Abstract
Golden Retriever (GR) muscular dystrophy is an inherited degenerative muscle disease that provides an excellent model for Duchenne muscular dystrophy in humans. This study defined the histopathologic lesions, including the distribution of type I and II muscle fibers (FTI and FTII), in 12 dystrophic and 3 nondystrophic dogs between 7 and 15 months of age. The authors were interested in studying the influence on disease phenotype from crossing the base GR breed with Yellow Labrador Retrievers. The dystrophic dogs were divided according to breed: GRs and Golden Labrador Retrievers (GLRs). On hematoxylin and eosin staining, histopathologic lesions were more severe in GRs than GLRs. Six of eight GR muscles (75%) had a severe lesion grade (grade 3). In contrast, seven GLR muscles (87.5%) had mild lesions (grade 2), and only one had severe lesions (grade 3). Changes in fiber-type distribution were more pronounced in GRs versus GLRs. FTI:FTII ratio inversion was observed in three dystrophic GRs but only one GLR. The mean diameter of FTI and FTII was smaller in GRs and GLRs than in nondystrophic dogs (P < .01). The FTI of five GR muscles (62.5%) were larger than those of GLRs, whereas only one GLR muscle was larger (P < .05). The differential was less pronounced for FTII, with four GR muscles being larger and three GLR being larger. Observations indicate that crossing the base GR breed with Labrador Retrievers lessened the severity of the GR muscular dystrophy phenotype.
Keywords
Duchenne muscular dystrophy (DMD) is an X-linked recessive disorder caused by mutations in the 2.4 MB DMD gene, with an associated absence of dystrophin, a cytoskeletal protein that helps to maintain the structural integrity of muscle during the contraction process. 8,14,22,26 The animal model most used for DMD is the MDX C57BI/10ScSn mouse. However, there are morphofunctional differences between the muscles of affected mice and those of DMD patients. 5,8 Other experimental models have been identified in dogs, cats, fish, and invertebrates. Whereas numerous dog breeds with dystrophin deficiency have been described, the Golden Retriever muscular dystrophy (GRMD) model has been studied most widely. Affected dogs have genotypic and phenotypic manifestations similar to DMD. 8,9
The first descriptions of DMD referred to hypertrophy of the calf muscles as one of its most prominent characteristics. 11,14 Similarly, GRMD dogs have hypertrophy of several muscles, including the tongue, diaphragm, cranial sartorius, and esophagus, whereas most other muscles are atrophied. 18,19,28 In neonates, the flexor musculature is severely affected owing to greater use during this phase of life. 30 Thus, the effects of dystrophin deficiency vary among and within species. 12 The reasons for the phenotypic variation among dystrophin-deficient muscles are still poorly understood. 15
In general, muscles contain a mixture of myofiber types forming a mosaic pattern, with one type of muscle fiber often predominating over another. 17 The myosin ATPase (mATPase) histochemical reaction is commonly used to distinguish slowly and rapidly contracting type I and type II fibers (FTI and FTII), respectively. 17 With histochemical microscopy, FTI stain palely whereas FTII stain darkly. 10 Several studies have defined the fiber-type distribution in normal canine muscles. 2 –4,20 Descriptions of the functional and histochemical patterns of the musculature in GRMD are still scarce. In the initial report of GRMD, the mean diameter of fibers from two of three muscles evaluated was increased over controls, with fiber-type grouping and a tendency toward an increase in the proportion of FTI. 19 In a later study, the mean fiber diameter of the cranial sartorius muscle was increased in younger but not older GRMD dogs versus normal dogs. 18
All GRMD dogs worldwide were derived from the same founder dog, 19 which was used to establish a colony at Cornell University. 9 The original Golden Retriever (GR) was crossed with other breeds. 28,29 Accordingly, GRMD is not a disease of purebred GRs. However, in our breeding colonies in Brazil and at the University of North Carolina–Chapel Hill, we have maintained the GR as the base breed. The GR mutation has been further outbred to Beagles in Japan, resulting in a phenotype termed CXMDJ, which has features similar to GRMD. 27,31 In our experience, the level of inbreeding is a critical factor in determining phenotype, with highly inbred dogs being more severely affected (J. N. Kornegay and J. R. Bogan, unpublished). We were interested in whether crossbreeding the base GR breed with comparably sized Labrador Retrievers would influence the GRMD phenotype.
Thus, the aim of this study was to help fill the knowledge gaps that currently exist, by means of histochemical and morphometric analyses to characterize the distribution patterns of FTI and FTII in GRMD dogs and to determine the effect of crossbreeding on the overall phenotype.
