Abstract
The goal of this study was to morphologically characterize a ligated ileal loop model of Salmonella enterica serotype Typhimurium infection in rhesus macaques (Macaca mulatta) and to verify the occurrence of Salmonella-induced cell death in vivo. Eight adult healthy male rhesus macaques were used for ligated ileal loop surgery. Four macaques had been intravenously inoculated with simian immunodeficiency virus (SIV) mac251. Ileal ligated loops were inoculated with wild-type (WT) S. Typhimurium strain IR715 (ATCC14028 nal r), an isogenic noninvasive mutant strain (ATCC14028 nal r ΔsipAΔsopABDE2), or sterile Luria Bertani broth. Loops were surgically removed at 2, 5, and 8 hours post-inoculation (hpi). Intestinal samples were processed for histopathology, immunohistochemistry for detecting Salmonella, terminal deoxynucleotidyl transferase-mediated dUTP-biotin nick end labeling (TUNEL), and transmission electron microscopy. Combined histopathology scores were similar between SIV-infected and control macaques. As expected, the invasion-deficient mutant was less pathogenic than WT S. Typhimurium. Neutrophil infiltrate in the intestinal mucosa correlated with bacterial loads (r = 0.7148; P < .0001) and fluid accumulation (r = 0.6019; P < .0001) in the lumen of the intestinal loops. Immunolabeled WT S. Typhimurium was observed in the epithelium and lamina propria at the tip of the villi at 2 hpi, progressing toward deeper lamina propria at 5–8 hpi. Most TUNEL-positive cells localized to the lamina propria, and some had morphological features of macrophages. Ultrastructurally, bacteria were observed intracellularly in the lamina propria as well as within apoptotic bodies. This study provides morphological evidence of Salmonella-induced cell death in vivo in a relevant nonhuman primate model.
Nontyphoidal Salmonella serotypes (NTSs) are a major cause of food-borne infections worldwide. NTS infection is primarily characterized by enterocolitis and only rarely results in bacteremia. 23 Once in the intestinal lumen, NTSs readily invade the intestinal epithelium, including M cells and enterocytes, quickly reaching the lamina propria, where they are internalized by phagocytes. 22 During this invasive process, NTSs trigger a strong acute inflammatory reaction resulting in extensive tissue damage. 24 It has been demonstrated that the invasive phenotype of NTS is dependent on 5 effector proteins secreted through the type III secretion system encoded by the Salmonella pathogenicity island 1 (SPI-1). 35 Although systemic dissemination of NTS or bacteremia is a rare complication in immunocompetent individuals, it occurs commonly among patients with human immunodeficiency virus (HIV) infection, resulting in up to 47% mortality. 10 We have recently demonstrated that SIV infection in rhesus macaques, a model for HIV infection in humans, promotes systemic dissemination of Salmonella enterica serotype Typhimurium (S. Typhimurium) after intestinal invasion. 19
Early studies have demonstrated that S. Typhimurium–induced macrophage death requires a functional SPI-1 encoded type III secretion system.13,14,25 Recently, it has been demonstrated that flagellin secreted through the SPI-1 encoded type III secretion system is responsible for triggering cell death by acting as an agonist of the intracellular nucleotide-binding and oligomerization domain (NOD)–like receptor Ipaf, leading to activation of caspase-1.7,15 Since interleukin (IL)-1β and IL-18 are cleaved and activated by caspase 1, this Salmonella-induced macrophage death is considered a proinflammatory mechanism, and therefore it has been designated pyroptosis. 2 Although several studies have addressed the molecular mechanisms of Salmonella-induced macrophage death in vitro,7,15 its significance in a relevant animal model remains to be demonstrated. A previous study on S. Typhimurium infection of bovine ligated ileal loops did not report an increase in Salmonella-induced cell death 26 in this animal model of NTS-induced enterocolitis. 28
The goal of this study was to morphologically characterize a ligated ileal loop model of S. Typhimurium infection in rhesus macaques and investigate the occurrence of Salmonella-induced cell death in vivo.
Material and Methods
Animals and Experimental Design
All experiments were approved by the Institutional Animal Care and Use Committee at the University of California, Davis. Eight healthy, Salmonella-free (as determined by fecal culture), 2- to 4-year-old male rhesus macaques from a colony known to be free of SIV were used for ligated ileal loop surgery in this study (Table 1). The animals were kept in individual cages, receiving water ad libitum and commercial monkey chow. Four randomly selected macaques (macaque Nos. 1, 2, 4, and 8) were intravenously inoculated with 1000 TCID50 (tissue culture infective dose) previously titrated frozen stocks of SIV mac251 grown on peripheral blood mononuclear cells.
SIV Infection Status of Individual Macaques Used in this Study a
a Duration between SIV inoculation and Salmonella enterica serotype typhimurium infection.
Plasma viral loads were determined as previously described. 9 Briefly, plasma samples were subjected to quantitative reverse transcription–polymerase chain reaction (RT-PCR) analyses to determine the level of SIV infection. Primers and probes specific to the SIV RNA sequence were used. Probes were tagged with a fluorescent reporter dye at the 5′ end and a quencher dye at the 3′ end. Fluorescence signal was detected with an ABI Prism 7700 sequence detector (PE Applied Biosystems). Data were captured and analyzed with Sequence Detector Software. Viral copy number was determined by plotting computed tomographic values obtained from the plasma samples against a standard curve derived from samples with known viral copy numbers. 9
At 5–10 weeks after SIV inoculation (Table 1), SIV-infected macaques were anesthetized with ketamine (10 mg/kg intramuscularly; Parke-Davis), followed by placement of an endotracheal tube and maintenance of anesthesia with isoflurane (1–2%). When needed, ventilation was maintained with a positive-pressure respirator. Vital signs including heart rate, respiratory rate, and blood pressure were monitored. A laparotomy was performed, exposing the ileum and ligating 13 loops with an average of 4 cm in length, leaving 1-cm spacer loops in between. The loops were inoculated by intraluminal injections of 1 ml of either sterile Luria Bertani (LB) broth, a logarithmically grown culture containing 1 × 109 colony-forming units (CFUs) of wild-type S. Typhimurium (IR715), 19 or an isogenic invasion-deficient mutant (sipA sopA sopB sopD sopE2 mutant) of S. Typhimurium (strain ZA21). 35 Loops were surgically removed at 2, 5, or 8 hours after inoculation.19,35 An additional 4 loops were inoculated with another S. Typhimurium strain that was not included in this study. Samples for bacteriology were collected from the antimesenteric side of the intestinal mucosa using 6-mm biopsy punches. Fragments of the intestinal mucosa were incubated for 1 hour in phosphate-buffered saline (PBS) containing gentamicin at 50 mg/1 (to inactivate extracellular bacteria), homogenized, serially diluted, and plated on LB agar plates containing the appropriate antibiotics. The volume of fluid in each loop was estimated by the difference in the weight of loops before and after draining all the luminal fluid contents. None of the SIV-infected macaques developed clinical signs associated with immunodeficiency prior to surgery.
