spindle cells). Yet, losses to follow-up, failure to conduct necropsies, and lack of sufficiently long follow-up periods may underestimate metastatic rate. This is especially concerning for STS because time to metastasis may be longer than 3 years in some cases and metastases may be clinically silent.
22,32,46,48
Furthermore, to confirm the significance of metastasis, future studies should be designed such that the relationship between survival and metastasis can be evaluated. If survival is evaluated as the prognostic outcome, the magnitude of difference in median survival time among grade groups should be measured. Research that aims to confirm or improve on the present grading system might consider a comparison of 2 grading schemes (with and without “differentiation”) because of the questionable reproducibility of this category and because multivariable analysis indicates that MI and necrosis are the only statistically significant prognostic elements of the grading scheme.
32
MI and Proliferation Markers
MI, independent of grade, provides important prognostic information. High MI has been inconsistently defined among STS studies; the lowest cutoff value for high MI used was 9 mitotic figures per 10 high-power fields. 7,40,49 High MI is associated with recurrence, reduced tumor-free interval, metastasis, and reduced survival time. 7,23,32,40,49 For MI ≥ 9, median survival reportedly ranges from 150 to 343 days, whereas for MI < 9, median survival ranges from 826 to 1138. 7,49 Median survival was 236 days for MI ≥ 20, 532 days for MI between 10 and 19, and 1,444 days for MI < 10. 32 These numbers must be generalized with caution because each was derived from small and diverse study populations with differing treatment approaches and natural disease history. Even so, greater prognostic information has been demonstrated from grade, especially in terms of predicting recurrence. 28,40
Increased scores for other markers of cellular proliferation, including AgNOR and Ki-67, are also prognostic for decreased survival time; however, the magnitude of difference in median survival time among different scores was not adequately compared. 23 These techniques provide additional insight into the state of proliferation at the time of a tumor’s evaluation and are routinely used to gain prognostic information for several human neoplasms. 54 However, for canine STS, it is unclear whether these other markers provide a level of prognostic information that, in a clinical setting, has an advantage over MI or grade alone, especially with their additional cost including time. These other markers have yet to be widely adopted.
Other Factors
The potential for several other variables as prognostic factors remains to be resolved.
Concerns exist that recurrent STSs have a decreased survival time and higher metastatic potential relative to primary STSs, 33 but this has yet to be clearly demonstrated in dogs. Initial excision may disrupt or contaminate tissue planes, making subsequent complete surgical excision more difficult. 46 Reports describe conflicting trends for recurrence rates of reexcised STSs. 3,46
Tumor size or volume may preclude complete excision, depending on the anatomic location, and may be an important negative predictive factor, especially for local recurrence. Large size is generally considered a poor predictive factor. 19,33 However, in several studies examining tumor size, it was not significantly prognostic for tumor recurrence, survival, or disease-free interval. 3,7,11,37,46 These studies may not have statistical power to detect true differences. Other studies describe trends suggesting that large tumor size (> 5.5 cm or ≥ 4 cm, longest dimension) predicts decreased survival time or poorer response to radiation. 4,24,32,38 Because STSs resected in general practice tend to be smaller than those resected in referral centers, 11,32 source may be an important confounder to account for.
Some STSs involving the oral cavity have a low-grade histologic appearance but an aggressive clinical course 13 and poorer survival rates as compared to dogs with STS at other sites. 25 For this reason, oral STS are often not included in the conventional canine STS grouping. Apart from perioral location, there are no sites where canine cutaneous or subcutaneous STSs are considered to be more aggressive, and tumor location has not consistently been identified as a significant prognostic factor. Tumor location may affect resectability and ability to achieve complete excision. Yet several studies did not identify differences in survival time or local recurrence rate based on tumor location. 3,4,7,11 However, limited statistical power must again be considered in this instance. Studies describe trends suggesting that localization to the limbs may be prognostic for longer survival, lower metastasis, and better response to treatment. 7,8,32
Tumor mobility is an indication of invasion into underlying tissues. Invasiveness can be assessed via palpation, during surgery macroscopically, or by diagnostic imaging. Although STSs that are clearly invasive may behave more aggressively and invasive tumors may be more difficult to completely excise, reports describing prognostic trends for invasiveness conflict. 5,11,46,50
Further study is needed to determine if previously treated tumors, large tumor dimensions, certain tumor locations, and clinically evident invasiveness impart inferior prognosis. Such research will inevitably require multivariable analysis to account for the effect of more dominant prognostic factors and a sufficient number of events relative to number of variables examined.
A staging system modified from the American Joint Committee on Cancer staging system for human STS has been described for the dog. 33 However, staging systems are not consistently used in the clinical management of canine STS. To our knowledge, no studies have attempted to validate the prognostic significance of staging systems for canine STS.