Materials and Methods
Animals
A total of 12 male dogs between 7 and 15 months of age were examined. Six dogs aged 7 to 13 months were dystrophic GRs coming from the Brazilian GRMD colony at the University of São Paulo. The other 3, aged 10 to 11 months, were produced by breeding dystrophic GRs with nondystrophic and not myopathic Yellow Labrador Retrievers, resulting in Golden Labrador Retrievers (GLRs). The Yellow Labrador Retrievers were from the Brazilian Association of Friends of Muscular Dystrophy colony based in Ribeirão Preto, São Paulo, Brazil. Finally, the 3 nondystrophic GRs, aged 14 to 15 months, were from the University of North Carolina–Chapel Hill.
The animals were classified as nondystrophic or dystrophic according to two simultaneous complementary criteria: genotypic analysis and serum creatine kinase (CK) levels.
Serum CK Concentration
Serum samples were obtained by means of venipuncture, starting just after the dogs' birth and continuing monthly until their death. The CK analysis was determined by means of an enzymatic kit (Sigma Diagnostics, St Louis, MO). 21,24
Analysis of Genomic DNA
The genomic DNA was analyzed at the Human Genome Study Center at University of São Paulo. To perform the analyses, DNA was extracted from blood samples collected from young pups, using a commercial kit (GFX Genomic Blood DNA Purification Kit, Amersham Pharmacia Biotech, Milwaukee, WI). The genotypes of the dystrophic and nondystrophic dogs were determined in accordance with the method of Sharp et al 25 and Honeyman et al, 16 using the primers GF2 and GR1.
Collection and Processing of Muscle Samples
Muscle samples of nondystrophic and dystrophic dogs were collected just after death. The dystrophic animals used in the present work died naturally. The nondystrophic dogs were euthanized (Euthasol, Virbac Animal Health, Inc, Fort Worth, TX), as recommended in the 2001 ethics code of the American Veterinary Medical Association for scientific research with animals.
Sections were collected from the medial portion of the masseter, costal part of the diaphragm, biceps brachii, long head of the triceps brachii, semitendinosus, semimembranosus, superficial head of the biceps femoris, and cranial sartorius muscles. Each sample was immersed in n-hexane (Labsynth Co, Diadema, São Paulo, Brazil), followed by freezing and storage in liquid nitrogen. Subsequently, serial transversal sections were cut (thickness of 8 μm) in a cryostat at –20°C and mounted on glass slides using 10% polylysine (Sigma Aldrich Diagnostics, St Louis, MO).
To evaluate the distribution pattern of the muscle fiber types and their morphometry, the mATPase reaction was used with alkaline preincubation (pH 9.4). FTII stain black whereas FTI remain colorless at pH 9.4, thus indicating that the mATPase activity is stable in FTII and labile in FTI at this alkalinity. For this reaction, adenosine 5′-triphosphate was used as the enzymatic substrate (Sigma Aldrich Diagnostics).
Histologic Features
The sections of the different muscles were mounted on histologic slides and stained using hematoxylin and eosin (HE) and modified Gomori trichrome to analyze the microscopic alterations present. Modified Gomori trichrome is particularly used for staining “ragged red fibers” and connective tissue. 13,23 To characterize the histopathologic changes, a qualitative analysis method was used taking into consideration the percentage of altered muscle fibers in relation to the overall muscle cross section. 22 Four lesion grades were defined according to the extent of muscle fiber damage presented on the sections stained with HE and modified Gomori trichrome (Table 1 ).
Lesion Grades for Dystrophic Muscles Stained With Hematoxylin and Eosin and Modified Gomori Trichrome

Histomorphometric Analysis
By using the mATPase reaction (pH 9.4), the percentage of muscle fiber types was determined for each of the eight muscles evaluated. 10 Quantitative analysis was performed using an image analysis system (Image J 1.410, National Institutes of Health, Bethesda, MD), from which the proportion of FTI and FTII for the dystrophic groups was evaluated.
The images were obtained with a video camera coupled to a trinocular microscope (Eclipse 80i, Nikon) and connected to a microcomputer. Given that the muscle fibers were displayed in cross-sectional form, the minimal diameter (μm) of FTI and FTII was determined on muscle sections stained using the mATPase histochemical reaction (pH 9.4). 10 For each fiber type (FTI and FTII), a total of 100 fibers were randomly evaluated to establish the mean minimal diameter for each muscle sample from the animals in dystrophic and nondystrophic groups.
Statistical Analysis
The results were statistically analyzed by means of the Mann–Whitney method at a significance level of 1%. When the results were not significant at a 1% level, we analyzed at a significance level of 5%. The calculations and analyses were performed using SPSS 12.0.
Results
CK Levels
Table 2 details the results from the CK evaluation and their distribution between nondystrophic and dystrophic dogs.