Histopathology and Immunohistochemistry
Fragments from the Peyer’s patches were fixed by immersion in 10% buffered formalin for 24–48 hours, embedded in paraffin, sectioned at 5-μm thickness, and stained with hematoxylin and eosin. Neutrophil infiltration was scored from 0 to 3 according to the following criteria: 0, no infiltration; 1, mild diffuse infiltration of neutrophils at the tips of absorptive villi; 2, moderate diffuse infiltration of neutrophils in the mucosa; and 3, severe diffuse infiltration of neutrophils in the mucosa and mild to moderate infiltration in the submucosa. Similarly, other histopathological changes were scored, including hemorrhage (0, absence of hemorrhage; 1, mild focal hemorrhage; 2, moderate multifocal hemorrhage; 3, diffuse hemorrhage), cell death (necrosis and/or apoptosis), for which the scoring system was based on the number of cells with morphological features of cell death, particularly chromatin condensation and fragmentation (0, absence of cells with morphological features of cell death; 1, a few necrotic/apoptotic cells; 2, moderate numbers of apoptotic/necrotic cells; 3, large numbers of apoptotic/necrotic cells in all villi), blunting of villi (0, normal villi; 1, some of the villi mildly blunted; 2, most of the villi moderately blunted; 3, all villi blunted with most villi severely blunted), epithelial loss (0, absence of epithelial loss; 1, detachment of a few enterocytes at the tip of a few villi; 2, detachment of some enterocytes in some villi; 3, detachment of several enterocytes at the tip of most villi), and edema (0, absence of edema; 1, mild edema characterized by mild and/or focal accumulation of interstitial fluid; 2, moderate edema with moderate and/or multifocal to coalescent accumulation of interstitial fluid with separation of collagen fibers and dilation of lymphatic vessels; 3, severe edema characterized by diffuse and severe accumulation of interstitial fluid with marked separation of collagen fibers and dilation of lymphatic vessels). All slides were coded and evaluated by a veterinary pathologist (R. L. S.) without knowledge of their infection status.
Immunohistochemistry was performed as previously described. 21 Serotype Typhimurium was detected in tissue sections by using a purified polyclonal antibody reactive to Salmonella lipopolysaccharide (Biodesign International, Saco, ME). A commercial avidin–biotin–peroxidase kit (LSAB1 Kit, DAKO Corporation, Carpinteria, CA) was used as detection system according to the manufacturer’s instructions. Briefly, 5-μm sections were hydrated and incubated in 4% hydrogen peroxide in PBS (0.01 M, pH 7.2), incubated with skim milk (2.6% in distilled water) as blocking antibody, and then incubated with the primary antibody (1:10,000 dilution) for 15–16 hours at 40°C in a humid chamber. After washing in PBS, the slides were incubated with biotinylated secondary antibody for 20 minutes at room temperature, washed in PBS again, and then incubated with streptavidin–peroxidase complex for 20 minutes at room temperature. The reaction was developed with a 0.024% diaminobenzidine (Sigma, St. Louis, MO) solution and 0.16% hydrogen peroxide. The slides were counterstained with Harris' hematoxylin. For negative controls, we used tissues known to be free of Salmonella with the primary antibody replaced by PBS. Tissue distribution of immunolabeled S. Typhimurium in different components of the intestinal mucosa and submucosa was qualitatively determined (ie, presence or absence of organisms, regardless of the number or intensity of staining).
TUNEL Staining
A TUNEL assay 8 was used for in situ detection of cells undergoing cell death. Five-micrometer sections of the Peyer’s patches were deparaffinized, hydrated, and treated with a proteinase K (Sigma, St. Louis, MO) solution (20 mg/ml) and 0.5% Triton X-100 (Sigma). In situ detection of cells undergoing cell death was performed using a commercial kit (Apoptag Plus kit; Intergen, Purchase, NY) according to the manufacturer’s instructions. Sections of normal female rodent mammary gland, obtained 3–5 days after weaning of rat pups, were used as positive controls. Negative controls were obtained by replacing the TdT with buffer on the same tissues used as positive control. Scores were attributed to the intensity of TUNEL staining as follows: absence of staining = 0; mild = 1; moderate = 2; and intense = 3.
Transmission Electron Microscopy
Small fragments of intestinal mucosa were fixed by immersion overnight at 4°C in a solution of 5% glutaraldehyde and 4% paraformaldehyde in 0.1 M sodium cacodylate buffer. Tissues were then washed 3 times with 0.1 M sodium cacodylate buffer and postfixed for 2 hours at 4°C in 1% osmium tetroxide in 0.1 M sodium cacodylate buffer. For transmission electron microscopy, the samples were stained overnight at 4°C in a saturated uranyl acetate solution. Tissues were dehydrated in a graduated series of ethanol solutions and propylene oxide and embedded in Epon Araldite. Sections (0.5 μm) were stained with toluidine blue and examined under a light microscope for selection of the microscopic fields. The blocks were trimmed, and thin sections (60–90 nm) were cut, mounted onto copper grids, stained with uranyl acetate and lead citrate, and examined with a Philips CM120 Biotwin Lens transmission electron microscope (Eindhoven, The Netherlands).
Statistical Analysis
Semiquantitative data were analyzed by the nonparametric method of Kruskal-Wallis. CFU numbers underwent logarithmic transformation prior to analysis of variance (ANOVA) and mean comparisons using the Tukey test. Selected sets of data were subjected to Pearson’s correlation analysis. Data are presented as mean ± standard error of the mean. P values were considered significant when P < .05. Statistical analyses were performed using the Graphpad Instat software 3.05 (Graphpad Software, Inc., San Diego, CA).
