Abstract
Purpose
Patients with HER2+ breast cancer (BC) can receive pertuzumab/trastuzumab (PT) subcutaneous (SC) or intravenous (IV) with similar efficacy. This study explores patients and healthcare professionals (HCPs) perceptions post-PT-SC implementation, alongside a pharmacoeconomic analysis.
Methods
This multicentre observational study included semi-structured interviews with HER2+ BC patients (SC or IV) and HCPs from six Lombardy (Italy) BC units plus a pharmacoeconomic analysis of organisational and cost impact. Data were analysed using descriptive statistics and thematic analysis to identify key themes: safety perception, treatment comfort, and trust issues.
Results
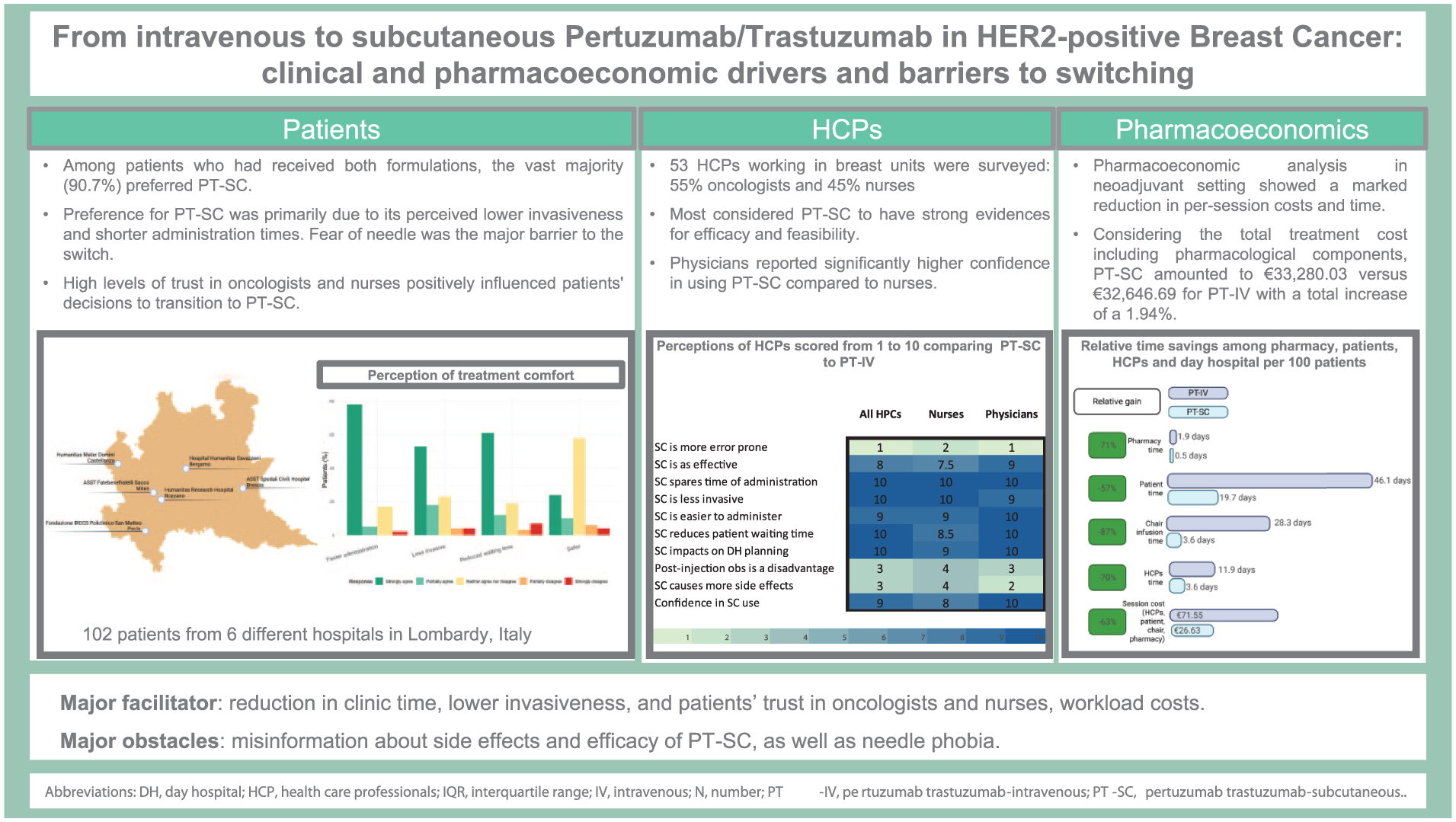
Among 102 patients, 10.8% received only IV, 12.7% only SC, and 76% both; 3.8% of SC patients discontinued due to side effects. Trust scores were high for oncologists and nurses, positively influencing switching. SC was perceived as quicker (81.4%), less invasive (69.8%) and timesaving (71.2%), with over half (56.9%) perceiving equal safety. Among patients exposed to both, 90.7% preferred SC due to lower invasiveness. SC was perceived to be associated with fewer gastrointestinal side effects but more local reactions. IV-preferring patients cited fear of side effects, reduced efficacy, and lack of HCP recommendation. Most HCPs rated SC as faster and workflow-improving; observation and error risk were not major barriers. Non-drug costs were 63% lower with SC, while total treatment costs were comparable (+1.9%).
Conclusion
Both patients and HCPs showed high acceptance of PT-SC, offering clear benefits in time, cost, and workflow efficiency. High trust in oncologists and nurses supported its adoption. Time savings may enhance hospital efficiency and patient experience.
Get full access to this article
View all access options for this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