Breed, Age, and Creatine Kinase Values of the Nondystrophic and Dystrophic Dogs
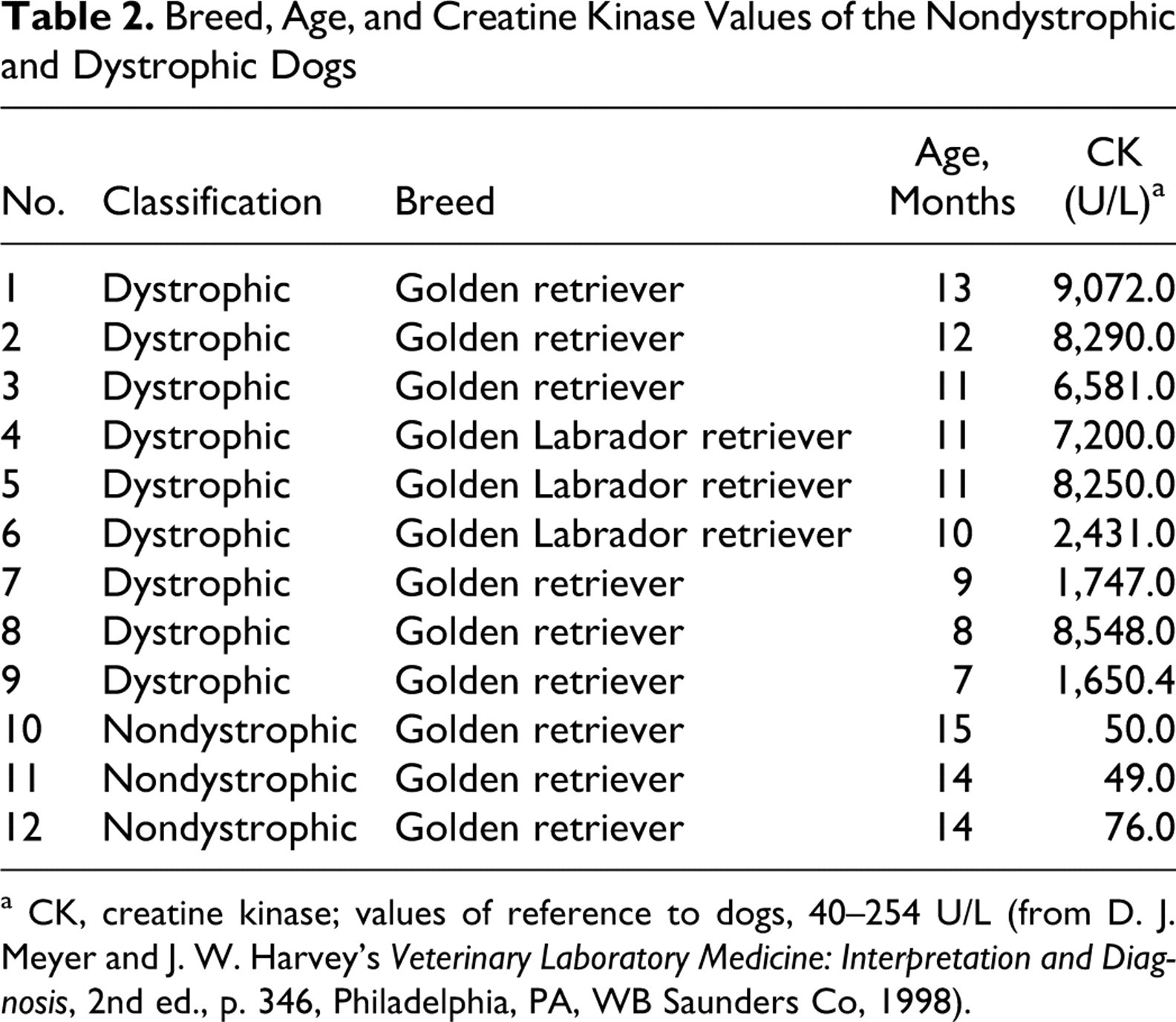
a CK, creatine kinase; values of reference to dogs, 40–254 U/L (from D. J. Meyer and J. W. Harvey’s Veterinary Laboratory Medicine: Interpretation and Diagnosis, 2nd ed., p. 346, Philadelphia, PA, WB Saunders Co, 1998).