Results
S. Typhimurium-Induced Intestinal Histopathology
All animals inoculated with SIV became infected as demonstrated by quantitative RT-PCR. Plasma viral loads of SIV-infected animals are demonstrated in Fig. 1. Histopathology scores for hemorrhage, cell death, blunting of villi, epithelial loss, edema, and neutrophil infiltration are presented in Fig. 2. The combined histopathology scores were similar between SIV-infected and control macaques throughout the time course of infection (Fig. 2). As expected, the invasion-deficient mutant (strain ZA21) was less pathogenic (ie, induced a lower combined histopathology score) compared with the wild-type S. Typhimurium (strain IR715) (Fig. 2). A marked diffuse neutrophilic infiltration was observed at 5 and 8 hours post-inoculation (hpi), which was associated with other secondary inflammatory changes such as edema and blunting of villi (Figs. 3, 4). Histopathological changes, particularly neutrophil infiltration, correlated well with bacterial loads (correlation of score of neutrophil infiltration and log of CFU numbers: r = 0.7148; P < .0001) and fluid accumulation (r = 0.6019; P < .0001) in the lumen of the intestinal loops. Bacterial numbers were consistently higher in the intestinal mucosa of loops infected with wild-type S. Typhimurium (strain IR715) compared with the mucosa of loops inoculated with the invasion-deficient mutant (strain ZA21), in both control and SIV-infected macaques (Fig. 5). Fluid accumulation increased over the time course of infection, with larger volumes of fluid accumulating in loops infected with the wild-type strain (strain IR715) compared with loops infected with the invasion-deficient mutant (strain ZA21). Loops inoculated with sterile LB broth accumulated no fluid. SIV-infected macaques tended to accumulate more fluid in response to S. Typhimurium infection than non–SIV-infected macaques, but this difference was not significant (Fig. 6).
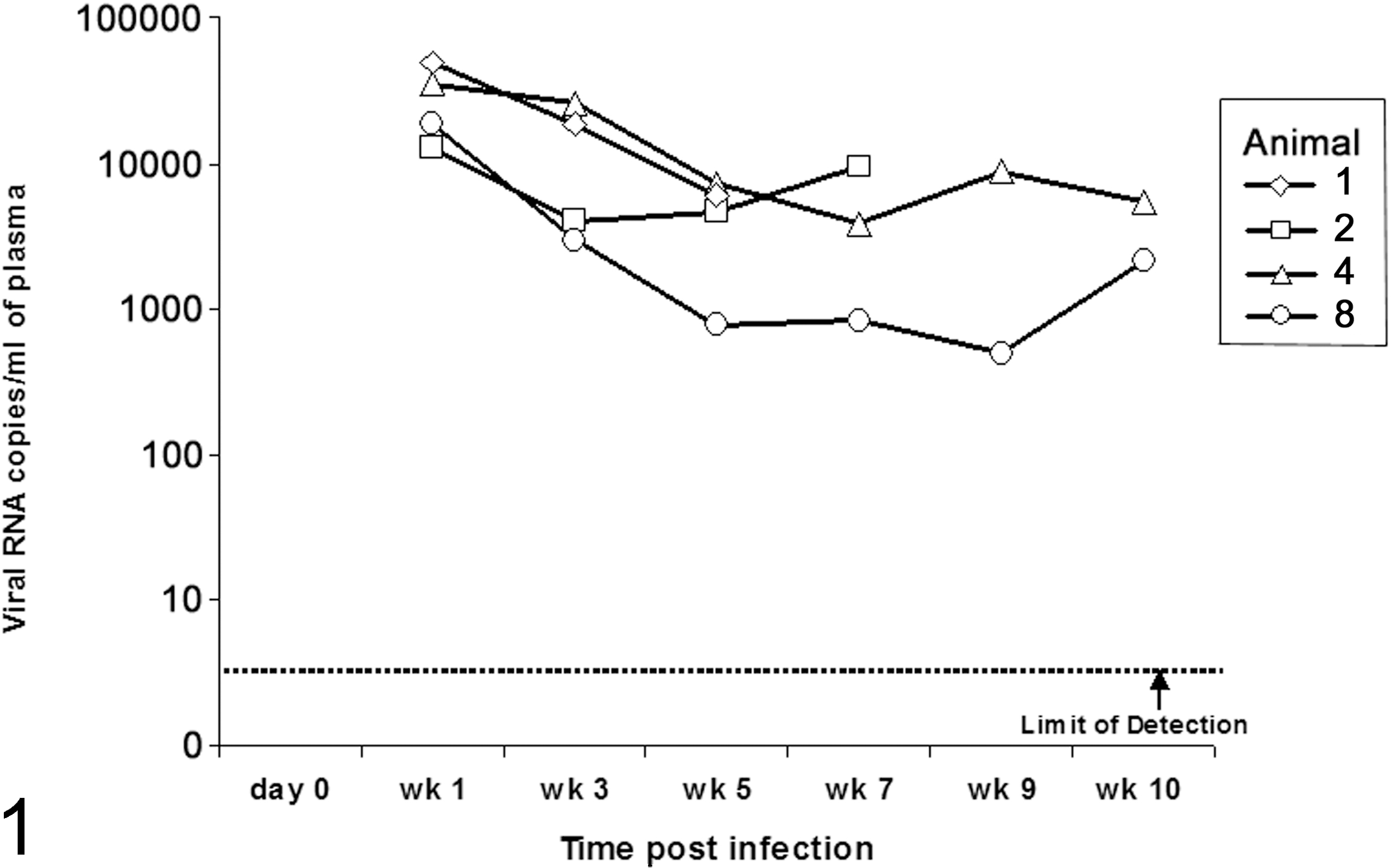
Plasma viral loads of SIV-infected rhesus macaques, animal Nos. 1, 2, 4, and 8, based on quantitative RT-PCR as determined in (19).