Intratumoral microvessel density (IMD), determined through quantitative assessment of factor VIII–related antigen and CD31 immunostaining, was recently examined as a method of predicting behavior of cutaneous and visceral STS. 34 Because IMD is a measure of tumor angiogenesis, it may play an important role in tumor invasion and metastasis, and there is a biological basis for increased malignant potential. A positive association between IMD and the occurrence of clinically evident metastasis was shown. 34 However, because there was an association between IMD and histologic grade, 34 it is presently unclear if IMD provides any clinically useful prognostic information beyond that provided by grading.
In humans, a combination of clinical features, histologic pattern, immunohistochemistry, and molecular cytogenetics is routinely used to accurately diagnose STS type, although histogenesis remains controversial. 30 Specific genetic alterations—including chromosomal translocations resulting in fusion genes, specific oncogenic mutations, and complex karyotypic abnormalities—have been identified in human STS, shedding light on mechanisms of tumorigenesis and promoting the development of targeted therapy for specific types of STS. Some specific cytogenetic abnormalities have a prognostic role in certain STS types. 1 To our knowledge, no studies have yet explored the utility of molecular cytogenetics in canine STS diagnosis and prognostication.
Consensus on Reporting of Prognostic and Predictive Factors for Canine STS of the Skin and Subcutis
Pathologists ideally describe several of the reviewed prognostic factors in the diagnostic report for a STS, including histologic type, histologic grade, MI, and completeness of surgical margins. Until accurate methods for differentiating STS of the skin and subcutis of dogs become available, affordable, and prognostically significant, there is little incentive to specifically subclassify these tumors in a diagnostic setting. However, histomorphologic features (Table 1) may be used in many instances. Because MI imparts important information regarding survival, it should always be provided in a histopathology report. However, grade is more informative regarding the likelihood of tumor recurrence; 40 thus, MI should not be used as a substitute for grade. Standard processing and trimming procedures, which include inking and more than one sectioning technique, should be used to evaluate the margins of excised cutaneous and subcutaneous masses. 31 In describing completeness of surgical margins, best practice includes the technique (or techniques) of margin evaluation used, the number of tissue planes evaluated that constitute surgical margins, the sites of the sample from which sections were prepared, and the minimum thickness (eg, distance between neoplastic cells and surgically created section edge) and composition (eg, adipose, fibrous, necrotic, inflamed) of tissue that borders the tumor. To strive for consistent interpretation of margin results, we recommend avoiding ambiguous terms such as close and complete as a substitute for aforementioned parameters. Pathologists may explain the prognostic significance of the factors reported on without providing expected rates for recurrence or metastasis, because precise estimates have not been reported (Table 3 ).
Prognosis for Canine Cutaneous and Subcutaneous Soft Tissue Sarcoma as Indicated by Histologic Grade and Completeness of Surgical Margins a

a Precise estimates for rates of metastasis and recurrence, as related to histologic grade and completeness of surgical margins, have not been reported.
Conclusion
In conclusion, STSs are common neoplasms of the skin and subcutis of the dog that are difficult to differentiate from one another based on hematoxylin and eosin–stained sections. Research has identified valuable prognostic information from histologic grading, MI, and completeness of surgical margins. Complete margins predict nonrecurrence. Recurrence appears to increase with grade, and metastasis is uncommon for grade I and II tumors and most likely for grade III tumors. High MI (> 9 mitotic figures per 10 high-power fields) is prognostic for reduced survival. Further research is needed to establish the relationship between survival and degree of resection and/or completeness of surgical margins, to determine more precise estimates for recurrence rates as related to completeness of surgical margins, to assess proper methods for STS margin evaluation, and to delineate potential differences in metastatic rate and median survival time between grades. Other factors—including markers of cellular proliferation, tumor dimension, tumor location, histologic type, invasiveness, and cytogenetic profiles—may be useful indicators of prognosis but presently require additional investigation. As ancillary molecular techniques continue to rapidly develop, veterinary pathologists and oncologists will evaluate new markers to aid in STS prognostication. Future research examining prognostic factors should seek creative ways to maintain large sample sizes, ensure thorough and long-term follow-up, accurately diagnose STS type, maintain consistency of STS grouping with other studies, and overcome biases associated with retrospective studies.
Footnotes
Acknowledgements
This study represents an initiative of the American College of Veterinary Pathologists' Oncology Committee. The manuscript has also been reviewed and endorsed by the World Small Animal Veterinary Association. We thank the ACVP for its support and guidance.
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
The authors declared that they received no financial support for their research and/or authorship of this article.