Histopathologic Features
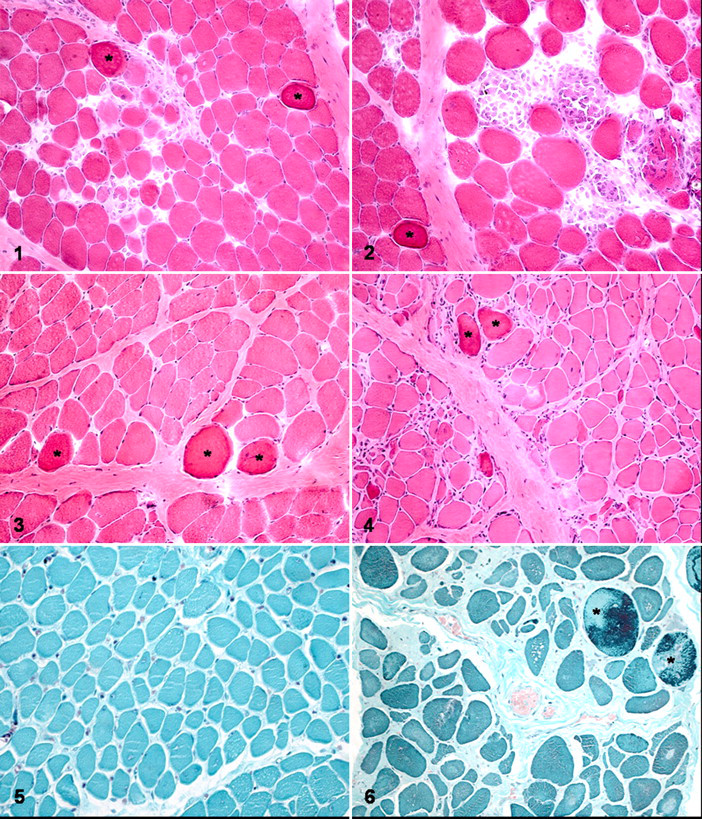
Histopathologic alterations were identified in all muscles from dystrophic dogs. With HE staining, degenerative changes ranging from dark (hyalinized) to disintegrating myofibers were most common (Figs. 1–4). Isolated or grouped myofiber necrosis was present (Fig. 2). Inflammatory cells—particularly, mononuclear cells—were associated with some degenerating myofibers and distributed throughout the endomysium or surrounding perimysium (Figs. 1, 2). There was fiber size variation. Small-diameter fibers often had basophilic cytoplasm and large vesicular nuclei, suggestive of regeneration. Some myofibers had undergone dystrophic calcification. Interfascicular fatty infiltration was observed in some muscles.
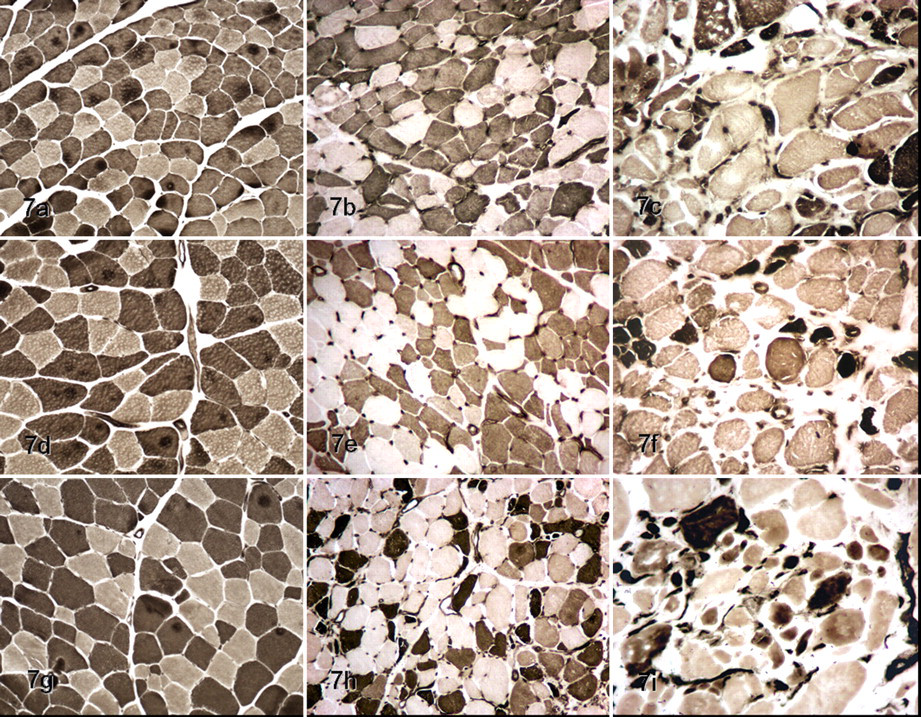
Fiber types in normal muscles (left column; a, d, g; dog Nos. 11, 10, and 12, respectively), dystrophic Golden Labrador Retriever muscles (middle column; b, e, h; dog Nos. 4, 5, 6, respectively); and Golden Retriever muscles (right column; c, f, i; dog Nos. 1, 2, 3, respectively). Top row (a, b, c) is semitendinosus muscle; middle row (d, e, f) is biceps femoris muscle; and bottom row (g, h, i) is cranial sartorius muscle. Type 1 fibers are pale; type 2 are dark. Normal muscle fibers are uniform and polyhedral. Dystrophic muscle fibers are no longer polyhedral or of uniform size. Dystrophic changes are more pronounced in the Golden Retrievers. mATPase reaction (pH 9.4).