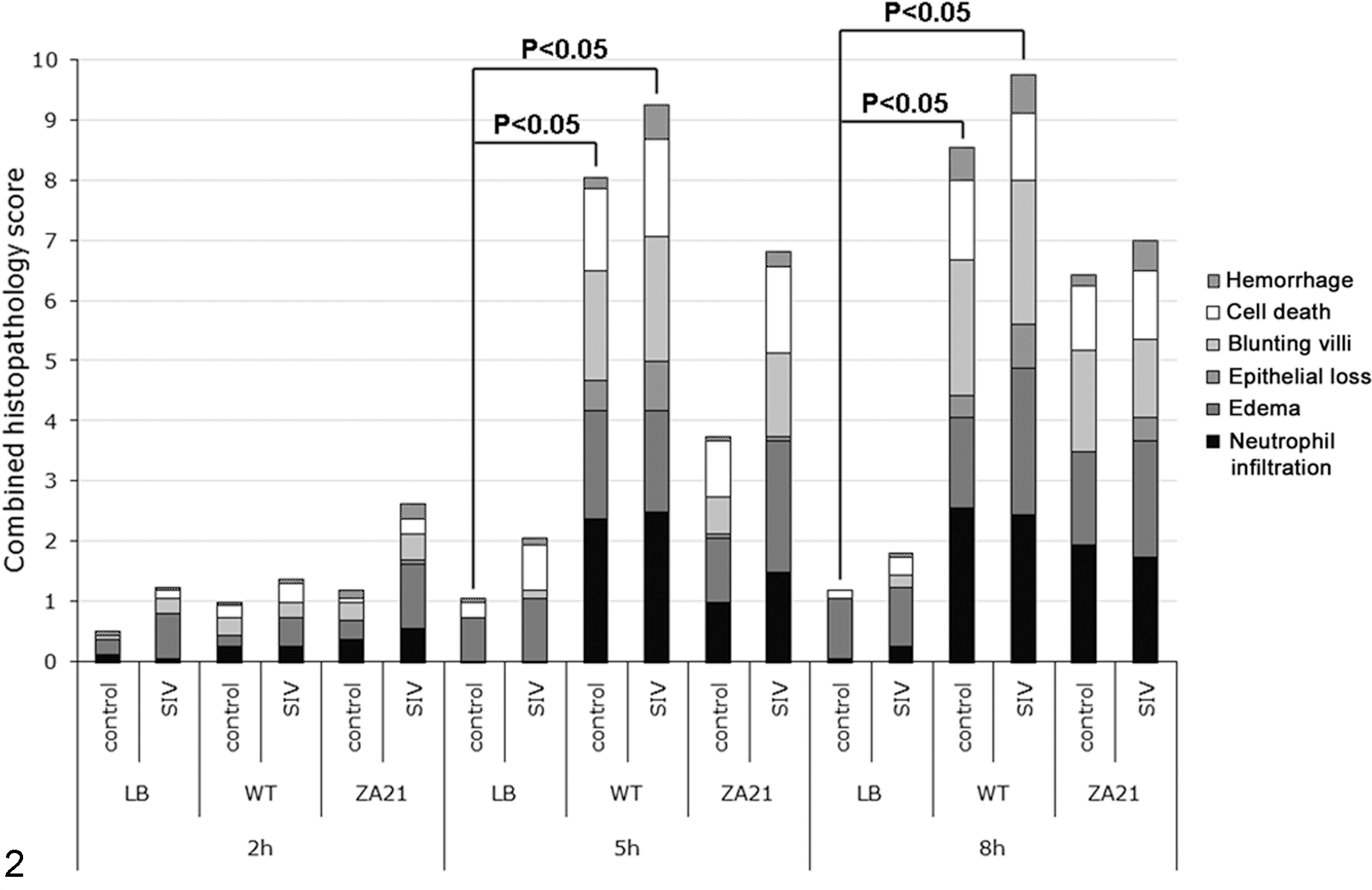
Combined histopathology scores. The scores for histologic parameters (0 for absent or 1–3 for mild to severe, respectively) were added, and the data shown are the average combined score from the 4 macaques in each group. Macaques were SIV-infected (n = 4) or control (n = 4). Intestinal loops were inoculated with sterile broth (LB), wild-type S. Typhimurium (WT), or the noninvasive mutant of S. Typhimurium (ZA21). Combined scores were compared by the Kruskal-Wallis test, and statistically significant differences are indicated (P < .05).
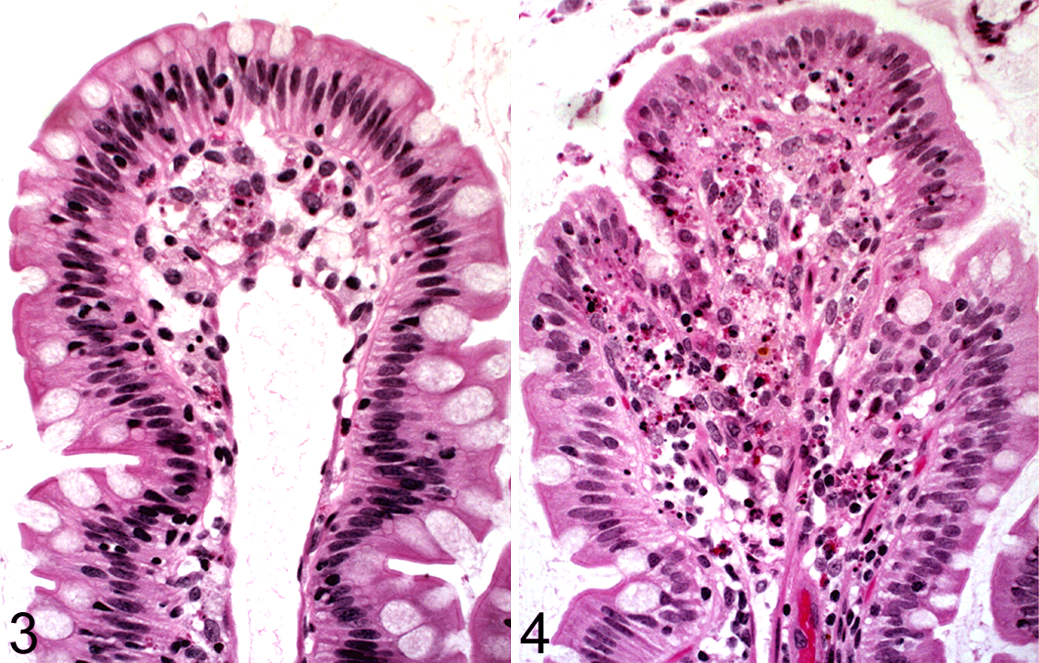
Small intestine; rhesus macaque, animal No. 6 (not infected with SIV) at 5 hours post-infection of the intestinal loop with sterile LB broth (no infection of the intestinal loop with S. Typhimurium). Intestinal villus with no significant histological changes. HE.
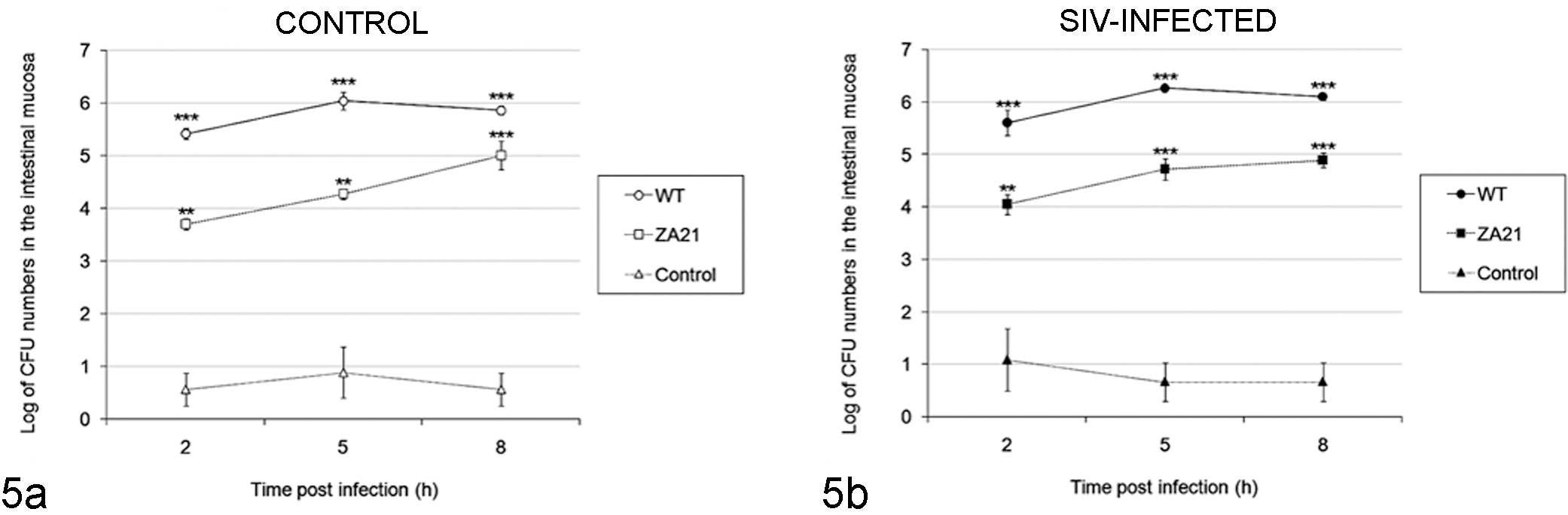
Bacterial numbers in intestinal loops of rhesus macaques in response to inoculation with S. Typhimurium. (a) SIV-noninfected (control) macaques; (b) SIV-infected macaques. Intestinal loops were inoculated with sterile broth (control), wild-type S. Typhimurium (WT), or the noninvasive mutant of S. Typhimurium (ZA21). CFU numbers that are statistically different from those in control loops not infected with S. Typhimurium are indicated (**P < .01; ***P < .001; ANOVA with Tukey’s comparison of means).
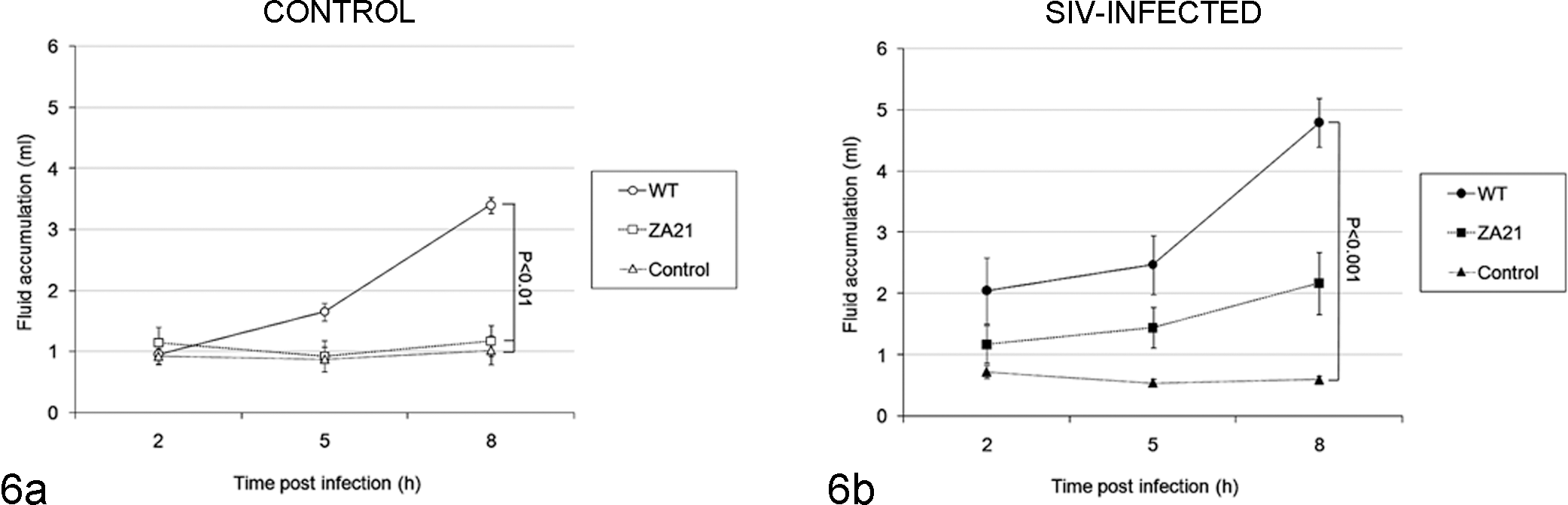
Fluid accumulation in intestinal loops of rhesus macaques in response to inoculation with S. Typhimurium. (a) SIV-noninfected (control) macaques; (b) SIV-infected macaques. Intestinal loops were inoculated with sterile broth (control), wild-type S. Typhimurium (WT), or the noninvasive mutant of S. Typhimurium (ZA21). Statistically significant differences are indicated (ANOVA with Tukey’s comparison of means).