Modified Gomori trichrome staining revealed moderate to pronounced perimysial and endomysial connective tissue in GLR dogs versus GR dogs, respectively (Figs. 5, 6 ). There were fewer ragged-red fibers, with a granular mottled red appearance in GLR dogs, compared with GR dogs (Fig. 6). 13,26
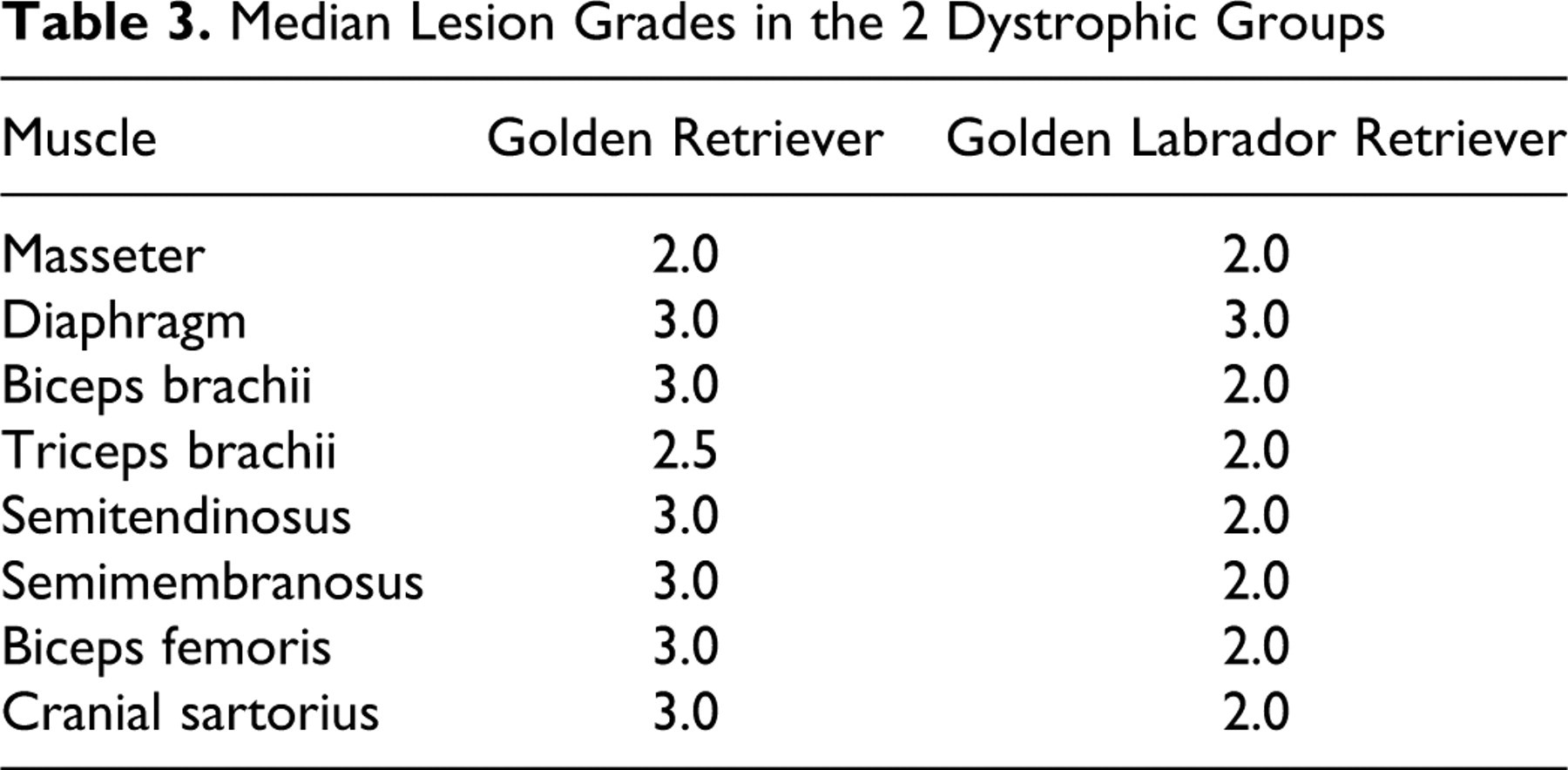
The lesions in dystrophic muscles were more severe in GRs than in GLRs. In GR dogs, six of eight muscles (75%) had a severe lesion grade (grade 3) (Figs. 2, 4, 6). In contrast, seven GLR muscles (87.5%) had mild lesions (grade 2) (Figs. 1, 3, 5), and only one (diaphragm) had severe lesions (grade 3) (Table 3 ).
Median Lesion Grades in the 2 Dystrophic Groups
Proportions of Muscle Fiber Types
We found differences for FTI between nondystrophic and both GR and GLR dystrophic dogs (Table 4 ; Fig. 7 ). In GR dogs, six muscles (75%) had an increased proportion of FTI, compared with the muscles of nondystrophic dogs. For GLR dogs, only three muscles (37.5%) had an increase of FTI. The other muscles (62.5%) had a decrease of FTI in relation to nondystrophic muscles.