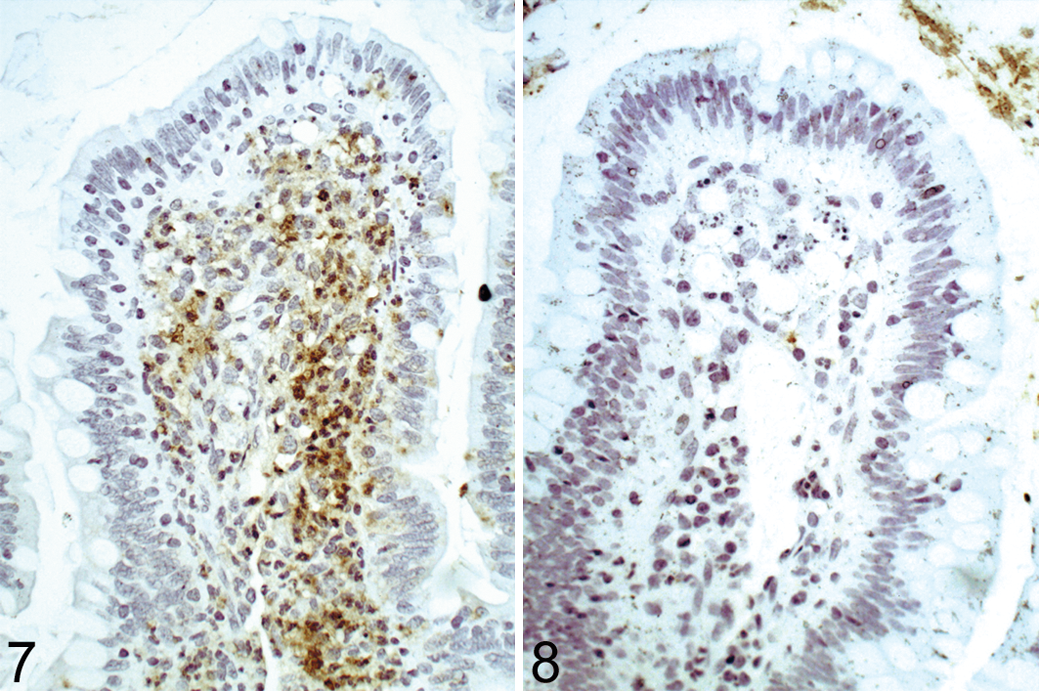
Small intestine; rhesus macaque, animal No. 7 (not infected with SIV) at 8 hours post-infection of the intestinal loop with wild-type S. Typhimurium (strain IR715). Intestinal villus with intense and diffuse immunolabeling of Salmonella sp. in the lamina propria. Polyclonal antibody against S. Typhimurium lipopolysaccharide (LPS), counterstained with hematoxylin.
Tissue Distribution of S. Typhimurium
The kinetics of tissue invasion were evaluated by immunohistochemical labeling of S. Typhimurium. Immunolabeled wild-type S. Typhimurium was observed in the epithelium and lamina propria at the tip of the villi at 2 hpi, progressing toward deeper lamina propria at 5–8 hpi and reaching the submucosa and lymphoid follicles at 8 hpi in animal No. 5 (Table 2), in which immunolabeled S. Typhimurium were observed in lymphoid follicles at earlier time points. Similar kinetics of invasion were observed in SIV-infected macaques, although the number of animals with immunolabeled S. Typhimurium tended to be higher in deeper tissues in these animals compared with control macaques (Table 2). As expected, the invasion-deficient mutant (strain ZA21) remained mostly restricted to the lumen and apical epithelial surface throughout the time course of the study (Table 2, Figs. 7, 8).
Summary of Immunolabeling of Salmonella enterica Serotype Typhimuriu m in Ligated Intestinal Loops of Uninfected Controls or SIV-Infected Rhesus Macaques Inoculated With the Wild-Type Strain or an Invasion-Defective Mutant (ZA21)
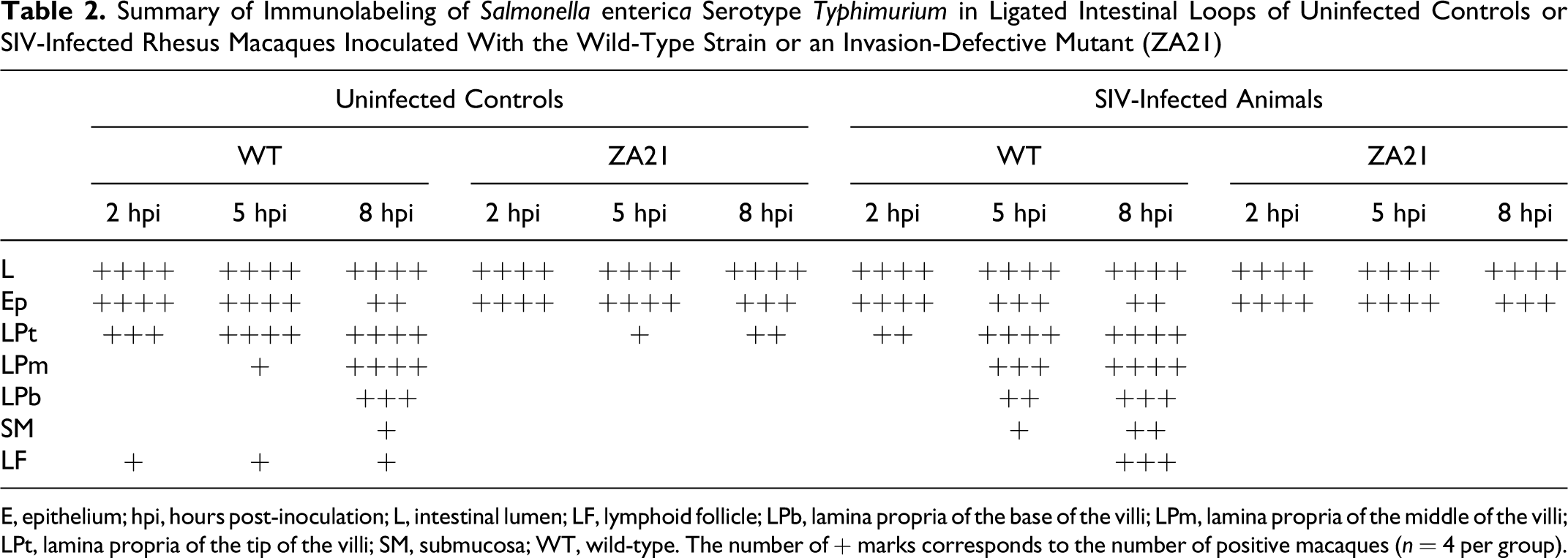
E, epithelium; hpi, hours post-inoculation; L, intestinal lumen; LF, lymphoid follicle; LPb, lamina propria of the base of the villi; LPm, lamina propria of the middle of the villi; LPt, lamina propria of the tip of the villi; SM, submucosa; WT, wild-type. The number of + marks corresponds to the number of positive macaques (n = 4 per group).