Proportions of Type I and II Fibers in the Nondystrophic and Dystrophic Groups
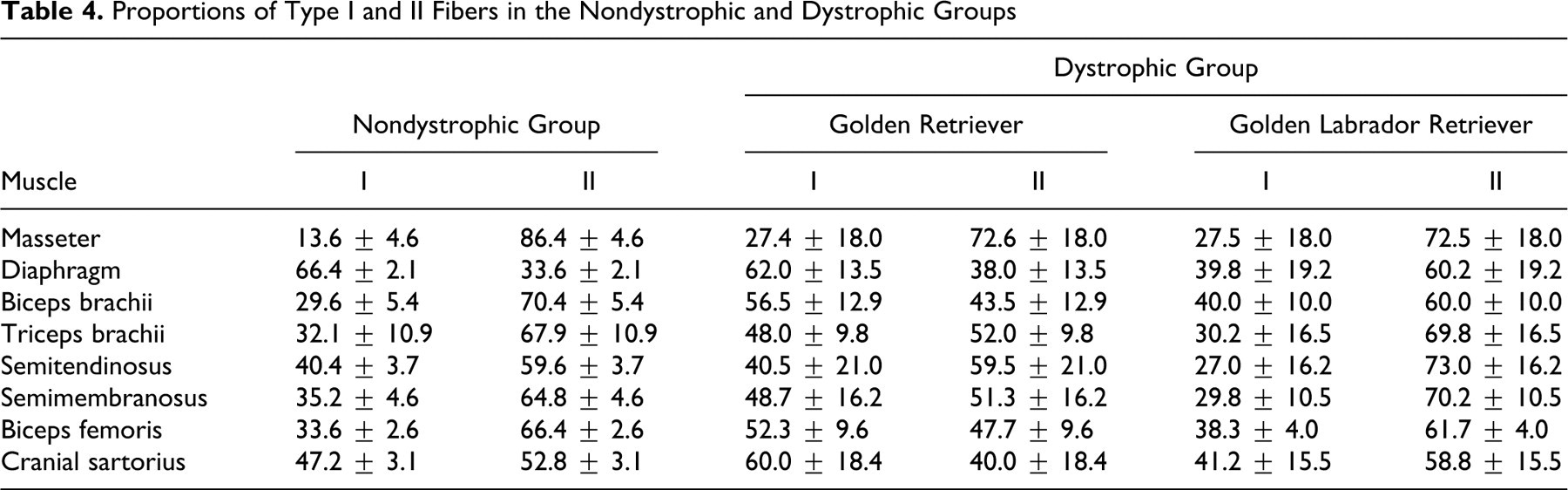
In addition, three GR dystrophic muscles (biceps brachii, biceps femoris, and cranial sartorius) had an inversion in the FTI:FTII ratio, whereby FTI predominated (Table 4; Fig. 7f, 7i). For GLR dystrophic muscles, there was an opposite inversion in only one muscle (diaphragm), in which FTII predominated. No inversion was observed in the masseter, triceps brachii, semitendinosus, and semimembranosus muscles of the dystrophic dogs (Fig. 7b).
Histomorphometric Analysis
Dystrophic muscle fibers lost their typical polyhedral shape and size uniformity, showing a range of sizes and irregular margins (Fig. 7). The mean minimum diameter of both FTI and FTII was smaller in GR and GLR dogs than in the nondystrophic dogs and showed significant difference (P < .01 for both) (Tables 5 and 6 ; Fig. 7). In addition, the FTI in five muscles (62.5%) of GR dystrophic dogs were larger than in GLR (P < .05) (Table 5). The FTI were larger in GLR versus GR dystrophic dogs in only the triceps brachii (P < .05). We did not find any significant difference for masseter and semimembranosus muscles (P < .01).