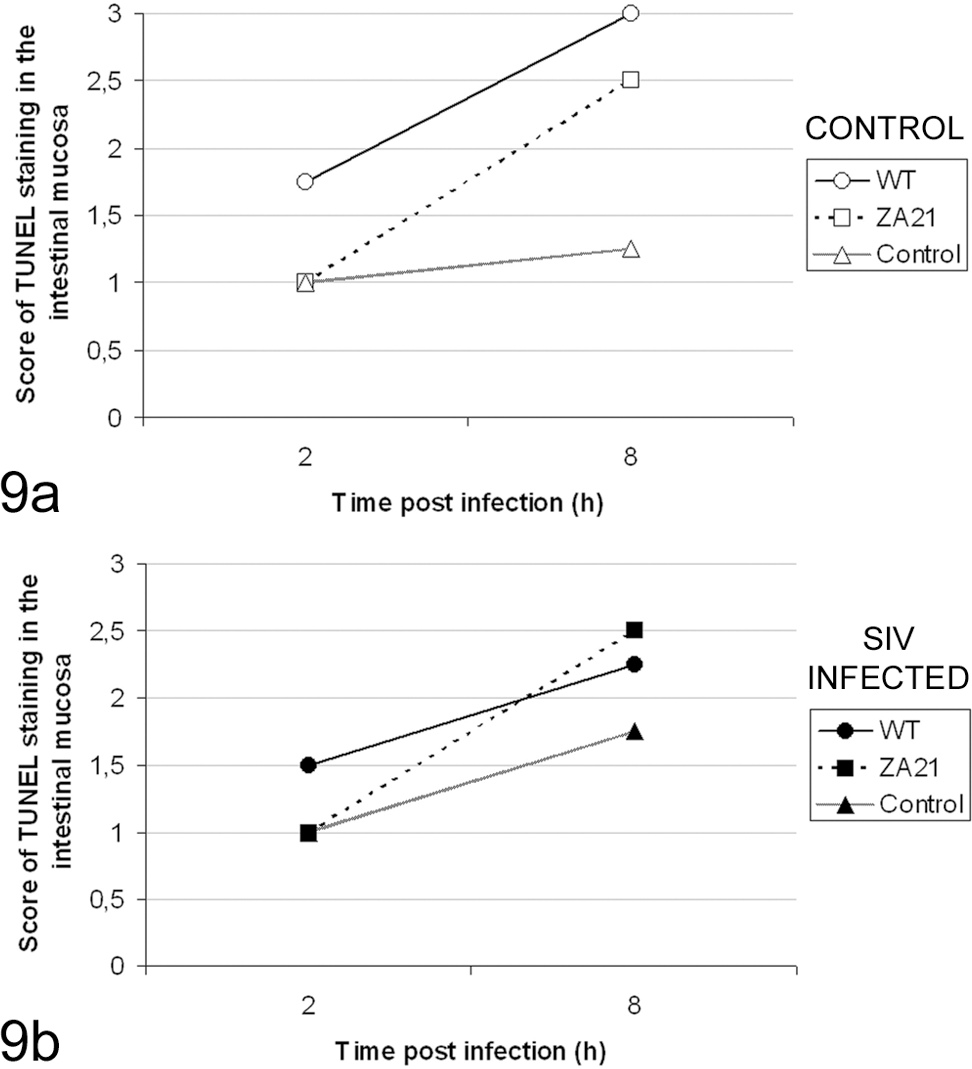
Average scores of TUNEL staining in the intestinal mucosa of loops from control (a, n = 4) or SIV-infected (b, n = 4) rhesus macaques inoculated with S. Typhimurium, either wild-type (strain IR715) or the noninvasive mutant (strain ZA21), and uninfected controls. There were no statistically significant differences (P > .05).
S. Typhimurium-Induced Cell Death
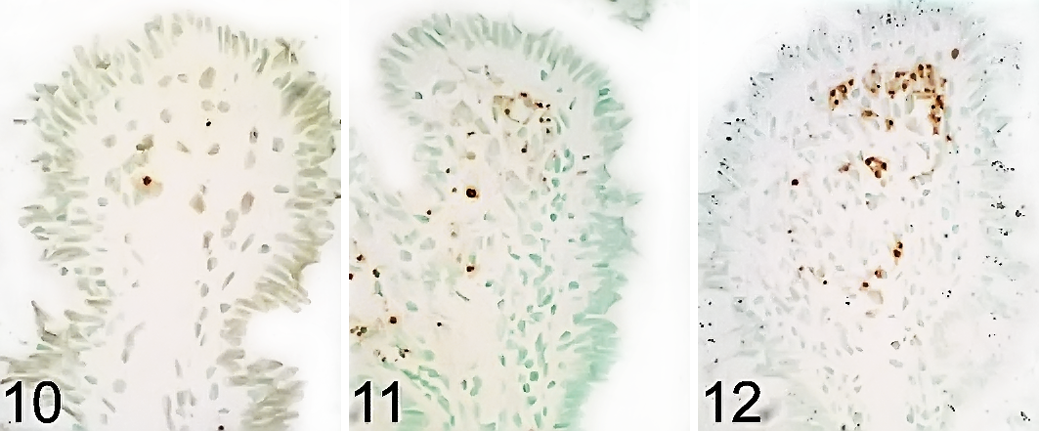
In this study, cells in the intestinal mucosa were labeled by the TUNEL technique, and morphological features of apoptotic cell death were examined by transmission electron microscopy. Wild-type S. Typhimurium (IR715) infection resulted in an increase (P < .05) in cell death in the intestinal mucosa as assessed by TUNEL (Fig. 9). Most of the TUNEL-positive cells localized to the lamina propria (Figs. 10–12). The intensity of TUNEL labeling was similar between control and SIV-infected macaques. A significant correlation was observed between the scores for neutrophil infiltration and TUNEL labeling in the intestinal mucosa (r = 0.7103; P < .0001). Ultrastructurally, bacteria were observed within the cytosol of macrophages in the lamina propria as well as within apoptotic bodies (Fig. 13).