Morphometric Data Relating to the Minimum Diameter of Fiber Type I in the Nondystrophic and Dystrophic Groups
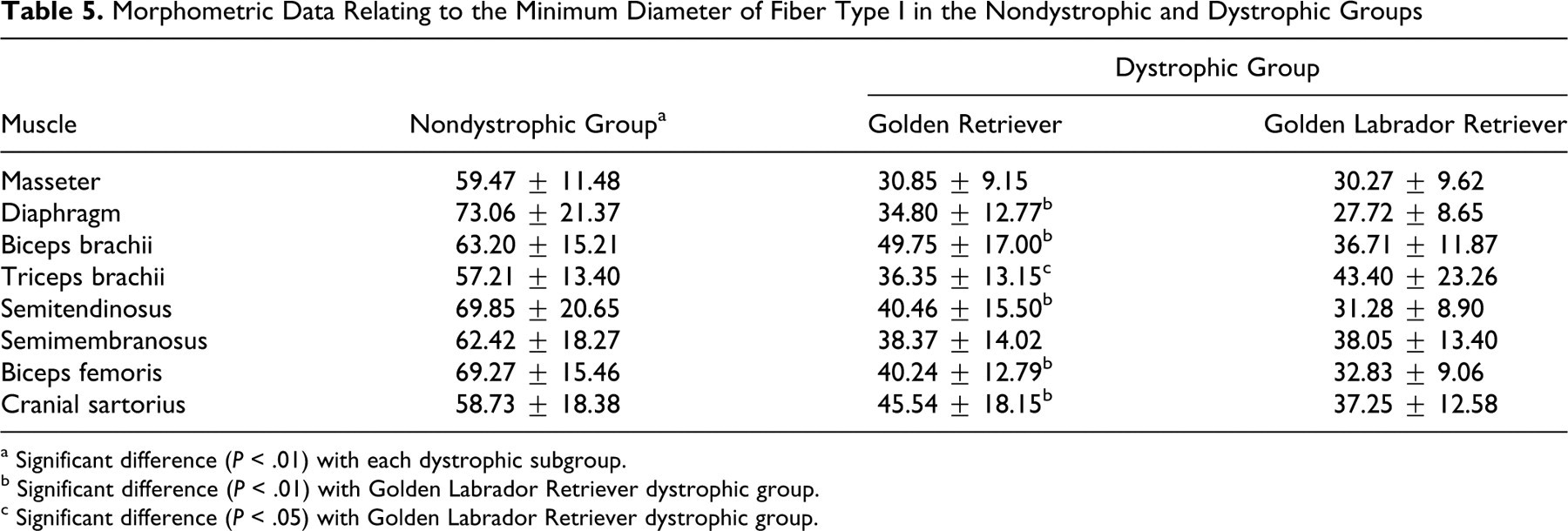
a Significant difference (P < .01) with each dystrophic subgroup.
b Significant difference (P < .01) with Golden Labrador Retriever dystrophic group.
c Significant difference (P < .05) with Golden Labrador Retriever dystrophic group.
Morphometric Data Relating to the Minimum Diameter of Fiber Type II in the Nondystrophic and Dystrophic Groups
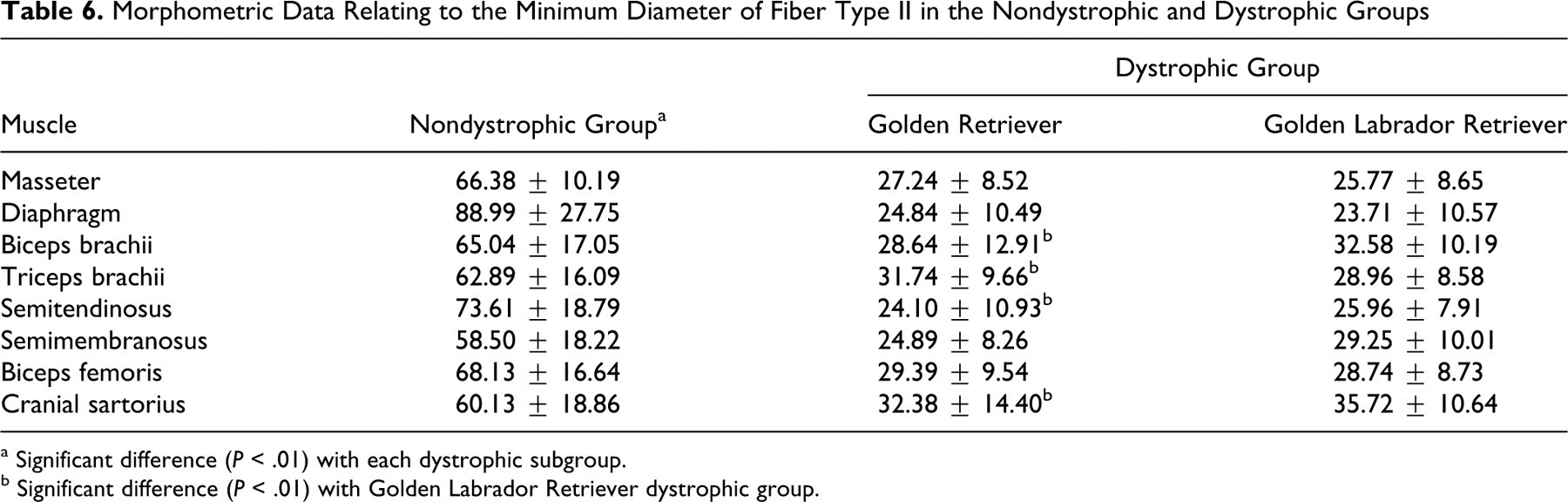
a Significant difference (P < .01) with each dystrophic subgroup.
b Significant difference (P < .01) with Golden Labrador Retriever dystrophic group.
Minimum diameter of FTII differed between GR and GLR (P < .01) in four muscles, being bigger in the GLR biceps brachii, semitendinosus, and cranial sartorius and the GR triceps brachii (Table 6; Fig. 7h, 7i). We did not find any significant difference for masseter, diaphragm, biceps femoris, and semimembranosus muscles (P < .01).