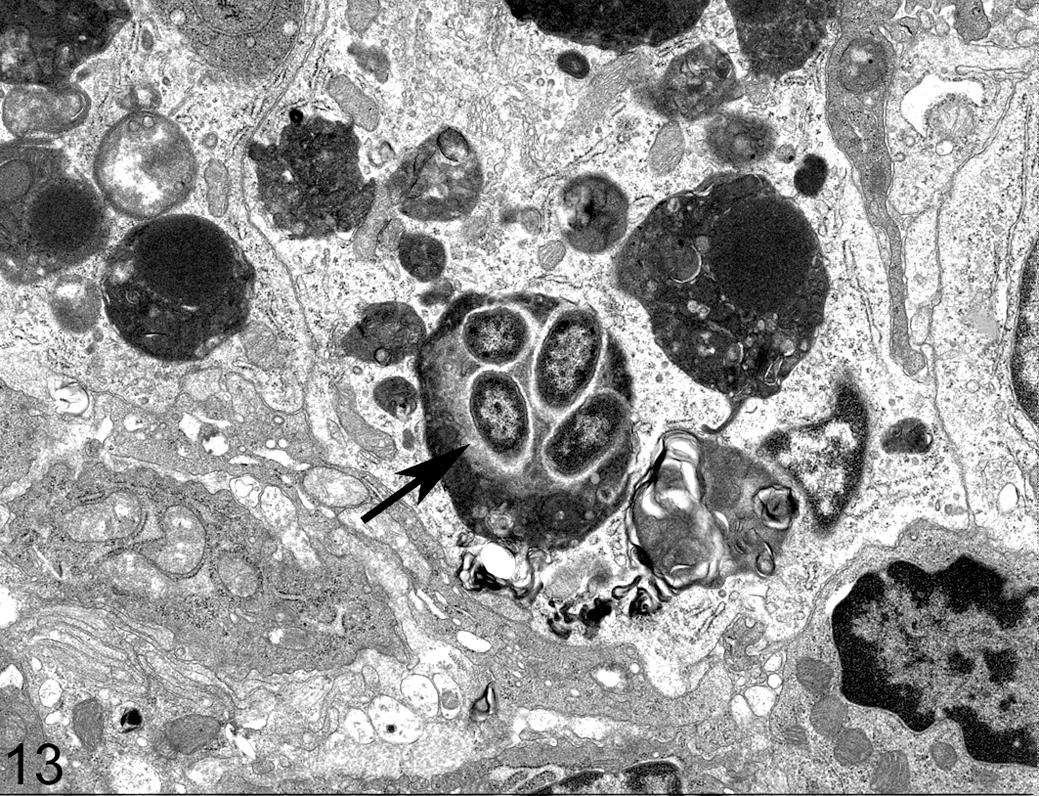
Small intestine, lamina propria; rhesus macaque, animal No. 1 (SIV-infected macaque) at 5 hours post-infection of the intestinal loop with wild-type S. Typhimurium (strain IR715). Bacterial organisms with morphology compatible with Salmonella sp., characterized by multiple bacilli within an apoptotic body in the lamina propria (arrow). Transmission electron microscopy.
Discussion
This is the first in vivo study providing evidence for a direct interaction of S. Typhimurium with cells undergoing cell death in a relevant animal model for NTS. Previous studies have demonstrated the ability of S. Typhimurium to induce macrophage cell death in vitro.4,5,6,14,17,25,31,32 This effect is dependent on a functional SPI-1 encoded type III secretion system, and it is mediated by caspase 1. However, it only recently became clear that activation of caspase 1 is mediated by Ipaf upon recognition of flagellin that is translocated into the host cell cytosol through the SPI-1 type III secretion system.7,15,30,31 It has also been recently reported that S. Typhimurium can induce caspase 1–mediated death of stromal cells. 18 Considering that the proinflammatory cytokines IL-1 and IL-18 are substrates of caspase 1, being cleaved and activated during the caspase 1–mediated macrophage cell death, this mechanism of Salmonella-induced macrophage death is thought to be a significant proinflammatory mechanism during S. Typhimurium infection, and therefore it has been named pyroptosis, which can be triggered not only by S. Typhimurium but also by several infectious or noninfectious pathological stimuli. 2
The role of caspase 1 during S. Typhimurium infection has been studied in the mouse.16,20 However, this animal model has limitations for studying enterocolitis since mice develop a systemic disease but not diarrhea in response to S. Typhimurium infection 28 and the induction of enterocolitis in mice requires artificial conditions in which the intestinal flora is depleted prior to infection. 1 Previous attempts to demonstrate the occurrence of this phenomenon in the calf, a relevant animal model for Salmonella-induced diarrhea, did not provide evidence for S. Typhimurium–induced cell death in vivo. 26 A recent study demonstrates that recognition of flagellin by the host innate immune system contributes to a proinflammatory response in the calf, which is a suitable model for NTS-induced diarrhea, but not in the mouse.33, 34
Our results support the notion that the presence of bacteria in proximity to the host target cell is not the only mechanism responsible for cell death in the intestinal mucosa during Salmonella infection. This conclusion is supported by the immunohistochemistry data that have proven to be appropriate for studying tissue distribution of Salmonella in the intestine. 21 This approach demonstrated that an invasion-deficient S. Typhimurium mutant (strain ZA21) 35 induced an increase in TUNEL-labeled cells in the intestinal mucosa despite the fact that it remained mostly associated with the epithelial layer throughout the course of infection, with minimal invasion of the lamina propria. However, considering the association of S. Typhimurium with cells undergoing cell death in the lamina propria and the proinflammatory nature of this mechanism as demonstrated in cultured macrophages,7,15 it is likely that Salmonella-induced cell death plays a proinflammatory role in vivo in this nonhuman primate model.
Nonhuman primates have been used in the past as animal models for Salmonella infection. 12 The adaptation of the intestinal ligated loop model to rhesus macaques provided a useful animal model for studying enteric pathogens, resulting in a significant reduction in the number of animals required for a given experiment. The pattern of host response observed in this study characterized by an acute inflammatory response with abundant neutrophilic infiltration is similar to that previously described in the calf in which the ileal ligated loop model has been extensively used.26,27
Some of our observations are consistent with the idea that SIV infection impairs the integrity of the intestinal epithelial barrier, although the differences between noninfected and SIV-infected macaques in this study were not statistically significant. First, S. Typhimurium elicited increased fluid accumulation (a surrogate measure of diarrhea) in the intestinal lumen of SIV-infected compared with control animals. Second, spreading of wild-type S. Typhimurium into the deeper layers of the mucosa was more evident in SIV-infected than in control animals. These observations are in good agreement with previous studies demonstrating that SIV infections result in an impairment of intestinal mucosal integrity, 11 which may favor bacterial translocation from the intestinal lumen. 3 Previous work suggests that the mechanism for SIV-mediated defects of mucosal barrier function is a depletion of TH17 cells. 19 SIV-induced changes in intestinal barrier and absorption are not associated with intestinal bacterial overgrowth, 29 which could interfere with the experimental model employed in this study.
In conclusion, here we describe the intestinal host response to S. Typhimurium. This study also provides the first in vivo evidence of the association of S. Typhimurium with cells undergoing cell death in the intestinal mucosa in a relevant biological model.
Footnotes
Acknowledgements
We thank G. Adamson (Electron Microscopy Laboratory, Department of Medical Pathology and Laboratory Medicine–UC Davis) for technical assistance.
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
Work in RLS’s laboratory is supported by CNPq (Conselho Nacional de Desenvolvimento Científico e Tecnológico, Brazil) and FAPEMIG (Fundação de Amparo a Pesquisa do Estado de Minas Gerais, Brazil). RLS, TAP, and MNX are recipients of fellowships from CNPq (Brazil). RLS is a fellow of the John Simon Guggenheim Memorial Foundation. Work in AJB’s laboratory is supported by Public Health Service Grants AI040124, AI044170, AI073120, AI076246 and AI079173.