Discussion
Characteristic histopathologic changes were seen in both GR and GLR dogs. However, these changes were more severe in the GR dogs, suggesting that cross breeding could lessen the disease phenotype. Six of eight (75%) GR muscles had a severe lesion grade (grade 3). In contrast, seven of the GLR muscles (87.5%) had mild lesions (grade 2) and only one muscle had severe lesions (grade 3).The fiber-type composition of dystrophic dogs was also altered. In GR muscle, the fast contracting FTII fibers were replaced by slow contracting FTI in six muscles (masseter, semimembranosus, biceps femoris, cranial sartorius, biceps, and triceps brachii). In contrast, most muscles in GRL dogs showed the opposite pattern, in that the slow contracting FTI fibers were replaced by fast contracting FTII fibers.
Fiber types of the skeletal musculature are controlled by the nerve activity through specific signaling pathways. The calcineurin (Cn)-NFAT pathway is the principal signaling agent responsible for maintaining activity of genes that produce slow-contraction fibers in adult muscles. Induction of this activity also occurs during regeneration of slow-contraction muscles. 26 Lines of dystrophic MDX mice with enhanced activity of the Cn pathway present a more attenuated form of the disease and greater resistance to injury during the contraction processes. 6,29 This beneficial effect of Cn may occur because FTI express greater quantities of utrophin, a membrane protein that compensates for lack of dystrophin. 7
We previously reported two GRMD dogs (Ringo and Suflair) with an unusually mild disease course, despite complete muscle dystrophin deficiency. Both are descendants of a dog named Beth, a GRMD female carrier provided to our group in Brazil by one of the authors (J.N.K.), who directs the parent GRMD colony at University of North Carolina–Chapel Hill. Interestingly, histopathologic and immunohistochemical findings from biopsies of biceps femoris in these dogs (Ringo and Suflair), including fiber-type distribution, were similar to those seen in GRMD dogs with a more severe clinical phenotype. In addition, dystrophin was absent in both, and utrophin was overexpressed in a pattern similar to that observed in severely affected dogs. 1,32 Most muscles of the GR dogs studied here had a higher proportion of FTI despite having more severe histopathologic lesions. Thus, taken together, these data do not support a palliative role for FTI predominance in GRMD.
The mean fiber diameter of GR and GLR dystrophic dogs was consistently less than that of normal dogs for both FTI and FTII, presumably because of the effects of myofiber atrophy and regeneration. These findings contrast with those from earlier studies showing increased mean fiber diameter. In particular, in a previous study of 4- to 66-month-old GRMD dogs, there was evidence of cranial sartorius hypertrophy in the initial stages of the disease, followed by a reduction in myofiber diameter and an increase in the endomysial space. 20 In the present study (of 7- to 13-month-old GR dogs), the mean fiber diameter of FTI and FTII in GR dystrophic dogs was reduced, compared with normal. We might have seen larger fibers had we sampled dogs earlier. However, the mean fiber diameter of FTI was higher than that of FTII, which suggests that the cranial sartorius muscle responds to early necrosis by increasing not just the number of FTI but also the size.
In an early study of GRMD, Valentine et al suggested that smaller dogs, as produced by breeding GRs and Beagles, had a less severe phenotype. 29 More recent studies in Japan suggested that breeding affected GRs to Beagles (CXMDJ) may lessen the disease phenotype affecting skeletal and cardiac muscle. 27,31 A relative reduction in body size could contribute to this amelioration of signs. However, we believe that outbreeding itself plays a greater role than that of size in ameliorating phenotype, with highly inbred dogs being more severely affected. With this in mind, the less severe phenotype seen with GRMD dogs bred to Beagles could occur because of reduced inbreeding rather than reduced body size. Thus, we were interested in contrasting the phenotype in GRs outbred to comparably sized Labrador Retrievers. That the dogs crossbred to Labradors had a less severe histopathologic phenotype, as evidenced by degree of myofiber necrosis and fiber-type differentiation, supports the notion that outbreeding can lessen disease severity, perhaps by reducing the coinheritance of potentially deleterious modifier genes.
Footnotes
Acknowledgments
We thank the State of São Paulo Research Foundation (FAPESP) for the financial support; the Veterinary Surgery Department, University of São Paulo, Brazil; the School of Medicine, University of North Carolina; José Alexandre M. Pigatto, Maria Ines Y. de Campos, Francisca A. Ardison, and Dan Bogan for technical assistance.
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
The authors declare that their research was funded by grant FAPESP (Brazil) number 07/58360-1 and grants 1U24NS059696-01A1 (NINDS), Cooperative Program in Translational Research: Proposal for Establishment of the National Center for Canine Models of Duchenne Muscular Dystrophy (NCDMD) (Kornegay) and Muscular Dystrophy Association Infrastructure Grant to the Translational Research Advisory Committee (TRAC), Natural history, immunologic parameters in the GSHPMD dog (Kornegay).
